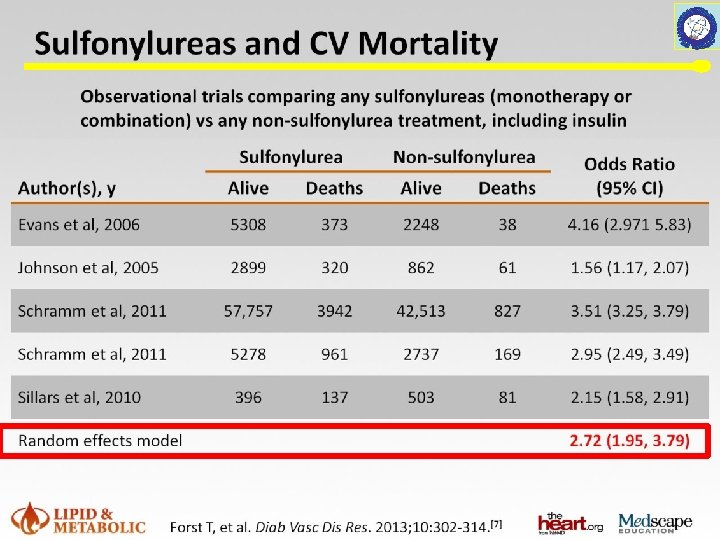
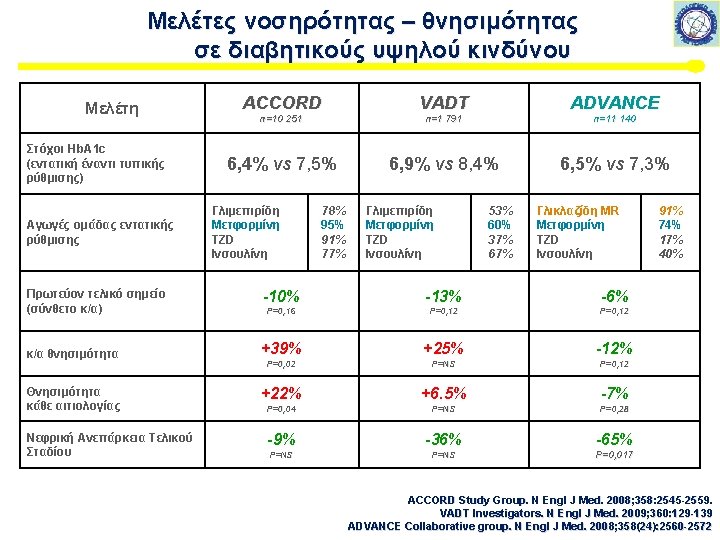
Metformin RCTs UK Prospective Diabetes Study UKPDS Group



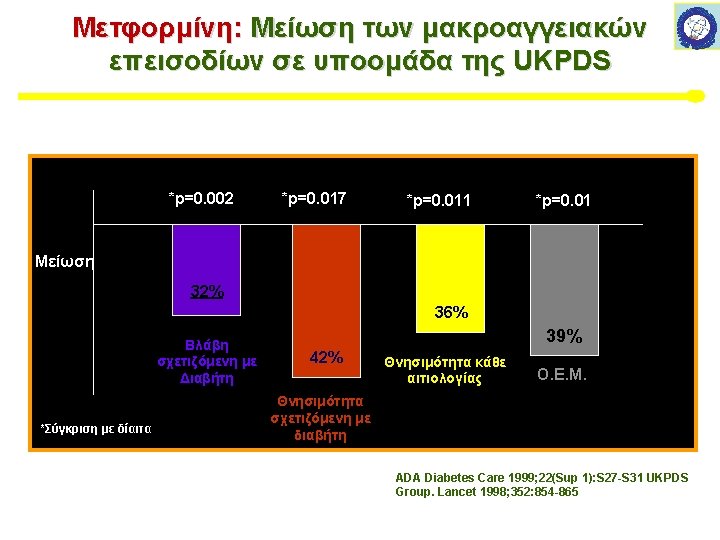
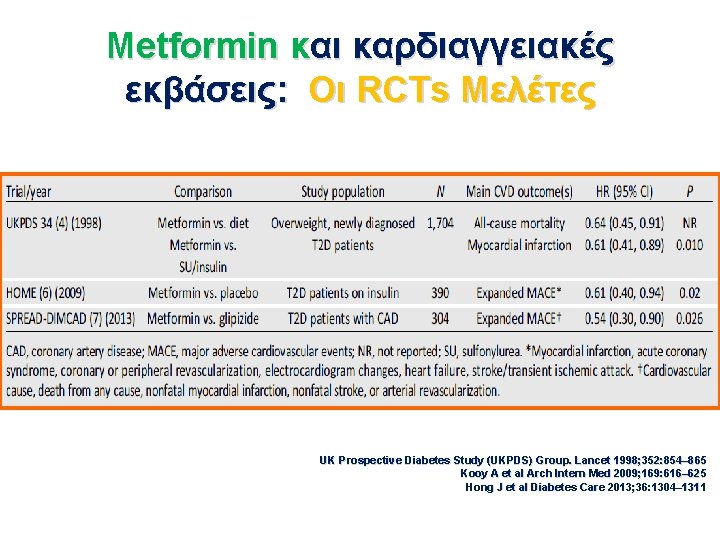
Metformin και καρδιαγγειακές εκβάσεις: Οι RCTs Μελέτες UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998; 352: 854– 865 Kooy A et al Arch Intern Med 2009; 169: 616– 625 Hong J et al Diabetes Care 2013; 36: 1304– 1311





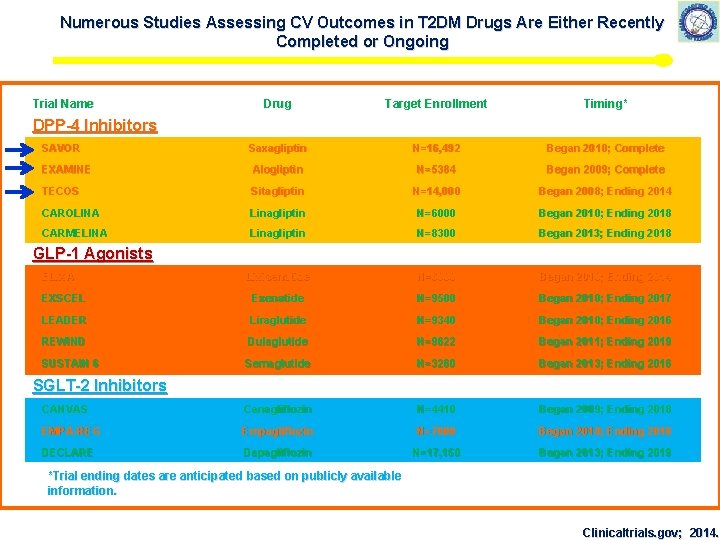
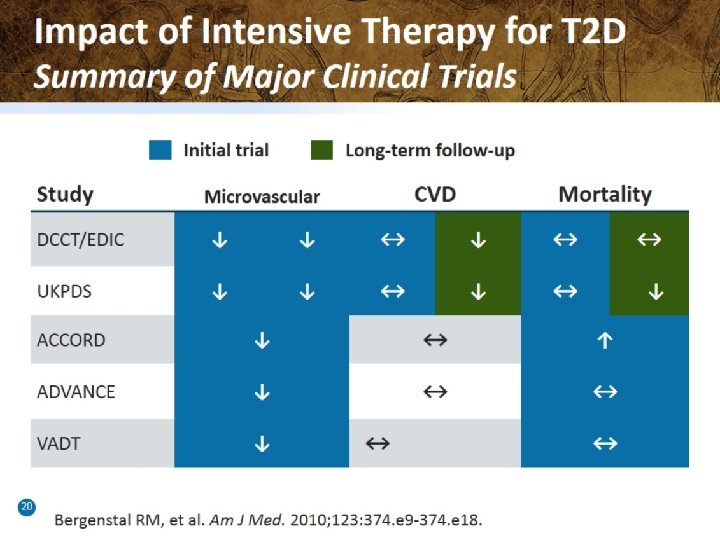
Numerous Studies Assessing CV Outcomes in T 2 DM Drugs Are Either Recently Completed or Ongoing Trial Name Drug Target Enrollment Timing* Saxagliptin N=16, 492 Began 2010; Complete EXAMINE Alogliptin N=5384 Began 2009; Complete TECOS Sitagliptin N=14, 000 Began 2008; Ending 2014 CAROLINA Linagliptin N=6000 Began 2010; Ending 2018 CARMELINA Linagliptin N=8300 Began 2013; Ending 2018 Lixisenatide N=6000 Began 2010; Ending 2014 EXSCEL Exenatide N=9500 Began 2010; Ending 2017 LEADER Liraglutide N=9340 Began 2010; Ending 2016 REWIND Dulaglutide N=9622 Began 2011; Ending 2019 SUSTAIN 6 Semaglutide N=3260 Began 2013; Ending 2016 CANVAS Canagliflozin N=4410 Began 2009; Ending 2018 EMPA-REG Empagliflozin N=7000 Began 2010; Ending 2018 DECLARE Dapagliflozin N=17, 150 Began 2013; Ending 2019 DPP-4 Inhibitors SAVOR GLP-1 Agonists ELIXA SGLT-2 Inhibitors *Trial ending dates are anticipated based on publicly available information. Clinicaltrials. gov; 2014.

