Metric system Measurements Significant figures Relationships Factor Label

§Metric system §Measurements §Significant figures §Relationships § Factor Label Method

SYSTEM OF MEASUREMENTS (SI)
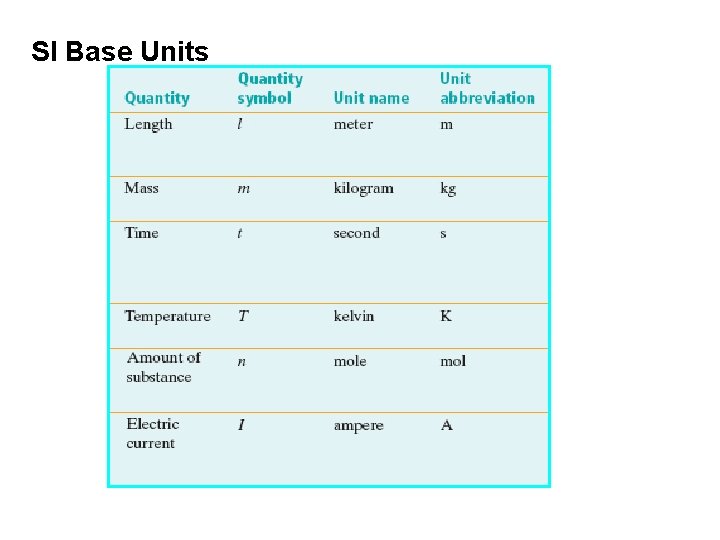
SI Base Units
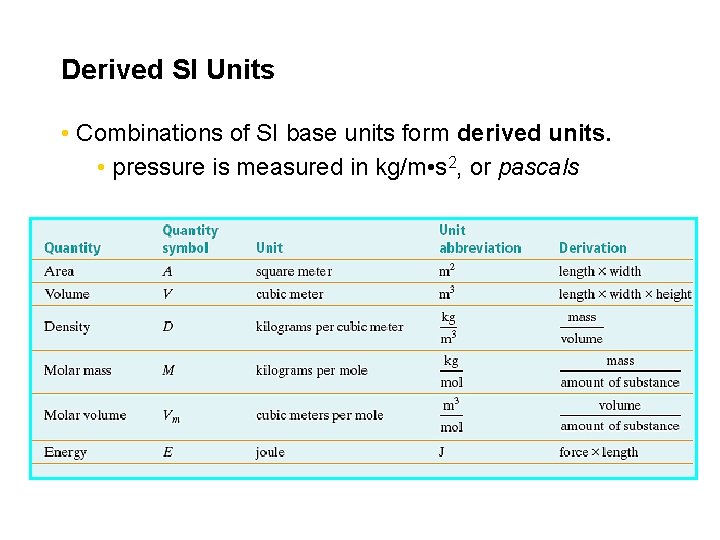
Derived SI Units • Combinations of SI base units form derived units. • pressure is measured in kg/m • s 2, or pascals
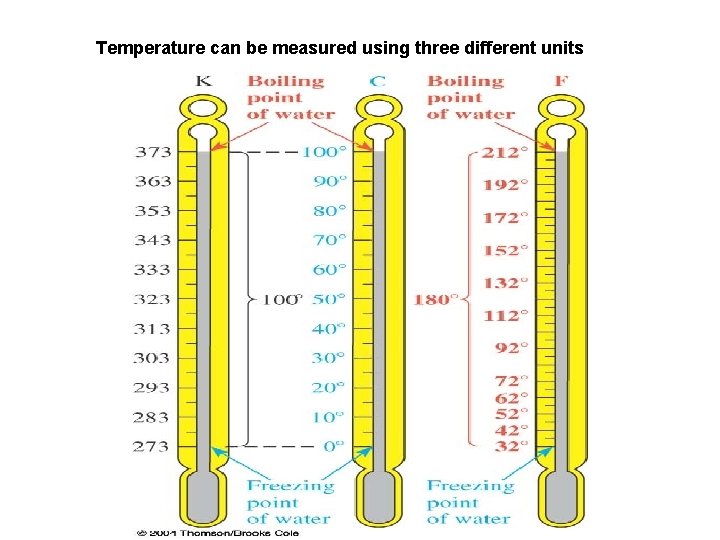
Temperature can be measured using three different units

