METHANIMINE AT HIGH SPATIAL RESOLUTION IN SGR B

METHANIMINE AT HIGH SPATIAL RESOLUTION IN SGR B 2: IMPLICATIONS FOR THE FORMATION OF CYANOMETHANIMINE Amanda Steber 1, Joanna F. Corby 2, Nathan A. Seifert 1, Cristobal Perez 1, Anthony Remijan 3, Brooks H. Pate 1 1 Department of Chemistry, The University of Virginia, Charlottesville, VA 22904 2 Department of Astronomy, The University of Virginia, Charlottesville, VA 22904 3 AL MA, National Radio Astronomy Observatory, Charlottesville, VA 22904
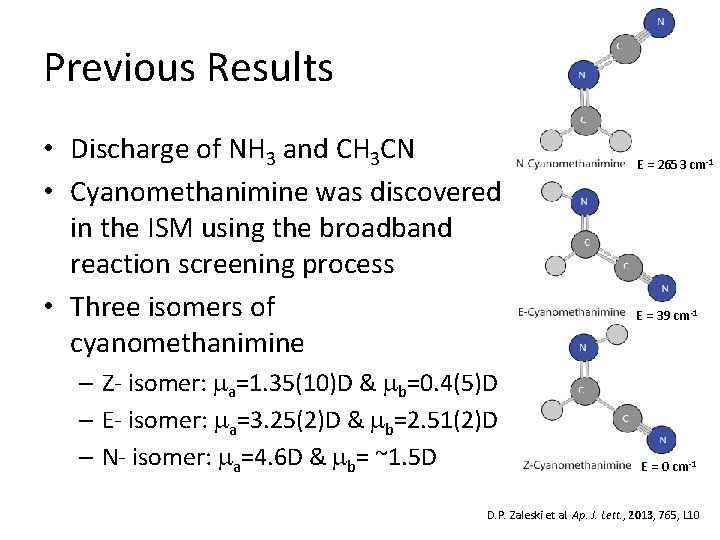
Previous Results • Discharge of NH 3 and CH 3 CN • Cyanomethanimine was discovered in the ISM using the broadband reaction screening process • Three isomers of cyanomethanimine – Z- isomer: ma=1. 35(10)D & mb=0. 4(5)D – E- isomer: ma=3. 25(2)D & mb=2. 51(2)D – N- isomer: ma=4. 6 D & mb= ~1. 5 D E = 2653 cm-1 E = 39 cm-1 E = 0 cm-1 D. P. Zaleski et al. Ap. J. Lett. , 2013, 765, L 10
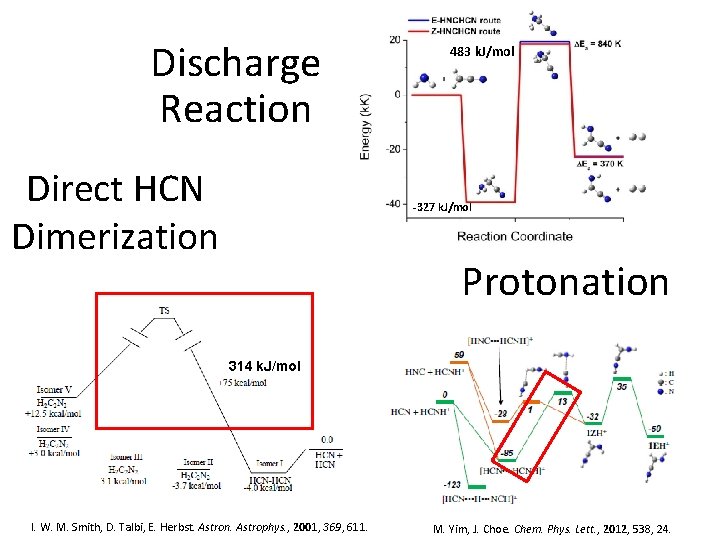
Discharge Reaction Direct HCN Dimerization 483 k. J/mol -327 k. J/mol Protonation 314 k. J/mol I. W. M. Smith, D. Talbi, E. Herbst. Astron. Astrophys. , 2001, 369, 611. M. Yim, J. Choe. Chem. Phys. Lett. , 2012, 538, 24.

• CN addition • The Kaiser group (among others) has focused on the formation of cyanides through the reaction of • CN with olefins – Crossed molecular beam experiments – Methylacetylene, allene, ethylene, dimethylacetylene, etc… • The radical attacks the p electron density of the hydrocarbon • They have been able to determine that the neutral – radical reaction occurs with out an activation barrier N. Balucani et al. Astrophys. J. , 2000, 545, 892.