Πληθυσμοί Ασθενών που Εντάχθηκαν σε Μελέτες ΚΑ Εκβάσεων των DPP-4 Αναστολέων Παράγοντες Κινδύνου Σταθερή ΣΝ-ΚΑΝ-ΠΑΝ Ασθενείς μετά ΟΣΣ Αλογλιπτίνη EXAMINE (N=5380)1 Σαξαγλιπτίνη SAVOR-TIMI (N=16. 492)2 ΟΣΣ εντός15– 90 ημερών Προϋπάρχουσα ΚΑΝ ή πολλαπλοί παράγοντες κινδύνου για ΚΑΝ Σιταγλιπτίνη TECOS (N=~14. 000)3 Προϋπάρχουσα ΚΑΝ Λιναγλιπτίνη CARMELINA (N=8300)4 Προϋπάρχουσα ΚΑΝ + λευκωματινουρία ή έκπτωση νεφρικής λειτουργίας Παρουσιάστηκ Σεπ 2013 Ολοκλήρωση Δεκ 2014 Ολοκλήρωση Ιαν 2018 Δεν υπάρχει εξελισσόμενη μελέτη ΚΑ εκβάσεων για τη βιλνταγλιπτίνη ΚΑ = καρδιαγγειακός, DPP-4 = διπεπτιδυλική πεπτιδάση-4, , ΣΝ = στεφανιαία νόσος, ΚΑΝ = καρδιαγγειακή νόσος, ΠΑΝ = περιφερική αρτηριακή νόσος, ΟΣΣ = οξύ στεφανιαίο σύνδρομο, EXAMINE = Examination of Cardiovascular Outcomes: Alogliptin vs Standard of Care in Patients With Type 2 Diabetes Mellitus and Acute Coronary Syndrome, SAVOR-TIMI = Saxagliptin Assessment of Vascular Outcomes Recorded in Patients With Diabetes Mellitus Trial. Thrombolysis in Myocardial Infarction, TECOS = Trial Evaluating Cardiovascular Outcomes With Sitagliptin, CARMELINA = Cardiovascular and Renal Microvascular Outcome Study With Linagliptin in Patients With Type 2 Diabetes Mellitus at High Vascular Risk. 1. White W et al. N Engl J Med. 2013; 369: 1327– 1335. 2. Scirica BM et al. N Engl J Med. 2013; 369: 1317– 1326. 3. Green JB et al. Am Heart J 2013; 166: 983– 989. e 7. 4. CARMELINA: Cardiovascular and renal microvascular outcome study with linagliptin in patients with type 2 diabetes mellitus at high vascular risk. Clinical. Trials. gov web site. http: //clinicaltrials. gov/ct 2/show/ NCT 01703298. Accessed September 12, 2014.
