Metabolisme Protein Lecture 9 l Amino acid digestion

Metabolisme Protein

Lecture 9 l Amino acid digestion cont… l Amino acids classification l Intestinal absorption and utilization of amino acids

Trypsin Inhibitors in the Diet l Diets can contain protease inhibitors – l Trypsin inhibitors in soybeans Resistance of the soy protein to digestion also contributes significantly to increases in: – – Pancreatic protein Trypsin Chymotrypsin Pancreatic enlargement

Soy Protein l The major soy storage protein glycinin and trypsin inhibitors are rich in disulfide bonds l Dietary soy bean trypsin inhibitor causes the release of cholecystokinin (CCK) – Stimulates pancreatic enzyme secretion and pancreatic enlargement

Enteropeptidase Trypsinogen -------------- Trypsin Chymotrypsin Carboxypeptidases

l Disruption of the disulfide bonds may change three-dimensional structure of the proteins so that they lose their bioactivity (such as trypsin inhibition activity or potential allergenicity) l Become more susceptible to digestive enzymes
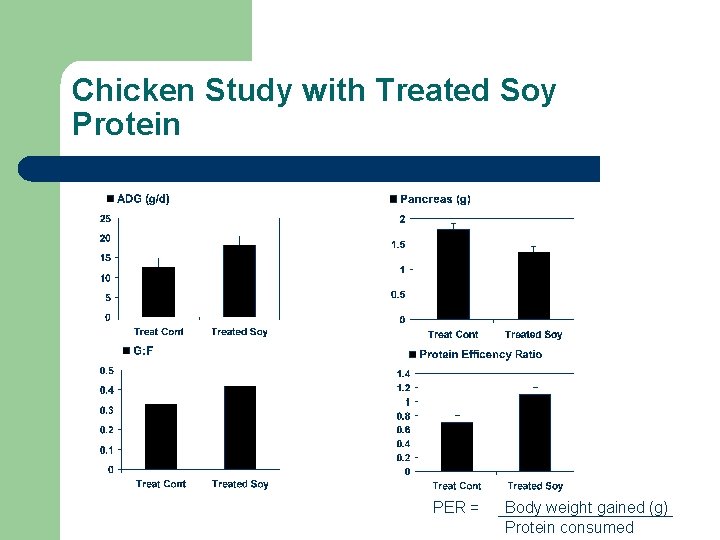
Chicken Study with Treated Soy Protein PER = Body weight gained (g) Protein consumed

Amino Acids l Amino acids can be classified by side its side groups or by the presence pr absence of net charge – – – Neutral such as Alanine, Glycine, Valine etc… +ve such as Arginine, Histidine, Lysine -ve such as Aspartic acid, Glutamic acid Aromatic such as Tyrosine and Tryptophan etc… Acidic such as Glutamine, Asparagine etc… Basic such as Lysine and Histidine etc…

Essential vs Non-essential amino acids l l What is essentiality? – Amino acids that must be supplied by the diet – Cannot be synthesized at a rate sufficient to meet the nutritional requirements of the animal 20 different amino acids commonly incorporated into proteins, but not all are essential

Non-essential Amino Acids l Some amino acids can be synthesized by the body and don’t need to be provided via the diet – Non-essential Alanine Asparagine Aspartic acid Cysteine Glutamine Glycine Proline Serine Glutamic acid Tyrosine

Essential Amino Acids Arginine Histidine Isoleucine Leucine Methionine Phenylalanine Threonine Tryptophan Lysine Valine l l MATT HILL VP Histidine and Arginine not essential for maintenance of most adult mammals Arginine essential for birds Poultry also require glycine and proline for feathers and rapid growth

