Metabolism Transformations Interactions Chapter 7 Metabolism n Metabolism

Metabolism: Transformations & Interactions Chapter 7

Metabolism n Metabolism ¨The sum total of all the chemical reactions that go on in living cells n Energy Metabolism ¨Includes all the reactions by which the body obtains & spends energy from food

The Site of Metabolic Reactions n Metabolic reactions take place inside cells, especially liver cells Anabolism is the building up of body compounds and requires energy. n Catabolism is the breakdown of body compounds and releases energy. n
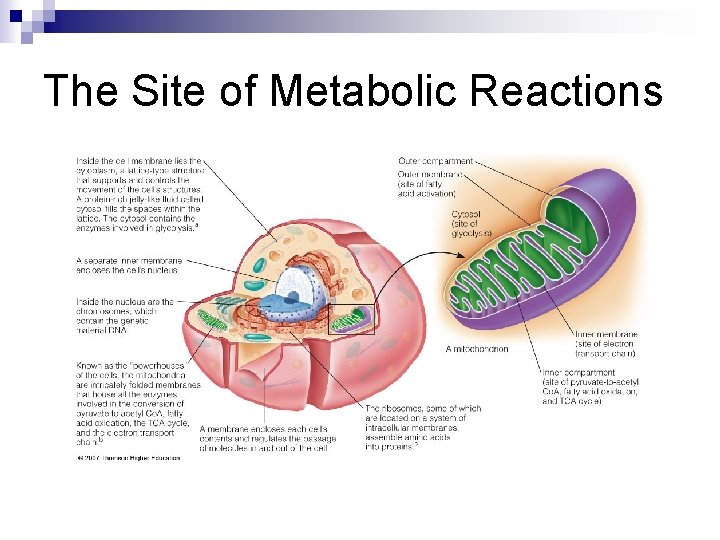
The Site of Metabolic Reactions

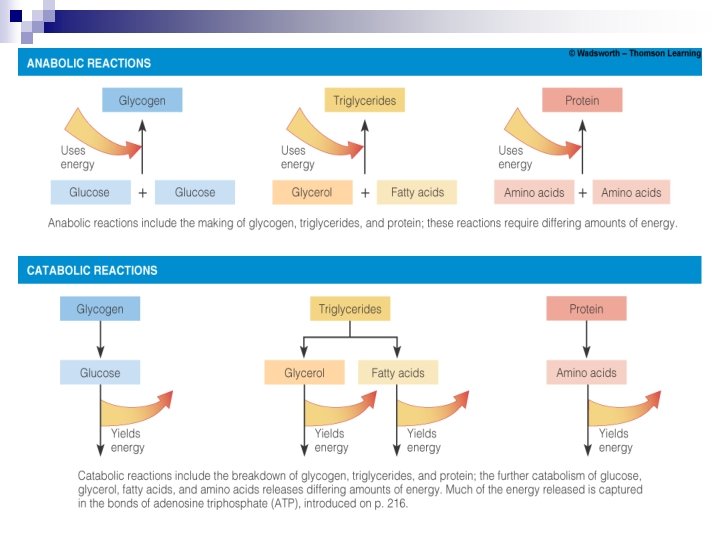
Energy Yielding Nutrients n From CHO → glucose n From fats → glycerol & FA n From proteins → AA n 4 basic units used in metabolic pathway n Alcohol can disrupt normal metabolic pathways (Highlight 7)