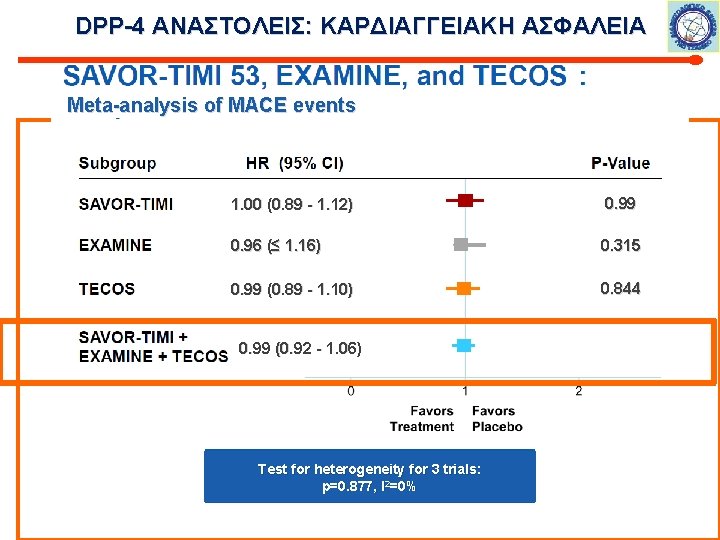
DPP-4 ΑΝΑΣΤΟΛΕΙΣ: ΚΑΡΔΙΑΓΓΕΙΑΚΗ ΑΣΦΑΛΕΙΑ Meta-analysis of MACE events 1. 00 (0. 89 - 1. 12) 0. 99 0. 96 (≤ 1. 16) 0. 315 0. 99 (0. 89 - 1. 10) 0. 844 0. 99 (0. 92 - 1. 06) Test for heterogeneity for 3 trials: p=0. 877, I 2=0%
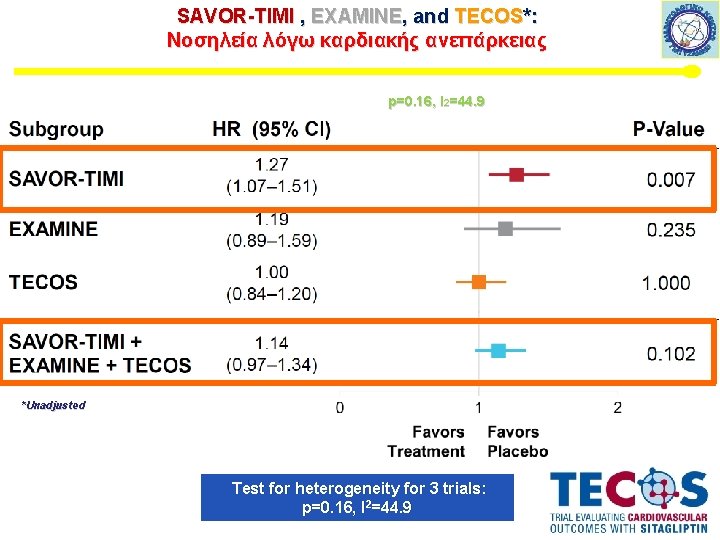
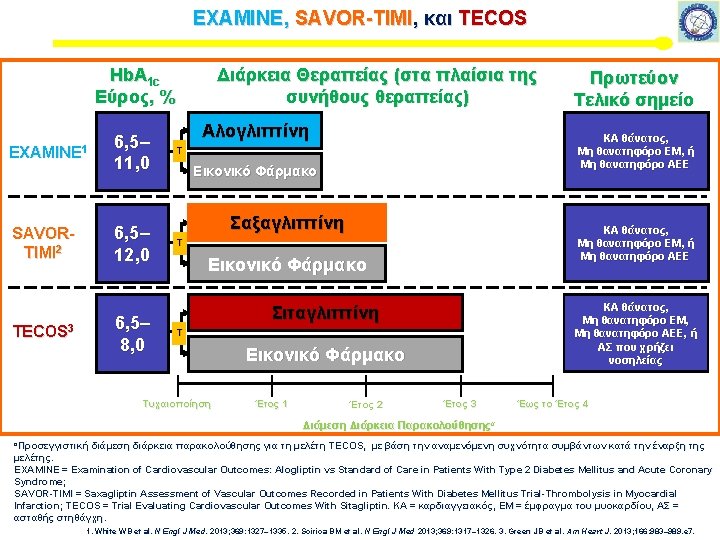
SAVOR-TIMI , EXAMINE, and TECOS*: Νοσηλεία λόγω καρδιακής ανεπάρκειας p=0. 16, I 2=44. 9 *Unadjusted Test for heterogeneity for 3 trials: p=0. 16, I 2=44. 9

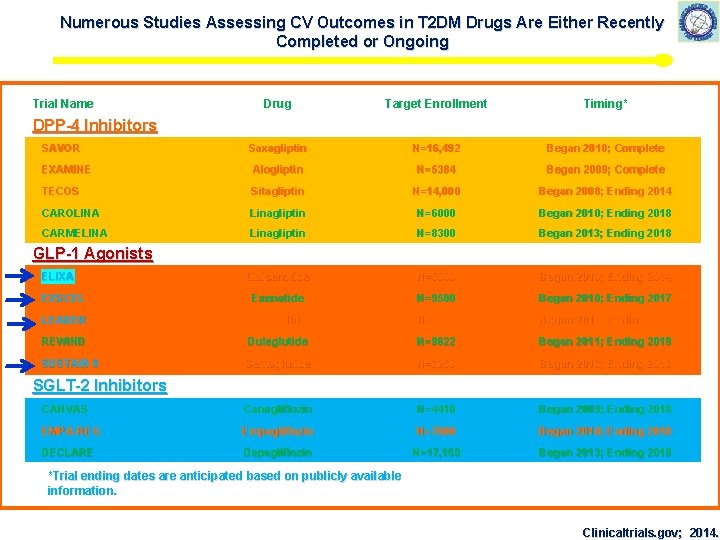
Numerous Studies Assessing CV Outcomes in T 2 DM Drugs Are Either Recently Completed or Ongoing Trial Name Drug Target Enrollment Timing* Saxagliptin N=16, 492 Began 2010; Complete EXAMINE Alogliptin N=5384 Began 2009; Complete TECOS Sitagliptin N=14, 000 Began 2008; Ending 2014 CAROLINA Linagliptin N=6000 Began 2010; Ending 2018 CARMELINA Linagliptin N=8300 Began 2013; Ending 2018 Lixisenatide N=6000 Began 2010; Ending 2014 EXSCEL Exenatide N=9500 Began 2010; Ending 2017 LEADER Liraglutide N=9340 Began 2010; Ending 2016 REWIND Dulaglutide N=9622 Began 2011; Ending 2019 SUSTAIN 6 Semaglutide N=3260 Began 2013; Ending 2016 CANVAS Canagliflozin N=4410 Began 2009; Ending 2018 EMPA-REG Empagliflozin N=7000 Began 2010; Ending 2018 DECLARE Dapagliflozin N=17, 150 Began 2013; Ending 2019 DPP-4 Inhibitors SAVOR GLP-1 Agonists ELIXA SGLT-2 Inhibitors *Trial ending dates are anticipated based on publicly available information. Clinicaltrials. gov; 2014.