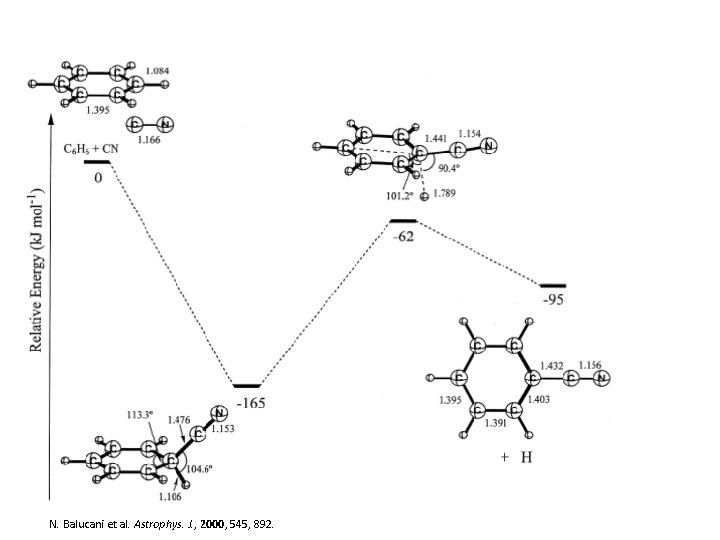
N. Balucani et al. Astrophys. J. , 2000, 545, 892.
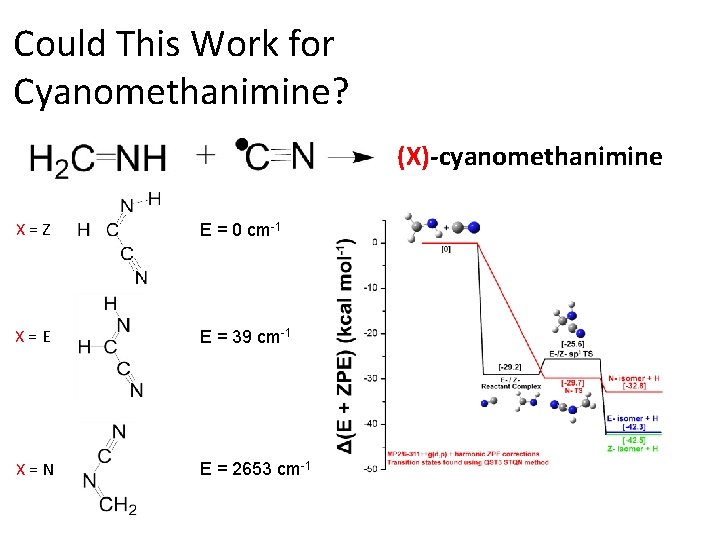
Could This Work for Cyanomethanimine? (X)-cyanomethanimine X=Z E = 0 cm-1 X=E E = 39 cm-1 X=N E = 2653 cm-1