The Transfer of Energy in Reactions - ATP n ATP – adenosine triphosphate ¨ High-energy storage compound ¨ Captures some energy released during breakdown of glucose, glycerol, fatty acids, and amino acids
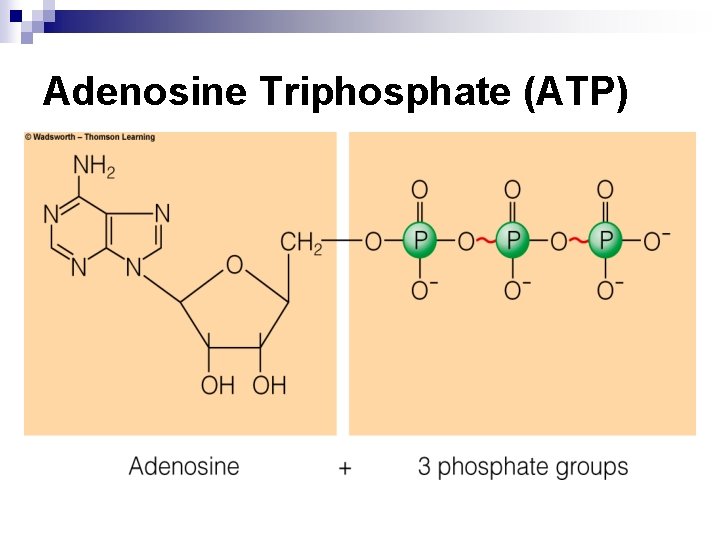
Adenosine Triphosphate (ATP)

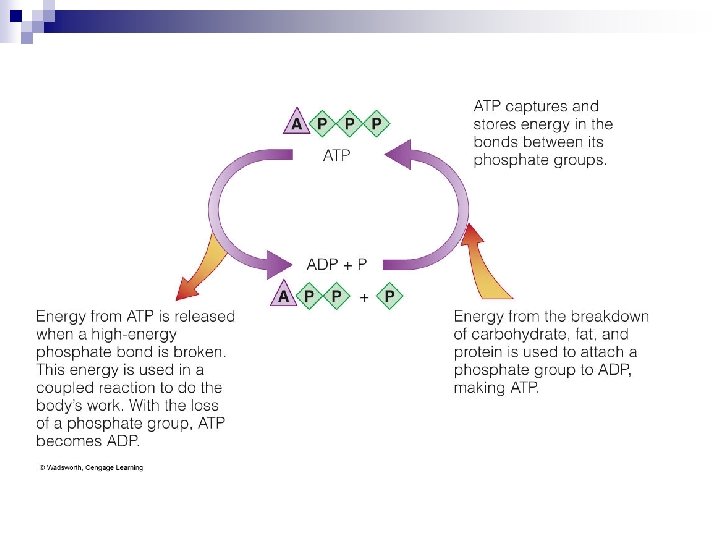
The Transfer of Energy in Reactions - ATP n Hydrolysis of ATP occurs simultaneously with reactions that will use that energy ¨ Coupled reactions ¨ Energy released from breakdown of one compound used to create bond in formation of another

Enzymes & Co-enzymes – Helpers in Metabolism n Co-enzymes are organic molecules that work with enzymes to facilitate their activity. n Enzymes & co-enzymes are helpers in metabolic reactions. n Some B vit serve as coenzymes to enzymes that release energy from glucose, glycerol, FA & AA.

