micro RNA and lung cancer Gabriella Sozzi Lung

micro. RNA and lung cancer Gabriella Sozzi

Lung Cancer Screening by LDCT : critical issues Recent clinical trials results (DANTE, 2009, DLCST , 2012, MILD 2012; ~10. 000 subjects overall; NLST 2011, 53. 454 persons), indicate that LDCT screening in high risk subjects may reduce lung cancer mortality. The high false positive rates of LDCT, leading to multiple screening rounds, over-diagnosis, unnecessary (harmful? )diagnostic follow –up and costs underscore the need for non-invasive complementary biomarkers for standardized use.
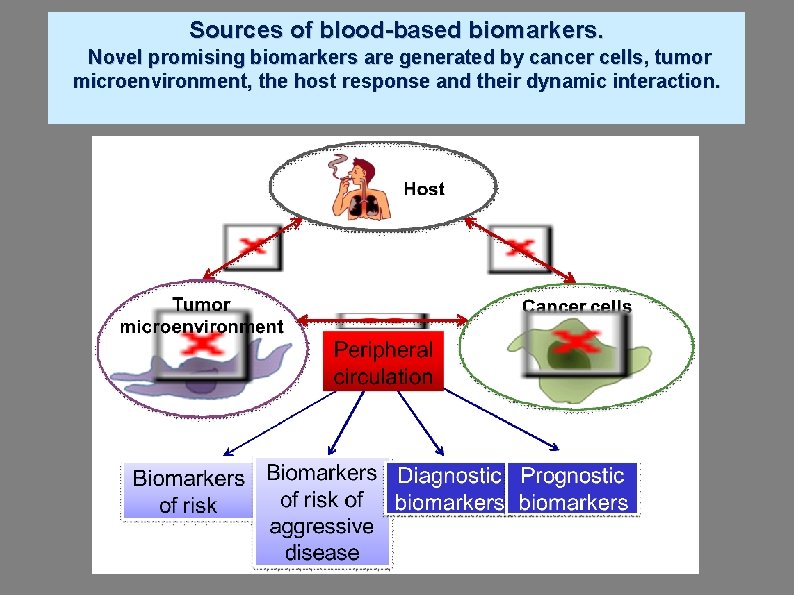
Sources of blood-based biomarkers. Novel promising biomarkers are generated by cancer cells, tumor microenvironment, the host response and their dynamic interaction.

Plasma biomarkers studies at INT (Milan) Microsatellite changes, KRAS and p 53 mutation in casecontrol series (Sozzi G. Cancer Research 1999; Andriani F. IJC 2004) Methylation markers plasma samples in case-control series (Bearzatto A. Clin Cancer Res. 2002 ) Circulating plasma DNA (h. TERT) quantification in casecontrol series and in pilot INT-IEO and MILD screening trial (Sozzi G. , Can Res 2001; Sozzi G. , J Clin Oncol. 2003; Sozzi G. , AJRCCM 2009; Roz L. Lung Cancer 2009) Highthroughput micro. RNAs expression profiles in plasma samples in independent screening trials (INT-IEO, MILD) (Boeri M. et al. , PNAS 2011; Sozzi G. Cell Cycle 2011; Boeri M. et al. , Cancer J 2012)