Dietary Limiting Amino Acids l The first limiting amino acid (essential amino acid) is the amino acid present at the lowest amount in the diet relative to the animal’s amino acid requirement l The amount of this amino acid will determine the level of production l All or nothing attitude for amino acids
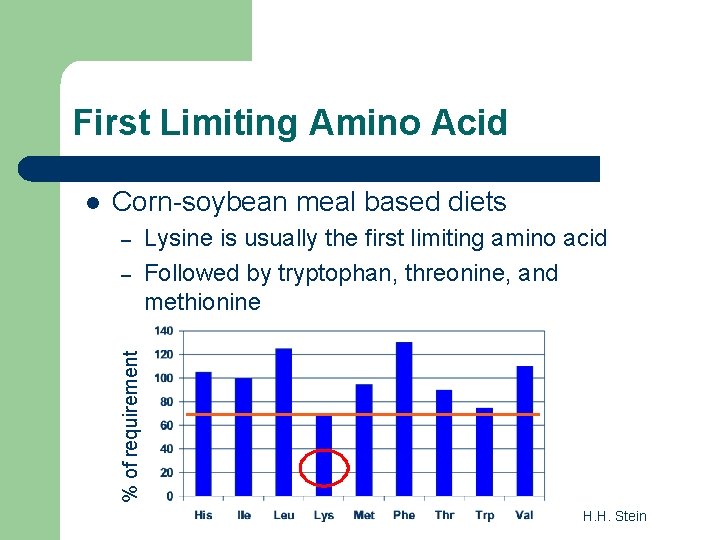
First Limiting Amino Acid Corn-soybean meal based diets – – Lysine is usually the first limiting amino acid Followed by tryptophan, threonine, and methionine % of requirement l H. H. Stein

Cats l Higher crude protein requirement – l Inability of nitrogen catabolic enzymes in the cat’s liver to adapt to changes in dietary protein intake – – l Increased needs for maintenance of normal body tissue Low conservation of nitrogen under low CP diets Liver enzymes function at high rates of activity high catabolism of protein Dietary Arginine, Taurine, sulfur amino acids and aromatic amino acids (eg. phenylalanine, tryptophan, and tyrosine) are in high demand

Protein Deficiency l Reduced appetite and feed intake l Reduced birth weights and growth l Reduced colostrum and milk production l Decreased hormonal production l Decreased fertility

Intestinal Absorption and Amino Acid Transport l After digestion, the resulting amino acids and peptides (di and tri) are efficiently absorbed by enterocytes l Occurs along the entire small intestine – l Most proximal (upper) small intestine Absorption requires carriers (active transport) to bring the amino acid across the brush border membrane, but some passive absorption occurs – Active transport is important when lumen amino acid levels are low

Amino Acid Transport Systems l These carrier systems traditionally been designated using a lettering system – Upper case for sodium-dependence – Lower case for sodium-independence (more passive transport) – Some exceptions (eg. System L, does not require Na+)

