Metals and Ores 26 December 2021 Key words

Metals and Ores 26 December 2021 Key words: Metal, ore, copper, electrolysis, anode, cathode Starter What is the brown substance on the car?

Metals and Ores 26 December 2021 Key words: Metal, ore, copper, electrolysis, anode, cathode Starter What is the brown substance on the car? Rust How does rust form?

Learning Outcomes • Recall that unreactive metals are found in the Earth as metals and more reactive metals are found as ores • Describe that most metals are found as compounds that need chemical reactions to extract the metal • Explain that metals less reactive than carbon can be extracted by heating the metal ore with carbon

We will focus on: Independent enquirers Self - managers

Activity Date written and underlined Rusting Metal Ores Extracting methods and reactivity series Copper and electrolysis Approach to learning and SPLATS Tick when done

Corrosion • Rust forms when iron reacts with the oxygen and water. • This has a fancy name called iron oxide

Preventing corrosion • Galvanising prevents rusting by coating steel with a coat of zinc. • Instead of the oxygen reacting with the iron in the steel the zinc reacts with the oxygen
Activity Date written and underlined Rusting Metal Ores Extracting methods and reactivity series Copper and electrolysis Approach to learning and SPLATS Tick when done

Unreactive Metals • Very unreactive metals (such as silver and gold) do not react (combine) with elements and compounds around them, so they are found in the ground as a pure metal.

Metal Ores • Most metals are found in the Earth’s crust combined with other elements in rocks known as ores. • For example, iron is found combined with oxygen in ores. • Metals need to be extracted from ores before they can be turned into useful products, such as cars or cutlery. • The extraction of metals and minerals is the fifth-largest industry in the world.

How do you get to the metal ores? • Mining for ores is expensive and so is only carried out where minerals are abundant enough for this to be profitable. • In industry, an ore is a rock that contains enough metal to make mining and extraction economical • For example, rocks containing only 5% copper would have been considered unprofitable in the 19 th century. Today, most copper comes from ores containing 0. 4 to 1% copper. Why do you think this percentage has changed?
Activity Date written and underlined Rusting Metal Ores Extracting methods and reactivity series Copper and electrolysis Approach to learning and SPLATS Tick when done

Extraction • Extraction methods vary for different metals (whether they are reactive or unreactive) • Gold and other unreactive metals occur in the ground as uncombined elements do not require further extraction. • Most metals are found combined with other elements, as compounds in ores. These metals need to be separated from the other elements that they are combined with using chemical reactions. • There are two main ways of extracting metals from their ores: – Burning ores with carbon (reduction) – Electrolysis

Reactivity Series Metals above carbon in the reactivity series must be extracted using electrolysis. Electrolysis can also be used to purify copper. Metals less reactive than carbon can be extracted from their ores by reduction using carbon, coke or charcoal. Platinum, gold, and silver can occur as pure metals and do not need to be extracted.

Reduction • Metals are often found combined with oxygen as oxides. To obtain the metal, the oxygen must be removed. • The removal of oxygen from a substance is called reduction metal oxide (in ore) reduction metal • https: //www. youtube. com/watch? v=fx. BIgb. RT 8 f w

Reduction • Carbon can be used to extract metals by reduction. lead oxide Pb. O + + carbon C lead Pb + + carbon monoxide CO • In this reaction, the carbon removes oxygen from lead oxide. This occurs because carbon is more reactive than lead. • The addition of oxygen to a substance is called oxidation

What can copper be used for? • Coins • Water pipes • Electrical wires
Activity Date written and underlined Rusting Metal Ores Extracting methods and reactivity series Copper and electrolysis Approach to learning and SPLATS Tick when done

Why use copper? • Copper is an excellent conductor and does not corrode quickly. These properties make it a good material for wiring and plumbing. • Only pure copper can be used for electric wires. Even a very low level of impurities will reduce copper’s conductivity.