Conversion between T units °F = 9/5 °C + 32 OR °F = 1. 8 °C + 32 K = °C + 273. 15
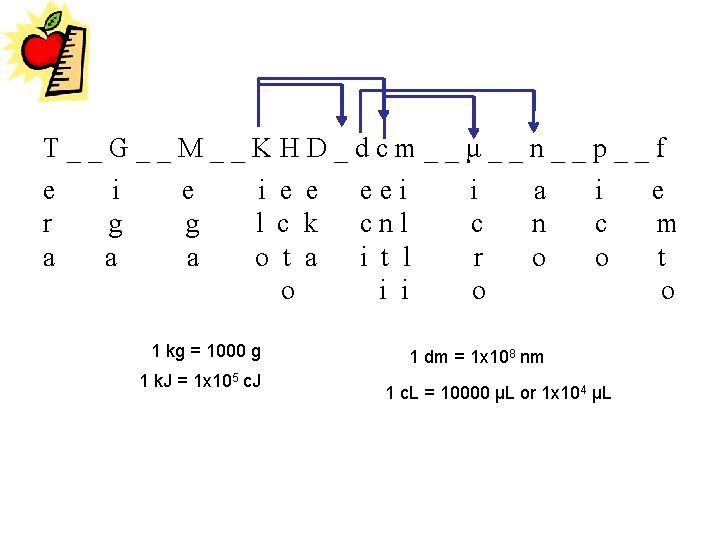
T__G__M__KHD_dcm__μ__n__p__f e i e e eei i a i e r g g l c k cnl c n c m a a a o t a i t l r o o t o i i o o 1 kg = 1000 g 1 k. J = 1 x 105 c. J 1 dm = 1 x 108 nm 1 c. L = 10000 μL or 1 x 104 μL
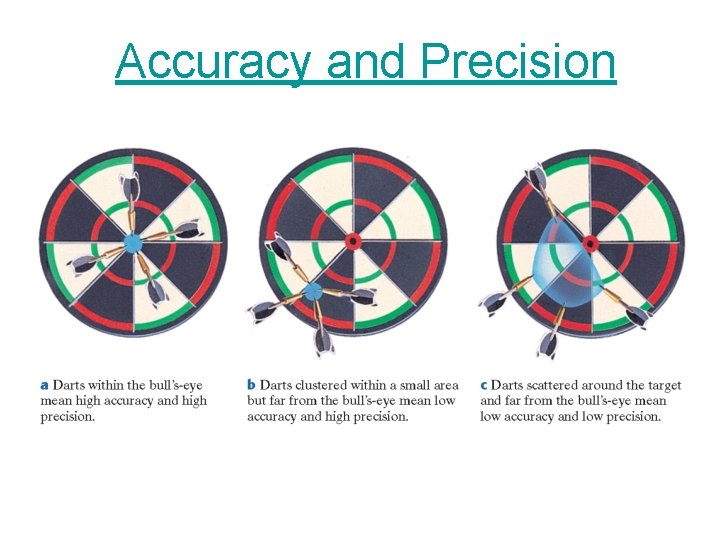
Accuracy and Precision
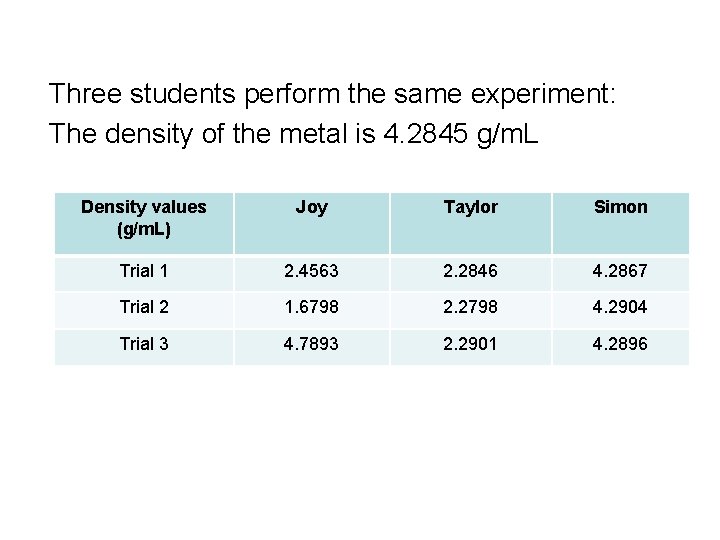
Three students perform the same experiment: The density of the metal is 4. 2845 g/m. L Density values (g/m. L) Joy Taylor Simon Trial 1 2. 4563 2. 2846 4. 2867 Trial 2 1. 6798 2. 2798 4. 2904 Trial 3 4. 7893 2. 2901 4. 2896

Accuracy and Precision, continued Percentage Error • Percentage error is calculated by subtracting the accepted value from the experimental value, dividing the difference by the accepted value, and then multiplying by 100.

• Sample Problem • A student measures the mass and volume of a substance and calculates its density as 1. 40 g/m. L. The correct, or accepted, value of the density is 1. 30 g/m. L. What is the percentage error of the student’s measurement?

Use of Numbers • Exact numbers – 1 dozen = 12 things for example • Accuracy – how closely measured values agree with the correct value • Precision – how closely individual measurements agree with each other

Use of Numbers • Significant figures – digits believed to be correct by the person making the measurement • Exact numbers have an infinite number of significant figures 12. 00000000 = 1 dozen because it is an exact number!!!!

