Metallic and Ionic Nanoparticles Extendable Structures Reactivity Catalysis

Metallic and Ionic Nanoparticles Extendable Structures: Reactivity, Catalysis, Adsorption

Nanoparticles Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 2

Nanoparticles Think Coordination Numbers! Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 3

Nanoparticles Attracted to lots of nearest neighbors High Coordination Numbers Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 4

Nanoparticles Attracted to few nearest neighbors Low Coordination Numbers Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 5

Nanoparticles Surface atoms have Higher energy! E (surface - E (interior atoms) = E (surface) Surface Energy! Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 6
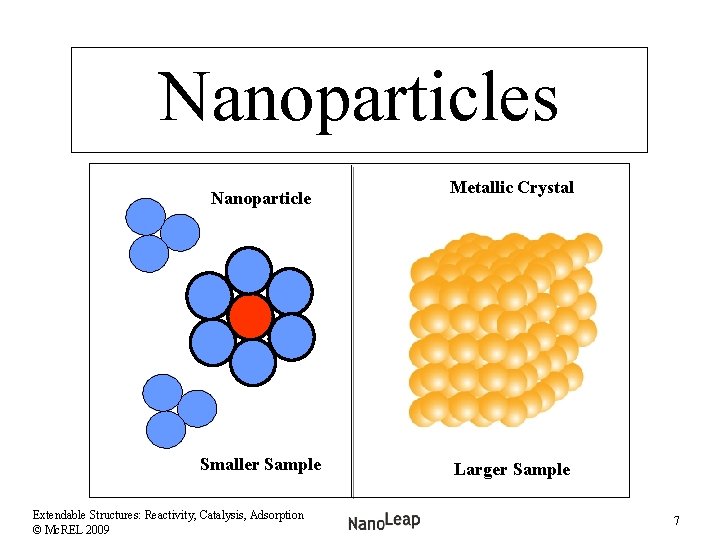
Nanoparticles Nanoparticle Smaller Sample Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 Metallic Crystal Larger Sample 7

Nanoparticles …is where the interactions that result in changes in physical and chemical properties occur. …is where chemical reactions take place. Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 8

Nanoparticles Factors that affect the RATE of chemical reactions 1. Size of reaction particles and increasing surface area 2. Concentrations of reactants 3. Temperature 4. Presence of a catalyst Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 9

Nanoparticles What factors account for the increase in reaction rates of chemical processes at the nanoscale level? As the size of nanoscale particles decreases, the surface area to volume ratio increases. Therefore, the surface energy increases! Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 10

Nanoparticles Adsorption takes place at the surface. Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 11

Nanoparticles 7 grams of nanoparticles (four nm) have a surface area equivalent to a football field Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 12

Nanoparticles Catalytic reactions takes place at the surface. Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 13

Nanoparticles Nanowires Gold Catalytic Surface Scanning Electron Microscop (SEM) showing the vertical growth of nanowires on a gold catalytic surface for electronic devices (Stanford Nanofabrication Facility) Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 14

Metallic and Ionic Nanoparticles Ø Decreases in the size of particles Ø Increases in surface area Increase the rates of some chemical reactions Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 15

Nanoparticles Iron nanoparticles in YOUR backyard? http: //www. phschool. com/science_news/articles/special_treatment. html Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 16

Making Connections 1. What chemical properties were most effected by surface energy? 2. How were the chemical properties affected by surface energy? 3. What physical properties might be affected by surface energy? Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 17
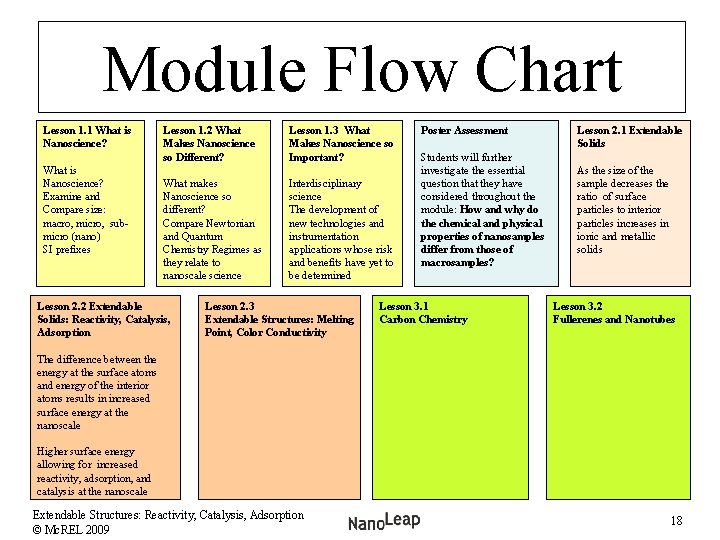
Module Flow Chart Lesson 1. 1 What is Nanoscience? Examine and Compare size: macro, micro, submicro (nano) SI prefixes Lesson 1. 2 What Makes Nanoscience so Different? Lesson 1. 3 What Makes Nanoscience so Important? What makes Nanoscience so different? Compare Newtonian and Quantum Chemistry Regimes as they relate to nanoscale science Interdisciplinary science The development of new technologies and instrumentation applications whose risk and benefits have yet to be determined Lesson 2. 2 Extendable Solids: Reactivity, Catalysis, Adsorption Lesson 2. 3 Extendable Structures: Melting Point, Color Conductivity Poster Assessment Students will further investigate the essential question that they have considered throughout the module: How and why do the chemical and physical properties of nanosamples differ from those of macrosamples? Lesson 3. 1 Carbon Chemistry Lesson 2. 1 Extendable Solids As the size of the sample decreases the ratio of surface particles to interior particles increases in ionic and metallic solids Lesson 3. 2 Fullerenes and Nanotubes The difference between the energy at the surface atoms and energy of the interior atoms results in increased surface energy at the nanoscale Higher surface energy allowing for increased reactivity, adsorption, and catalysis at the nanoscale Extendable Structures: Reactivity, Catalysis, Adsorption © Mc. REL 2009 18
- Slides: 18