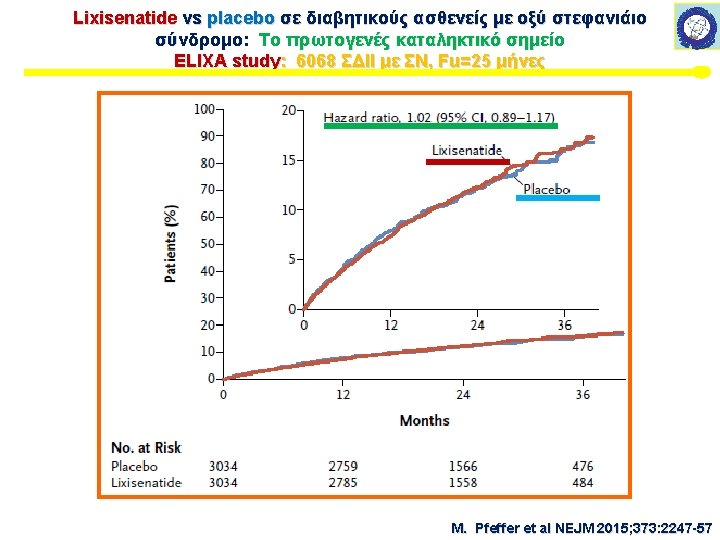
ELIXA study: 6068 ΣΔΙΙ με ΣΝ, Fu=25 μήνες

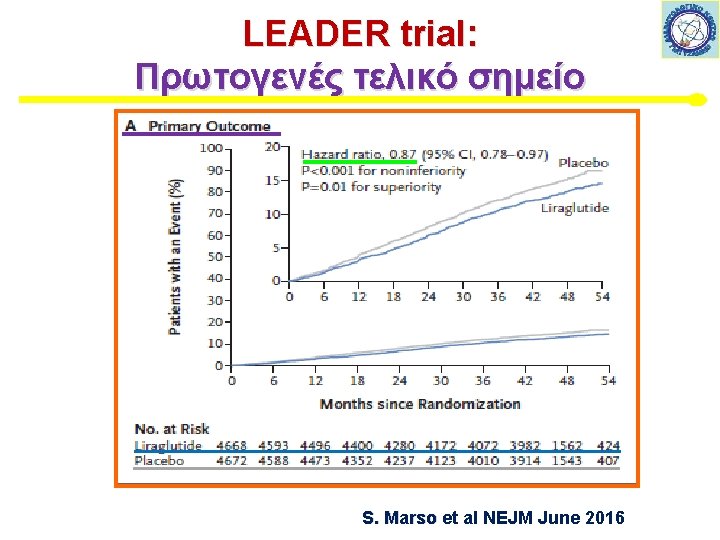
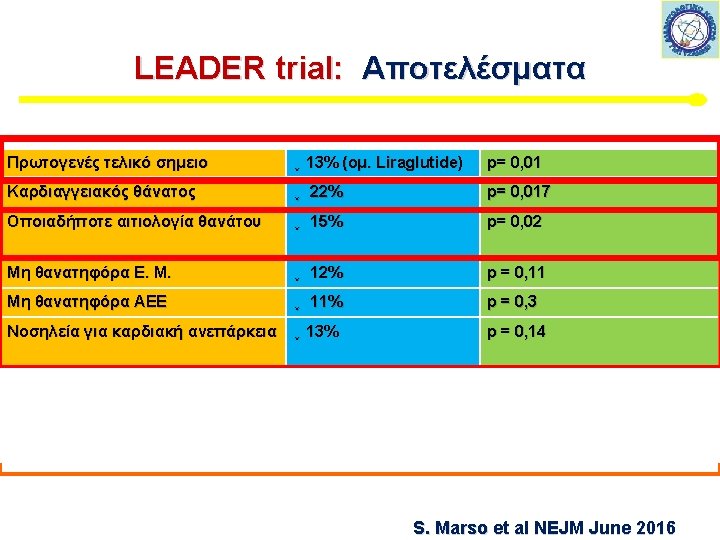
LEADER trial: Πρωτογενές τελικό σημείο S. Marso et al NEJM June 2016

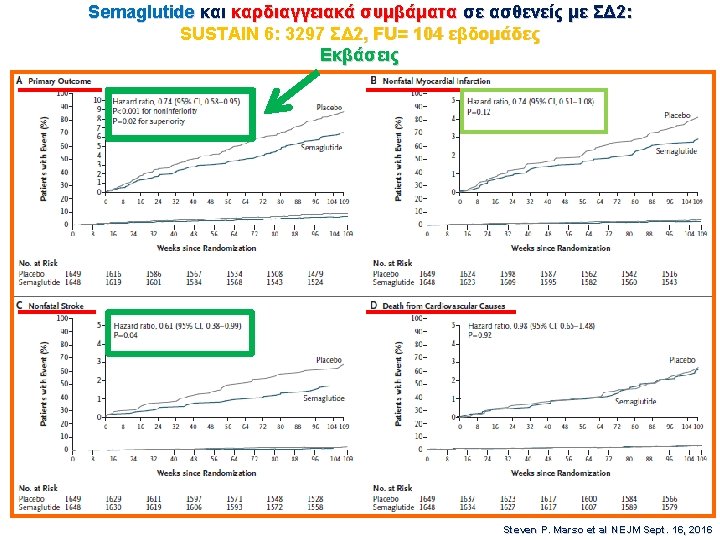
EASD 2016 SUSTAIN-6 3297 ΣΔ 2, FU= 104 εβδομάδες

EXSCEL Primary Results Presentation EASD 2017 Lisbon, Portugal 14752 ΣΔ 2 FU=3, 2 έτη EASD 2017 NEJM 2017
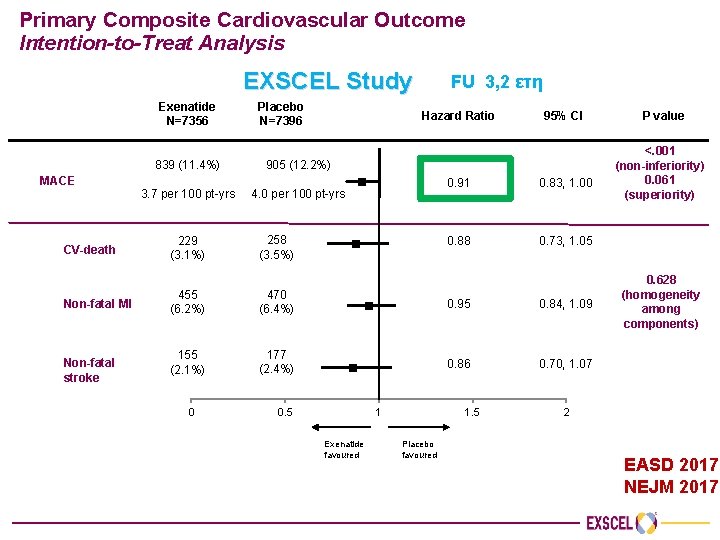
Primary Composite Cardiovascular Outcome Intention-to-Treat Analysis EXSCEL Study Exenatide N=7356 MACE Placebo N=7396 FU 3, 2 ετη Hazard Ratio 839 (11. 4%) 905 (12. 2%) 3. 7 per 100 pt-yrs 4. 0 per 100 pt-yrs 95% CI 0. 91 0. 83, 1. 00 0. 88 0. 73, 1. 05 CV-death 229 (3. 1%) 258 (3. 5%) Non-fatal MI 455 (6. 2%) 470 (6. 4%) 0. 95 0. 84, 1. 09 155 (2. 1%) 177 (2. 4%) 0. 86 0. 70, 1. 07 Non-fatal stroke 0 0. 