Metal in medicine Medicinal inorganic chemistry Biomedical inorganic


Metal in medicine Medicinal inorganic chemistry Biomedical inorganic chemistry 2
History 3000 BC : Egyptians used Cu to sterilize water 2500 BC : Chinese empire uses Au in a variety of medicine 400 BC : Hippocrates used Hg 1600 s : Paracelsus pioneered the use of minerals in medicine using Sb, As, Mg salt Early 1900 s : Metals started making an impact on modern medicine K[Au(CN)2] used for tuberculosis Salvarsan for the treatment of syphilis Jaouen, G. Bioorganometallics, 2006, 1 st Ed. pp. 1 -32 Orvig, C. Abrams, M. J. Chem. Rev. 1999, 2201

Outline 1. Traditional applications of inorganic compounds: - Chelation - Imaging properties 2. Inorganic compounds that utilize reactivity of metals 3. Inorganic compound that utilizes both the structure of metal and their reactivity in biological system 4. Inorganic compounds that utilize the unique structural opportunities of metals 4
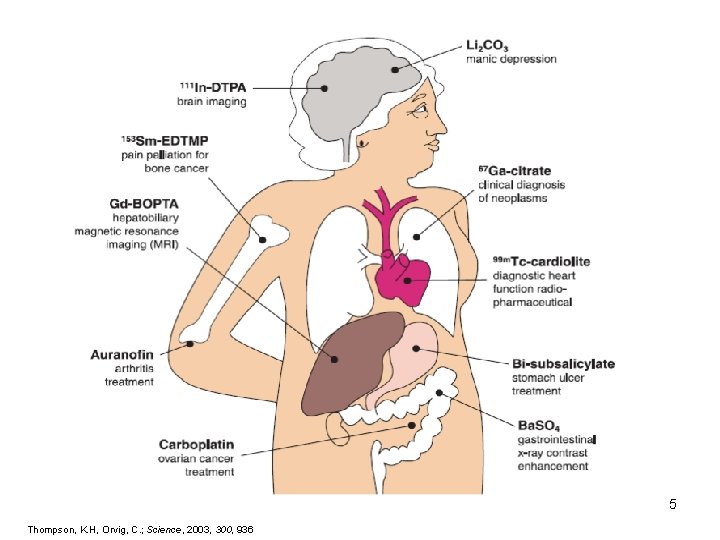
5 Thompson, K. H, Orvig, C. ; Science, 2003, 300, 936
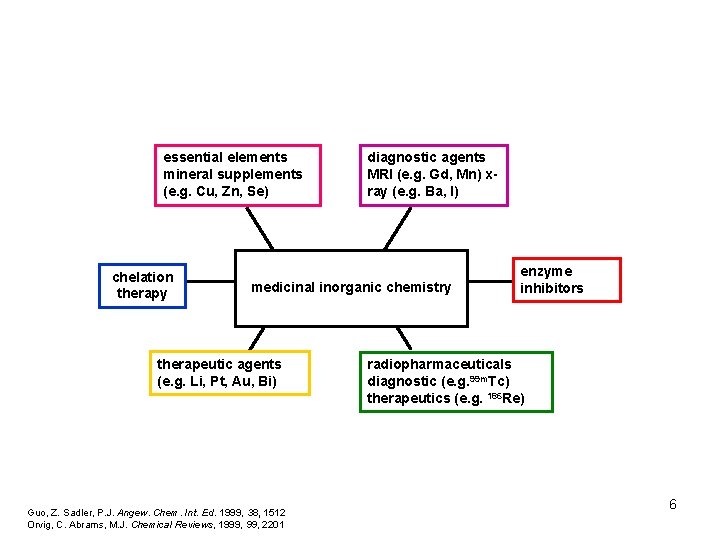
essential elements mineral supplements (e. g. Cu, Zn, Se) chelation therapy diagnostic agents MRI (e. g. Gd, Mn) xray (e. g. Ba, I) medicinal inorganic chemistry therapeutic agents (e. g. Li, Pt, Au, Bi) Guo, Z. Sadler, P. J. Angew. Chem. Int. Ed. 1999, 38, 1512 Orvig, C. Abrams, M. J. Chemical Reviews, 1999, 2201 enzyme inhibitors radiopharmaceuticals diagnostic (e. g. 99 m. Tc) therapeutics (e. g. 186 Re) 6
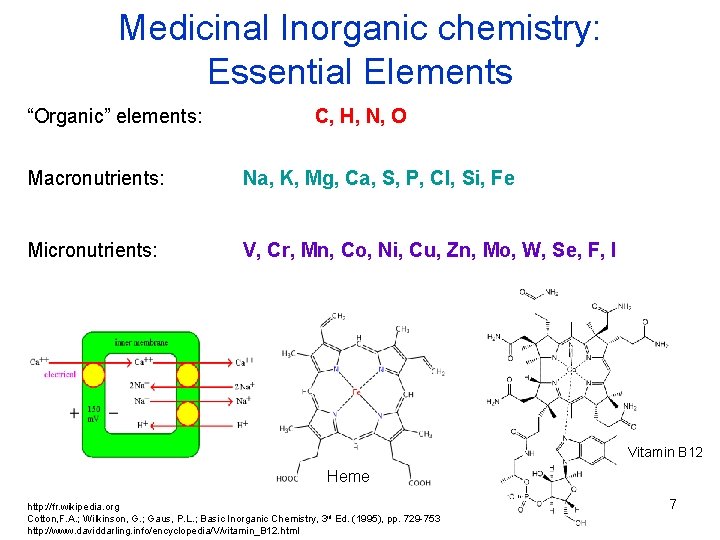
Medicinal Inorganic chemistry: Essential Elements “Organic” elements: C, H, N, O Macronutrients: Na, K, Mg, Ca, S, P, Cl, Si, Fe Micronutrients: V, Cr, Mn, Co, Ni, Cu, Zn, Mo, W, Se, F, I Vitamin B 12 Heme http: //fr. wikipedia. org Cotton, F. A. ; Wilkinson, G. ; Gaus, P. L. ; Basic Inorganic Chemistry, 3 rd Ed. (1995), pp. 729 -753 http: //www. daviddarling. info/encyclopedia/V/vitamin_B 12. html 7

Medicinal Inorganic chemistry: Chelation Therapy Used for metal intoxication 1941: Citrate is used for acute lead intoxication Since then, other chelating agents have come into clinical use: EDTA TETA DMSA 8 Andersen, O. Chem. Rev. 1999, 2683
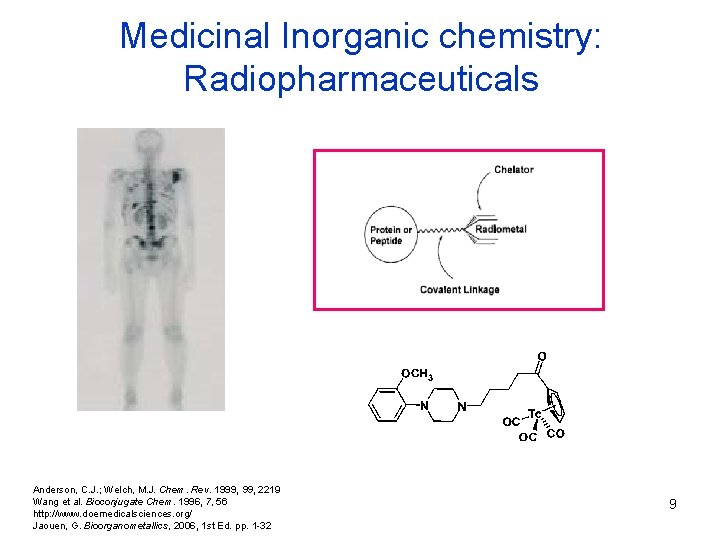
Medicinal Inorganic chemistry: Radiopharmaceuticals Anderson, C. J. ; Welch, M. J. Chem. Rev. 1999, 2219 Wang et al. Bioconjugate Chem. 1996, 7, 56 http: //www. doemedicalsciences. org/ Jaouen, G. Bioorganometallics, 2006, 1 st Ed. pp. 1 -32 9
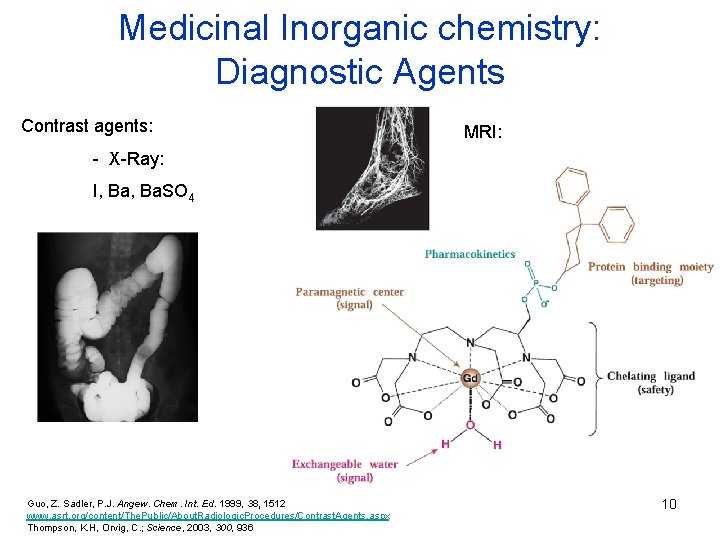
Medicinal Inorganic chemistry: Diagnostic Agents Contrast agents: MRI: - X-Ray: I, Ba. SO 4 Guo, Z. Sadler, P. J. Angew. Chem. Int. Ed. 1999, 38, 1512 www. asrt. org/content/The. Public/About. Radiologic. Procedures/Contrast. Agents. aspx Thompson, K. H, Orvig, C. ; Science, 2003, 300, 936 10
Physical properties Behavior in magnetic field Role of metals Coordination Half life and energy of isotopic decay 11 Guo, Z. Sadler, P. J. Angew. Chem. Int. Ed. 1999, 38, 1512 Orvig, C. Abrams, M. J. Chem. Rev. 1999, 2201
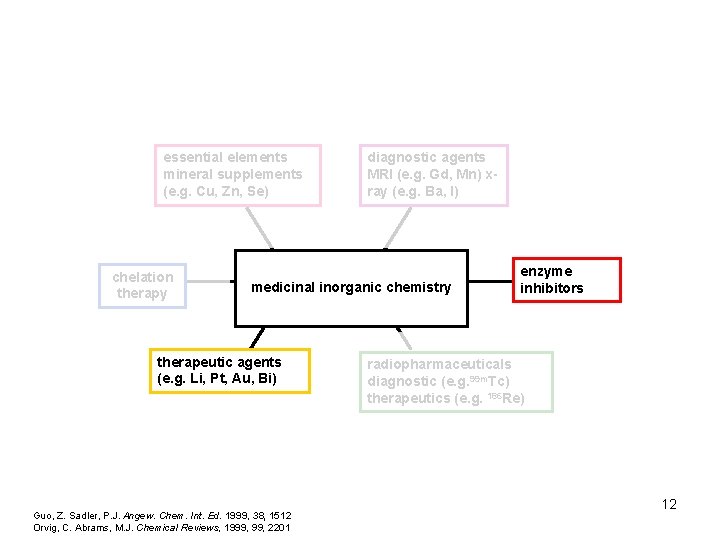
essential elements mineral supplements (e. g. Cu, Zn, Se) chelation therapy diagnostic agents MRI (e. g. Gd, Mn) xray (e. g. Ba, I) medicinal inorganic chemistry therapeutic agents (e. g. Li, Pt, Au, Bi) Guo, Z. Sadler, P. J. Angew. Chem. Int. Ed. 1999, 38, 1512 Orvig, C. Abrams, M. J. Chemical Reviews, 1999, 2201 enzyme inhibitors radiopharmaceuticals diagnostic (e. g. 99 m. Tc) therapeutics (e. g. 186 Re) 12

