Metals Model of Bonding Link to Bozeman Science

Metals: Model of Bonding Link to Bozeman Science

STAGES OF TECHNOLOGY IN HUMAN CIVILIZATION

Stone Age

Bronze Age

Middle Ages

Modern Times

Modern Times l

Introduction – Properties of Metals/ Importance in Civilization List at least 5 Properties of Metals l What do the terms “Stone Age” / “Bronze Age” and “Iron Age” mean in the context of civilizations? Why are metals so important in human history? l Copper age video

Properties of Metals l Luster (Shine) l Malleable l Ductile l Excellent Conductor of Heat and Electricity l Can form Alloys
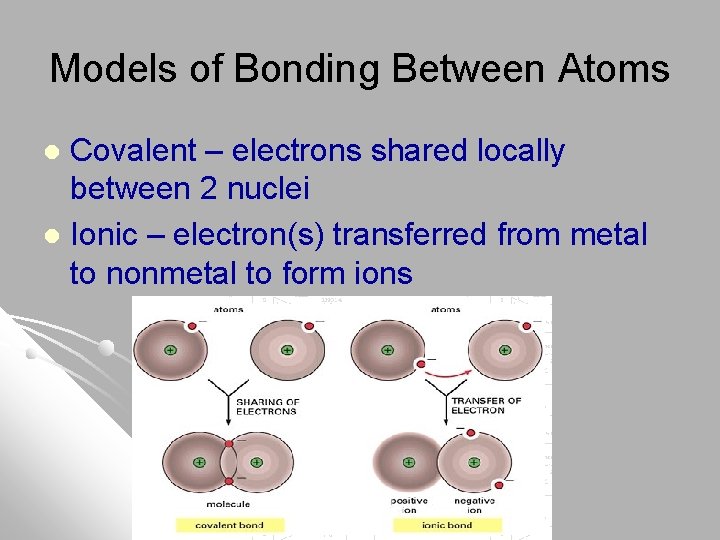
Models of Bonding Between Atoms Covalent – electrons shared locally between 2 nuclei l Ionic – electron(s) transferred from metal to nonmetal to form ions l

Metallic Bonding – “Sea of Electrons Model” l In Metal crystal – electrons are mobile and delocalized (constantly moving throughout entire metal crystal). The valence electrons in metals do not belong to any particular nucleus, but rather are shared by all the different nuclei.
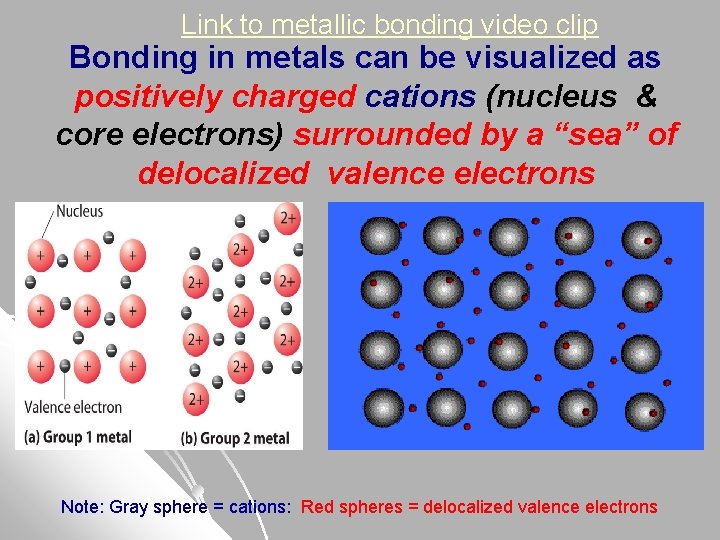
Link to metallic bonding video clip Bonding in metals can be visualized as positively charged cations (nucleus & core electrons) surrounded by a “sea” of delocalized valence electrons Note: Gray sphere = cations: Red spheres = delocalized valence electrons

Properties of Metals 1) LUSTER (polished surfaces reflect light). Most metals appear silvery white (“metallic shine”) because they reflect essentially all wavelengths of light.

Explanation of Luster l Explanation: Electrons constantly moving in different positions near many different nuclei experience a hugh range of energies and hence can make nearly every possible quantum energy transition possible which releases light in the visible range of light. l

