Mhendislik Fakltesi Kimya Mhendislii Blm KMB 236 Mesleki

Mühendislik Fakültesi Kimya Mühendisliği Bölümü KMB 236 -Mesleki İngilizce Dr. Öğr. Üyesi, İsa DEĞİRMENCİ

Solutions KMB 236 -Mesleki İngilizce Hafta-6

Solutions

Solutions are homogeneous mixtures. Solvent: A matter that dissolves a solid, liquid, or gaseous solute, resulting in a solution. Solvent is the largest component of a solution. Solute: The other solution components which are dissolved in the solution.

Types of Solutions are homogeneous mixtures. The major component is called solvent, and the minor components are called solute. When gas or solid material dissolve in a liquid, the gas or solid material is called the solute. When two liquids dissolve in each other, the major component is called the solvent and the minor component is called the solute Many chemical reactions are carried out in solutions, and solutions are also closely related to our every day lives. The air we breathe, the liquids we drink, and the fluids in our body are all solutions. Furthermore, we are surrounded by solutions such as the air and waters (in rivers, lakes and oceans).

Types of Solutions • Material exists in three states: solid, liquid, and gas. Solutions exist in all these states: • Gaseous mixtures are usually homogeneous and all gases mixtures are gas-gas solutions. The air is a natural gas solution, but its water and carbon dioxide contents may vary depends on the temperature and places. http: //www. science. uwaterloo. ca/~cchieh/cact/c 120/solution. html

Types of Solutions When molecules of gas, solid or liquid are dispersed and mixed with those of liquid, the homogeneous (uniform) states are called liquid solutions. Solid, liquid and gas dissolve in liquid to form liquid solutions. Gases and liquid solutions have attracted the attention of most chemists, while material scientists and engineers are more interested in the manufacture and properties of solid solutions. Many alloys, ceramics, and polymer blends are solid solutions. Within certain range, copper and zinc dissolve in each other and harden to give solid solutions called brass. Silver, gold, and copper form http: //www. science. uwaterloo. ca/~cchieh/cact/c 120/solution. html many different alloys with unique colors and

Alloys and solid solutions are important in the world of materials. The research, development, manufacture, and production of these material are big business. Some examples for alloys Brass (prinç): Copper-Zinc (Cu-Zn, BakırÇinko) Bronze : Copper-Tin (Cu-Sn, Bakır-Kalay) http: //www. science. uwaterloo. ca/~cchieh/cact/c 120/solution. html

Solubility • • • The maximum amount of a substance dissolved in a given volume of solvent is called solubility. Often, the solubility in water is expressed in gram/100 m. L. A solution reached the maximum solubility is called a saturated solution. Often, another phase such as gas, liquid, or solid of the solute is present and in contact with in the solution. In case another phase is not present, a solution may be unsaturated or supersaturated. When promoted by a seed, a solution may start to precipitate quickly. When a seeding crystal is present or started due to surface of another medium, the entire solution will solidify. During the crystallization process, heat is evolved, and the solution becomes warm. One useful classification of materials is polarity. Substances such as H 2, O 2, N 2, CH 4, CCl 4 etc are called non-polar compounds, whereas H 2 O, NH 3, CH 3 OH, NO, CO, HCl, H 2 S, PH 3 etc are called polar compounds. Regarding solubility, a useful rule is Like dissolves like. Polar materials dissolve polar materials whereas non-polar materials will mix and become true solutions. An additional factor to consider is the hydrogen bonding. For example, ethanol and water are completely miscible at any proportion due to the extensive hydrogen bonding among their molecules. http: //www. science. uwaterloo. ca/~cchieh/cact/c 120/solution. html

Properties of Solutions • Properties of a solution are very different from those of pure forms of its components. For example, only pure water freezes or solidifies at 273 K, and boils at 373 K. Freezing point of the ocean water is lower. Would you expect the boiling point of a salt solution to be lower? Actually, it's higher! • In Canada, they use a solution of equal volumes of ethylene glycol and water as the engine coolant, because such a solution will not freeze until the temperature drops to really low. http: //www. science. uwaterloo. ca/~cchieh/cact/c 120/solution. html

Solution Concentration • Mass percent. • Volume percent. • Mass/volume percent. (m/m) (v/v) (m/v) • Isotonic saline is prepared by dissolving 0. 9 g of Na. Cl in 100 m. L of water and is said to be: 0. 9% Na. Cl (mass/volume)

ppm, ppb and ppt • Very low solute concentrations are expressed as: ppm: parts per million ppb: parts per billion ppt: parts per trillion ( g/g, mg/L) (ng/g, g/L) (pg/g, ng/L) ppm, ppb, and ppt are properly m/m or v/v.