Ø Bioactivity is at the metal center Cisplatin Ø Bioactivity is related to reaction caused by the metal center Tamoxifen Ø Metal is the structural scaffold Pyridocarbazole ruthenium complexes 13
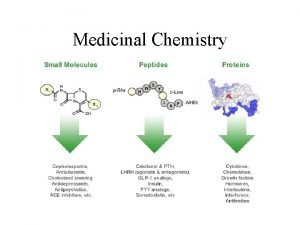
Therapeutic Agents Ø Pharmaceutical industry usually dominated by organic drugs Ø Certain Inorganic drugs have proven their utility: Li, Bi Most important inorganic pharmaceuticals on the market: Cisplatin • Discovered by chance by Rosenberg • Used in the treatment of various cancers (testicular and ovarian) • Approved for Clinical use in 1978 • World wide sales are around 2 billion U. S $ Guo, Z. Sadler, P. J. Angew. Chem. Int. Ed. 1999, 38, 1512 Fricker, S. P. Dalton Trans. , 2007, 4903– 4917 Alderden et al. Journal of Chemical Education 2006, 83 14
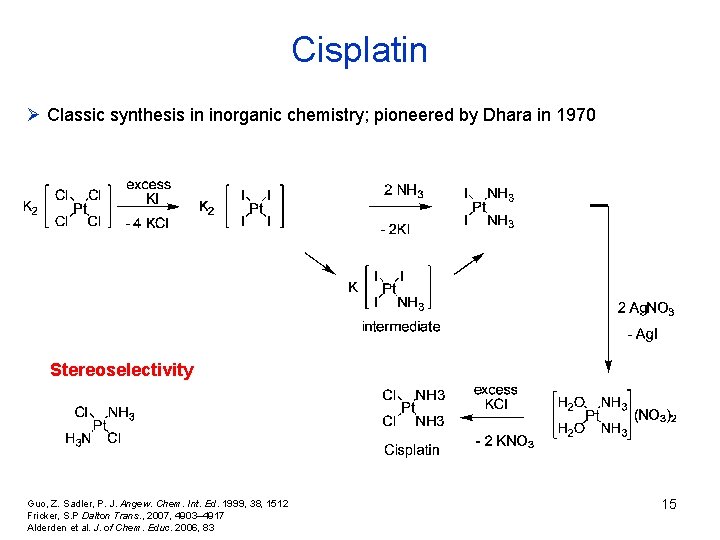
Cisplatin Ø Classic synthesis in inorganic chemistry; pioneered by Dhara in 1970 Stereoselectivity Guo, Z. Sadler, P. J. Angew. Chem. Int. Ed. 1999, 38, 1512 Fricker, S. P Dalton Trans. , 2007, 4903– 4917 Alderden et al. J. of Chem. Educ. 2006, 83 15
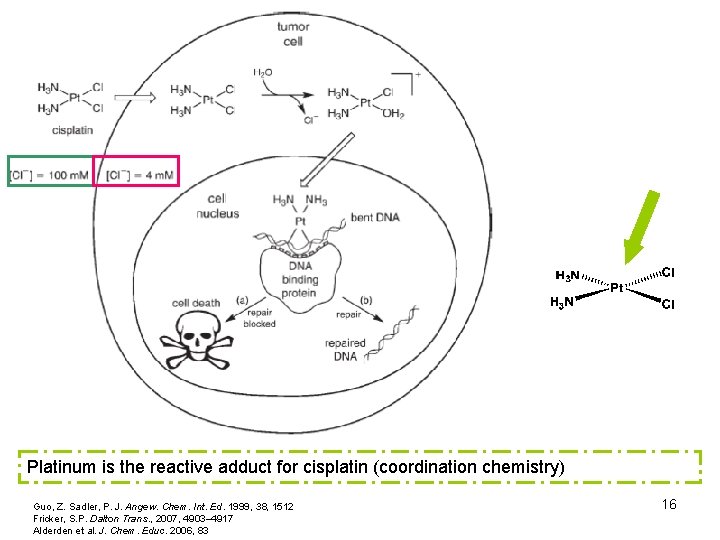
Platinum is the reactive adduct for cisplatin (coordination chemistry) Guo, Z. Sadler, P. J. Angew. Chem. Int. Ed. 1999, 38, 1512 Fricker, S. P. Dalton Trans. , 2007, 4903– 4917 Alderden et al. J. Chem. Educ. 2006, 83 16
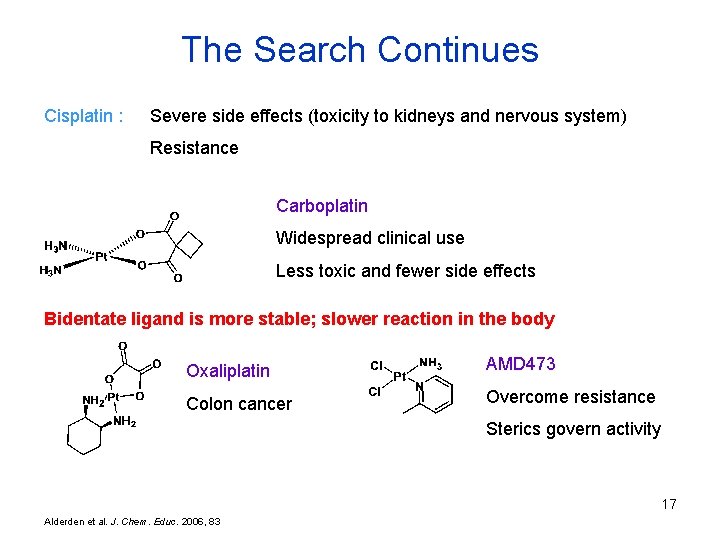
The Search Continues Cisplatin : Severe side effects (toxicity to kidneys and nervous system) Resistance Carboplatin Widespread clinical use Less toxic and fewer side effects Bidentate ligand is more stable; slower reaction in the body Oxaliplatin AMD 473 Colon cancer Overcome resistance Sterics govern activity 17 Alderden et al. J. Chem. Educ. 2006, 83

