Metabolomic Reprogramming Detected by 1 HNMR Spectroscopy in

Metabolomic Reprogramming Detected by 1 H-NMR Spectroscopy in Human Thyroid Cancer Tissues Zainab Zubair

Overview • Thyroid cancer cells demonstrate an increase in oxidative stress and decreased antioxidant action, but its effects on cell function are unknown. • Focus: identify changes in the metabolism of thyroid cancer cells caused by oxidative stress using 1 H-NMR spectroscopy

Introduction • Oxidative stress: attack of free radicals on healthy cells • Can lead to cell damage • Downstream effects on biochemical and metabolic pathways • EPR spectroscopy showed increased oxidative stress in blood of patients with thyroid diseases. • Reduced expression of antioxidant seleno-enzymes: GPx 1 and Trx. R 1 in thyroid cancer • Reduce H 2 O 2 to H 2 O • Catalyze reduction of proteins

Methods • Study involved patients with thyroid diseases • Thyroidectomy • 11 tumor and 10 healthy thyroid tissue samples • NMR spectroscopy performed on tissues.

NMR Spectroscopy • Frozen tissue samples were homogenized with an ethanolic solution three times. • Ultrasonic disintegrator used on samples at 20 k. Hz • NMR analyses using a 60 -degree flip angle pulse

Comparative Analysis • Evaluate metabolic differences between the healthy and tumor tissues • To clarify biochemical and metabolic changes that occur in thyroid cancer and to evaluate their relationship with increased oxidative stress.
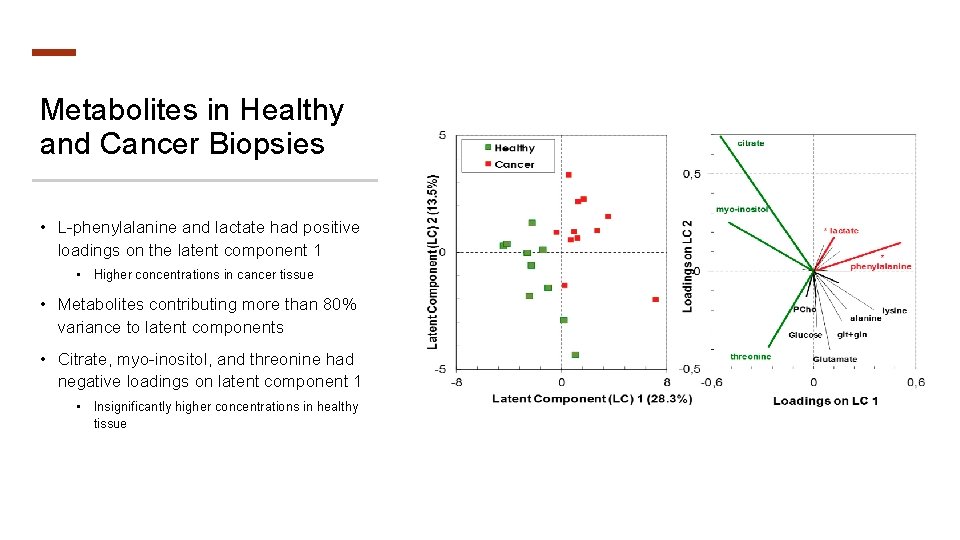
Metabolites in Healthy and Cancer Biopsies • L-phenylalanine and lactate had positive loadings on the latent component 1 • Higher concentrations in cancer tissue • Metabolites contributing more than 80% variance to latent components • Citrate, myo-inositol, and threonine had negative loadings on latent component 1 • Insignificantly higher concentrations in healthy tissue
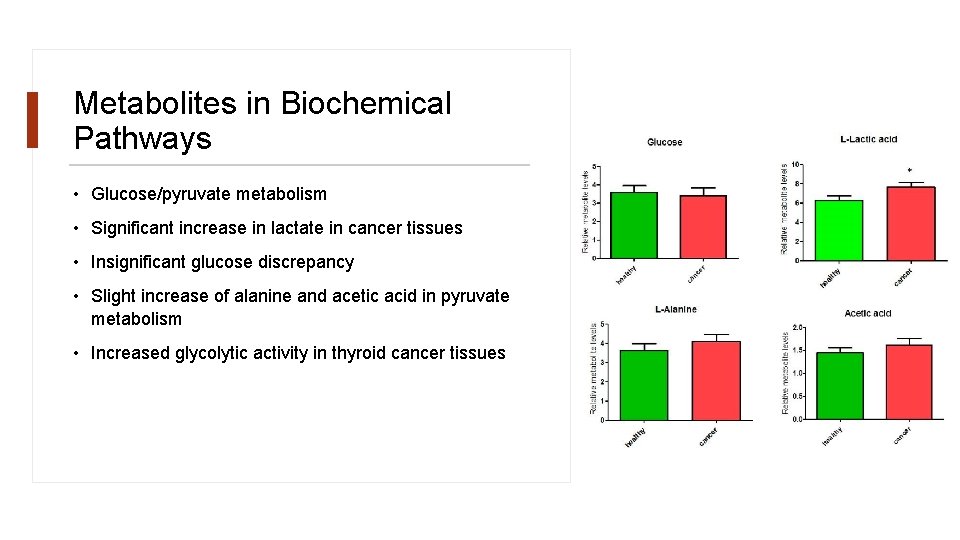
Metabolites in Biochemical Pathways • Glucose/pyruvate metabolism • Significant increase in lactate in cancer tissues • Insignificant glucose discrepancy • Slight increase of alanine and acetic acid in pyruvate metabolism • Increased glycolytic activity in thyroid cancer tissues

