Mhendislik Fakltesi Kimya Mhendislii Blm KMB 236 Mesleki

Mühendislik Fakültesi Kimya Mühendisliği Bölümü KMB 236 -Mesleki İngilizce Dr. Öğr. Üyesi, İsa DEĞİRMENCİ

Global Warming KMB 236 -Mesleki İngilizce Hafta-4

What Is Global Warming? Global warming is the warming of the earth through carbon dioxide (CO 2) being pumped into the atmosphere. The greenhouse effect is the process by which radiation from a planet's atmosphere warms the planet's surface. http: //www. worldviewofglobalwarming. or g/

What’s Happening “The impacts of warming temperatures in Antarctica are likely to occur first in the northern sections of the continent, where summer temperatures approach the melting point of water, 0 degrees C. http: //www. climatehotmap. or g/antarctica. html
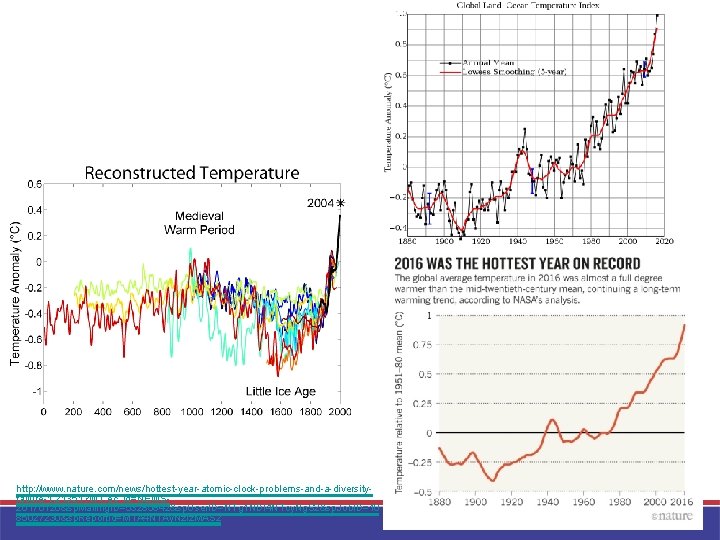
http: //www. nature. com/news/hottest-year-atomic-clock-problems-and-a-diversityfailure-1. 21351? WT. ec_id=NEWS 20170126&sp. Mailing. ID=53285842&sp. User. ID=NTg 1 NDI 4 NTcy. Ng. S 2&sp. Job. ID=10 85027230&sp. Report. Id=MTA 4 NTAy. Nz. Iz. MAS 2