Mole Fraction and Mole Percent = Amount of component i (in moles) Total amount of all components (in moles) 1 + 2 + 3 + … n = 1 Molarity and Molality Molarity (M) = Amount of solute (in moles) Volume of solution (in liters) Molality (m) = Amount of solute (in moles) Mass of solvent (in kilograms)
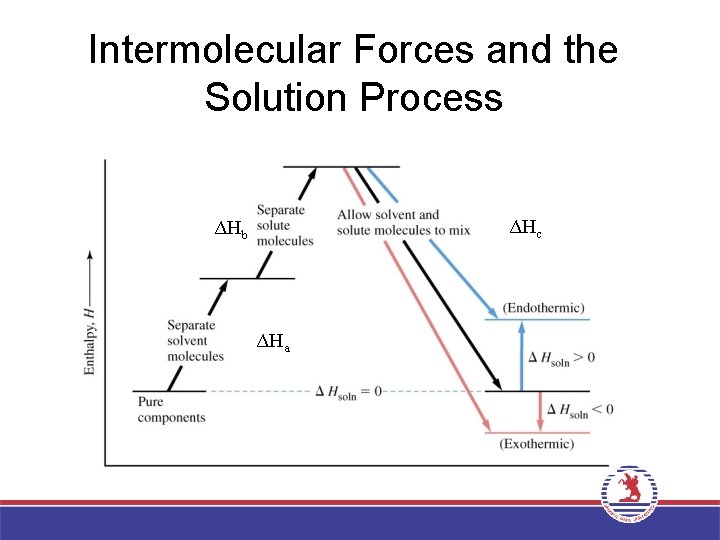
Intermolecular Forces and the Solution Process ΔHc ΔHb ΔHa
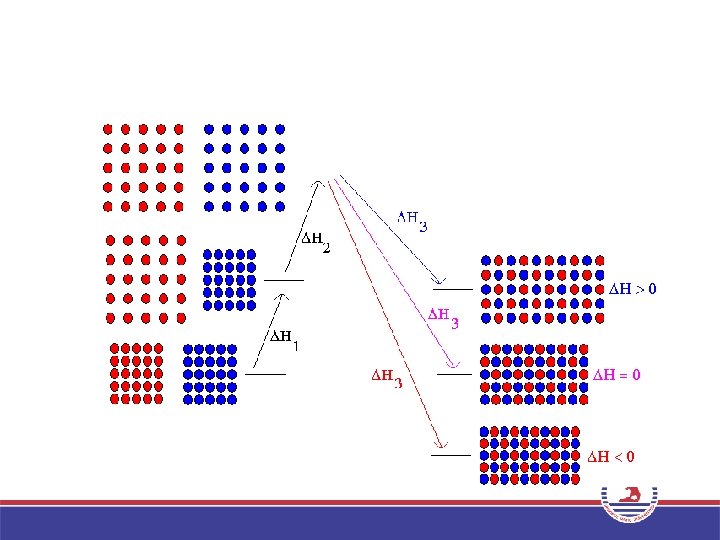
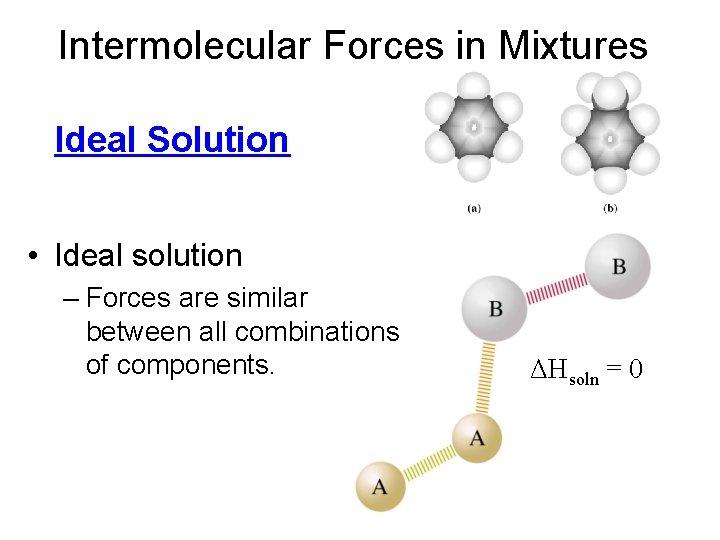
Intermolecular Forces in Mixtures Ideal Solution • Ideal solution – Forces are similar between all combinations of components. ΔHsoln = 0