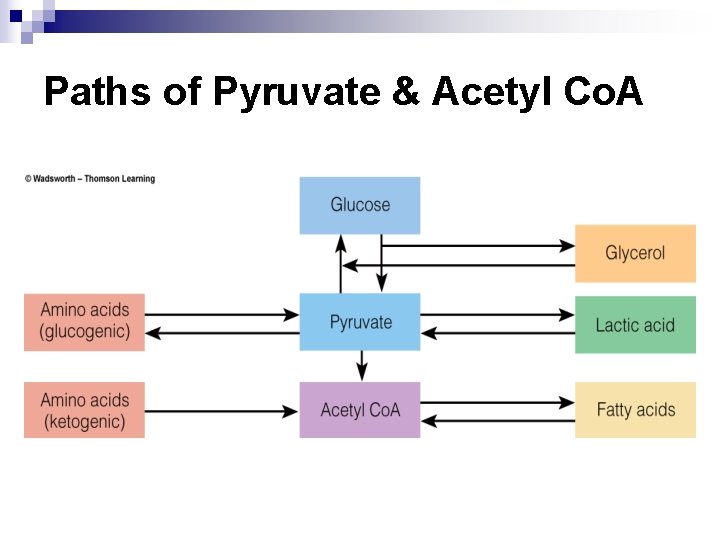
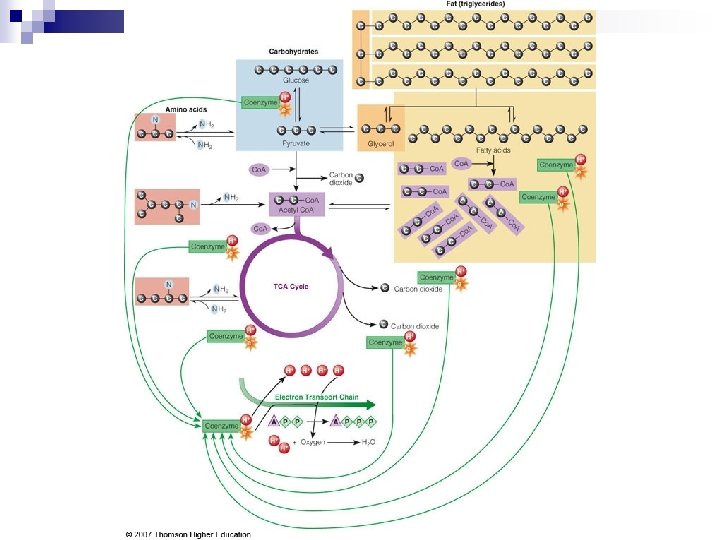
Breaking Down Nutrients for Energy n Basic units enter metabolic pathways. During catabolism, the body separates atoms of basic units. Glucose: 6 C ¨ Glycerol: 3 C ¨ FA: even number of C (commonly 16 or 18) ¨ AA: 2, 3 or more C, with N attached ¨ n While each starts down a different path – 2 by-products are common ¨ n pyruvate & acetyl-co. A Eventually all enter the TCA cycle & electron transport chain

Glycolysis n n Glucose splitting 6 -C Glucose → two 3 -C compounds → Pyruvate Glucose anaerobic Pyruvate
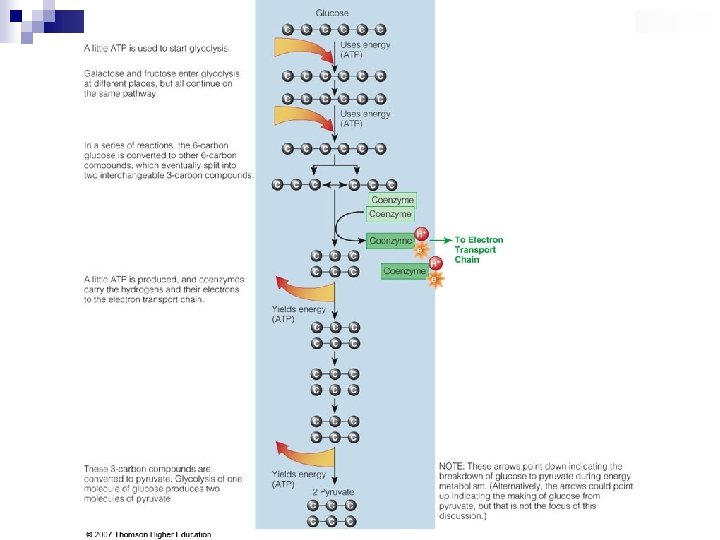

