metals lose valence electrons form cation ion nonmetals










![Lewis Diagram of calcium iodide (Ca. I 2) ·· -1 +2 [Ca] [: I: Lewis Diagram of calcium iodide (Ca. I 2) ·· -1 +2 [Ca] [: I:](https://slidetodoc.com/presentation_image_h2/3e2d142d3933d715b3b794c4612990be/image-17.jpg)

- Slides: 19


• metals: lose valence electrons – form cation (+ ion) • non-metals: gain electrons – form anion (- ion)

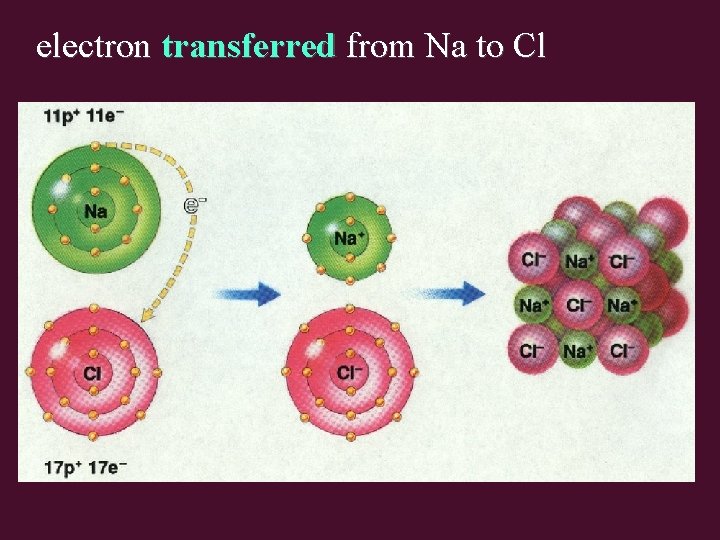
Ionic Bond • occurs between: metal element & non-metal element • involves transfer of electrons between metal & non-metal elements to form ions • # electrons lost by cation(s) = # electrons gained by anion(s)

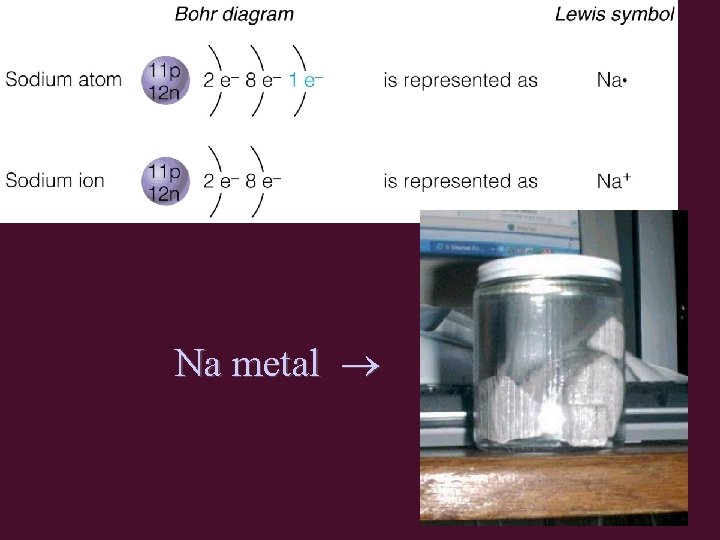
Metals are Losers! • Na atom configuration: 2 -8 -1 • Na loses 1 electron → Na+1 ion – Na+1 configuration is 2 -8 • same as Ne configuration (2 -8)

Na metal

elements from what group _? _ will easily take the one valence electron from Na? ? what group has 7 valence electrons so only need one more? group 1 elements will easily give up their one valence electron to the elements in group 17