5 1 Exenatide favoured 1. 5 Placebo favoured P value <. 001 (non-inferiority) 0. 061 (superiority) 0. 628 (homogeneity among components) 2 EASD 2017 NEJM 2017
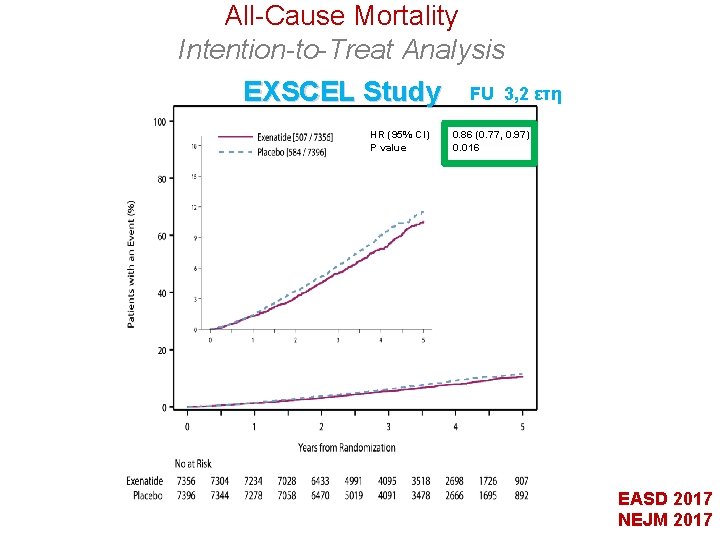
All-Cause Mortality Intention-to-Treat Analysis EXSCEL Study HR (95% CI) P value FU 3, 2 ετη 0. 86 (0. 77, 0. 97) 0. 016 EASD 2017 NEJM 2017

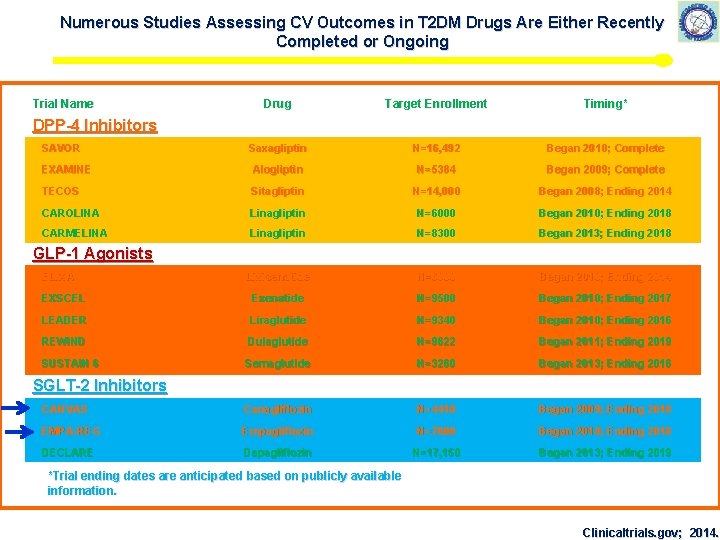
Numerous Studies Assessing CV Outcomes in T 2 DM Drugs Are Either Recently Completed or Ongoing Trial Name Drug Target Enrollment Timing* Saxagliptin N=16, 492 Began 2010; Complete EXAMINE Alogliptin N=5384 Began 2009; Complete TECOS Sitagliptin N=14, 000 Began 2008; Ending 2014 CAROLINA Linagliptin N=6000 Began 2010; Ending 2018 CARMELINA Linagliptin N=8300 Began 2013; Ending 2018 Lixisenatide N=6000 Began 2010; Ending 2014 EXSCEL Exenatide N=9500 Began 2010; Ending 2017 LEADER