Greenhouse Gases • By their percentage contribution to the greenhouse effect on Earth the five major gases are: • water vapor, 36– 70% • carbon dioxide, 9– 26% • methane, 4– 9% • ozone, 3– 7% • Nitrous oxide ? %
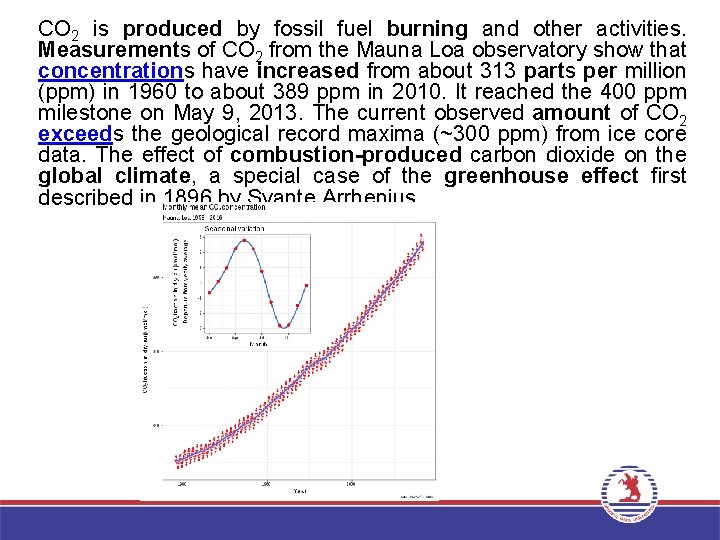
CO 2 is produced by fossil fuel burning and other activities. Measurements of CO 2 from the Mauna Loa observatory show that concentrations have increased from about 313 parts per million (ppm) in 1960 to about 389 ppm in 2010. It reached the 400 ppm milestone on May 9, 2013. The current observed amount of CO 2 exceeds the geological record maxima (~300 ppm) from ice core data. The effect of combustion-produced carbon dioxide on the global climate, a special case of the greenhouse effect first described in 1896 by Svante Arrhenius.
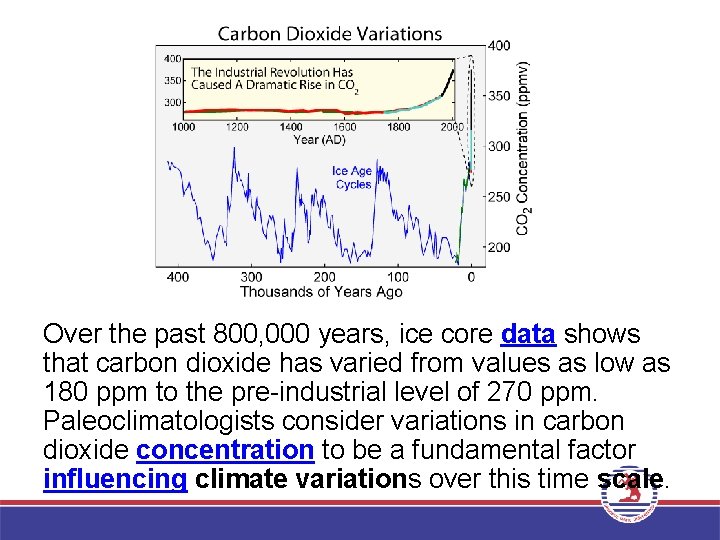
Over the past 800, 000 years, ice core data shows that carbon dioxide has varied from values as low as 180 ppm to the pre-industrial level of 270 ppm. Paleoclimatologists consider variations in carbon dioxide concentration to be a fundamental factor influencing climate variations over this time scale.

Greenhouse effect A representation of the exchanges of energy between the source (the Sun), Earth's surface, the Earth's atmosphere, and the ultimate sink outer space. The ability of the atmosphere to capture and recycle energy emitted by Earth's surface is the defining characteristic of the greenhouse effect.

What Will Happen “Rising global temperatures are expected to raise sea level, and change precipitation and other local climate conditions. Changing regional climate could alter forests, crop yields, and water supplies. It could also affect human health, animals, and many types of ecosystems. Deserts may expand into existing greenlands, and features of some of our national parks may be permanently altered. ” http: //yosemite. epa. gov/oar/globalwarming. nsf/con tent/impacts. html http: //www. nrdc. org/global. Warming/default. asp