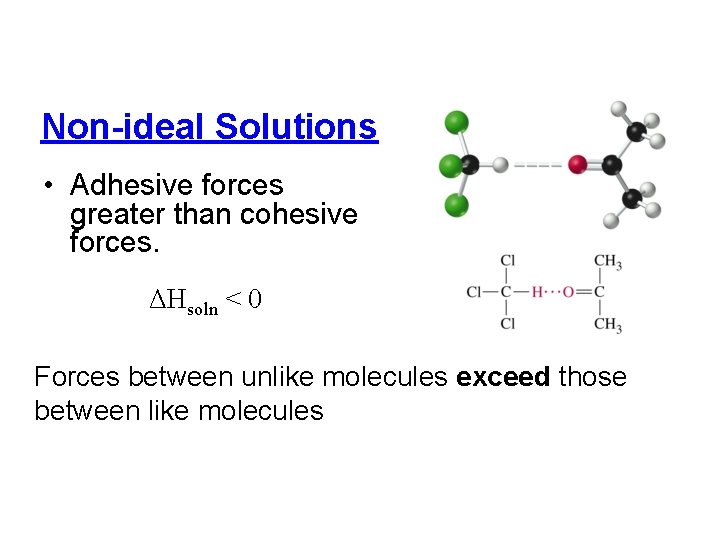
Non-ideal Solutions • Adhesive forces greater than cohesive forces. ΔHsoln < 0 Forces between unlike molecules exceed those between like molecules
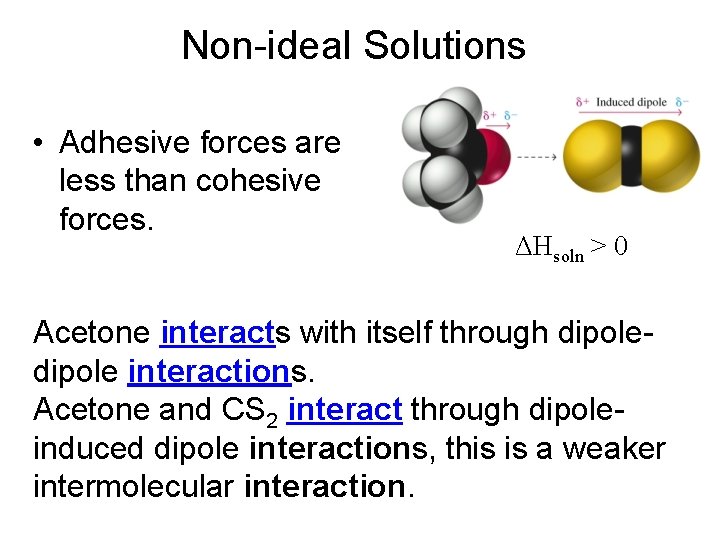
Non-ideal Solutions • Adhesive forces are less than cohesive forces. ΔHsoln > 0 Acetone interacts with itself through dipole interactions. Acetone and CS 2 interact through dipoleinduced dipole interactions, this is a weaker intermolecular interaction.