Liraglutide N=9340 Began 2010; Ending 2016 REWIND Dulaglutide N=9622 Began 2011; Ending 2019 SUSTAIN 6 Semaglutide N=3260 Began 2013; Ending 2016 CANVAS Canagliflozin N=4410 Began 2009; Ending 2018 EMPA-REG Empagliflozin N=7000 Began 2010; Ending 2018 DECLARE Dapagliflozin N=17, 150 Began 2013; Ending 2019 DPP-4 Inhibitors SAVOR GLP-1 Agonists ELIXA SGLT-2 Inhibitors *Trial ending dates are anticipated based on publicly available information. Clinicaltrials. gov; 2014.

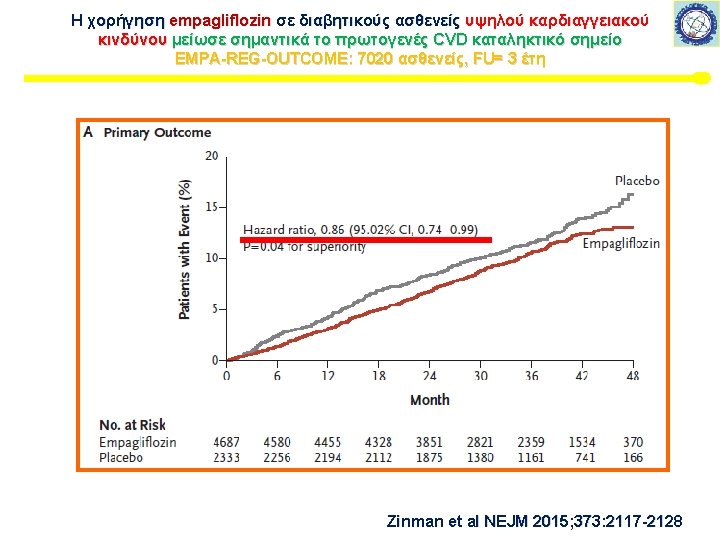
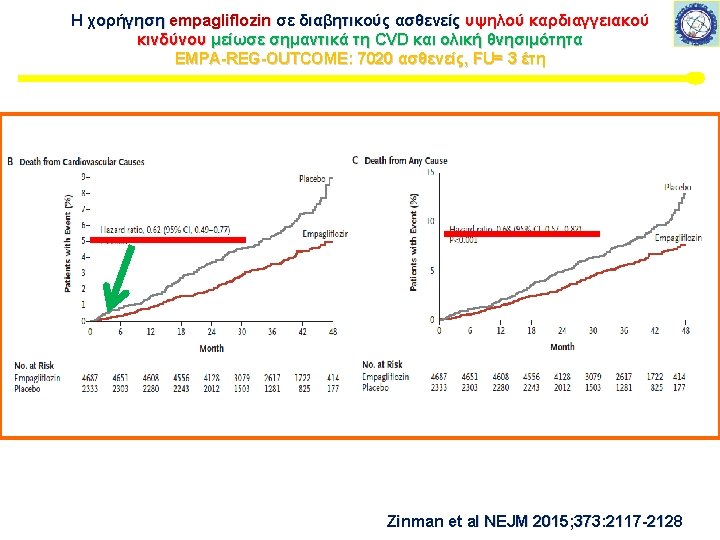
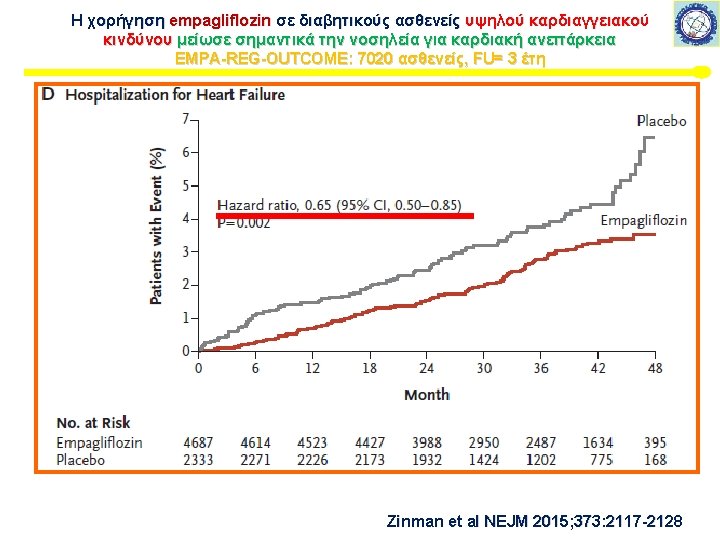
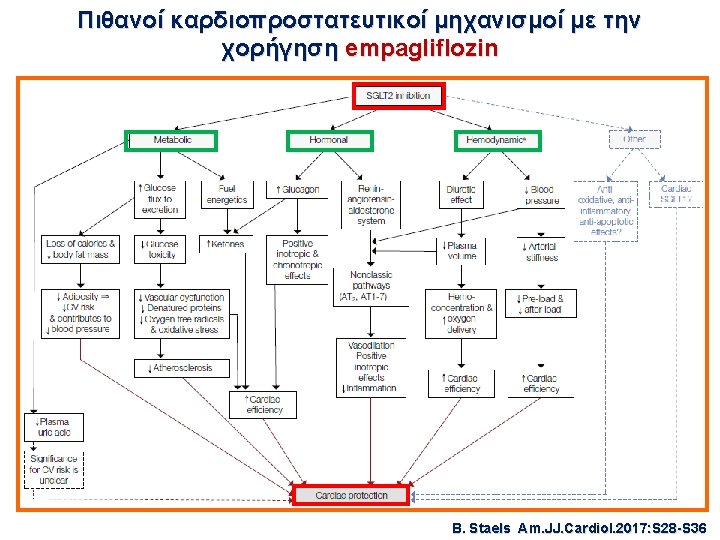
Μελέτη καρδιαγγειακής ασφάλειας με SGLT-2 i Εμπαγλιφλοζίνη – EMPA-REG This article was published on September 17, 2015, at NEJM. org. 7020 ασθενείς, FU= 3 έτη

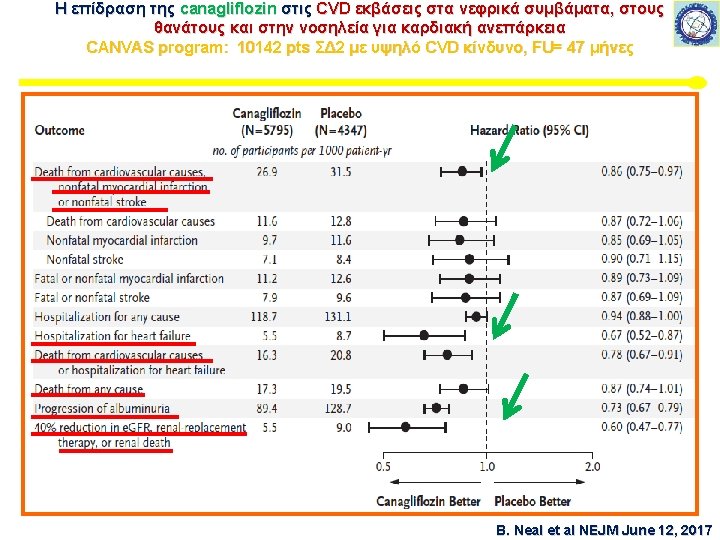