The yellow represents warming, melting glaciers, flooding and rising of sea level. The red represents the spread of disease, earlier springs, plant and animal shifts and population changes, coral reef bleaching, downpours, heavy snowfalls, and flooding, droughts and fires. http: //www. climatehotmap. org/
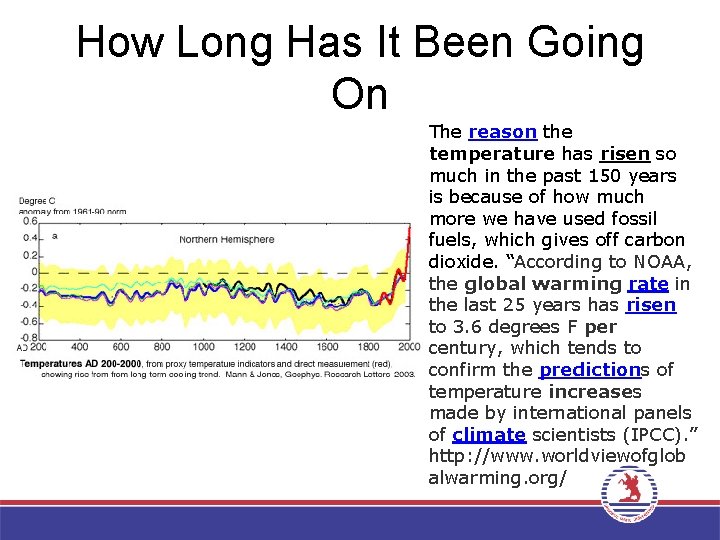
How Long Has It Been Going On The reason the temperature has risen so much in the past 150 years is because of how much more we have used fossil fuels, which gives off carbon dioxide. “According to NOAA, the global warming rate in the last 25 years has risen to 3. 6 degrees F per century, which tends to confirm the predictions of temperature increases made by international panels of climate scientists (IPCC). ” http: //www. worldviewofglob alwarming. org/
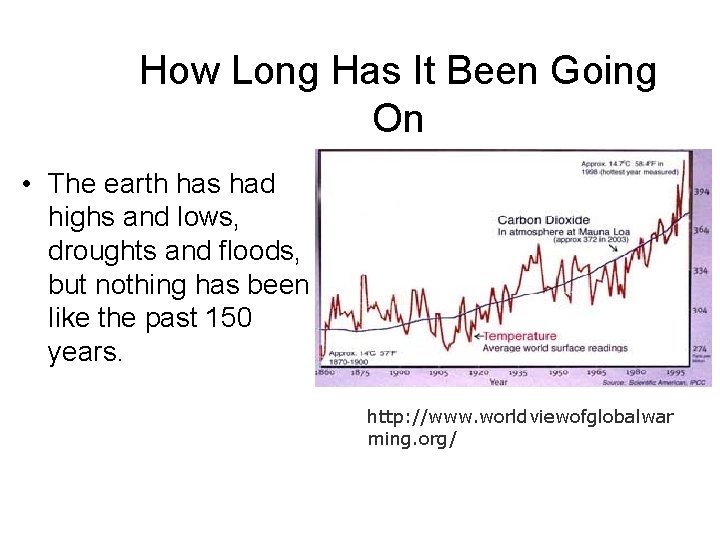
How Long Has It Been Going On • The earth has had highs and lows, droughts and floods, but nothing has been like the past 150 years. http: //www. worldviewofglobalwar ming. org/

What’s Happening to the Animals • “Penguin population decline. Adelie penguin populations have shrunk by 33 percent during the past 25 years in response to declines in their winter sea ice habitat. ”

What’s Happening to the Animals “Coral reef bleaching, the whitening of diverse invertebrate taxa, results from the loss of symbiotic zooxantheallae and/or a reduction in photosynthetic pigment concentrations in zooxanthellae residing within scleractinian corals. Coral reef bleaching is caused by various anthropogenic and natural variations in the reef environment including sea temperature, solar irradiance, sedimentation, xenobiotics, subaerial exposure, inorganic nutrients, freshwater dilution, and epizootics. Coral bleaching events have been increasing in both frequency and extent worldwide in the past 20 years. Global climate change may play a role in the increase in coral bleaching events, and could cause the destruction of major reef tracts and the extinction of many coral species. ” http: //www. marinebiology. org/coralbleaching. htm

Air Pollution Chemistry • Some air pollutants that are released into the atmosphere by man-made activities pose environmental and health risks directly. These primary pollutants include carbon monoxide, carbon dioxide, nitrogen oxides and lead, emitted from exhausts of road vehicles. • Additional impacts, however, result from the conversion of primary pollutants by a complex series of chemical reactions in the atmosphere, to secondary pollutants, many of which are potentially more harmful than their precursors. • Since much of the pollutant chemistry is driven by the presence of sunlight, the secondary products are commonly referred to as photochemical pollutants. http: //www. enviropedia. org. uk/Air_Quality/Air_Pollution_Chemistry. ph p

Air Pollution Chemistry • A well-known secondary photochemical pollutant is ozone (O 3). Its formation results from the sunlight-initiated oxidation (reaction with oxygen) of volatile organic compounds (VOCs) such as benzene in the presence of nitrogen oxides (NOx), mostly nitric oxide (NO) and nitrogen dioxide (NO 2). Once formed, ozone is scavenged by NO, and in the absence of other competing reactions, a "photostationary state" is formed where concentrations of NO, NO 2 and O 3 are all inter-related. • In rural areas away from major sources of NO, such as urban road transport, ozone scavenging by NO is lower, and consequently ozone concentrations in the atmosphere are higher. • The primary pollutants sulphur dioxide and nitrogen oxides also undergo chemical transformation as they are dispersed in the atmosphere, forming sulphuric acid and nitric acid respectively, which may be deposited as acid rain. http: //www. enviropedia. org. uk/Air_Quality/Air_Pollution_Chemistry. ph p