Ø Bioactivity is at the metal center Cisplatin Ø Bioactivity is related to reaction caused by the metal center Tamoxifen Ø Metal is the structural scaffold Pyridocarbazole ruthenium complexes 18
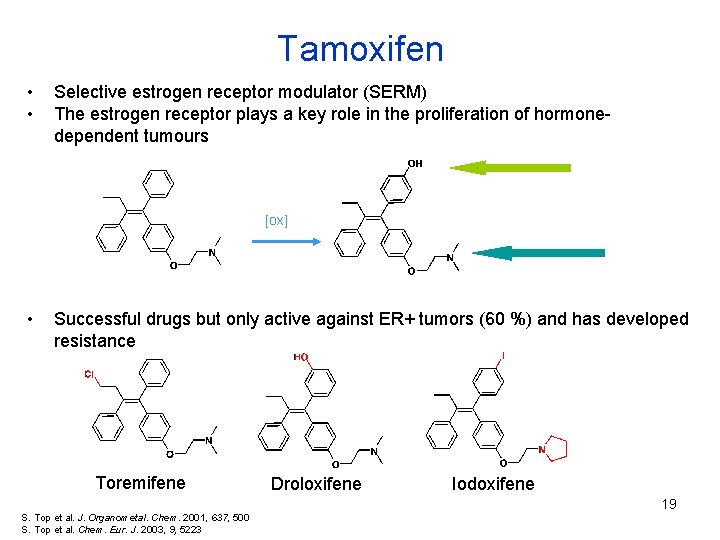
Tamoxifen • • Selective estrogen receptor modulator (SERM) The estrogen receptor plays a key role in the proliferation of hormonedependent tumours [ox] • Successful drugs but only active against ER+ tumors (60 %) and has developed resistance Toremifene Droloxifene Iodoxifene 19 S. Top et al. J. Organometal. Chem. 2001, 637, 500 S. Top et al. Chem. Eur. J. 2003, 9, 5223
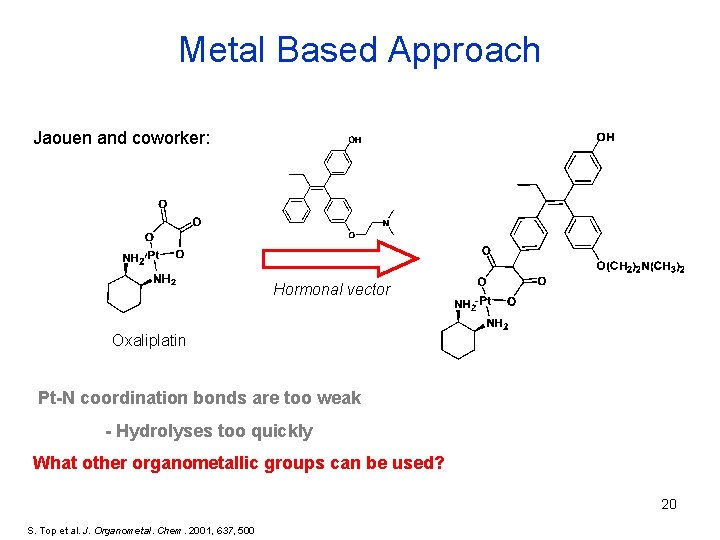
Metal Based Approach Jaouen and coworker: Hormonal vector Oxaliplatin Pt-N coordination bonds are too weak - Hydrolyses too quickly What other organometallic groups can be used? 20 S. Top et al. J. Organometal. Chem. 2001, 637, 500