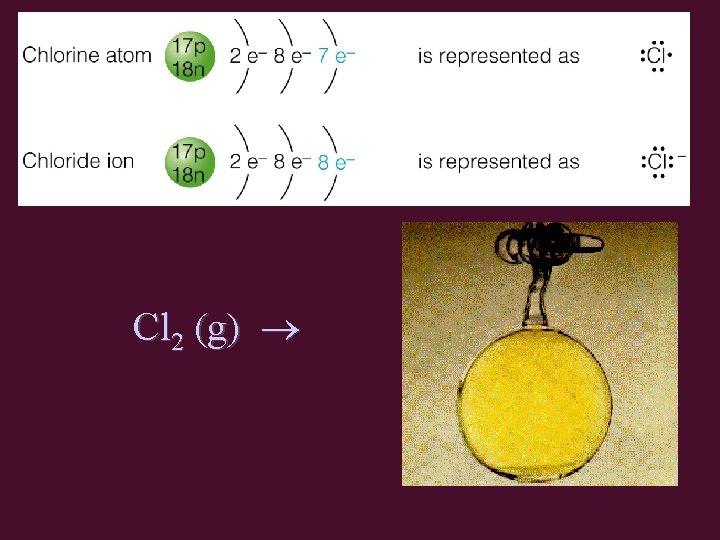
Non-metals are Winners! • Cl configuration: 2 -8 -7 • Cl gains 1 electron → Cl-1 ion – Cl-1 has the configuration 2 -8 -8 • same as Ar configuration (2 -8 -8)

Cl 2 (g)

electron transferred from Na to Cl


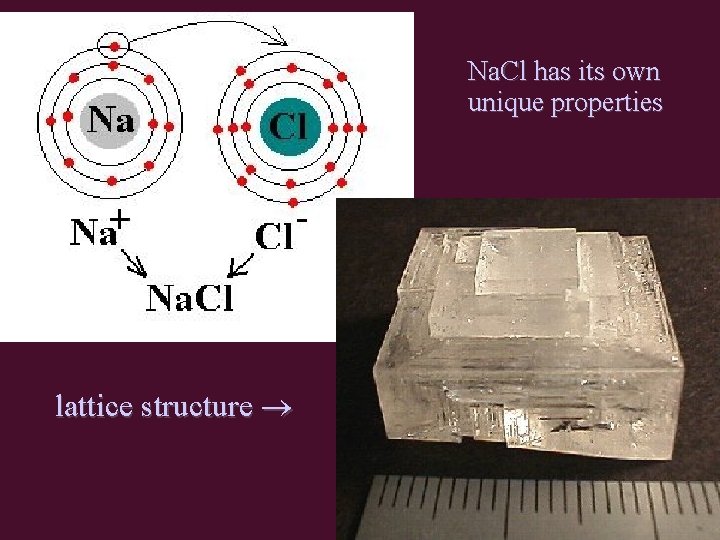
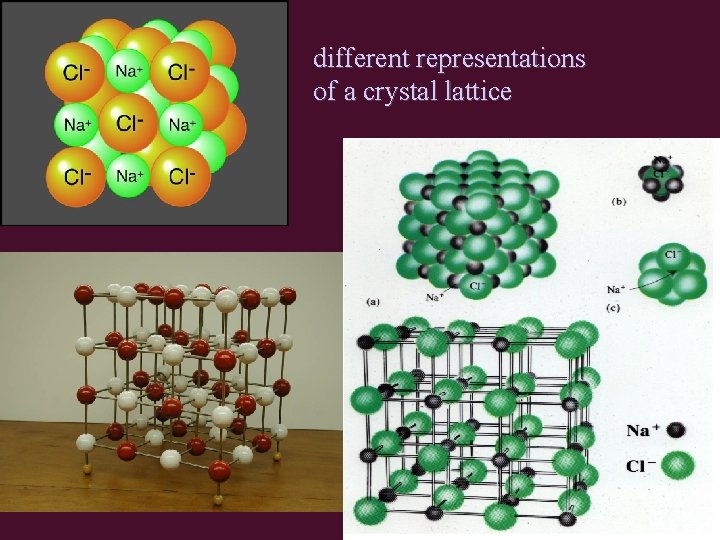
Structure of Ionic Compounds • oppositely charged ions are attracted to each other by strong electrostatic interactions (+/-) • (+/-) ions form crystal lattice – regular 3 -D pattern or array – ions held in fixed positions (solid state) • Unit Cell = smallest repetitive unit in lattice