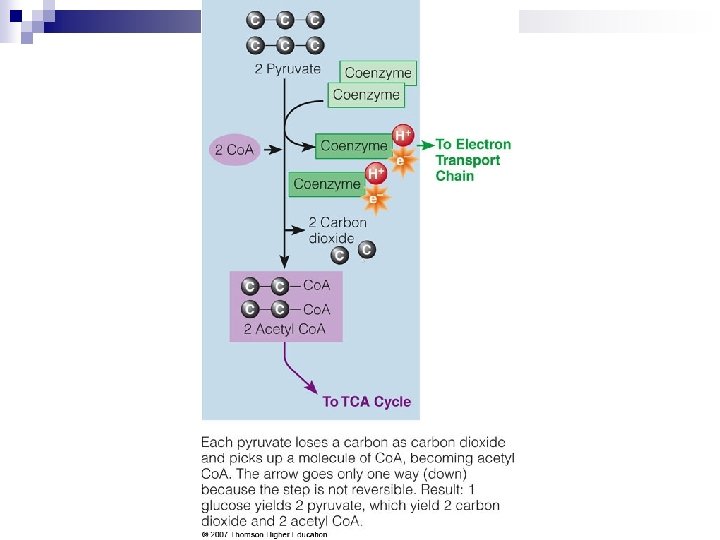
Pyruvate to Acetyl Co. A n If cell needs energy (& O 2 is available), it removes COOH group from pyruvate to produce 2 C compounds that bonds with Co. A to form Acetyl Co. A n Absence of sufficient O 2 (or mitochondria) pyruvate is converted to lactic acid Pyruvate anaerobic Pyruvate aerobic Lactate Acetyl Co. A n Lactate recycled to glucose in liver by Cori cycle
Paths of Pyruvate & Acetyl Co. A

Glycerol to Pyruvate n n 3 -C glycerol is easily converted to pyruvate Also easily converted to glucose Glycerol Pyruvate

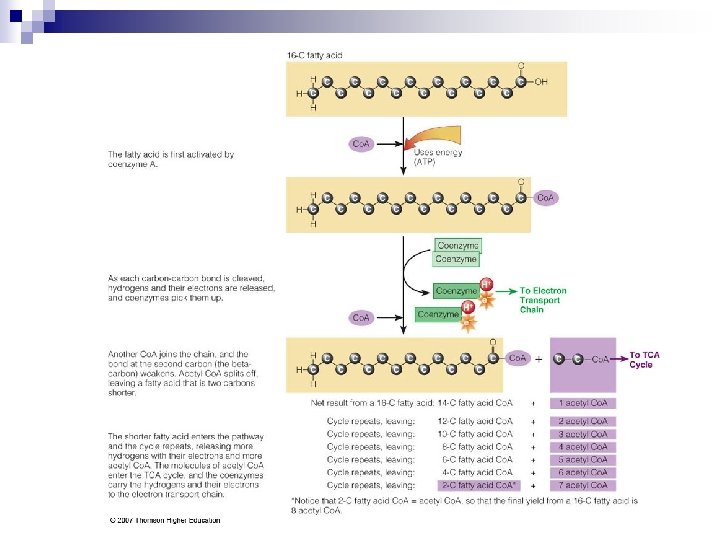
Fatty Acids to Acetyl Co. A n FA are taken apart in 2 -C units through fatty acid oxidation. n 2 -C units split off & combine with Co. A to form Acetyl Co. A. n If cell doesn’t need energy, acetyl Co. A molecules combine to create TG.

Amino Acids Breakdown n Deaminated first (lose N) n Catabolized in a variety of ways: Pyruvate glucose ¨ Acetyl Co. A more energy or body fat ¨ Directly into TCA Cycle generate energy ¨
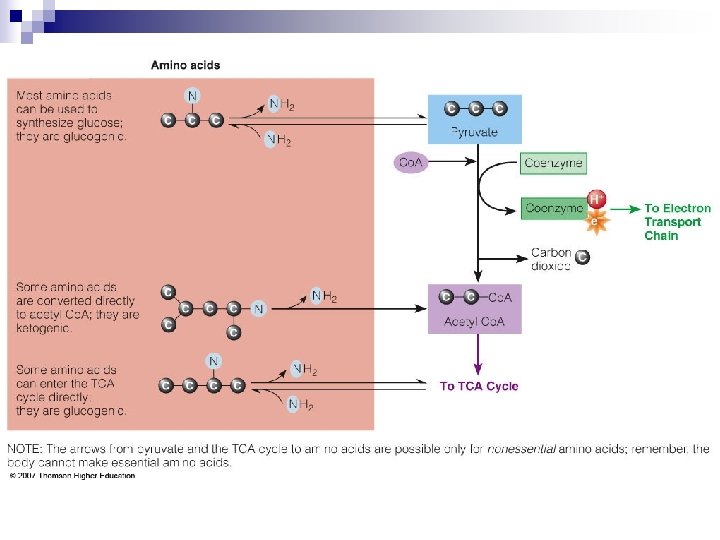