How is copper purified? • Copper is not very reactive and can occur native but it is rare to find pure copper. Usually, it is found combined with other elements in ores. • The copper extracted from compounds by reduction with carbon is impure. Electrolysis can actually be used at this stage to remove the impurities and obtain pure copper.
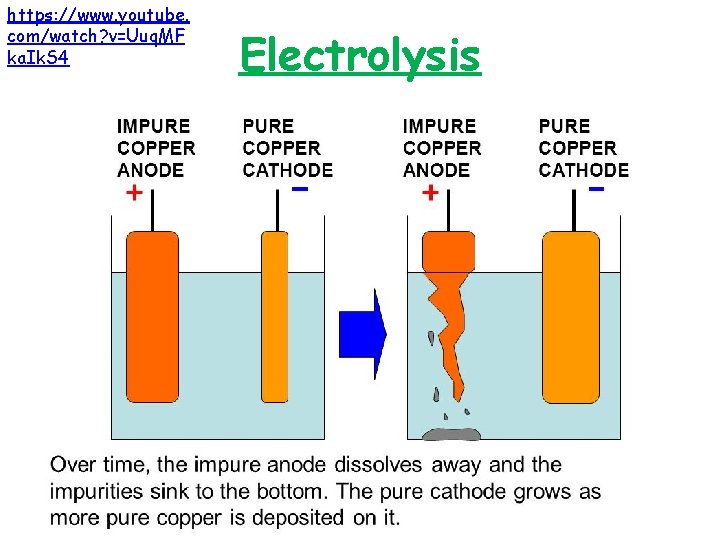
https: //www. youtube. com/watch? v=Uuq. MF ka. Ik. S 4 Electrolysis
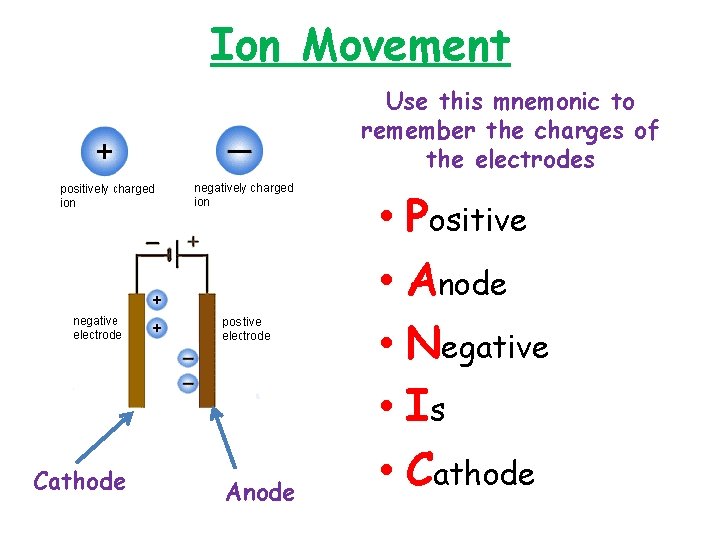
Ion Movement Use this mnemonic to remember the charges of the electrodes Cathode Anode • Positive • Anode • Negative • Is • Cathode

Summary questions 1. In what form are most metals found in the ground? 2. Describe what rust is. 3. Describe what oxidation is. 4. Describe what reduction is. 5. State uses of copper. 6. Explain how electrolysis works.
Activity Date written and underlined Rusting Metal Ores Extracting methods and reactivity series Copper and electrolysis Approach to learning and SPLATS Tick when done
Did we achieve our LOs? • Recall that unreactive metals are found in the Earth as metals and more reactive metals are found as ores • Describe that most metals are found as compounds that need chemical reactions to extract the metal • Explain that metals less reactive than carbon can be extracted by heating the metal ore with carbon

Did we achieve our focus: Independent enquirers Self - managers

Today’s Lesson: Independent enquirers Creative Thinkers Reflective Learners Team workers Self - managers Effective Participators 26 December 2021
- Slides: 27