METO 637 Lesson 16 Sulfur chemistry The abundances

METO 637 Lesson 16

Sulfur chemistry • The abundances, sources, budgets, and photochemistry of atmospheric sulfur compounds are poorly understood compared to carbon, nitrogen and oxygen species. • Sulfur can be converted to SO 2, SO 3, and H 2 SO 4. • Hence it acts as an aerosol precursor • Sulfate aerosols are of climatological importance as they can effect the radiative balance of the atmosphere (primary effect). • They are also very efficient cloud condensation nuclei • This in turn may have an effect on cloud production, which is also of climatological significance (secondary effect). • H 2 SO 4 can also lower the p. H of rainwater. • Anthropogenic sources are comparable with natural sources – principally from the burning of fossil fuels.

VOLCANIC ACTIVITY • MOST VOLCANOES EJECT DUST ETC. INTO THE TROPOPSHERE WHERE IT IS QUICKLY RAINED OUT. • HOWEVER LARGE VOLCANOES CAN EJECT GASES, ESPECIALLY SULFUR DIOXIDE, INTO THE STRATOSPHERE. • IN THE STRATOSPHERE THE SULFUR DIOXIDE TRANSFORMS INTO AEROSOLS, WHICH REMAIN IN THE STRATOSPHERE FOR ONE TO TWO YEARS. • THIS WILL TEND TO COOL THE TROPOSPHERE SCATTERS SOLAR RADIATION BACK TO SPACE. • ERUPTION OF MOUNT TAMBORA IN INDONESIA LED TO 'YEAR WITHOUT A SUMMER' • MOUNT PINATUBO, 1991, LOWERED TEMPERATURE BY 0. 5 C
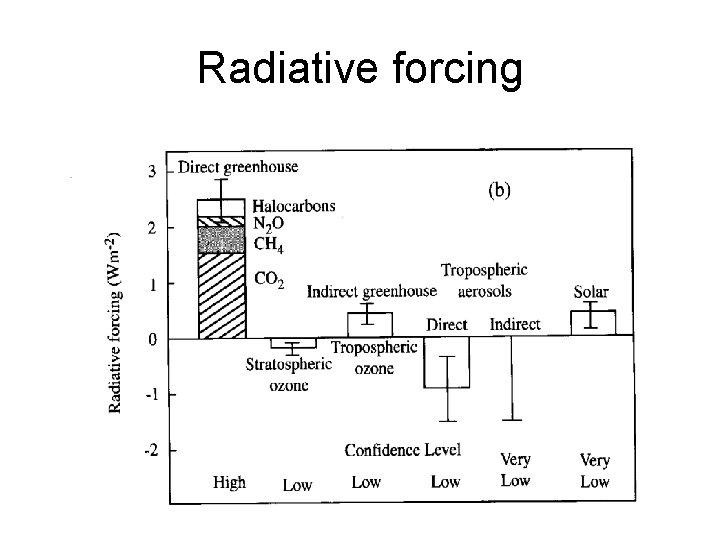
Radiative forcing

Sulfur chemistry • Sources: (1) Volcanic activity – 7 x 109 kg per year, principally as SO 2 (2) Decay of biogenic matter produces 58 x 109 kg per year over land 48 x 109 kg per year over the oceans, principally in the reduced forms of H 2 S, (CH 3)2 S (dimethyl sulfide, DMS) and (CH 3)2 S 2 (dimethyl disulfide, ) (3) Sea spray – 44 x 109 kg per year • 2 additional sulfur compounds found in the troposphere are COS (carbonyl sulfide) and CS 2 (carbon disulfide). • The lifetime of CS 2 is only a few weeks, converting to COS. • COS has a lifetime of more than a year, and is the most abundant. Largely comes from the oceans.

Sulfur chemistry • Oceanic source for COS estimated as 0. 15 x 109 kg per year. • Most sulfur gases emitted into the troposphere have a short lifetime. • Hence stratospheric sulfur comes mainly from two sources – volcanic injection and COS. • The origins of CS 2 and COS are really not known at this time. Not known how much is anthropogenic or natural. • COS is distributed fairly uniformly in the troposphere with a mixing ratio of about 0. 5 ppb