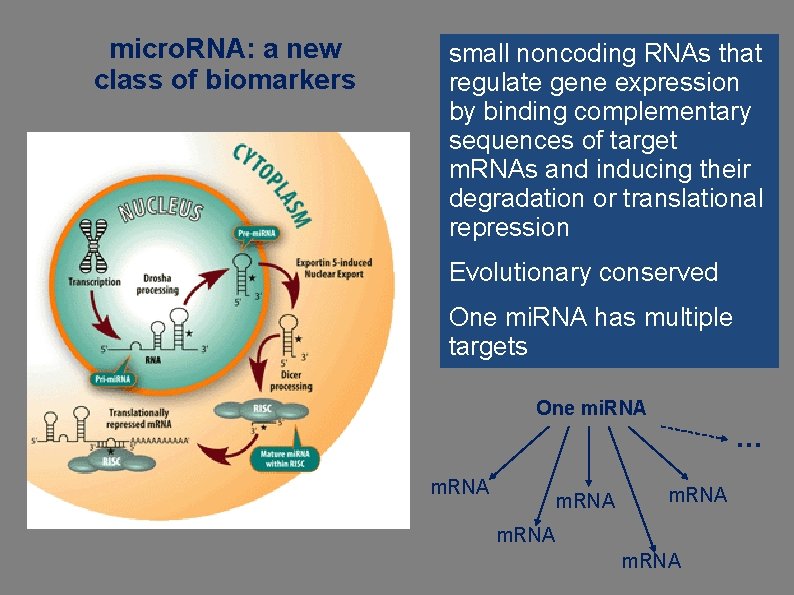
micro. RNA: a new class of biomarkers small noncoding RNAs that regulate gene expression by binding complementary sequences of target m. RNAs and inducing their degradation or translational repression Evolutionary conserved One mi. RNA has multiple targets One mi. RNA … m. RNA
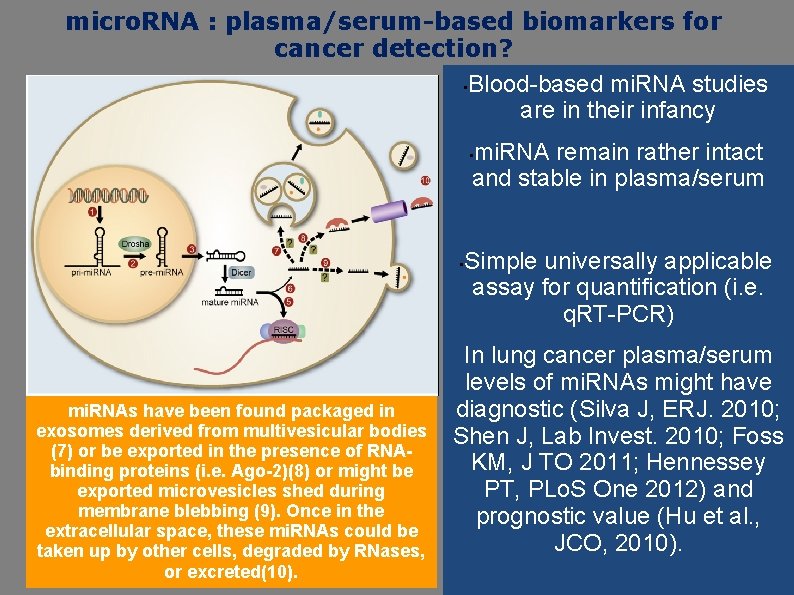
micro. RNA : plasma/serum-based biomarkers for cancer detection? • Blood-based mi. RNA studies are in their infancy mi. RNA remain rather intact and stable in plasma/serum • • mi. RNAs have been found packaged in exosomes derived from multivesicular bodies (7) or be exported in the presence of RNAbinding proteins (i. e. Ago-2)(8) or might be exported microvesicles shed during membrane blebbing (9). Once in the extracellular space, these mi. RNAs could be taken up by other cells, degraded by RNases, or excreted(10). Simple universally applicable assay for quantification (i. e. q. RT-PCR) In lung cancer plasma/serum levels of mi. RNAs might have diagnostic (Silva J, ERJ. 2010; Shen J, Lab Invest. 2010; Foss KM, J TO 2011; Hennessey PT, PLo. S One 2012) and prognostic value (Hu et al. , JCO, 2010).