Metabolites in Biochemical Pathways • Amino acid metabolism • Significant increase in aromatic amino acid levels of tyrosine and phenylalanine • Increase in branched-chain amino acid isoleucine (BCCA) in cancer tissues

Metabolites in Biochemical Pathways • Lipid metabolism metabolites in cancer vs healthy tissue • Insignificant differences between phosphatidylcholine and phosphatidylinositol levels • Increased myo-inositol levels in healthy tissue • Decreased citric acid levels in cancer tissue
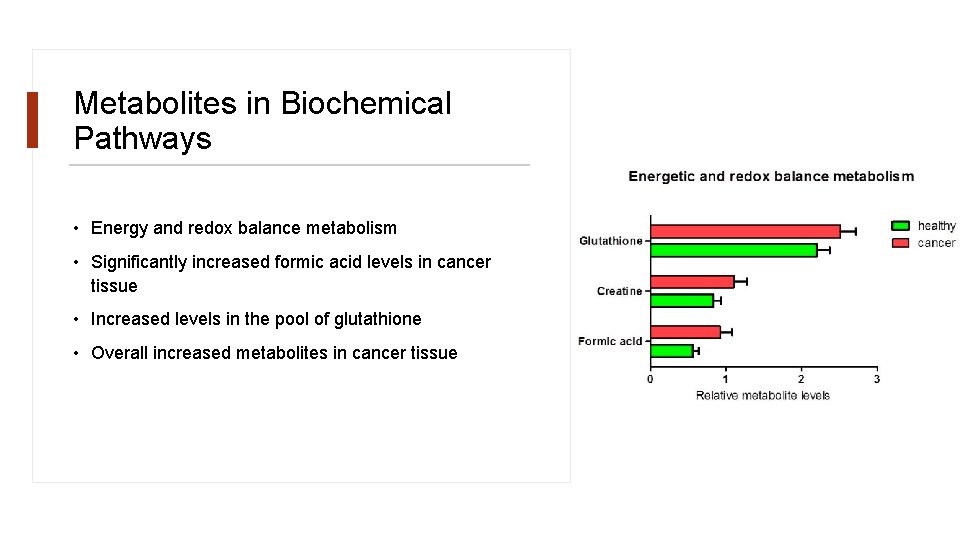
Metabolites in Biochemical Pathways • Energy and redox balance metabolism • Significantly increased formic acid levels in cancer tissue • Increased levels in the pool of glutathione • Overall increased metabolites in cancer tissue

