Metal speciation using ionselective electrodes Sbastien Sauv Department

Metal speciation using ion-selective electrodes Sébastien Sauvé Department of Chemistry Université de Montréal sebastien. sauve@ umontreal. ca © Sauvé 2002

Ion selective electrodes n Prejudiced against n Often, presumed unreliable n Very easy to use n Give a simple, direct measurement of free ionic activity n Commercial combined electrodes can be used with as little as ~5 m. L of solution sample n Cheap © Sauvé 2002

Avdeef et al. 1983 © Sauvé 2002

Prejudice n Too often, confusion over the speciation vs. concentration comparisons, i. e. , not accounting for complexation n The « limit of detection » in dilute salts given around 10 -7 M is close the background concentration expected in clean solutions (resulting in a standard addition type of plateau) © Sauvé 2002

Cupric Ion-Selective Electrodes n Linear, Nernstian response down to p. Cu 2+ of: w 7 in dilute copper salts solutions (60 µg·L-1) w 19 using solutions copper-buffered with ligands of known stability constants (10 -19 M or 60 ag·L-1) n Simple equipment n Extensive literature © Sauvé 2002

Cupric Ion-Selective Electrodes n Interferences w Ionic strength variations • Need a relatively uniform IS w Aluminum w Mercury w Chloride n Electrode surface is sensitive © Sauvé 2002

Cupric Electrode Calibration n Suggested Cu-IDA calibration solutions have: • 1· 10 -3 M IDA • 1· 10 -4 M Cu(NO 3)2 • 6· 10 -3 M Na. OH • 2. 5· 10 -3 M KHphthalate • 1· 10 -2 M Ca. Cl 2 (media) • p. H adjusted with HNO 3 w Use IDA stability constants reported in the literature, interpolated to 0. 02 ionic strength © Sauvé 2002
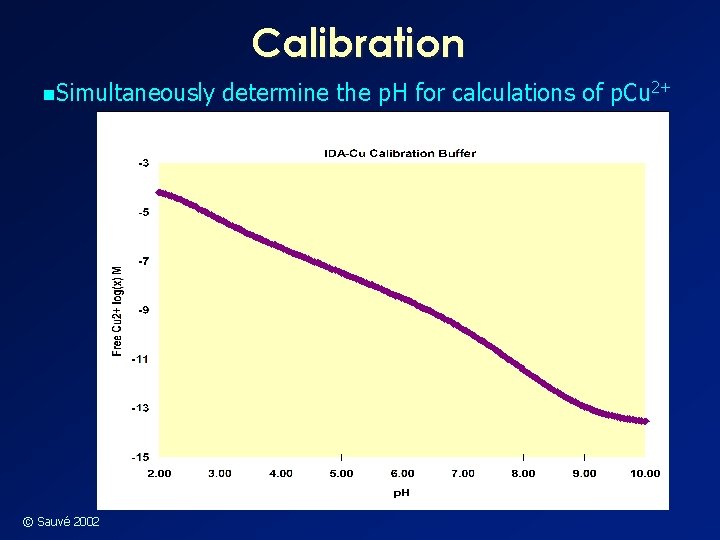
Calibration n. Simultaneously © Sauvé 2002 determine the p. H for calculations of p. Cu 2+
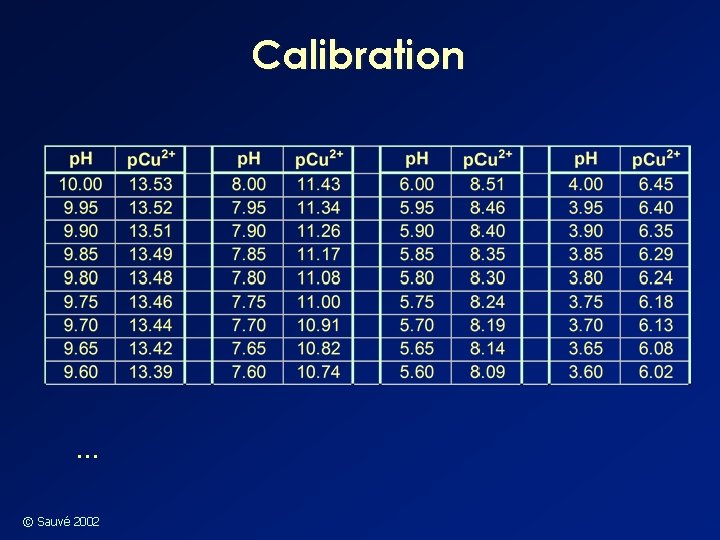
Calibration … © Sauvé 2002