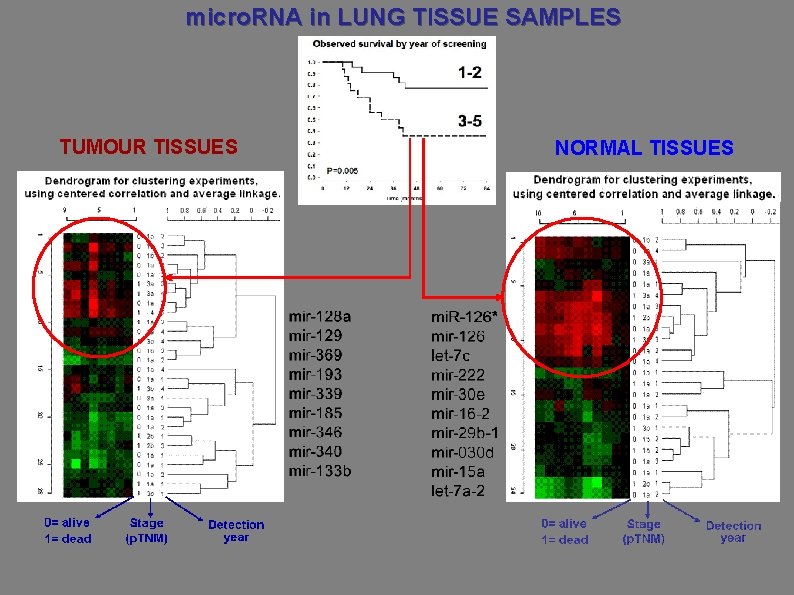
micro. RNA in LUNG TISSUE SAMPLES TUMOUR TISSUES NORMAL TISSUES
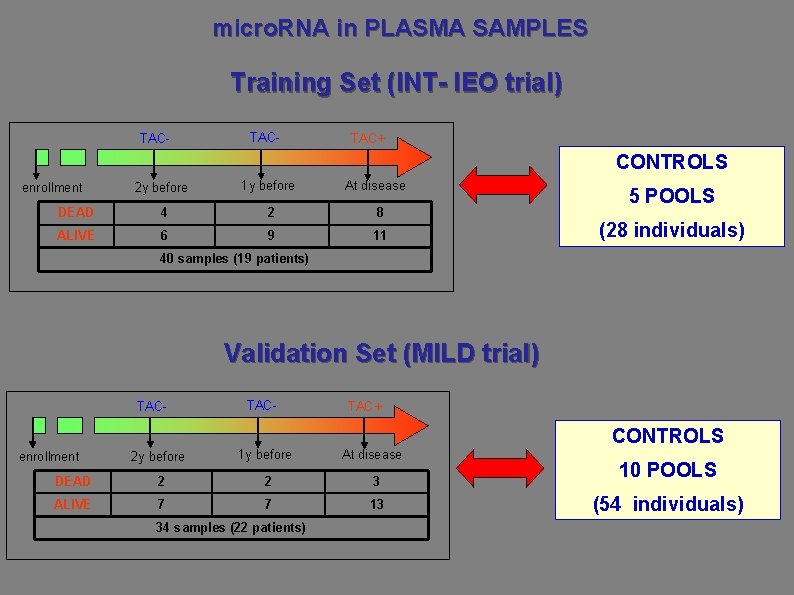
micro. RNA in PLASMA SAMPLES Training Set (INT- IEO trial) TAC- TAC+ CONTROLS At disease 2 y before 1 y before DEAD 4 2 8 ALIVE 6 9 11 enrollment 5 POOLS (28 individuals) 40 samples (19 patients) Validation Set (MILD trial) TAC- TAC+ CONTROLS 2 y before 1 y before DEAD 2 2 3 ALIVE 7 7 13 enrollment 34 samples (22 patients) At disease 10 POOLS (54 individuals)
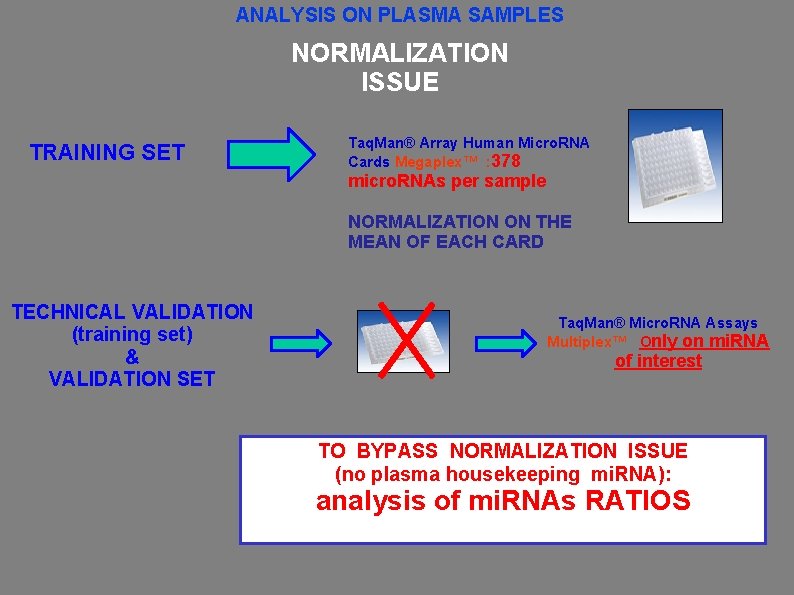
ANALYSIS ON PLASMA SAMPLES NORMALIZATION ISSUE TRAINING SET Taq. Man® Array Human Micro. RNA Cards Megaplex™ : 378 micro. RNAs per sample NORMALIZATION ON THE MEAN OF EACH CARD TECHNICAL VALIDATION (training