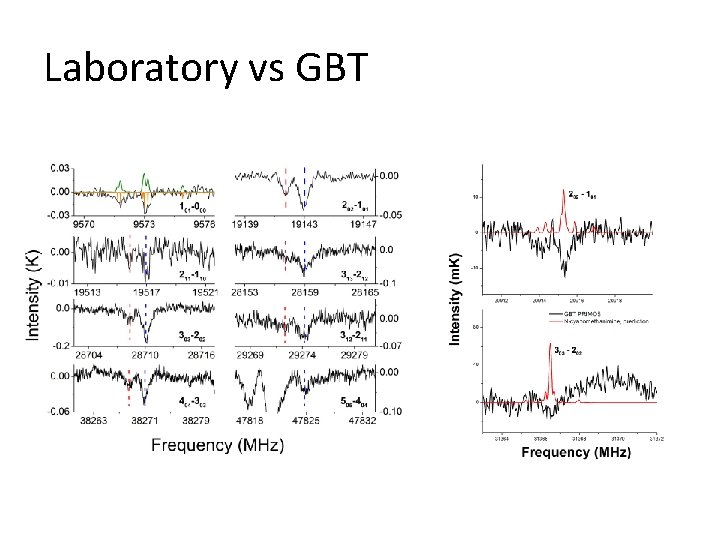
Laboratory vs GBT
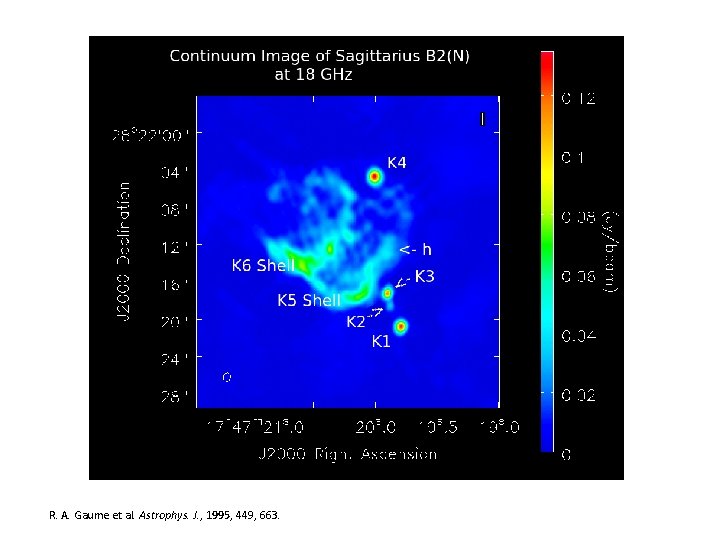
R. A. Gaume et al. Astrophys. J. , 1995, 449, 663.
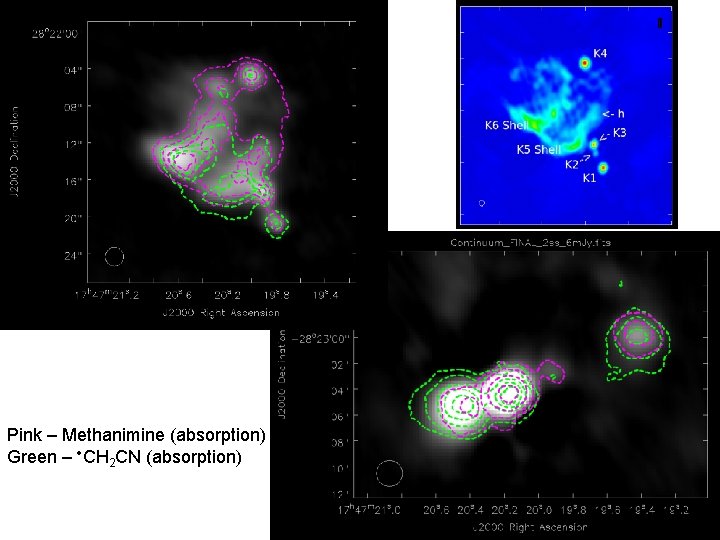
Pink – Methanimine (absorption) Green – CH 2 CN (absorption)
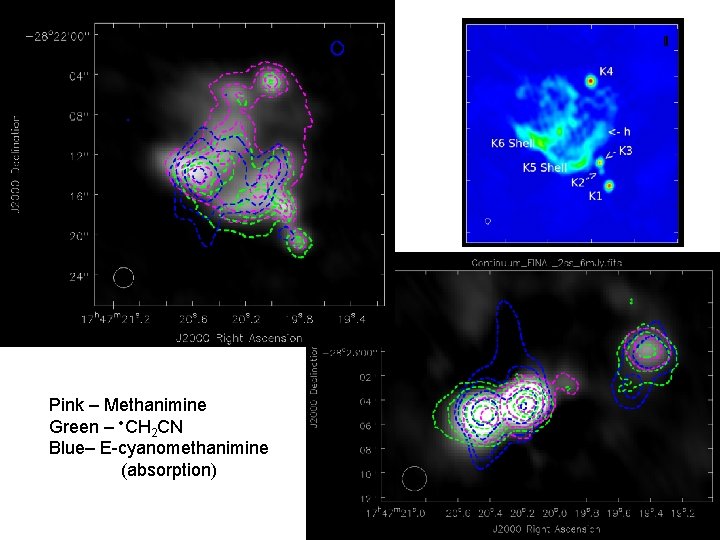
Pink – Methanimine Green – CH 2 CN Blue– E-cyanomethanimine (absorption)
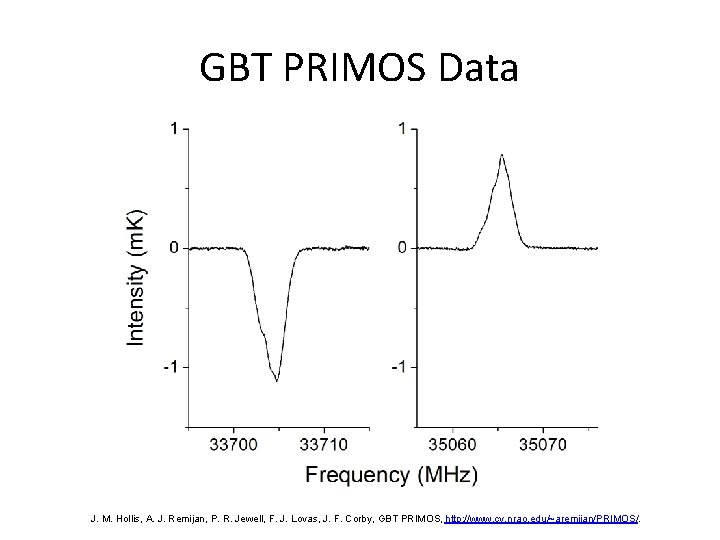
GBT PRIMOS Data J. M. Hollis, A. J. Remijan, P. R. Jewell, F. J. Lovas, J. F. Corby, GBT PRIMOS, http: //www. cv. nrao. edu/~aremijan/PRIMOS/.