Organometallic Approach: Metallocenes Organometallic chemistry: - Strong metal-carbon covalent bonds instead of weak coordination bonds Antitumor activity: - different mechanism from that of cisplatin complexes Ferrocene: - 18 electrons inert gas configuration: very stable - Chemistry is similar to ordinary aromatic compounds - Lipophilic S. Top et al. J. Organometal. Chem. 2001, 637, 500 S. Top et al. Chem. Eur. J. 2003, 9, 5223 21
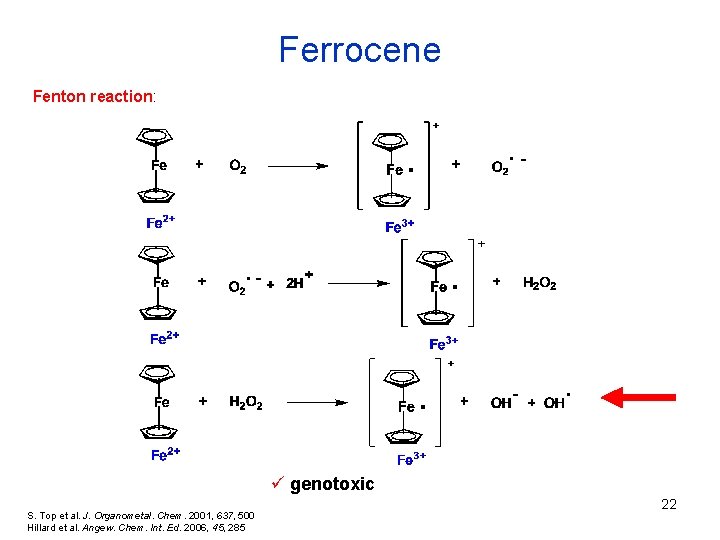
Ferrocene Fenton reaction: ü genotoxic S. Top et al. J. Organometal. Chem. 2001, 637, 500 Hillard et al. Angew. Chem. Int. Ed. 2006, 45, 285 22
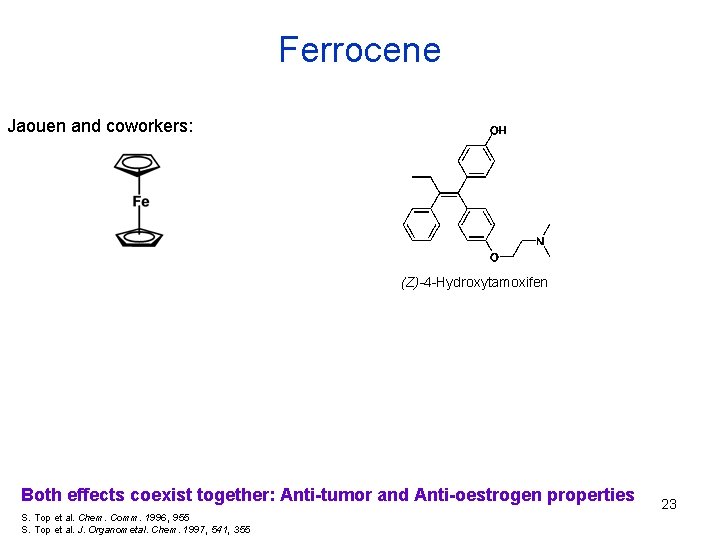
Ferrocene Jaouen and coworkers: (Z)-4 -Hydroxytamoxifen Both effects coexist together: Anti-tumor and Anti-oestrogen properties S. Top et al. Chem. Comm. 1996, 955 S. Top et al. J. Organometal. Chem. 1997, 541, 355 23
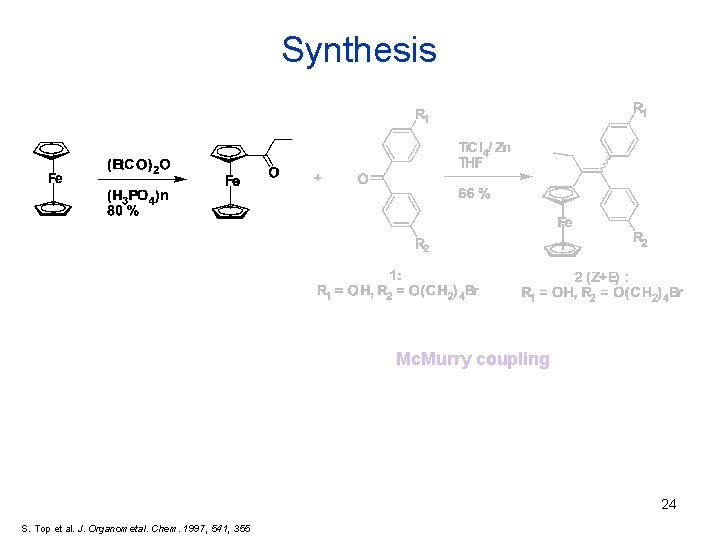
Synthesis Mc. Murry coupling 24 S. Top et al. J. Organometal. Chem. 1997, 541, 355
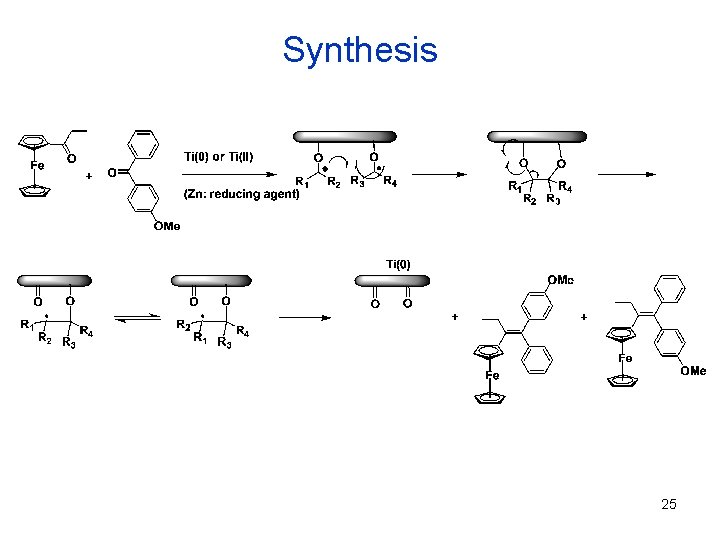
Synthesis 25
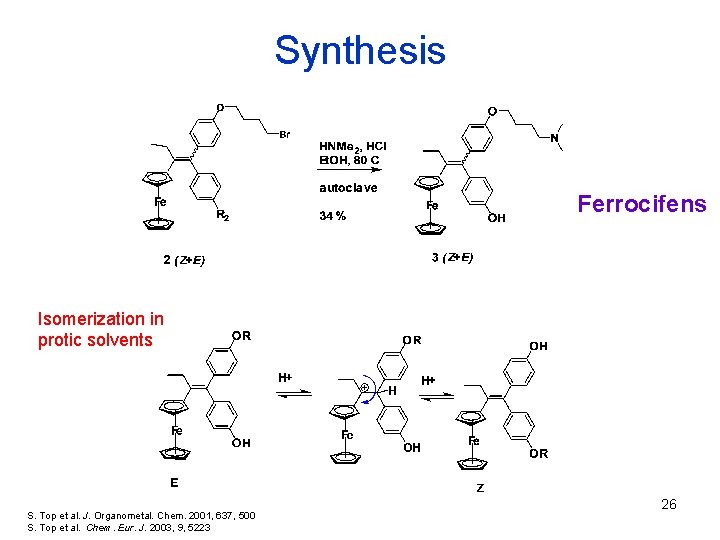
Synthesis Ferrocifens Isomerization in protic solvents S. Top et al. J. Organometal. Chem. 2001, 637, 500 S. Top et al. Chem. Eur. J. 2003, 9, 5223 26
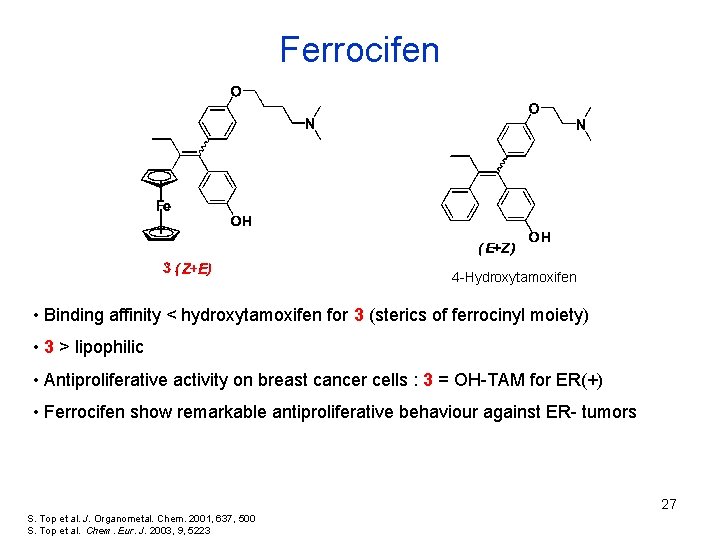
Ferrocifen 4 -Hydroxytamoxifen • Binding affinity < hydroxytamoxifen for 3 (sterics of ferrocinyl moiety) • 3 > lipophilic • Antiproliferative activity on breast cancer cells : 3 = OH-TAM for ER(+) • Ferrocifen show remarkable antiproliferative behaviour against ER- tumors 27 S. Top et al. J. Organometal. Chem. 2001, 637, 500 S. Top et al. Chem. Eur. J. 2003, 9, 5223
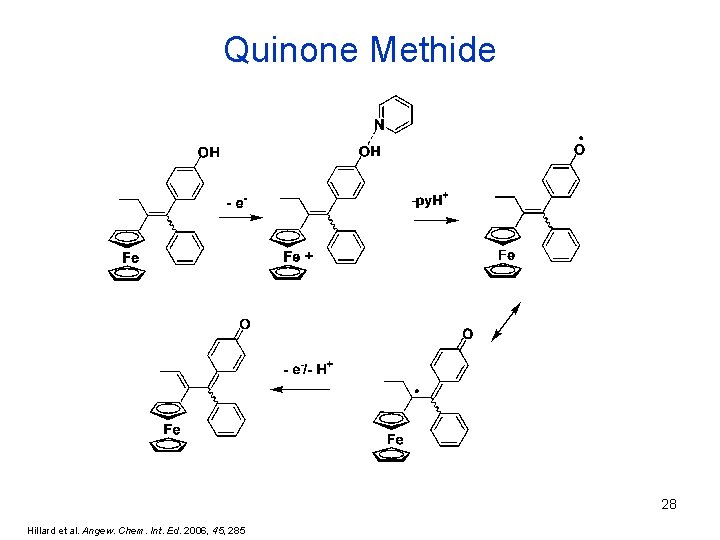
Quinone Methide 28 Hillard et al. Angew. Chem. Int. Ed. 2006, 45, 285
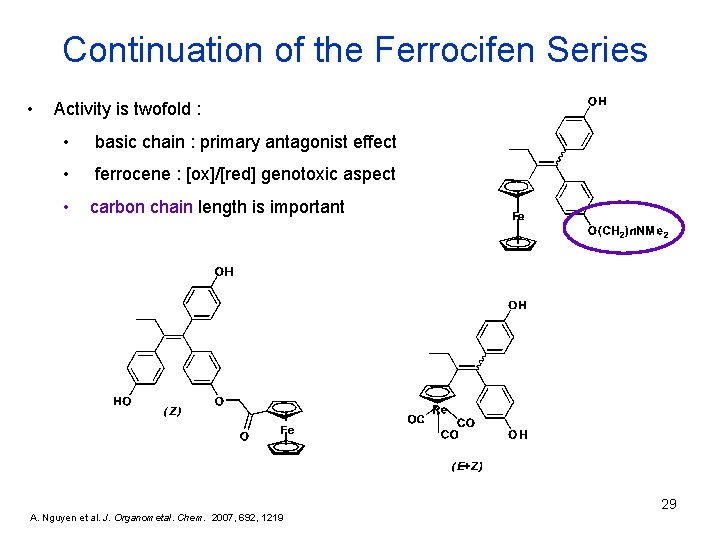
Continuation of the Ferrocifen Series • Activity is twofold : • basic chain : primary antagonist effect • ferrocene : [ox]/[red] genotoxic aspect • carbon chain length is important 29 A. Nguyen et al. J. Organometal. Chem. 2007, 692, 1219

Ø Bioactivity is at the metal center Cisplatin Ø Bioactivity is related to reaction caused by the metal center Tamoxifen Ø Metal as a structural scaffold Pyridocarbazole ruthenium complexes 30
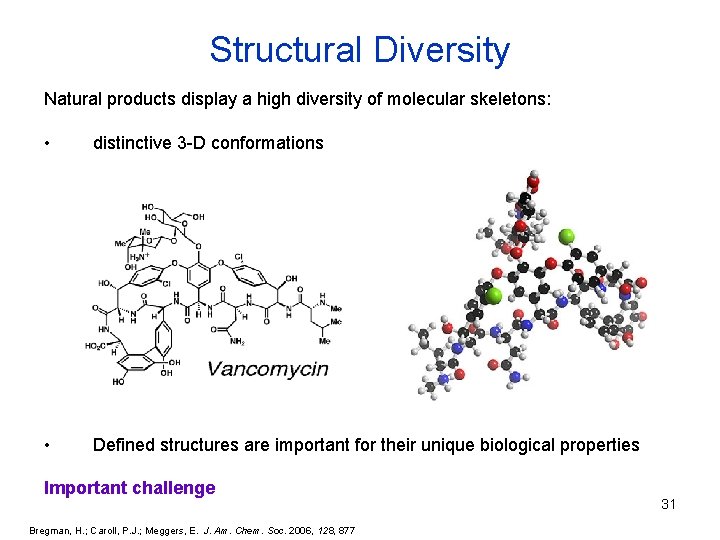
Structural Diversity Natural products display a high diversity of molecular skeletons: • distinctive 3 -D conformations • Defined structures are important for their unique biological properties Important challenge Bregman, H. ; Caroll, P. J. ; Meggers, E. J. Am. Chem. Soc. 2006, 128, 877 31