Electrode Calibration n I considered the electrode to be equilibrated when the potential stays within the same 0. 3 m. V range for 3 min w (Very slow equilibration time — about two hours in the lowest activity samples) n Calibration and samples are analyzed in order of increasing activities, otherwise a much longer equilibration time is neccessary (especially when there is a large decrease in activity between two samples) © Sauvé 2002
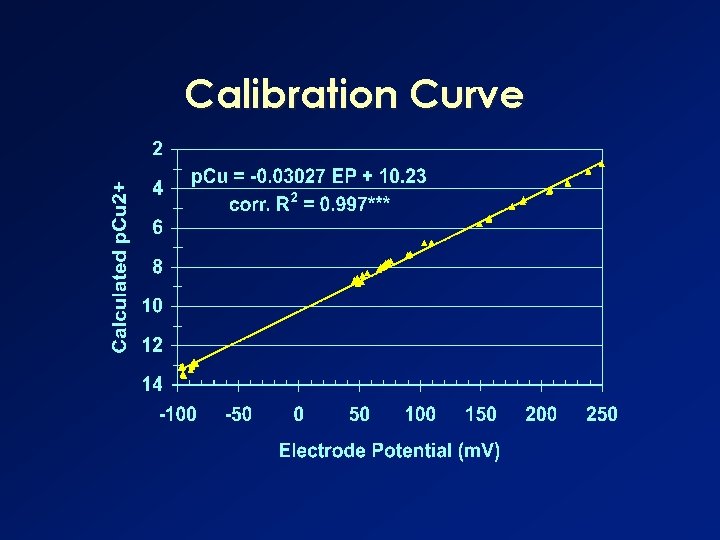
Calibration Curve
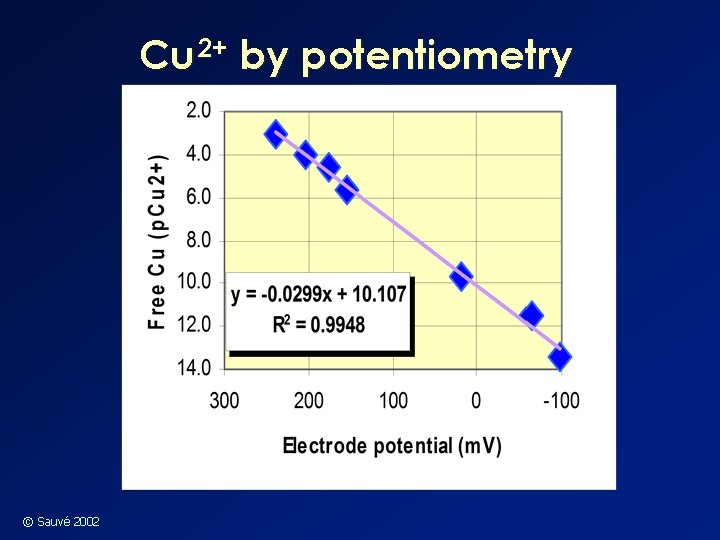
Cu 2+ by potentiometry © Sauvé 2002

Procedures n Soil preparation w Soil is air-dried and ground to 2 mm w Shake 5. 00 g of soil in 10. 00 m. L of 0. 01 M Ca. Cl 2 for 20 min w Centrifuge 10 min at 10000 g n Determination of p. Cu 2+ w Electrode potential measured in 20 -m. L polystyrene cups shaken by hand (or with stirrer, but systematically…) © Sauvé 2002