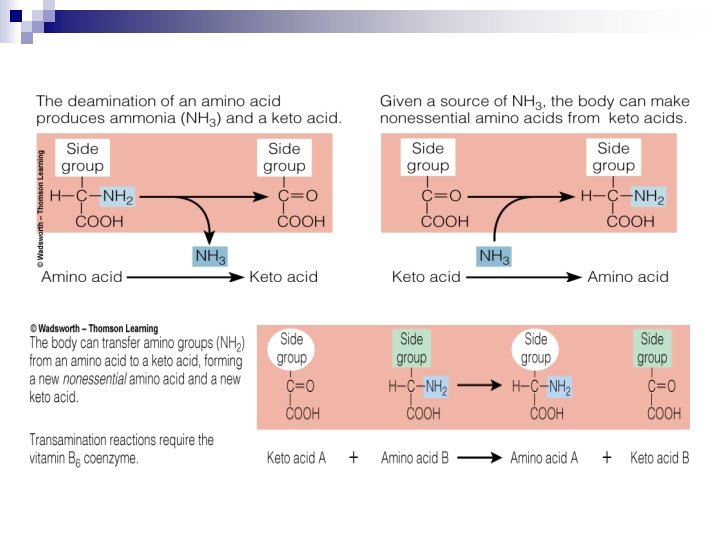
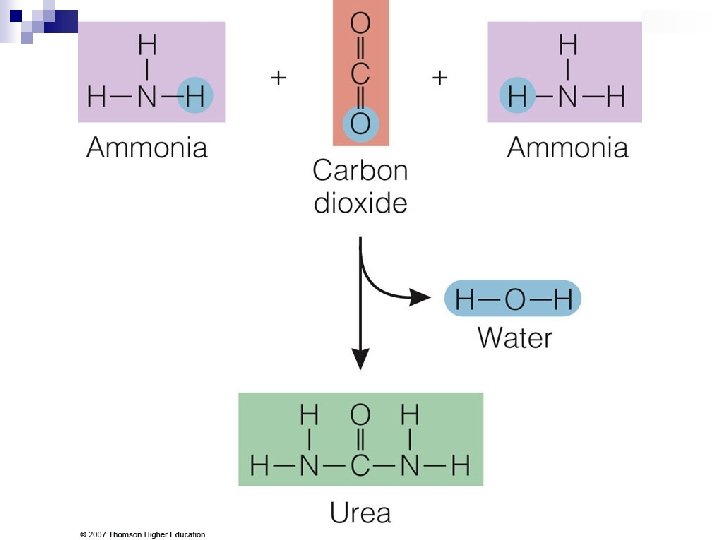
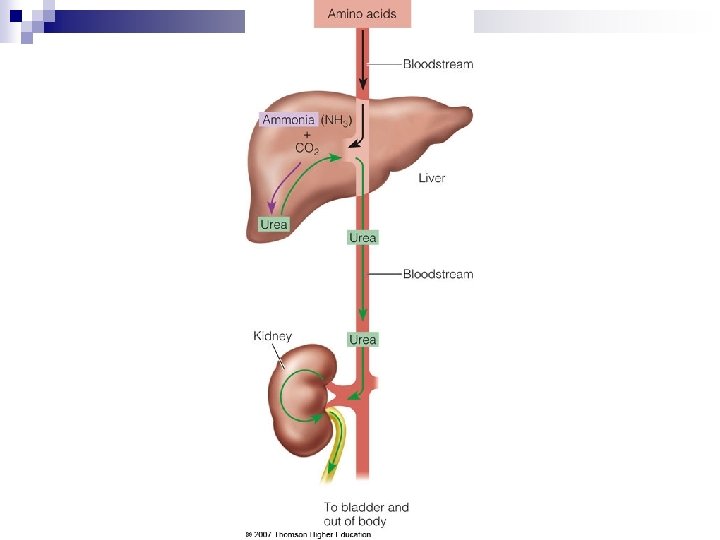
Amino Acids Breakdown n Amino Acids ¨ Deamination n n results in two products: Keto acid Ammonia ¨ Transamination is the transfer of the amino group from an amino acid to a keto acid. ¨ Ammonia is converted to urea—a much less toxic compound—in the liver. ¨ Urea is excreted through the kidneys to rid the body of unused nitrogen.