Solubility of Gases • Most gases are less soluble in water as temperature increases. Henry’s Law • Solubility of a gas increases with increasing pressure. C = k Pgas

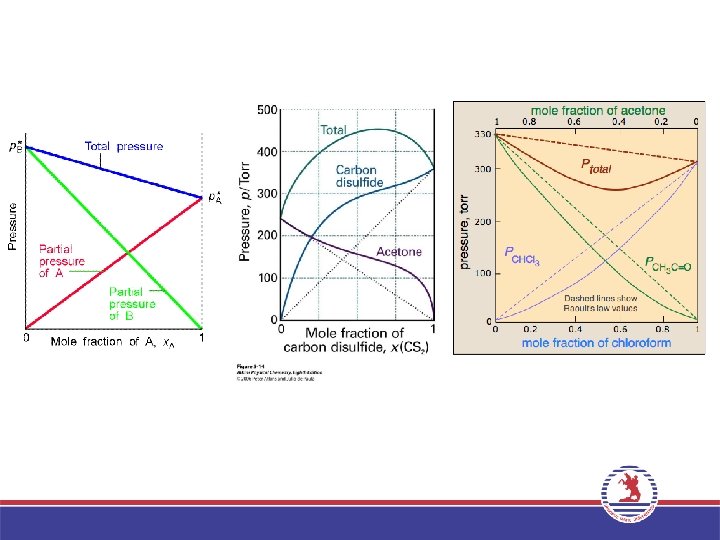
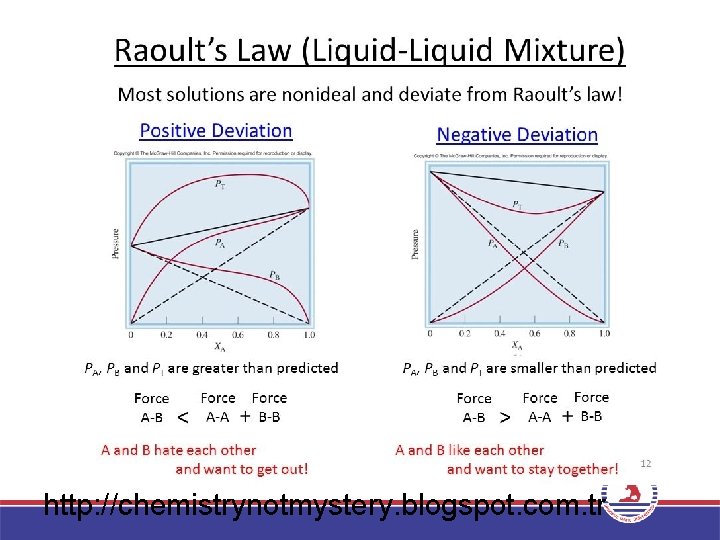
Vapor Pressures of Solutions PA = A P° • Roault, 1880 s. – Dissolved solute lowers vapor pressure of solvent. – The partial pressure exerted by solvent vapor above an ideal solution is the product of the mole fraction of solvent in the solution and the vapor pressure of the pure solvent at a given temperature.

Predicting vapor pressure of ideal solutions. The vapor pressures of pure benzene and toluene at 25°C are 95. 1 and 28. 4 mm Hg, respectively. A solution is prepared in which the mole fractions of benzene and toluene are both 0. 500. What are the partial pressures of the benzene and toluene above this solution? What is the total vapor pressure? Balanced Chemical Equation: Pbenzene = benzene P°benzene = (0. 500)(96. 1 mm Hg) = 47. 6 mm Hg Ptoluene = toluene P°toluene = (0. 500)(28. 4 mm Hg) = 14. 2 mm Hg Ptotal = Pbenzene + Ptoluene = 61. 8 mm Hg
http: //chemistrynotmystery. blogspot. com. tr
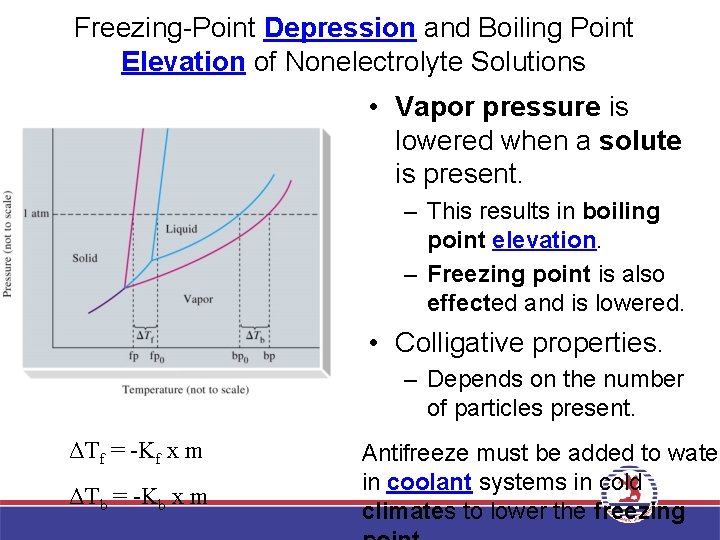
Freezing-Point Depression and Boiling Point Elevation of Nonelectrolyte Solutions • Vapor pressure is lowered when a solute is present. – This results in boiling point elevation. – Freezing point is also effected and is lowered. • Colligative properties. – Depends on the number of particles present. ΔTf = -Kf x m ΔTb = -Kb x m Antifreeze must be added to water in coolant systems in cold climates to lower the freezing
- Slides: 24