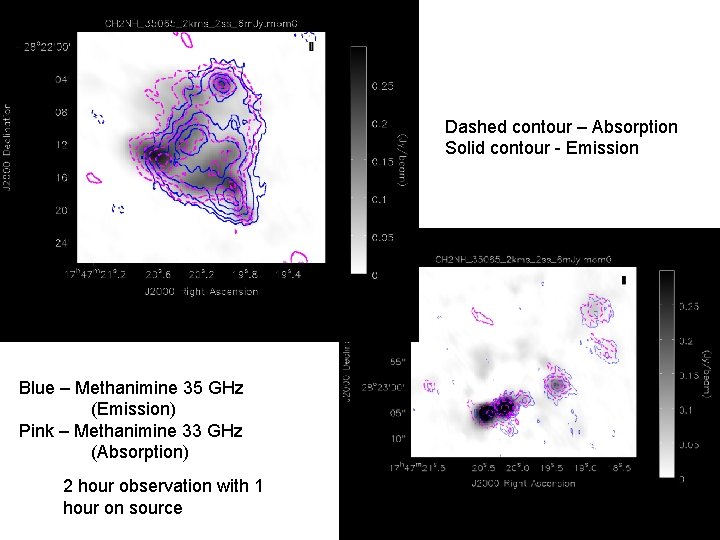
Dashed contour – Absorption Solid contour - Emission Blue – Methanimine 35 GHz (Emission) Pink – Methanimine 33 GHz (Absorption) 2 hour observation with 1 hour on source
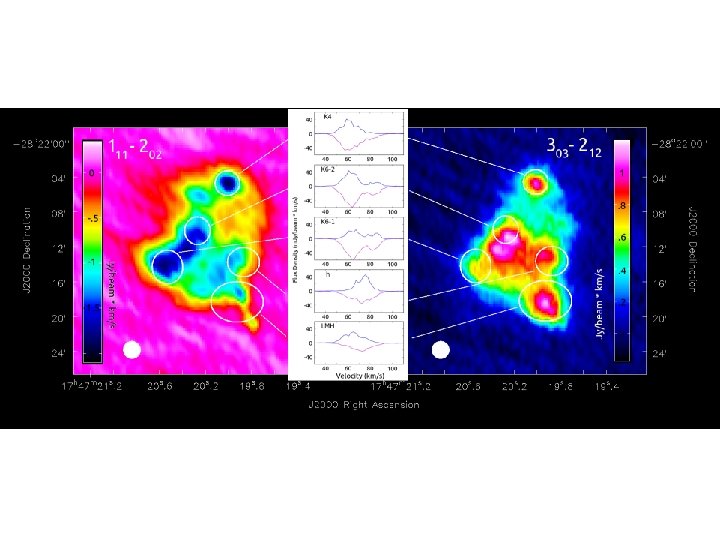


Conclusions • Previously proposed reaction mechanisms for the formation of cyanomethanimine in the interstellar medium are not very feasible • The addition of • CN to methanimine would yield cyanomethanimine + H through a barrierless reaction • The high resolution spatial maps show that this could be happening as all three species reside in the same spatial locations • The complex chemistry is suggested to be occurring outside of the hotcore (LMH)

Acknowledgements • Pate Lab • NRAO • NSF – Centers for Chemical Innovation Award Number 0847919 – Chemistry Award Number 1213200
- Slides: 16