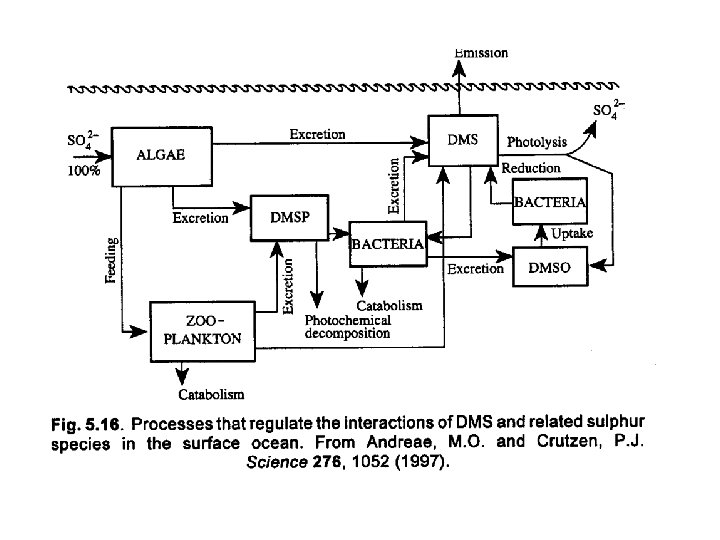
Sulfur chemistry • DMS constitutes the most important biogenic flux of sulfur compounds. • Biogenic inputs dominate in the Southern hemisphere, but anthropogenic inputs dominate in the Northern hemisphere. • The manmade sources are rapidly oxidized and removed by rainout and deposition. So their impact tends to be regional. • The longer lived biogenic compounds have a larger global impact.

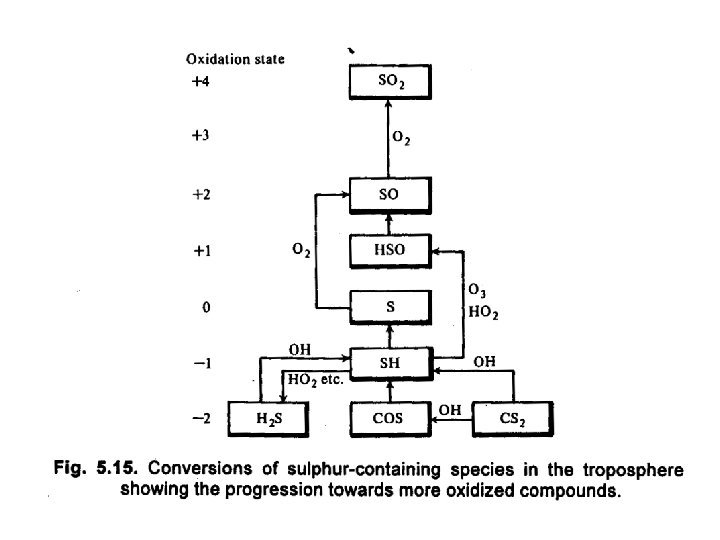
Sulfur chemistry • Oxidation of the inorganic reduced-sulfur oxidation is driven by reactions with OH: OH + CS 2 → COS + SH OH + COS → CO 2 + SH OH + H 2 S → H 2 O + SH • The SH radical is then oxidized to SO 2

Natural halogen containing species • Has long been recognized that the organic halides (e. g. methyl chloride and methyl bromide) can be found in he troposphere. • However there is increasing awareness that large amounts of inorganic chlorine and bromine might also be present and participate in tropospheric chemistry. • Vast quantities of inorganic halides are present as particles in the troposphere. • Wave action generates small airborne droplets of seawater which can evaporate to leave suspended particles of sea salt. • However, can these particles be activated in such a way that the halogens play a role in tropospheric chemistry?

Natural halogen containing species • The unexpected answer was yes. • This is because chlorine is highly reactive toward many organic compounds. As with OH it can abstract hydrogen Cl + CH 4 → CH 3 + HCl • This rate constant can be two orders of magnitude larger than the similar reaction with OH. Hence chlorine can be effective even if present at orders of magnitude lower concentrations. • Although atomic chlorine has not been detected molecular chlorine has. • Br. O has also been detected in the troposphere, especially in the polar regions. Up to 30 pptv in the Arctic