Properties of Metals 2) Malleable – can 3) Ductile – can be be shaped; (often requires melting, then resetting shape and allowing to cool). drawn into a wire l Examples: 1 gram of gold can drawn into a wire over 1 mile long.
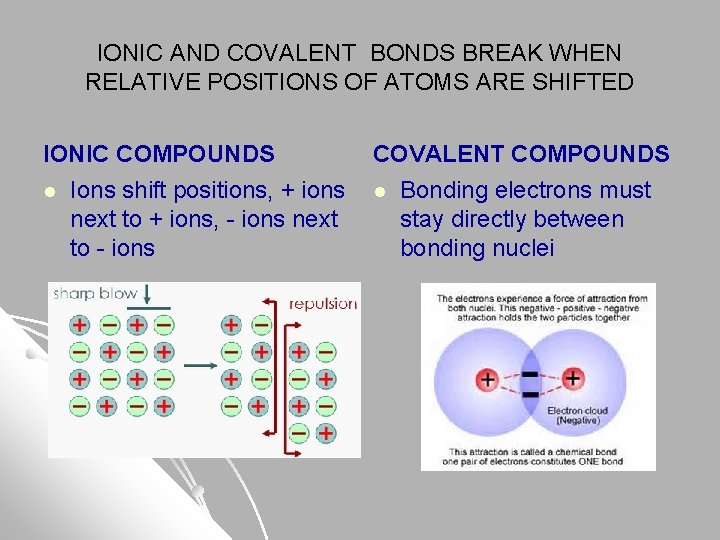
IONIC AND COVALENT BONDS BREAK WHEN RELATIVE POSITIONS OF ATOMS ARE SHIFTED IONIC COMPOUNDS COVALENT COMPOUNDS l Ions shift positions, + ions l Bonding electrons must next to + ions, - ions next stay directly between to - ions bonding nuclei
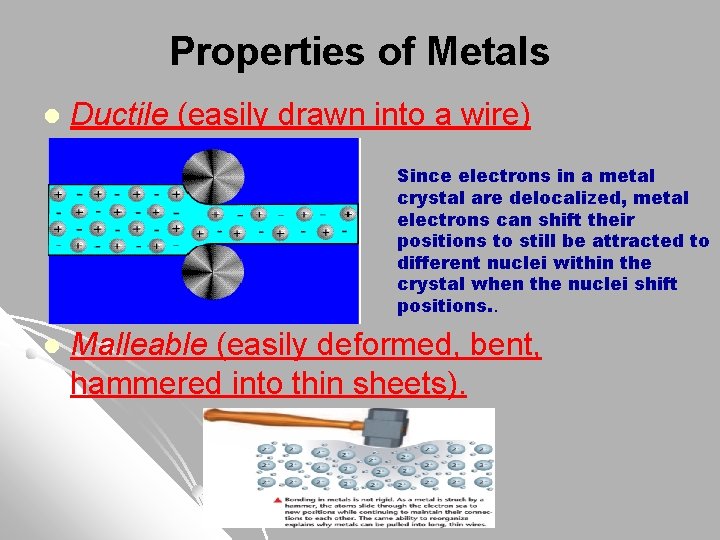
Properties of Metals l Ductile (easily drawn into a wire) Since electrons in a metal crystal are delocalized, metal electrons can shift their positions to still be attracted to different nuclei within the crystal when the nuclei shift positions. . l Malleable (easily deformed, bent, hammered into thin sheets).

Metals are EXCELLENT Conductors of Heat

4) HEAT CONDUCTION EXPLANATION: l MOBILE, DELOCALIZED, ELECTRONS rapidly carry the kinetic energy to different atoms throughout the metal crystal l

5) Metals are EXCELLENT Conductors of Electricity l Explanation: Electrical Current = Flow of Charge. Mobile (moving) Electrons carry current in metals. Electrons enter metal crystal at negative electrode and exit at the positive electrode. animation electrical conductivity
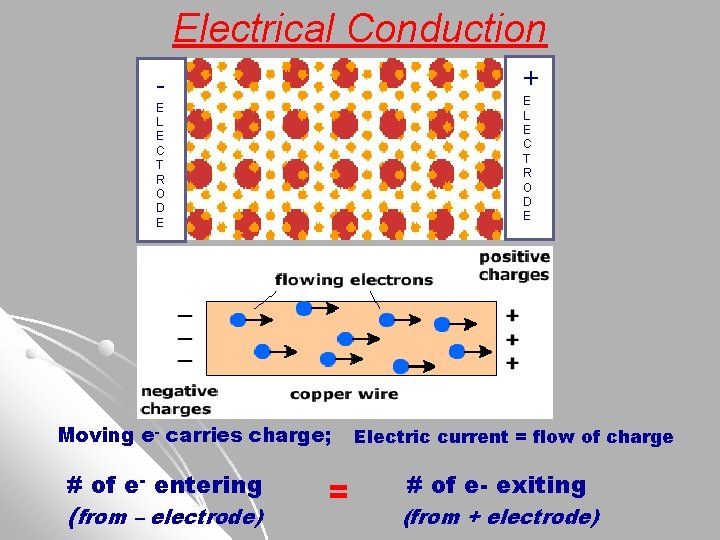
Electrical Conduction + - E L E C T R O D E Moving e- carries charge; # of e- entering (from – electrode) = Electric current = flow of charge # of e- exiting (from + electrode)

Alloys – stronger bonding due to addition of carbon or different metal Making Stuff Stronger Steel Substitute different metal to increase strength Add carbon to iron to increase strength of steel. Less carbon = more flexible steel
- Slides: 22