Mhendislik Fakltesi Kimya Mhendislii Blm KMB 236 Mesleki

Mühendislik Fakültesi Kimya Mühendisliği Bölümü KMB 236 -Mesleki İngilizce Dr. Öğr. Üyesi, İsa DEĞİRMENCİ

Common words for chemical engineers KMB 236 -Mesleki İngilizce Hafta-3

States of Matter There are three states of matter which exist in the environment of the Earth. The three states of matter most important to chemistry are solid, liquid and gas. If 5 -liters of liquid water is poured into a 10 -liter container, the liquid would occupy 5 -liters of the container and the other 5 liters would be empty. Suppose some water was frozen in the shape of a sphere and then put into a larger cubical shaped container. The spherical ball of ice would retain its spherical shape as well as retaining its volume even though it had been put into a container that was both larger and of a different shape.

Physical and Chemical Properties I. Physical Properties A physical property is any property that is measurable, whose value describes a state of a physical system. Physical properties are often characterized as intensive and extensive properties. An intensive property does not depend on the size of the system, nor on the amount of matter in the object, while an extensive property shows an additive relationship.
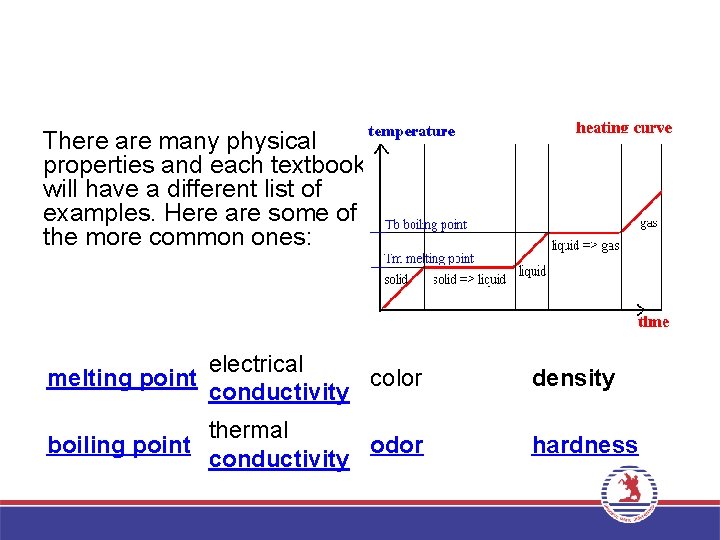
There are many physical properties and each textbook will have a different list of examples. Here are some of the more common ones: electrical melting point color conductivity density thermal boiling point odor conductivity hardness

II. Chemical Properties A chemical substance is a form of matter that has constant chemical composition and characteristic properties. It cannot be separated into components by physical separation methods, i. e. , without breaking chemical bonds. Chemical substances can be chemical elements, chemical compounds, ions or alloys.

Combustion • Combustion is the reaction of oxygen gas (O 2) with anything. • However, we will understand combustion as the reaction of oxygen with an compound containing carbon and hydrogen. A common synonym for combustion is burn.

CH 4 + O 2 ---> CO 2 + H 2 O C 2 H 6 + O 2 ---> CO 2 + H 2 O C 6 H 12 O 6 + O 2 ---> CO 2 + H 2 O C 2 H 5 OH + O 2 ---> CO 2 + H 2 O Notice that some compounds contain carbon, hydrogen AND oxygen. However, the products are all the same, in every reaction. We could vary it a bit by adding nitrogen (burns to form NO 2) to the compound formula or sulfur (burns to form SO 2). Like this: C 21 H 24 N 2 O 4 + O 2 ---> CO 2 + H 2 O + NO 2 C 2 H 5 SH + O 2 ---> CO 2 + H 2 O + SO 2 Balance the combustion reactions! Balance the chemical equations!

Gases • Determine the pressure change when a constant volume of an ideal gas at 1. 00 atm is heated from 20. 0 °C to 30. 0 °C. • A gas has a pressure of 0. 370 atm at 50. 0 °C. What is the pressure at standard temperature? • If a gas is cooled from 323. 0 K to 273. 15 K and the volume is kept constant what final pressure would result if the original pressure was 750. 0 mm Hg? • If a gas in a closed container is pressurized from 15. 0 atm to 16. 0 atm and its original temperature was 25. 0 °C, what would the final temperature of the gas be?

• A 30. 0 L sample of nitrogen inside a rigid, metal container at 20. 0 °C is placed inside an oven whose temperature is 50. 0 °C. The pressure inside the container at 20. 0 °C was at 3. 00 atm. What is the pressure of the nitrogen after its temperature is increased? • A sample of gas at 3. 00 x 103 mm Hg inside a steel tank is cooled from 500. 0 °C to 0. 00 °C. What is the final pressure of the gas in the steel tank? • Calculate the final pressure inside a scuba tank after it cools from 1. 00 x 103 °C to 25. 0 °C. The initial pressure in the tank is 130. 0 atm.

Converting between Celsius and Kelvin • If you have a Celsius temperature in the problem, you MUST change it to Kelvin, in order to use it in your problem. • Sometimes your teacher might put a Celsius temperature in the problem, but you really don't need to use it. Your teacher is doing what he or she is driven to do: confuse poor teenage kids. All teachers know this is really fun! OK, back to work. • You can convert between Celsius and Kelvin like this: Kelvin = Celsius + 273. 15. Often, the value of 273 is used instead of 273. 15. Check with your teacher on this point.

• Never, use a degree sign on the symbol for Kelvin. Do not do this: °K. This is what is known as an 'idiot indicator. ' You do that and your study partner will say "Didn't you learn anything in high school? You're an idiot. " • Also, never say 'degrees Kelvin. ' This value: 225 K is said "two hundred twenty five Kelvins. "

Here are some temperature conversion problems (no answers provided): • 1. What temperature scale must always be used when working gas law problems? _____ • 2. The boiling point of water on the Kelvin scale is _____. • 3. 0 K is also known as _____. • 4. 40. 0 °C is what temperature on the Kelvin scale? • 5. 298 K is what temperature in degrees Celsius? • 6. What is the temperature (give in both °C and K) at STP? • 9. Convert 20. 0 °C to K

Converting between Units of Pressure: atm. , mm. Hg and k. Pa There are three different units of pressure used in chemistry. This is an unfortunate situation, but we cannot change it. You must be able to use all three. Here they are: • atmospheres (symbol = atm) • millimeters of mercury (symbol = mm Hg) • Pascals (symbol = Pa) or, more commonly, kilo. Pascals (symbol = k. Pa)

Convert 0. 875 atm to mm. Hg. One atm. equals 760. 0 mm Hg, so there will be a multiplication or division based on the direction of the change. Solution: multiply the atm value by 760. 0 mm. Hg / atm.

Gas Density • Discussing gas density is slightly more complex than discussing solid/liquid density. Since gas volume is VERY responsive to temperature and pressure, these two factors must be included in EVERY gas density discussion. • By the way, solid and liquid volumes are responsive to temperature and pressure, but the response is so little that it can usually be ignored. • So, for gases, we speak of "standard gas density. " This is the density of the gas (expressed in grams per liter) at STP. If you discuss gas density at any other set of conditions, you drop the word standard and specify the pressure and temperature. • You can calculate the standard gas density fairly easily. Just take the mass of one mole of the gas and divide by the molar volume. • For nitrogen, we would have: • 28. 014 g mol¯ 1 / 22. 414 L mol¯ 1 = 1. 250 g/L • For water, we have: • 18. 015 g mol¯ 1 / 22. 414 L mol¯ 1 = 0. 8037 g/L
- Slides: 16