MG Testing Pilot Royal Victoria Hospital Belfast Dr

MG Testing Pilot Royal Victoria Hospital, Belfast Dr Laura Bell, Dr Stephen Megarity, Dr Carol Emerson Department of Genitourinary Medicine
Mycoplasma Genitalium • Mycoplasma genitalium was first isolated in 1981 • The smallest known self-replicating bacterium • No cell wall – does not gram stain • Culture is difficult and takes weeks

Relationship to Sexual Health • Prevalence of M. genitalium in men with NGU is 10 -20% • Non-chlamydial non- gonococcal urethritis (NCNGU) is 10 -35% (as compared to 1 -2% in the general population) • possible that sexually acquired reactive arthritis, epididymitis and proctitis may occur as a result of M. genitalium infection

Relationship to Sexual Health (2) • Data are lacking to support an association with prostatitis • M. genitalium is linked aetiologically to PID and accounts for 10 -13% of cases of PID • Majority of people infected with M. genitalium in the genital tract do not develop disease - hence symptomatic testing only

Testing Pilot in RVH • Launched Feb 2019 in RVH GUM • Aim to test as per BASHH guidelines – – – Non-gonococcal urethritis Pelvic inflammatory disease Muco- purulent cervicitis, particularly post-coital bleeding Epididymitis Sexually-acquired proctitis Current sexual partners of persons infected with M. genitalium • Review of pilot with hope to roll out regional access to testing
Testing • Males – First Void Urine • Females - Vulvo-vaginal Swab • Rectal swab • Cobas bottle for M. genitalium PCR and macrolide resistance mediating mutations (MRMM)
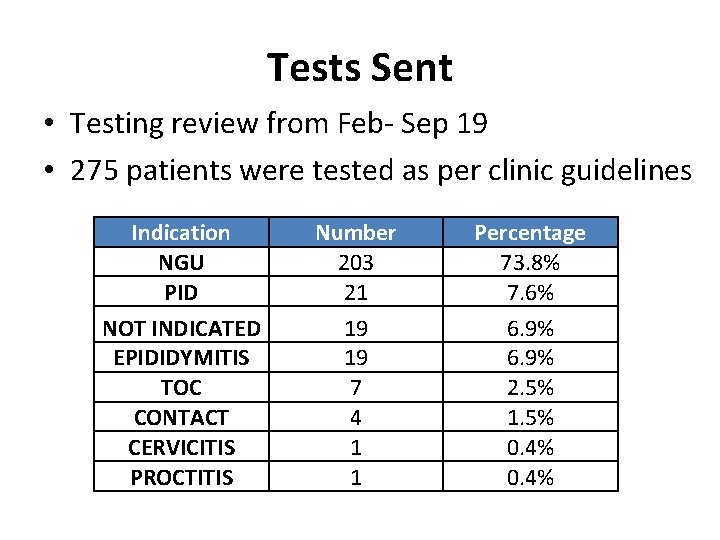
Tests Sent • Testing review from Feb- Sep 19 • 275 patients were tested as per clinic guidelines Indication NGU PID NOT INDICATED EPIDIDYMITIS TOC CONTACT CERVICITIS PROCTITIS Number 203 21 19 19 7 4 1 1 Percentage 73. 8% 7. 6% 6. 9% 2. 5% 1. 5% 0. 4%