Measuring Acid Rain The p. H (not PH) scale is used to measure the acidity or alkalinity of an aqueous solution and is determined by the hydrogen ion content (H+). This scale was invented by a Danish scientist called Sorenson in 1909. The p. H scale ranges from 0, which is strongly acid, to 14 which is strongly alkaline, the scale point 7 being neutral. Examples of solutions with differing p. H values include car battery acid (p. H 1), lemon juice (p. H 2), natural rain (p. H 5 -6), milk (p. H 6), seawater (p. H 8), milk of magnesia (p. H 10) and ammonia (p. H 12). http: //www. enviropedia. org. uk/Acid_Rain/Measuring. php

Measuring Acid Rain • The p. H scale is logarithmic rather than linear, and so there is a ten (!) fold increase in acidity with each p. H unit, such that rainfall with p. H 5 is ten times more acidic than p. H 6, rainfall with p. H 4 is 100 times more acidic than p. H 6 and rainfall with p. H 3 is 1000 times more acidic than p. H 6. • Rainfall acidity is measured in p. H units. Normal or unpolluted rainfall has a p. H of 5. 6. This is slightly acidic due to the presence of carbon dioxide in the atmosphere which forms weak carbonic acid in water. It is not uncommon for acidified rainwater to have a p. H of 4, about 30 times as acidic as normal rainwater. http: //www. enviropedia. org. uk/Acid_Rain/Measuring. php

UK Acid Rain In the 1980 s, the UK was described by Scandinavian countries as the dirty old man of Europe due to high emissions of sulphur dioxide from industrial sources causing transboundary acid rain. Emissions of sulphur dioxide and oxides of nitrogen have since been reduced, although the UK remains a considerable emitter of acidic pollution compared to other European countries. Most of the UK sulphur dioxide comes from power stations (65% in 1999) and other industries (22% in 1999) whilst the largest source of nitrogen oxides is road transport (44% in 1999) and power stations (21% in 1999). http: //www. enviropedia. org. uk/Acid_Rain/UK_Acid_Rain. php

UK Acid Rain • Total deposition of sulphur on the UK during 1998 was around 0. 33 million tonnes compared to 1. 19 million tonnes emitted in 1999. This highlights that the UK emits more sulphur pollution than is deposited in the UK. A significant proportion of sulphur and nitrogen pollutants are exported to other countries, mainly Germany, France, Norway, Sweden, the Netherlands and Russia. • The UK experiences typical impacts from acid rain. In 1995, the United Nations Economic Commission for Europe (UNECE) Forest Survey revealed that 42% of UK trees were healthy, 45% were slightly unhealthy and 13% were damaged. Freshwater acidification is a serious problem in susceptible parts of the UK. • Many historic monuments and buildings are affected by air pollution in the UK, in particular York Minster and Westminster Abbey. http: //www. enviropedia. org. uk/Acid_Rain/UK_Acid_Rain. php

UK Acid Rain The UK is committed to reducing sulphur emissions through the 1998 Gothenburg Protocol. This Protocol requires UK to reduce sulphur emissions by 85% and nitrogen emissions by 49% by year 2010 (from 1980 levels). To meet these requirements, emissions of sulphur dioxide in UK are being reduced, through the use of cleaner technology within the power generation industry, and the use of cleaner fuels and car engines in the transport sector. http: //www. enviropedia. org. uk/Acid_Rain/UK_Acid_Rain. php

Some effects of air pollution • Small marine organisms cannot survive in acidic lakes and rivers, and their depletion (reduced numbers) affects the larger fish who usually feed on them, and, ultimately, the entire marine-life food chain. • Snow from acid rain is also damaging; snowmelt has been known to cause massive, instant death for many kinds of fish. Some lakes in Scandinavia and New York's Adirondack Mountains are completely devoid of fish life. • Acid rain also eats away at buildings and metal structures. From the Acropolis in Greece to Renaissance buildings in Italy, ancient structures are showing signs of corrosion from acid rain. • In some industrialized parts of Poland, trains cannot exceed 40 miles (65 kilometers) per hour because the iron railway tracks have been weakened from acidic air pollution.

Conclusion Global warming is affecting plants, animals, humans and the earth. We need to learn how to conserve our use of fossil fuels to minimize carbon dioxide, sulfur dioxide and nitrogen oxide productions. This will slow down the effects of global warming.
- Slides: 24