Na. Cl has its own unique properties lattice structure

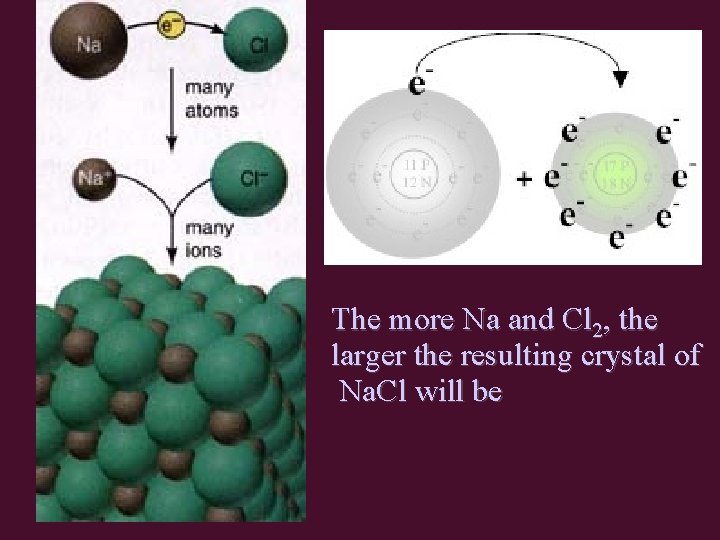
The more Na and Cl 2, the larger the resulting crystal of Na. Cl will be

different representations of a crystal lattice

Lewis Diagrams for Ionic Compounds Lewis structure for Na. Cl shows ions: • • • • -1 +1 [Na] [ • Cl • ] • • remember: Lewis diagrams show valence electrons!

Predicting Ionic Compounds • What is the ionic compound formed from calcium and iodine? • calcium: metal with 2 valence electrons - loses both electrons [Ca]+2 • iodine: non-metal with 7 valence electrons • • -1 - gains 1 electron [ • • I • • ] • • • how many iodine ions are needed to make a neutral compound? 2
![Lewis Diagram of calcium iodide Ca I 2 1 2 Ca I Lewis Diagram of calcium iodide (Ca. I 2) ·· -1 +2 [Ca] [: I:](https://slidetodoc.com/presentation_image_h2/3e2d142d3933d715b3b794c4612990be/image-17.jpg)
Lewis Diagram of calcium iodide (Ca. I 2) ·· -1 +2 [Ca] [: I: ] ·· ·· or ·· -1 +2 [: I: ] [Ca] [: I: ] ·· ·· note: total charge MUST add up to zero since compounds are neutral

Properties of Ionic Compounds • • • high melting points low vapor pressures tend to be hard and brittle solids do not conduct electricity molten (liquid) states do conduct electricity aqueous solutions do conduct electricity

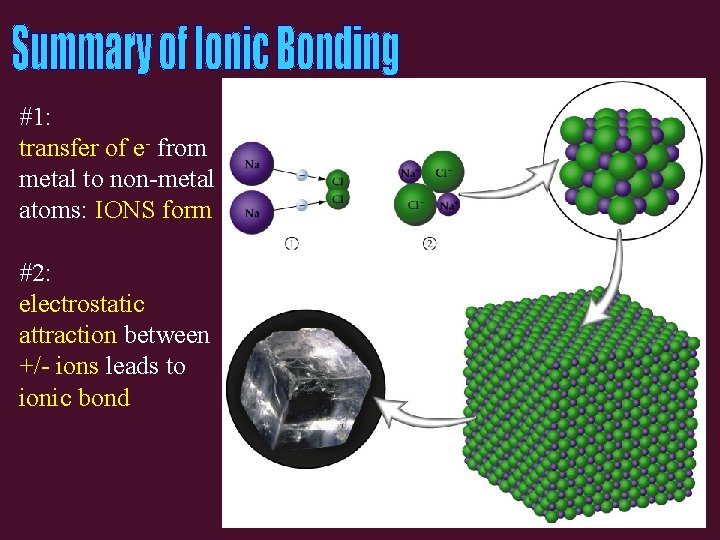
#1: transfer of e- from metal to non-metal atoms: IONS form #2: electrostatic attraction between +/- ions leads to ionic bond