Positive Results • Of those positive (total 24) – 23/24 (96%) Male – 18/24 (75%) Heterosexual • Indications for test – 21/24 (87. 4%) – NGU – 1/24 (4. 2%) - Epididymitis – 1/24 (4. 2%)- Proctitis – 1/24 (4. 2%)– Contacts
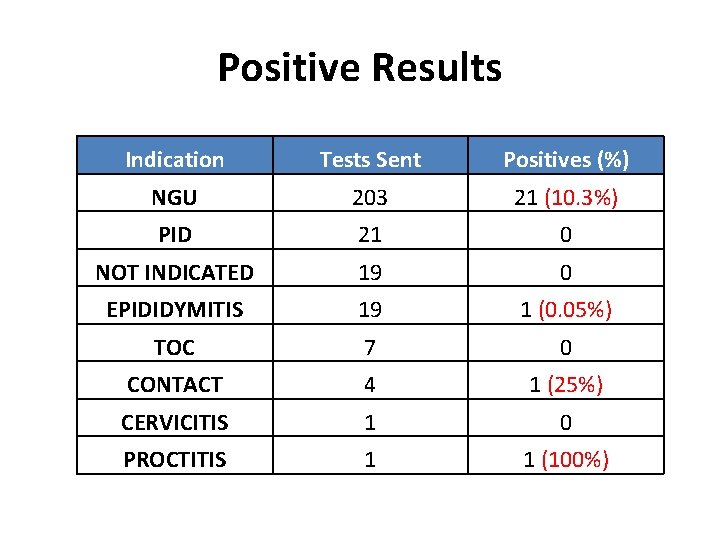
Positive Results Indication Tests Sent Positives (%) NGU 203 21 (10. 3%) PID 21 0 NOT INDICATED 19 0 EPIDIDYMITIS 19 1 (0. 05%) TOC 7 0 CONTACT 4 1 (25%) CERVICITIS 1 0 PROCTITIS 1 1 (100%)

Resistance • Macrolide resistance mediating mutations (MRMM) • 23 S mutation • Resistance 8/24 (33. 3%)

Treatment Doxycycline/Azithromycin 15/24 Doxycycline/Azithromycin and Moxifloxacin 1/24 Moxifloxacin 8/24 All patients have achieved TOC

Effect on management of patients e. g. NGU Management

Current NGU Management • Doxycycline 7 days • If recurrent or persisting – Azithromycin 1 g orally as a single dose then 500 mg orally once daily for 2 days plus metronidazole for 5 days (repeat 1 week doxycycline if >2 weeks since prev. completed) then – Moxifloxacin 400 mg once daily for 10 days
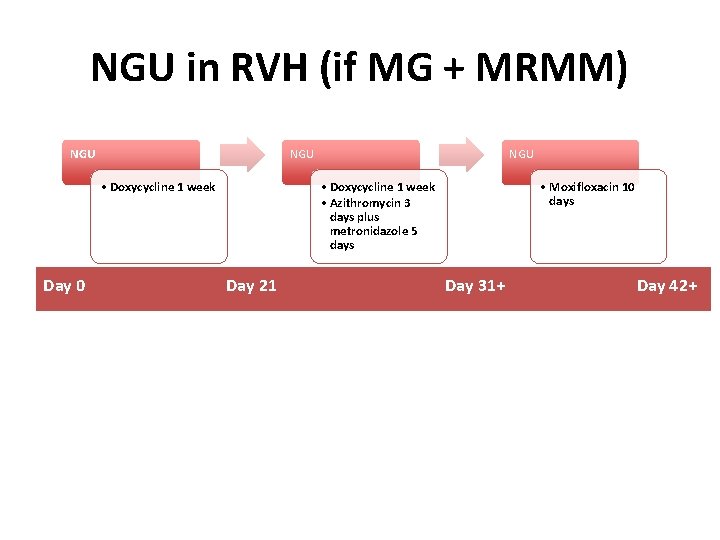
NGU in RVH (if MG + MRMM) NGU • Doxycycline 1 week Day 0 NGU • Doxycycline 1 week • Azithromycin 3 days plus metronidazole 5 days Day 21 Day 5 • Moxifloxacin 10 days Day 31+ Day 8 or 15 Day 42+
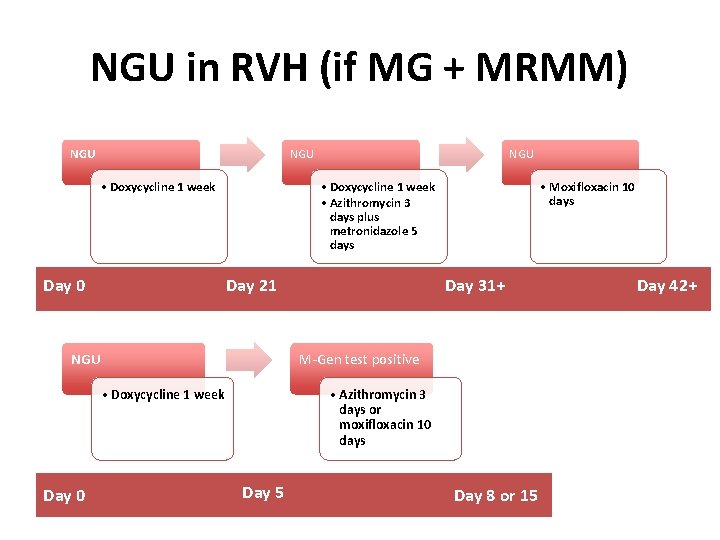
NGU in RVH (if MG + MRMM) NGU • Doxycycline 1 week Day 0 • Doxycycline 1 week • Azithromycin 3 days plus metronidazole 5 days Day 21 NGU • Moxifloxacin 10 days Day 31+ M-Gen test positive • Doxycycline 1 week Day 0 NGU • Azithromycin 3 days or moxifloxacin 10 days Day 5 Day 8 or 15 Day 42+