Outline 1. Target : Kinase; ATP binding site 2. Known inhibitor: Staurosporine 3. Metal scaffold 4. Synthetic approaches and development 5. Diversity oriented synthesis 32
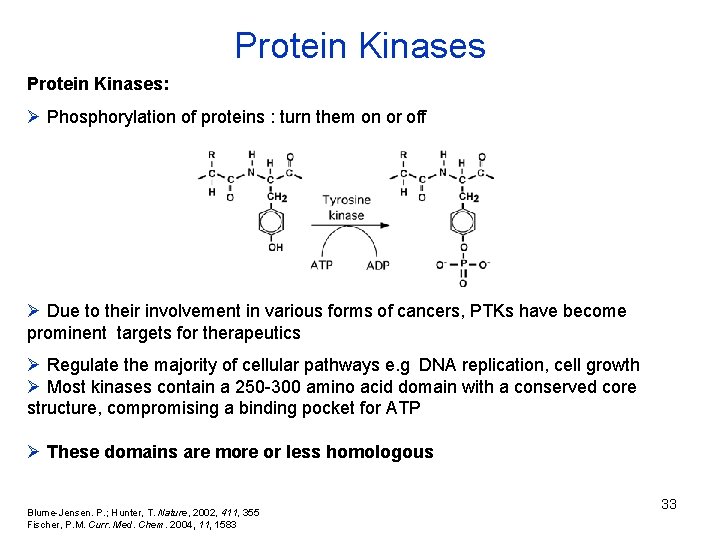
Protein Kinases: Ø Phosphorylation of proteins : turn them on or off Ø Due to their involvement in various forms of cancers, PTKs have become prominent targets for therapeutics Ø Regulate the majority of cellular pathways e. g DNA replication, cell growth Ø Most kinases contain a 250 -300 amino acid domain with a conserved core structure, compromising a binding pocket for ATP Ø These domains are more or less homologous Blume-Jensen. P. ; Hunter, T. Nature, 2002, 411, 355 Fischer, P. M. Curr. Med. Chem. 2004, 11, 1583 33
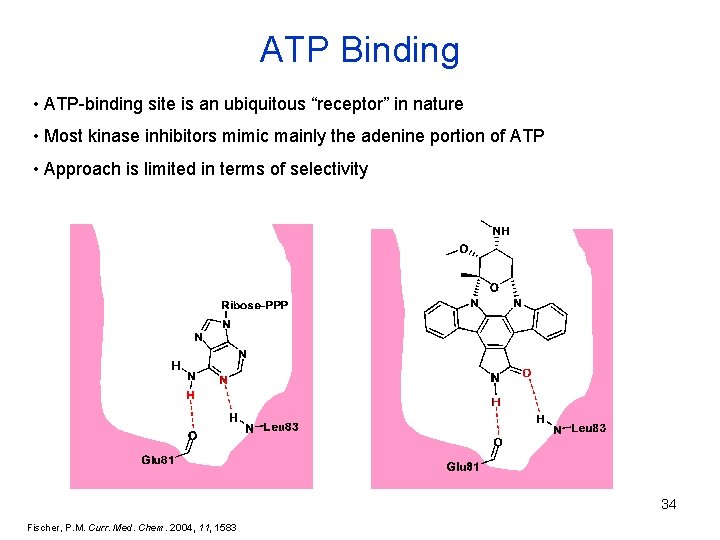
ATP Binding • ATP-binding site is an ubiquitous “receptor” in nature • Most kinase inhibitors mimic mainly the adenine portion of ATP • Approach is limited in terms of selectivity 34 Fischer, P. M. Curr. Med. Chem. 2004, 11, 1583

Bioorganometallic Chemistry: Staurosporine • discovered in 1977 while screening for microbials • has gained great interest since it was reported to be potent against protein kinases • Relatively potent; IC 50 in the nanomolar range Down side: Lacks specificity Derivatives with modulated specificities are in preclinical trials as anticancer drugs 35 Omura, S. et al. J. Antibiotics, 1994, 48, 535 M. Yang et al. Bioorg. Med. Chem. Lett. 2007, 17, 326
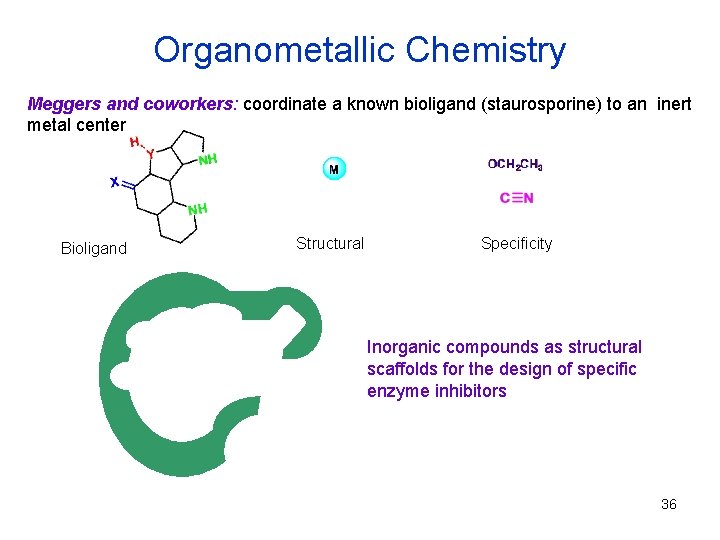
Organometallic Chemistry Meggers and coworkers: coordinate a known bioligand (staurosporine) to an inert metal center Bioligand Structural Specificity Inorganic compounds as structural scaffolds for the design of specific enzyme inhibitors 36
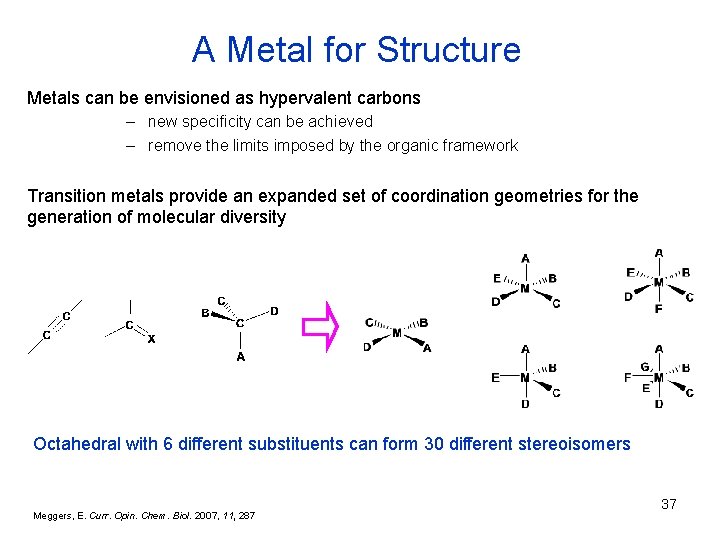
A Metal for Structure Metals can be envisioned as hypervalent carbons – new specificity can be achieved – remove the limits imposed by the organic framework Transition metals provide an expanded set of coordination geometries for the generation of molecular diversity Octahedral with 6 different substituents can form 30 different stereoisomers Meggers, E. Curr. Opin. Chem. Biol. 2007, 11, 287 37
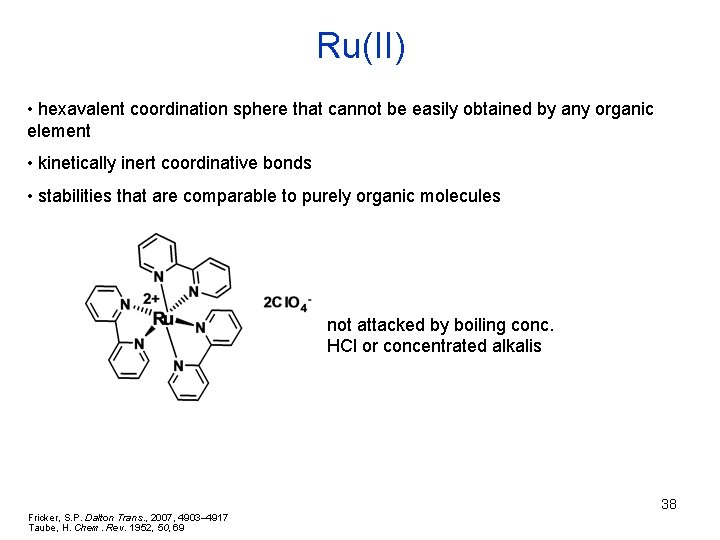
Ru(II) • hexavalent coordination sphere that cannot be easily obtained by any organic element • kinetically inert coordinative bonds • stabilities that are comparable to purely organic molecules not attacked by boiling conc. HCl or concentrated alkalis Fricker, S. P. Dalton Trans. , 2007, 4903– 4917 Taube, H. Chem. Rev. 1952, 50, 69 38
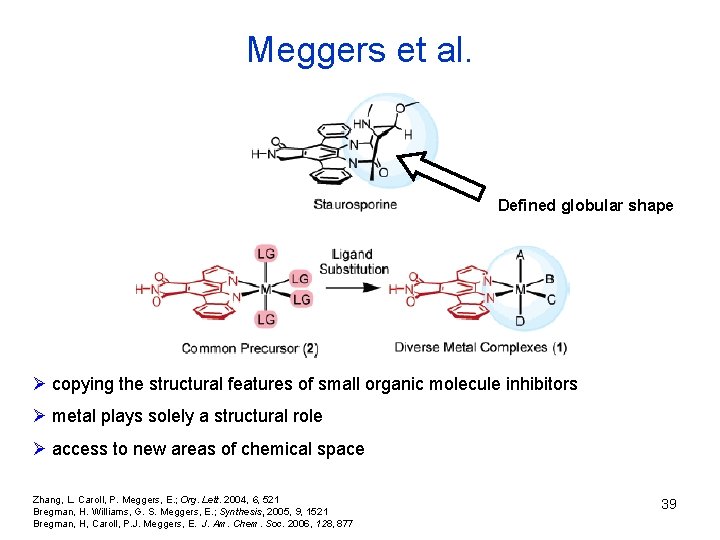
Meggers et al. Defined globular shape Ø copying the structural features of small organic molecule inhibitors Ø metal plays solely a structural role Ø access to new areas of chemical space Zhang, L. Caroll, P. Meggers, E. ; Org. Lett. 2004, 6, 521 Bregman, H. Williams, G. S. Meggers, E. ; Synthesis, 2005, 9, 1521 Bregman, H, Caroll, P. J. Meggers, E. J. Am. Chem. Soc. 2006, 128, 877 39
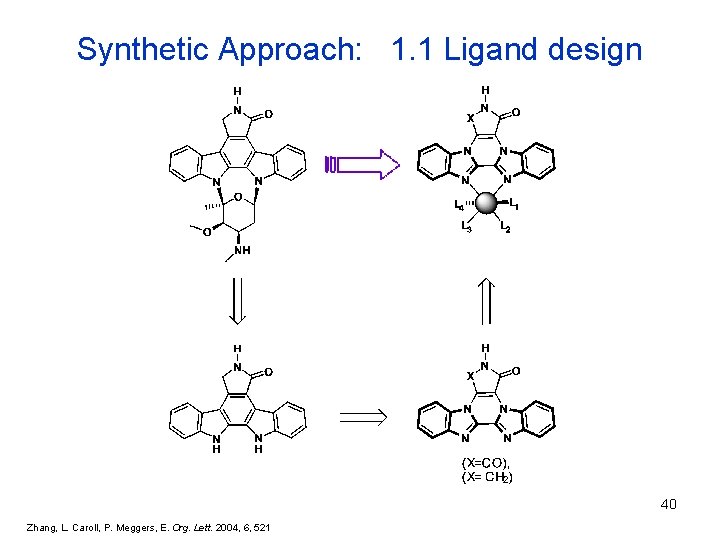
Synthetic Approach: 1. 1 Ligand design 40 Zhang, L. Caroll, P. Meggers, E. Org. Lett. 2004, 6, 521
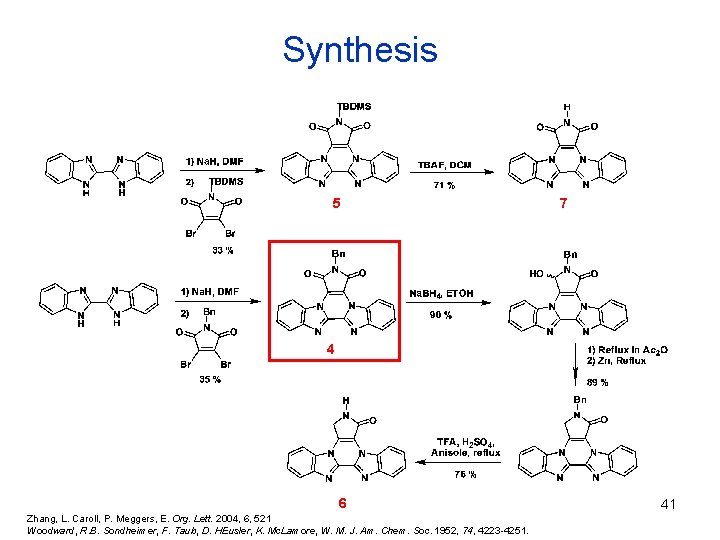
Synthesis 5 7 4 6 Zhang, L. Caroll, P. Meggers, E. Org. Lett. 2004, 6, 521 Woodward, R. B. Sondheimer, F. Taub, D. HEusler, K. Mc. Lamore, W. M. J. Am. Chem. Soc. 1952, 74, 4223 -4251. 41
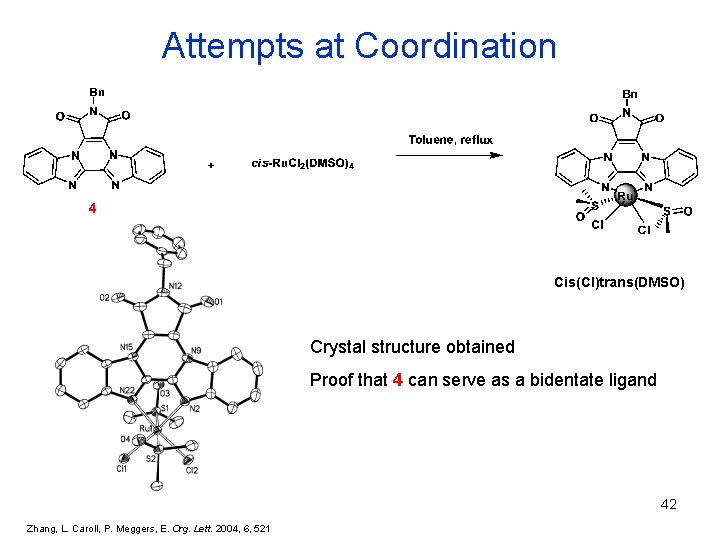
Attempts at Coordination 4 Cis(Cl)trans(DMSO) Crystal structure obtained Proof that 4 can serve as a bidentate ligand 42 Zhang, L. Caroll, P. Meggers, E. Org. Lett. 2004, 6, 521
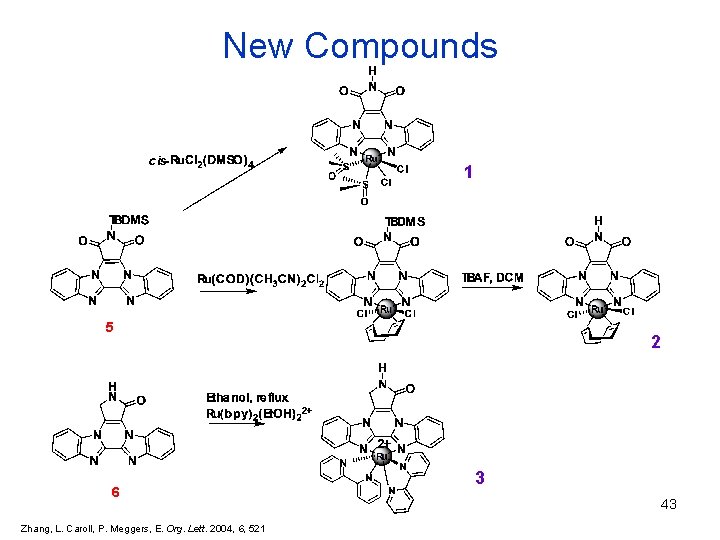
New Compounds 1 5 6 Zhang, L. Caroll, P. Meggers, E. Org. Lett. 2004, 6, 521 2 3 43