set) & VALIDATION SET Taq. Man® Micro. RNA Assays Multiplex™ Only on mi. RNA of interest TO BYPASS NORMALIZATION ISSUE (no plasma housekeeping mi. RNA): analysis of mi. RNAs RATIOS RECIPROCAL RATIOS AMONG mi. RNAs
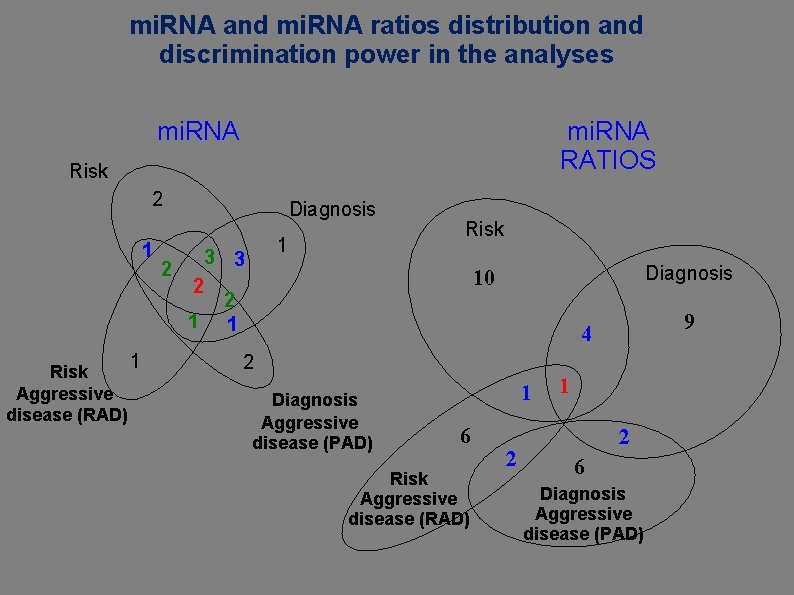
mi. RNA and mi. RNA ratios distribution and discrimination power in the analyses mi. RNA RATIOS Risk 2 1 2 Diagnosis 2 1 Risk Aggressive disease (RAD) 1 1 3 3 Risk Diagnosis 10 2 1 9 4 2 Diagnosis Aggressive disease (PAD) 1 6 Risk Aggressive disease (RAD) 2 1 2 6 Diagnosis Aggressive disease (PAD)
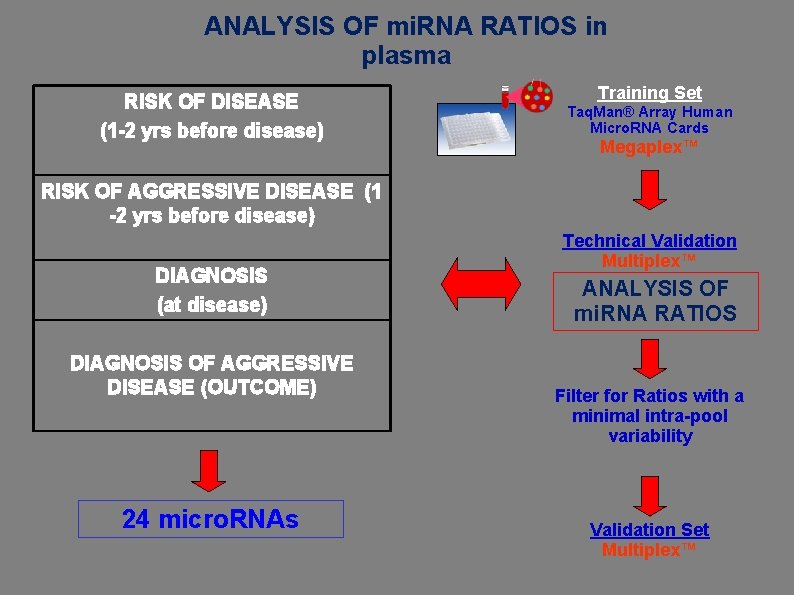
ANALYSIS OF mi. RNA RATIOS in plasma RISK OF DISEASE (1 -2 yrs before disease) Training Set Taq. Man® Array Human Micro. RNA Cards Megaplex™ RISK OF AGGRESSIVE DISEASE (1 -2 yrs before disease) DIAGNOSIS (at disease) DIAGNOSIS OF AGGRESSIVE DISEASE (OUTCOME) 24 micro. RNAs Technical Validation Multiplex™ ANALYSIS OF mi. RNA RATIOS Filter for Ratios with a minimal intra-pool variability Validation Set Multiplex™
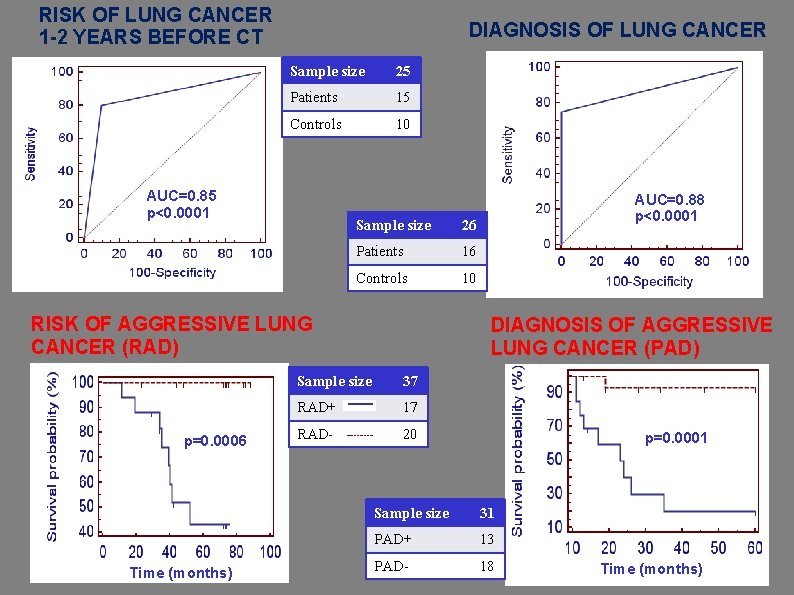
RISK OF LUNG CANCER 1 -2 YEARS BEFORE CT DIAGNOSIS OF LUNG CANCER Sample size 25 Patients 15 Controls 10 AUC=0. 85 p<0. 0001 Sample size 26 Patients 16 Controls 10 RISK OF AGGRESSIVE LUNG CANCER (RAD) p=0. 0006 Time (months) AUC=0. 88 p<0. 0001 DIAGNOSIS OF AGGRESSIVE LUNG CANCER (PAD) Sample size 37 RAD+ 17 RAD- 20 p=0. 0001 Sample size 31 PAD+ 13 PAD- 18 Time (months)
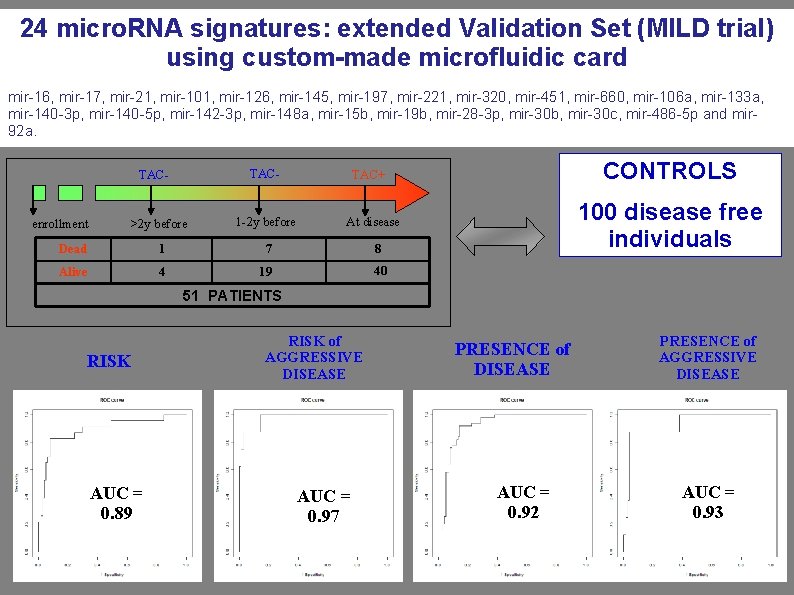