l Competition between transporter systems – The affinity (Km) of a carrier is influenced both by the hydrocarbon mass of the side chain of the amino acid and by the net electrical charge l l l Branch chain amino acids absorbed faster than smaller amino acids Neutral amino acids absorbed faster than basic or acidic amino acids Essential absorbed faster than non-essential
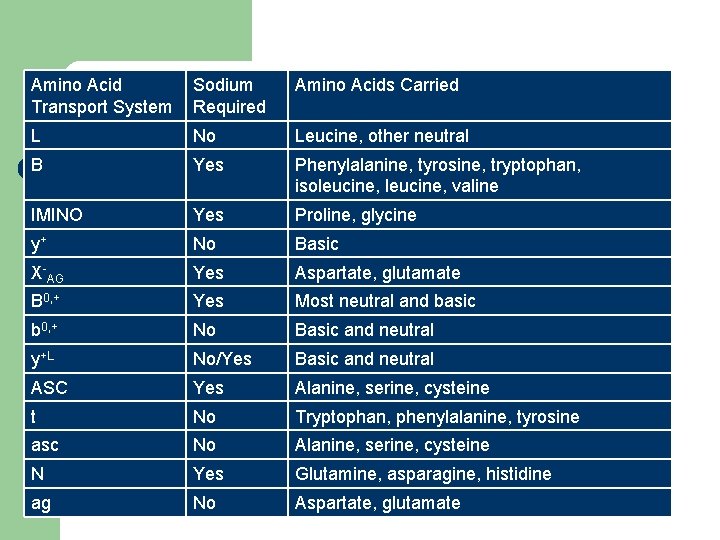
Amino Acid Transport System Sodium Required Amino Acids Carried L No Leucine, other neutral B Yes Phenylalanine, tyrosine, tryptophan, isoleucine, valine IMINO Yes Proline, glycine y+ No Basic X-AG Yes Aspartate, glutamate B 0, + Yes Most neutral and basic b 0, + No Basic and neutral y+L No/Yes Basic and neutral ASC Yes Alanine, serine, cysteine t No Tryptophan, phenylalanine, tyrosine asc No Alanine, serine, cysteine N Yes Glutamine, asparagine, histidine ag No Aspartate, glutamate
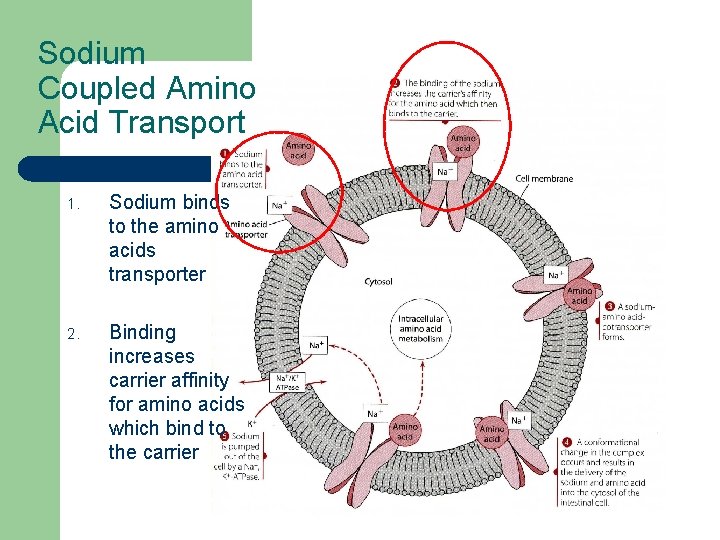
Sodium Coupled Amino Acid Transport 1. Sodium binds to the amino acids transporter 2. Binding increases carrier affinity for amino acids which bind to the carrier
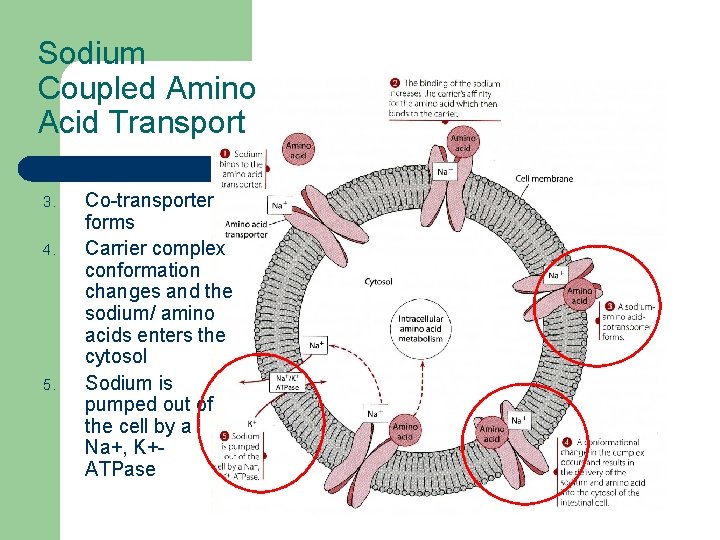
Sodium Coupled Amino Acid Transport 3. 4. 5. Co-transporter forms Carrier complex conformation changes and the sodium/ amino acids enters the cytosol Sodium is pumped out of the cell by a Na+, K+ATPase