Stability 1 2 3 • 3 is stable in a 1: 1 water/DMSO solution for 12 h • 3 can withstand a 2 -mercaptoethanol for 3 hours without decomposition • 1 and 2 slowly release bidentate ligand in 1: 1 water/DMSO solution , ½ life of 8 and 3 h respectively 44 Zhang, L. Caroll, P. Meggers, E. Org. Lett. 2004, 6, 521
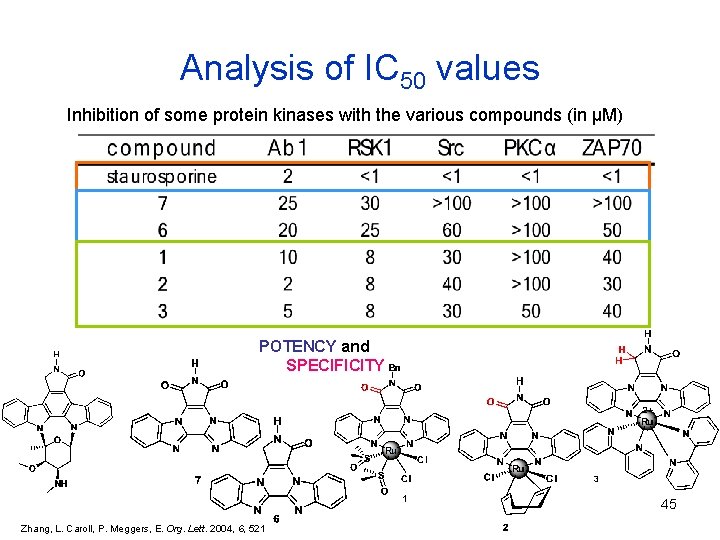
Analysis of IC 50 values Inhibition of some protein kinases with the various compounds (in μM) POTENCY and SPECIFICITY 3 45 Zhang, L. Caroll, P. Meggers, E. Org. Lett. 2004, 6, 521
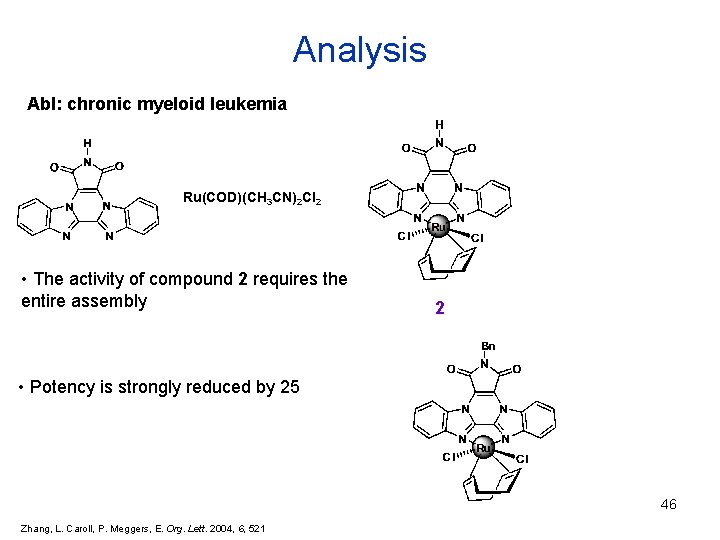
Analysis Abl: chronic myeloid leukemia Ru(COD)(CH 3 CN)2 Cl 2 • The activity of compound 2 requires the entire assembly 2 • Potency is strongly reduced by 25 46 Zhang, L. Caroll, P. Meggers, E. Org. Lett. 2004, 6, 521
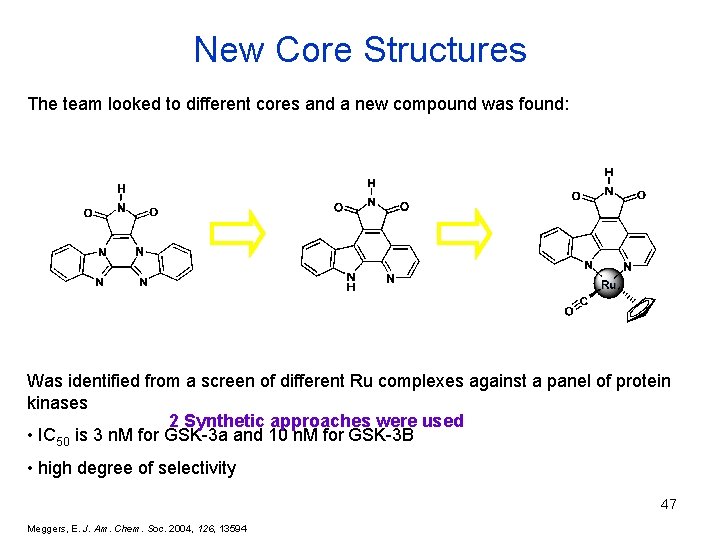
New Core Structures The team looked to different cores and a new compound was found: Was identified from a screen of different Ru complexes against a panel of protein kinases 2 Synthetic approaches were used • IC 50 is 3 n. M for GSK-3 a and 10 n. M for GSK-3 B • high degree of selectivity 47 Meggers, E. J. Am. Chem. Soc. 2004, 126, 13594
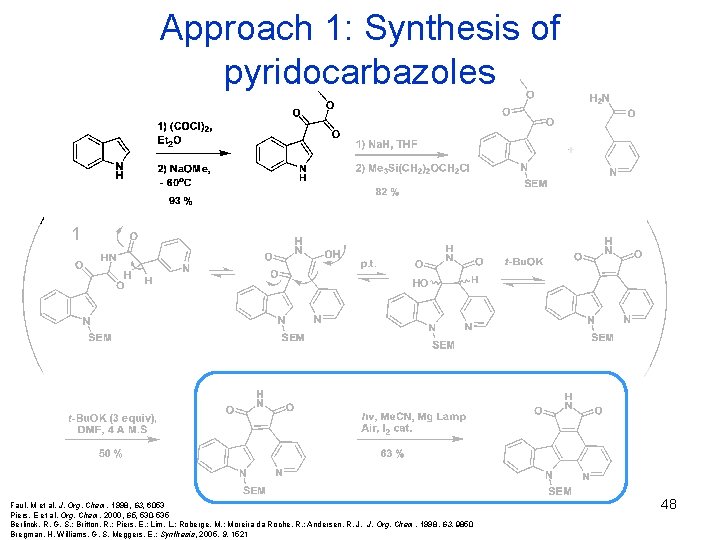
Approach 1: Synthesis of pyridocarbazoles 1 Faul. M et al. J. Org. Chem. 1998, 63, 6053 Piers. E et al. Org. Chem. 2000, 65, 530 -535 Berlinck, R. G. S. ; Britton, R. ; Piers, E. ; Lim, L. ; Roberge, M. ; Moreira da Roche, R. ; Andersen, R. J. J. Org. Chem. 1998, 63, 9850 Bregman, H. Williams, G. S. Meggers, E. ; Synthesis, 2005, 9, 1521 48
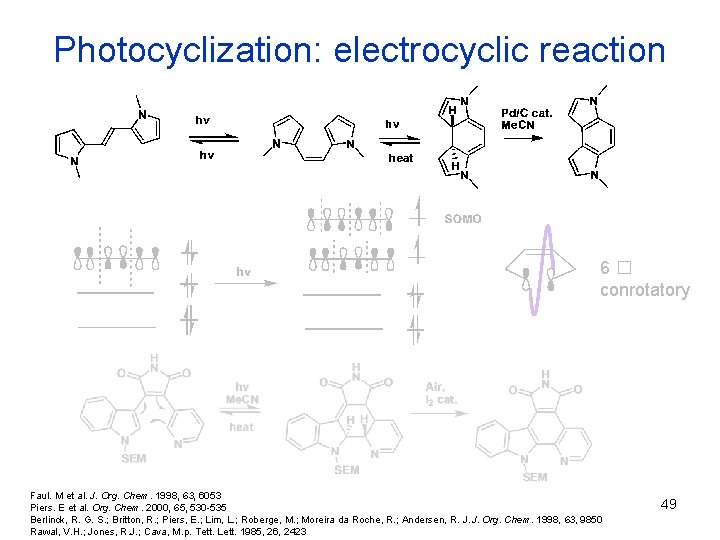
Photocyclization: electrocyclic reaction 6� conrotatory Faul. M et al. J. Org. Chem. 1998, 63, 6053 Piers. E et al. Org. Chem. 2000, 65, 530 -535 Berlinck, R. G. S. ; Britton, R. ; Piers, E. ; Lim, L. ; Roberge, M. ; Moreira da Roche, R. ; Andersen, R. J. J. Org. Chem. 1998, 63, 9850 Rawal, V. H. ; Jones, R. J. ; Cava, M. p. Tett. Lett. 1985, 26, 2423 49