CANVAS program 667 κέντρα – 30 χώρες CANVAS: 4330 ΣΔ 2 – CANVAS – R: 5812 CANVAS program: 9734 ΣΔ 2 Μέσο FU = 188, 2 εβδομάδες B. Neal et al NEJM June 12, 2017
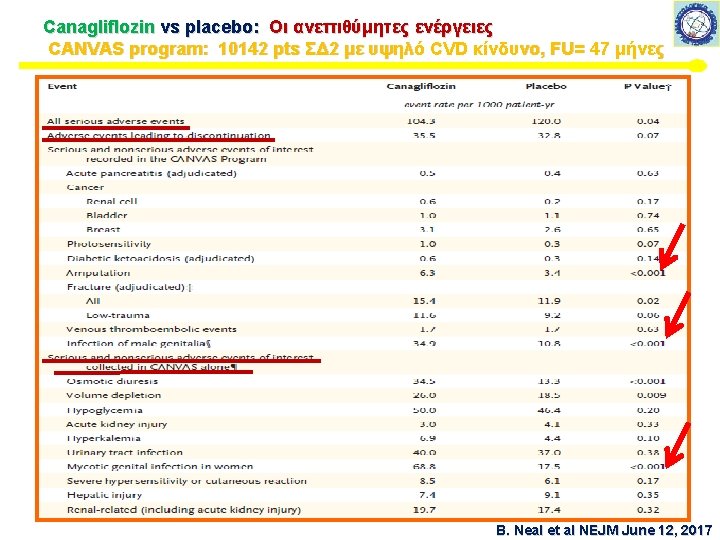
Canagliflozin vs placebo: Οι ανεπιθύμητες ενέργειες CANVAS program: 10142 pts ΣΔ 2 με υψηλό CVD κίνδυνο, FU= 47 μήνες 50 B. Neal et al NEJM June 12, 2017
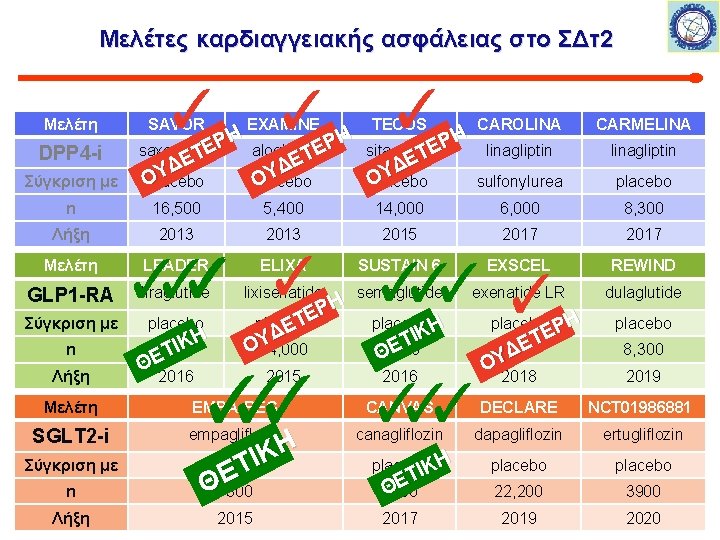
Μελέτες καρδιαγγειακής ασφάλειας στο ΣΔτ2 Μελέτη DPP 4 -i Σύγκριση με ✓ SAVOR ✓ EXAMINE ✓ TECOS Η Η Η Ρ Ρ Ρ Ε Ε Ε saxagliptin alogliptin sitagliptin Τ Τ ΕΤ Ε Ε Δ Δ Δ Υ Υ Υ Ο Ο placebo Ο CAROLINA CARMELINA linagliptin sulfonylurea placebo n 16, 500 5, 400 14, 000 6, 000 8, 300 Λήξη 2013 2015 2017 Μελέτη GLP 1 -RA Σύγκριση με n Λήξη ✓ ✓ ✓✓ LEADER ELIXA SUSTAIN 6 EXSCEL REWIND liraglutide lixisenatide semaglutide exenatide LR dulaglutide placebo Η Κ Ι 9, 340 Τ ΘΕ 2016 ΡΗ Ε Τ placebo Ε ΥΔ Ο 14, 000 placebo Η ✓✓✓ ✓ Η 2015 2016 Η Ρ ΤΕ Ε 5, 400 Δ ΟΥ 2018 placebo ΙΚ Τ Ε 6, 000 Θ placebo 8, 300 2019 Μελέτη EMPA-REG CANVAS DECLARE NCT 01986881 SGLT 2 -i empagliflozin canagliflozin dapagliflozin ertugliflozin Σύγκριση με placebo 22, 200 3900 2019 2020 n Λήξη ΙΚ Τ Ε Θ 7300 2015 placeboΙΚΗ Τ Ε Θ 4300 2017

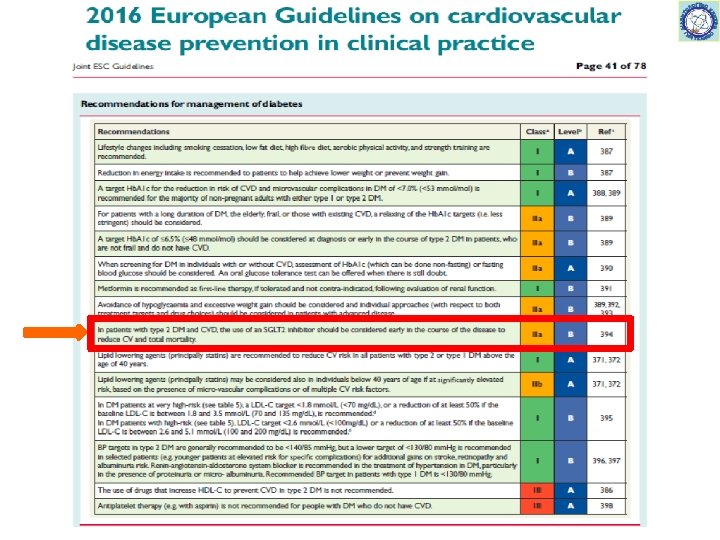
53
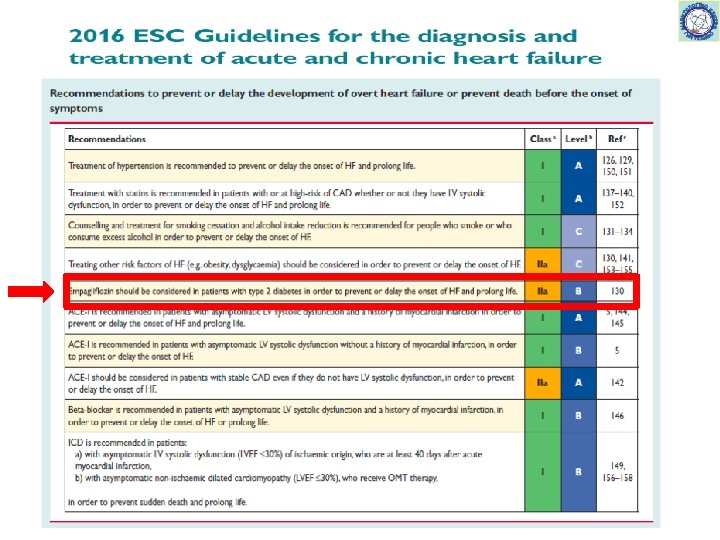
54

ADA Recommendations 2017 In patients with long standing type 2 diabetes and established atherosclerotic Cardiovascular disease, empagliflozin or liraglutide should be considered as they have been shown to reduce cardiovascular and all-cause mortality when added to standard care. Ongoing studies are investigating the cardiovascular benefits of other agents in these drug classes ( B ) Diabetes care 2017; 40(Supplement 1): S 64 -S 74 55
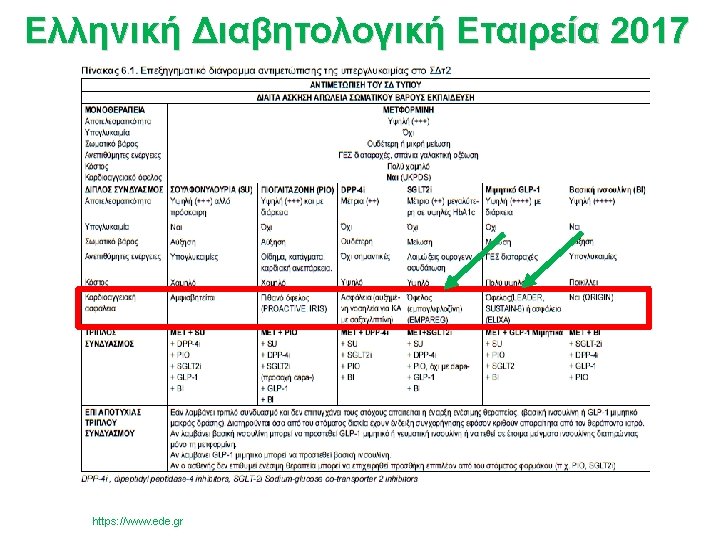


- Slides: 58