24 micro. RNA signatures: extended Validation Set (MILD trial) using custom-made microfluidic card mir-16, mir-17, mir-21, mir-101, mir-126, mir-145, mir-197, mir-221, mir-320, mir-451, mir-660, mir-106 a, mir-133 a, mir-140 -3 p, mir-140 -5 p, mir-142 -3 p, mir-148 a, mir-15 b, mir-19 b, mir-28 -3 p, mir-30 b, mir-30 c, mir-486 -5 p and mir 92 a. TAC- >2 y before 1 -2 y before Dead 1 7 8 Alive 4 19 40 enrollment CONTROLS TAC+ 100 disease free individuals At disease 51 PATIENTS RISK AUC = 0. 89 RISK of AGGRESSIVE DISEASE AUC = 0. 97 PRESENCE of DISEASE AUC = 0. 92 PRESENCE of AGGRESSIVE DISEASE AUC = 0. 93
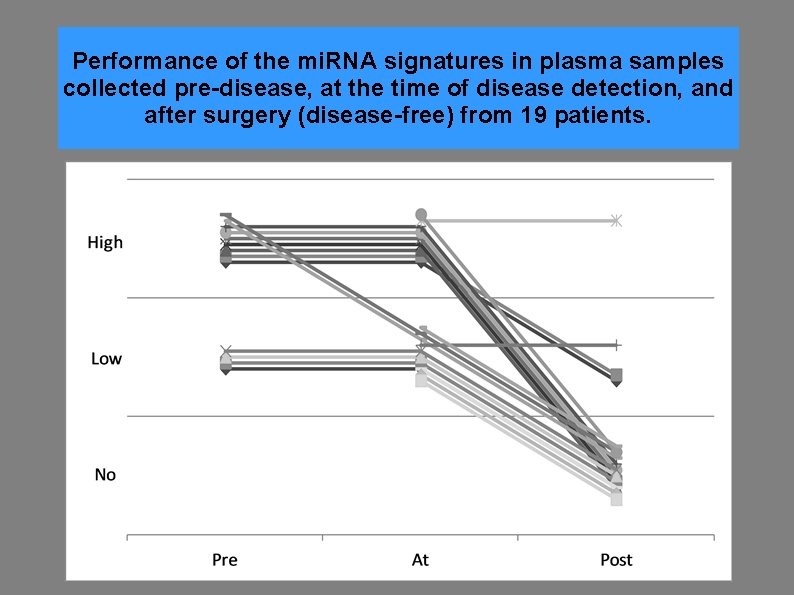
Performance of the mi. RNA signatures in plasma samples collected pre-disease, at the time of disease detection, and after surgery (disease-free) from 19 patients.