Approach 1: Synthesis of pyridocarbazoles No base is required, volatile side product Kita, Y. ; Haruta, J. ; Fujii, T. ; Segwawa, J. Synthesis 1981, 451 Bregman, H. Williams, G. S. Meggers, E. Synthesis, 2005, 9, 1521 50
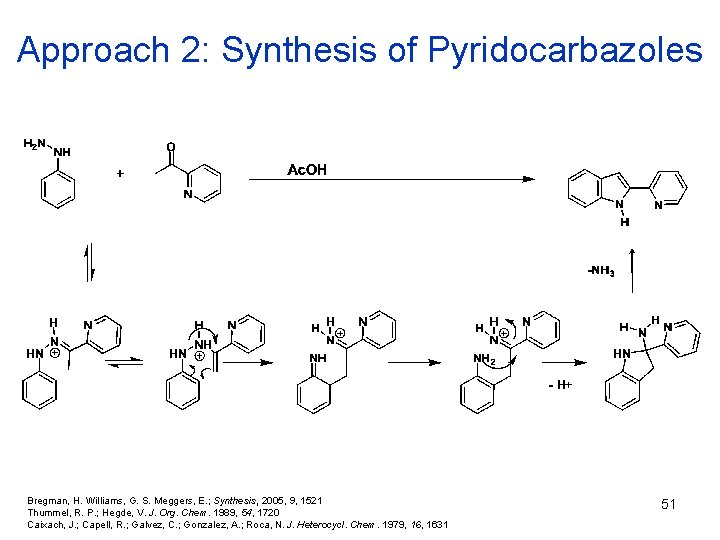
Approach 2: Synthesis of Pyridocarbazoles Bregman, H. Williams, G. S. Meggers, E. ; Synthesis, 2005, 9, 1521 Thummel, R. P. ; Hegde, V. J. Org. Chem. 1989, 54, 1720 Caixach, J. ; Capell, R. ; Galvez, C. ; Gonzalez, A. ; Roca, N. J. Heterocycl. Chem. 1979, 1631 51