NGU in RVH (if MG) NGU • Doxycycline 1 week No MG testing Day 0 Day 21 NGU • 3 clinic visits • Moxifloxacin 10 days • 32 days antibiotics • 31+ days until correct treatment initiated Day 45+ Day 35+ • Doxycycline 1 week • Azithromycin 3 days plus metronidazole 5 days M-Gen test positive • Doxycycline 1 week MG testing Day 0 NGU Day 5 • 2 clinic visits • 10 -17 days antibiotics • 5 days until correct treatment Day 8 orinitiated 15 • Azithromycin 3 days or moxifloxacin 10 days
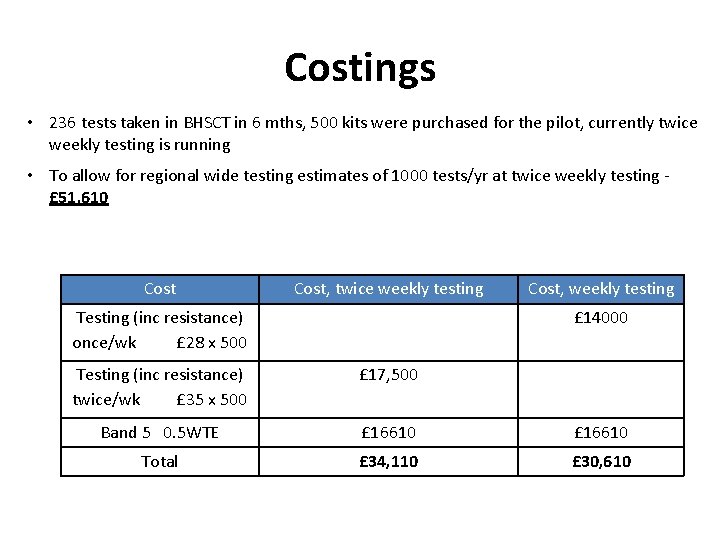
Costings • 236 tests taken in BHSCT in 6 mths, 500 kits were purchased for the pilot, currently twice weekly testing is running • To allow for regional wide testing estimates of 1000 tests/yr at twice weekly testing £ 51, 610 Cost, twice weekly testing Testing (inc resistance) once/wk £ 28 x 500 Cost, weekly testing £ 14000 Testing (inc resistance) twice/wk £ 35 x 500 £ 17, 500 Band 5 0. 5 WTE £ 16610 Total £ 34, 110 £ 30, 610

Summary • M. genitalium is a prevalent infection – 10. 3% of NGU • Prevalence in PID appears low but quoted as 10 -13% • Targeted symptomatic testing with macrolide mutation testing can reduce time to effective treatment, unnecessary antibiotics and clinic visits • Twice weekly laboratory testing provides satisfactory turnaround time for results allowing prompt treatment and meaning less time for onward transmission

Thank you for listening Questions
- Slides: 19