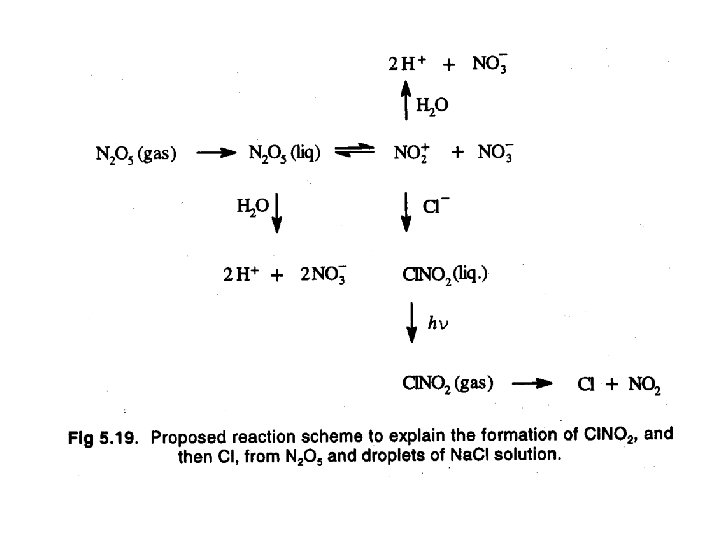
Natural halogen containing species • There is increasing evidence that the source of the halogens is sea salt. • Sea salt particles have been found to be deficient in chlorine and bromine. • This suggests that reactions between sea salt and atmospheric gases (acids, oxides of nitrogen, etc). For example: Na. Clcond + HNO 3 gas → HClgas + Na. NO 3 cond • The HCl does not photolyze in the troposphere, and the reaction with OH is slow. • Na. Clcond + N 2 O 5 gas → Cl. NO 2 gas + Na. NO 3 cond • Na. Clcond + Cl. ONO 2 gas → Cl 2 gas + Na. NO 3 cond
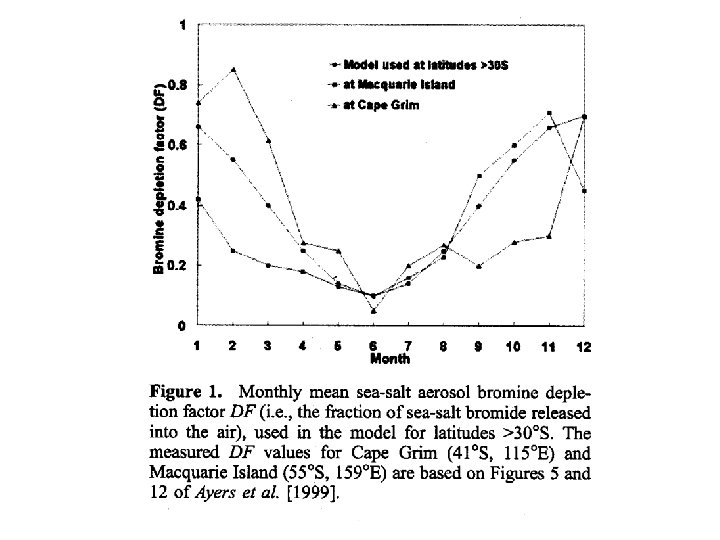
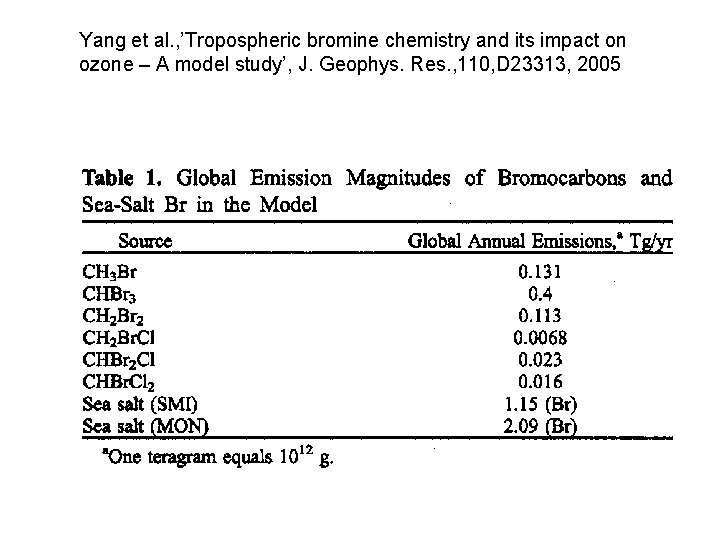
Yang et al. , ’Tropospheric bromine chemistry and its impact on ozone – A model study’, J. Geophys. Res. , 110, D 23313, 2005

Bromine chemistry • The detection of Br. O in the troposphere is of great interest. It has been suggested that Br. O is ubiquitous in the free troposphere at 0. 5 to 2 pptv. • In the troposphere the following reactions can occur: Br + O 3 → Br. O + O 2 Br. O + HO 2 → HOBr + O 2 HOBr + hν → Br + OH • Essentially we destroy ozone without the need for atomic oxygen. • Ozone loss due to Bromine has been estimated to be up to 18%.

Bromine chemistry • Yang et al. , found overall reductions in ozone of from 46% over most of the troposphere. • Larger reductions (up to 30%) at high latitudes. • Effect is due to two factors • (1) direct ozone loss by Br reactions • (2) NOx removal via bromine nitrate hydrolysis: Br. O + NO 2 → Br. ONO 2 + H 2 Oaq → HOBr + HNO 3
- Slides: 18