Findings • Increased free radical production • Increased oxidative stress • Physiological activities • Imbalanced oxidant/antioxidant system in cancer thyroid tissue • Decrease in GPx 1 and Trx. R 1 enzymes suggest antioxidant systems cannot counteract the effect of free radicals • Due to deficit of antioxidant systems or increased consumption of antioxidant cellular reserve

Findings • Metabolic reprogramming • Citrate involved in mitochondrial activities • Glucose/pyruvate metabolism • Decreased citrate and increased acetate levels support lipid biogenesis • Mitochondria regulate energy production processes, so altered/dysfunctional mitochondrial activity can lead to enhanced glycolysis in cancer tissue • Increased lactate levels show a metabolic shift towards glycolysis • Increase in glycolytic activity confirmed by lactate levels • May promote oxidative stress resistance • Lactate is a known factor in cancer cell mobility and immune suppression • Altered lipid profiles and fat concentrations • Hypothesize a connection between oxidative stress and the alteration of the metabolic profile in thyroid tumors • Reduced activity of the Krebs cycle • NMR spectroscopy results show reduced citrate, a Krebs cycle product, may be attributed to a decrease in oxidative metabolism • Impairment of oxidative metabolism explains reduction of acetyl Co. A
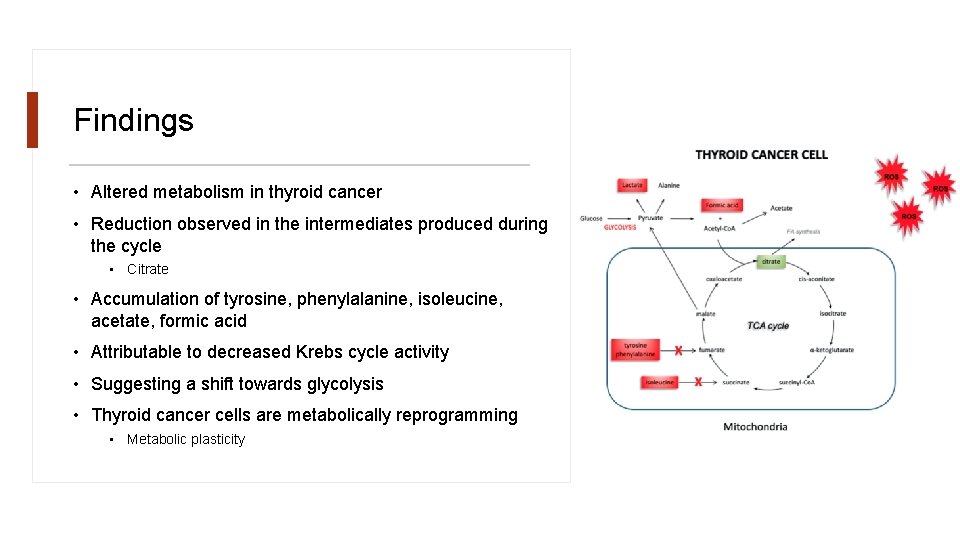
Findings • Altered metabolism in thyroid cancer • Reduction observed in the intermediates produced during the cycle • Citrate • Accumulation of tyrosine, phenylalanine, isoleucine, acetate, formic acid • Attributable to decreased Krebs cycle activity • Suggesting a shift towards glycolysis • Thyroid cancer cells are metabolically reprogramming • Metabolic plasticity

References • Alessio, et al. “Metabolomic Reprogramming Detected by 1 HNMR Spectroscopy in Human Thyroid Cancer Tissues. ” MDPI, Multidisciplinary Digital Publishing Institute, 27 May 2020, www. mdpi. com/2079 -7737/9/6/112.
- Slides: 15