Peptide Absorption (ie. PEPT 1) 1. Peptides are coupled to H+ and transported into the intestinal cell 2. H+ pumped back into the intestinal lumen in exchange for Na+ 3. Na+, K+ ATPase pumps Na+ out of the cell in exchange for K+ across the basolateral membrane Gropper et al 2008
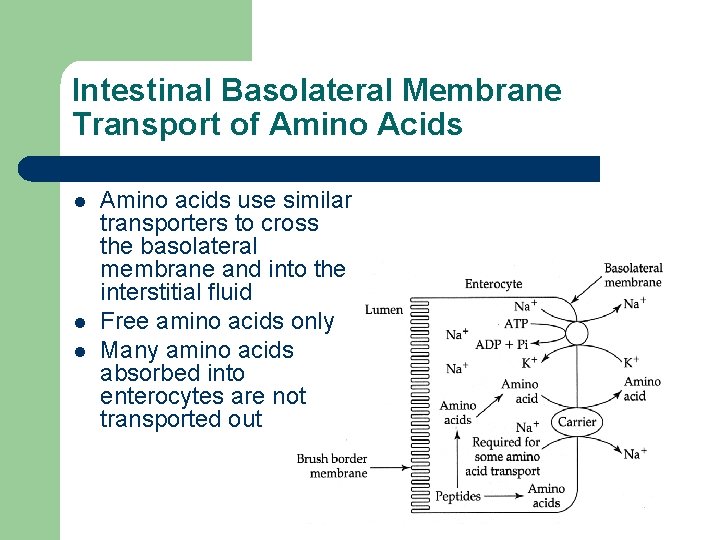
Intestinal Basolateral Membrane Transport of Amino Acids l l l Amino acids use similar transporters to cross the basolateral membrane and into the interstitial fluid Free amino acids only Many amino acids absorbed into enterocytes are not transported out
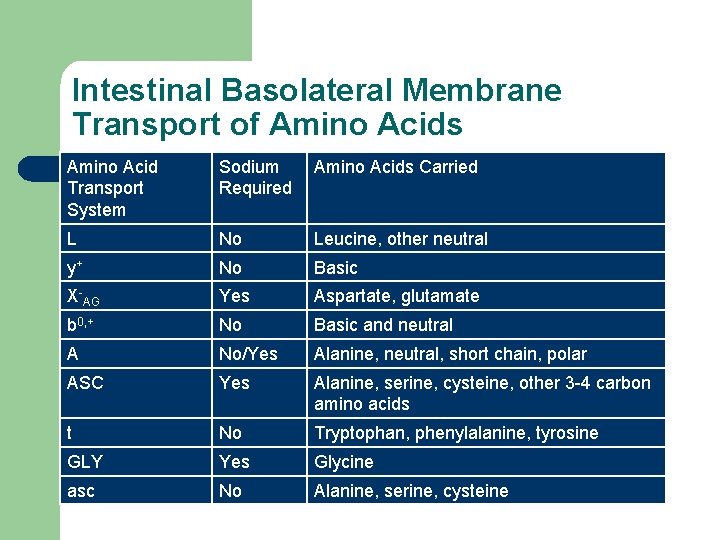
Intestinal Basolateral Membrane Transport of Amino Acids Amino Acid Transport System Sodium Required Amino Acids Carried L No Leucine, other neutral y+ No Basic X-AG Yes Aspartate, glutamate b 0, + No Basic and neutral A No/Yes Alanine, neutral, short chain, polar ASC Yes Alanine, serine, cysteine, other 3 -4 carbon amino acids t No Tryptophan, phenylalanine, tyrosine GLY Yes Glycine asc No Alanine, serine, cysteine

Protein Metabolism in the Enterocyte l Many amino acids absorbed following protein digestion are used by the intestinal cells for energy or protein synthesis – – – Structural proteins Nucleotides Apoproteins New digestive enzymes Hormones

l Amino acids may be partially metabolized to other amino acids or compounds, that may be released into portal blood l Intestines use 30 -40% and internal organs use up to 50% of the essential amino acids absorbed from the diet – 90% of glutamate absorbed is thought to be used by the intestines

Intestinal Metabolism- Glutamine l Glutamine not used for energy, may be partially catabolized to generate ammonia and glutamate – – Ammonia enters the portal blood for liver uptake or may be used by the intestine for carbamoyl phosphate synthesis Glutamate may undergo transamination (removal on amino group) to form alpha-ketoglutarate l l Intermediate in the Tricarboxylic acid (TCA) cycle Amino group is transferred to pyruvate to form alanine portal blood and to the liver
- Slides: 27