Approach 2: Synthesis of Pyridocarbazoles 52 Bregman, H. Williams, G. S. Meggers, E. ; Synthesis, 2005, 9, 1521
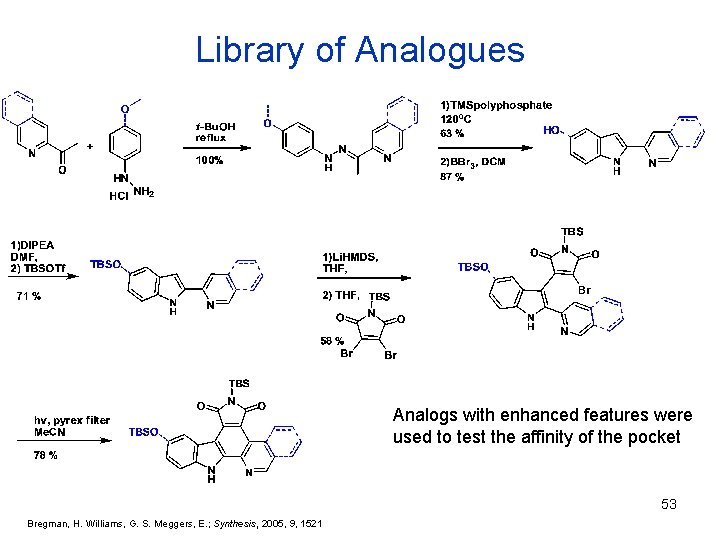
Library of Analogues Analogs with enhanced features were used to test the affinity of the pocket 53 Bregman, H. Williams, G. S. Meggers, E. ; Synthesis, 2005, 9, 1521
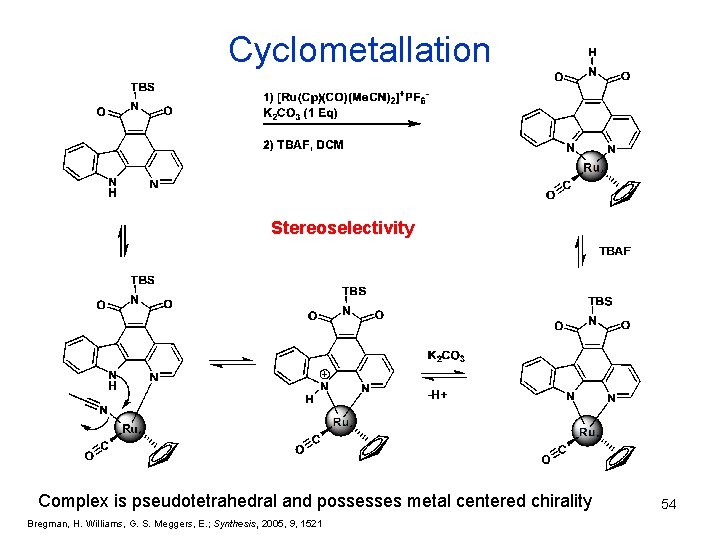
Cyclometallation Stereoselectivity Complex is pseudotetrahedral and possesses metal centered chirality Bregman, H. Williams, G. S. Meggers, E. ; Synthesis, 2005, 9, 1521 54
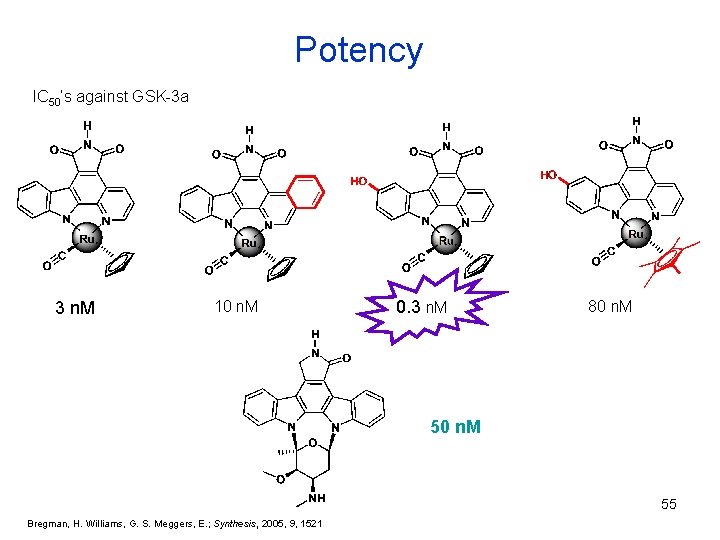
Potency IC 50’s against GSK-3 a 3 n. M 10 n. M 0. 3 n. M 80 n. M 55 Bregman, H. Williams, G. S. Meggers, E. ; Synthesis, 2005, 9, 1521
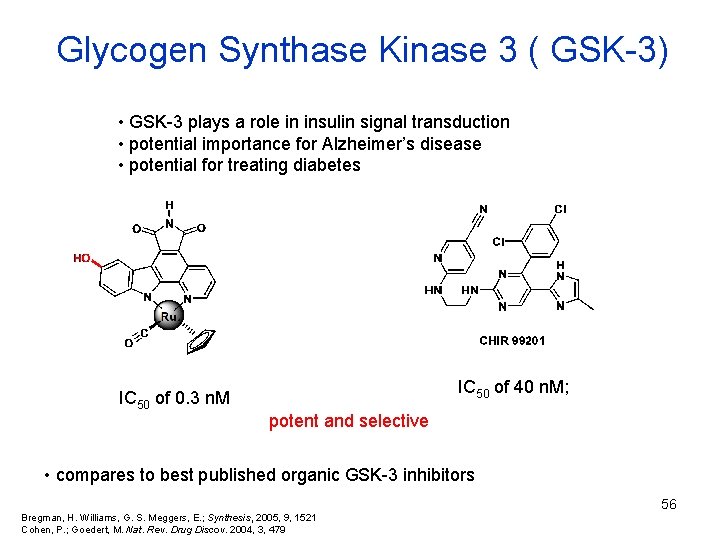
Glycogen Synthase Kinase 3 ( GSK-3) • GSK-3 plays a role in insulin signal transduction • potential importance for Alzheimer’s disease • potential for treating diabetes IC 50 of 0. 3 n. M IC 50 of 40 n. M; potent and selective • compares to best published organic GSK-3 inhibitors 56 Bregman, H. Williams, G. S. Meggers, E. ; Synthesis, 2005, 9, 1521 Cohen, P. ; Goedert, M. Nat. Rev. Drug Discov. 2004, 3, 479
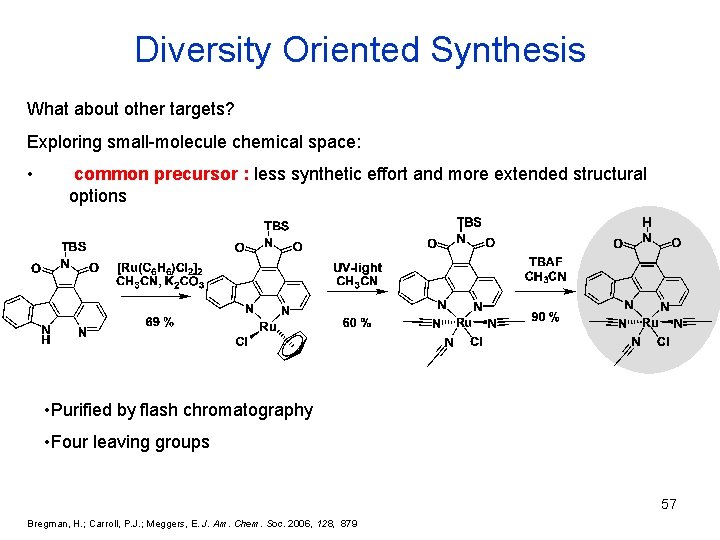
Diversity Oriented Synthesis What about other targets? Exploring small-molecule chemical space: • common precursor : less synthetic effort and more extended structural options • Purified by flash chromatography • Four leaving groups 57 Bregman, H. ; Carroll, P. J. ; Meggers, E. J. Am. Chem. Soc. 2006, 128, 879
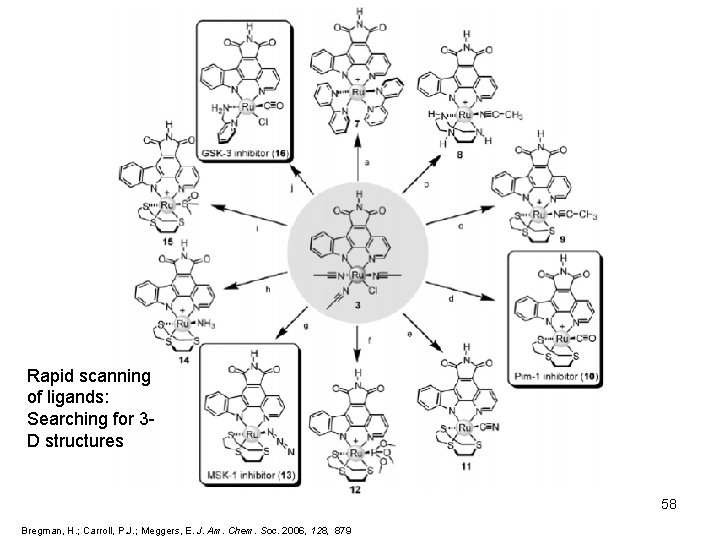
Rapid scanning of ligands: Searching for 3 D structures 58 Bregman, H. ; Carroll, P. J. ; Meggers, E. J. Am. Chem. Soc. 2006, 128, 879

To the Future 59 Bregman, H. ; Meggers, E. Org. Lett. 2006, 8, 5466

Conclusion THINK • Exploit the unique features of metallic elements • Metals are not always toxic • Metals can be used as hypervalent carbon • New ways to address problems that medicinal chemistry faces (NOT better!!!) 60

Acknowledgements Prof. Keith Fagnou Marc Lafrance Megan Ap. Simon Catherine Lebel Mégan Bertrand-Laperle Elisia Villemure Nicole Blaquiere Ho-Yan Sun Sophie Rousseaux Daniel Shore Derek Schipper David Stuart Doris Lee David Lapointe Daniel Black Benoît Liegault Chris Whipp Malcolm Huestis 61
- Slides: 61