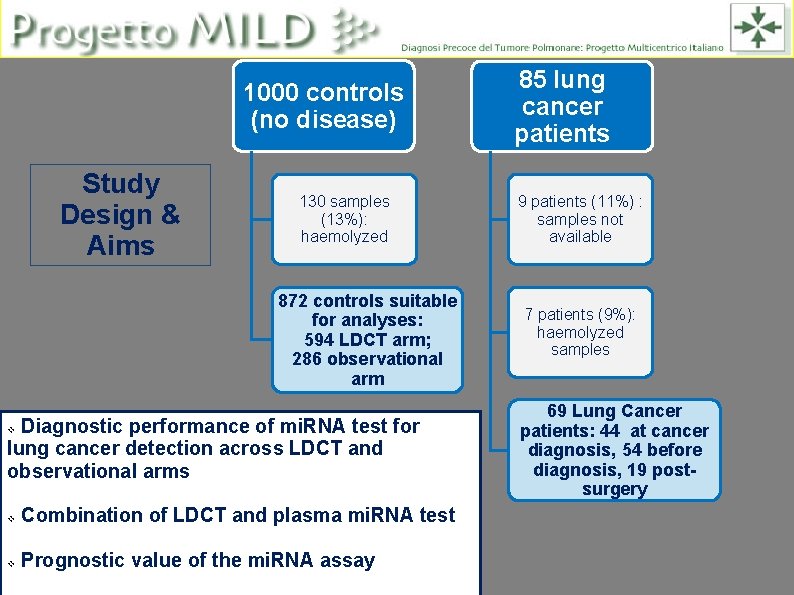
1000 controls (no disease) Study Design & Aims 130 samples (13%): haemolyzed 872 controls suitable for analyses: 594 LDCT arm; 286 observational arm Diagnostic performance of mi. RNA test for lung cancer detection across LDCT and observational arms v v Combination of LDCT and plasma mi. RNA test v Prognostic value of the mi. RNA assay 85 lung cancer patients 9 patients (11%) : samples not available 7 patients (9%): haemolyzed samples 69 Lung Cancer patients: 44 at cancer diagnosis, 54 before diagnosis, 19 postsurgery
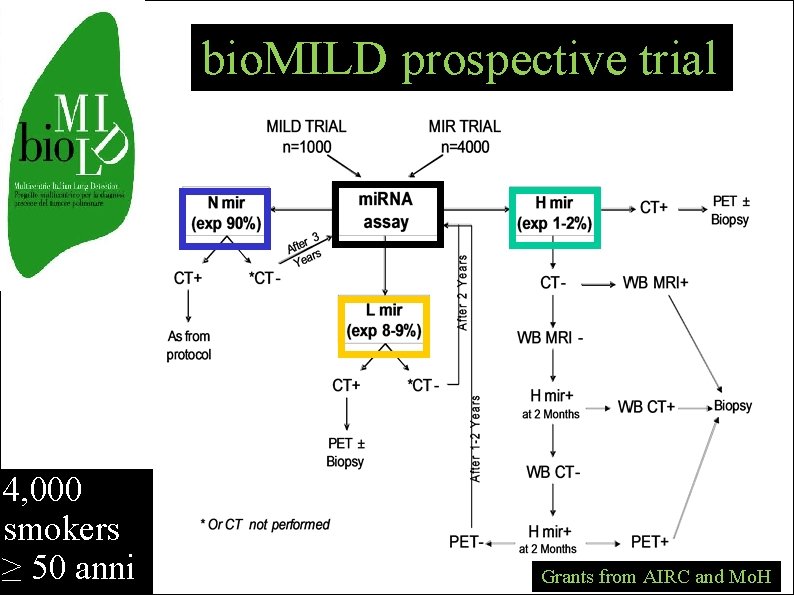
bio. MILD prospective trial 4, 000 smokers ≥ 50 anni Grants from AIRC and Mo. H

bio. MILD - 2011 biomarker-based prospective study validate mi. RNA profile combination of CT e mi. RNA CT dose according to risk Biology-based therapy Pharmacologic prevention

www. biomild. org ci servono 4000 volontari

TUMOR GENOMICS UNIT of THORACIC SURGERY Mattia Boeri Paola Suatoni Carla Verri Ugo Pastorino Davide Conte Mavis Mensah Luca Roz Federica Facchinetti Ohio State University Cancer Center, Columbus, OH, USA Carlo M. Croce Francesca Andriani Special Program “Innovative Tools for Cancer Risk Assessment and early Diagnosis”, 5 X 1000/5 per mille Supported by Italian Ministry of Heatlh Ricerca Finalizzata 20120
- Slides: 20