Use of Numbers Significant Figures - Rules • Leading zeroes are never significant 0. 000357 has three significant figures • Trailing zeroes only significant if after decimal point. 2. 7800 has five significant figures • Use scientific notation to remove doubt 2. 40 x 103 has 3 significant figures 2. 400 x 103 has 4 significant figures 2400 has only two significant figures.

Use of Numbers • Imbedded zeroes are always significant 3. 0604 has five significant figures

Use of Numbers • Multiplication & Division rule Easier of the two rules Product has the smallest number of significant figures of multipliers

Sample Problem. Calculate the density of a substance with a mass of 14. 78 g and a volume of 10. 3 m. L

Use of Numbers • Addition & Subtraction rule More subtle than the multiplication rule Answer contains smallest decimal place of the addends.

Sample problem Determine the perimeter of a piece of paper of 27. 94 cm long and 6. 92 cm wide.

• Sample Problem • How many significant figures are in each of the following measurements? • • • a. 28. 6 g b. 34. 40 cm c. 910 m d. 0. 046 04 L e. 0. 006 700 0 kg

Chapter 2 Direct Proportion • Two quantities are directly proportional to each other if dividing one by the other gives a constant value.
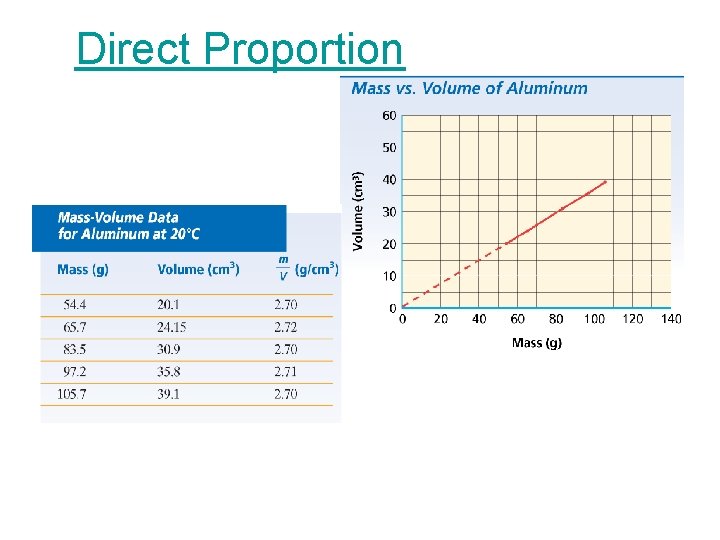
Chapter 2 Direct Proportion

Inverse Proportion Two quantities are inversely proportional to each other if multiplying one by the other gives a constant value.
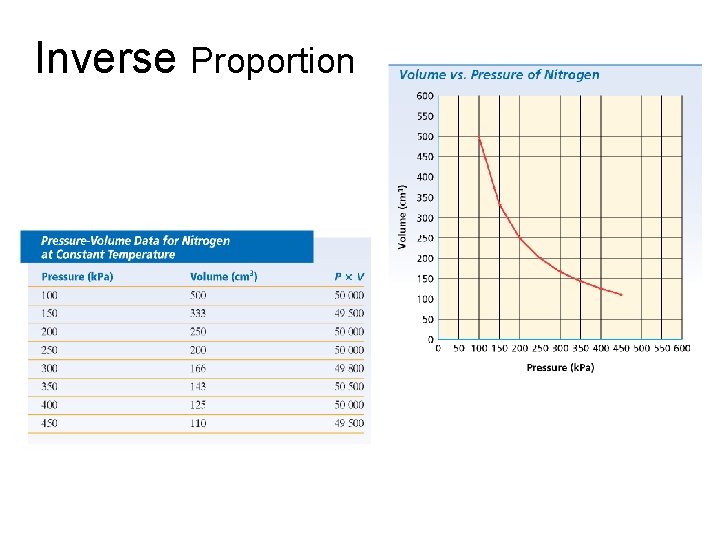
Chapter 2 Inverse Proportion

The Unit Factor Method • Simple but important method to get correct answers in word problems. • Method to change from one set of units to another.

The Unit Factor Method • fractions represent unit factors 1 ft = 12 in becomes or

The Unit Factor Method • Example: Express 9. 32 meters in micrometers.

The Unit Factor Method • Example: Express 627 milliliters in L.

The Unit Factor Method • Example: Express 45. 8 kg in mg.

Density conversions • Example: A 20. 0 g irregular solid is introduced in a graduated cylinder. The level of water inside the graduated cylinder when from 20. 0 m. L to 23. 3 m. L. Calculate the density of the solid.

Density conversions • Example: What volume will occupy a liquid with a mass of 15. 7 g and a density of 1. 34 g/m. L?

Density conversions • Example: Calculate the mass of a gas that occupies 1. 5 L and it has a density of 0. 00143 g/m. L?
- Slides: 32