Ionic Strength n Statistically significant but negligible ionic strength effect where EP is in m. V and IS is the ionic strength l © Sauvé 2002 The IS in the soil extracts is 0. 02± 0. 01 so, one SD = 0. 314 m. V (~0. 01 p. Cu 2+)
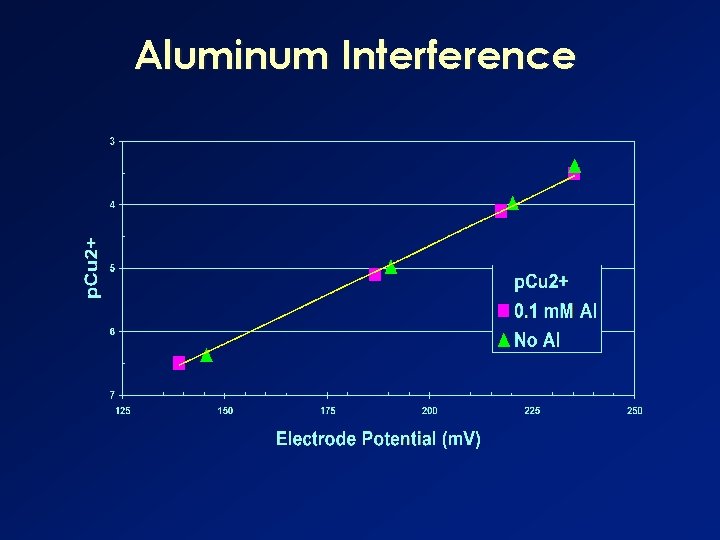
Aluminum Interference

Chloride Interference n Cu(II) is reduced at the electrode surface to Cu(I), which is stabilized by chloride complexation n The electrode the respond to a combination of Cu(II) and Cu(I), which also changes the Nernstian slope from 59 to 29 m. V/decade n Critical Cl concentration around 10 -1. 4 M (Westall et al. 1979), which prevents the use of the Cu ISE in seawater (~0. 5 M Cl) © Sauvé 2002
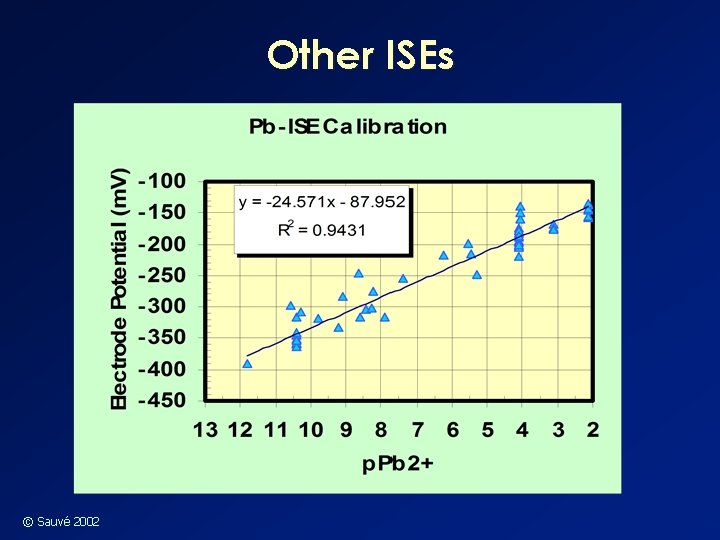
Other ISEs © Sauvé 2002

Other ISE’s n Cadmium and Lead w They are somewhat selective but could still possibly be used to measure Cu 2+… w Might be prone to interferences from natural organic matter and/or oxides w Will be useful in synthetic solutions of known composition © Sauvé 2002

Large selection n NH 3, NH 4+, Br+, Cd 2+, Ca 2+, CO 2, Cl-, Cl 2, Cu 2+, CN-, F-, I-, Pb 2+, NO 3 -, NO 2 -, NOx, O 2, Cl. O 4 -, K+, Redox, Ag+/S 2 -, Na+, SCN- n Analytical confidence needs confirmation, but many environmental applications could be better exploited © Sauvé 2002
- Slides: 19