Breaking Down Nutrients for Energy n In Summary ¨ Glucose and fatty acids are primarily used for energy, amino acids to a lesser extent. ¨ Glucose is made from all carbohydrates, most amino acids and the glycerol portion of fat. ¨ Protein is made from amino acids. ¨ Glucose can be made into nonessential amino acids if nitrogen is present. ¨ All energy-yielding nutrients consumed in excess can contribute to fat storage.

Breaking Down Nutrients for Energy – the final steps TCA Cycle – tricarboxylic acid cycle (aka Kreb’s cycle) n Oxaloacetate picks up acetyl Co. A & drops off 2 -C & returns to pick up another acetyl Co. A. n As acetyl Co. A breaks to CO 2, H atoms with their electrons are removed. n Coenzymes made from B vitamins take H & electrons & transfers them to ETC.
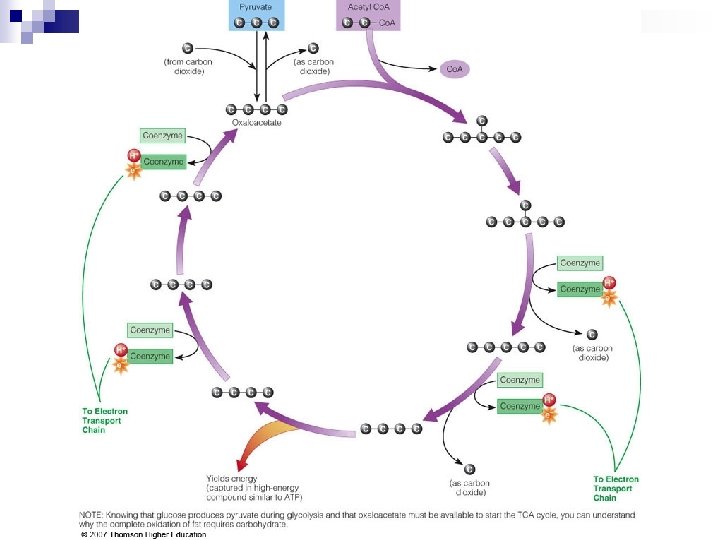

Breaking Down Nutrients for Energy – the final steps Electron Transport Chain (ETC) n Consist of a series of proteins that serve as electron carriers. These carriers are inside inner membrane of mitochondria. n In ETC, energy is captured in bonds of ATP molecules. n ATP leaves mitochondria & enters cytoplasm, where it can be used for energy.
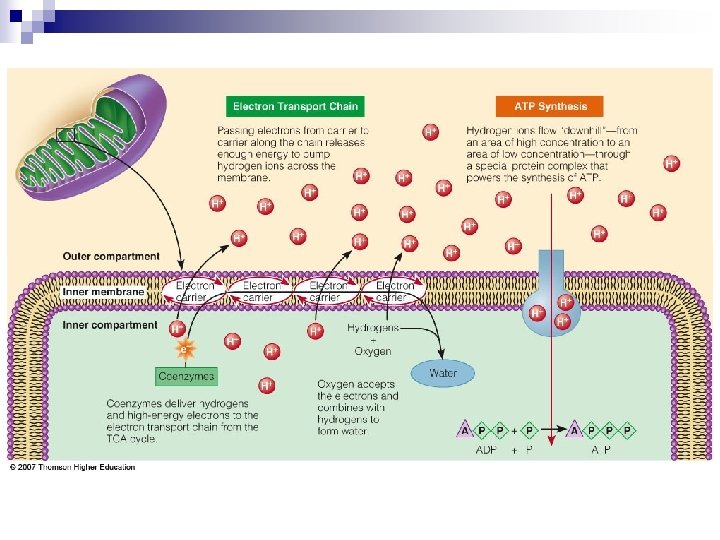

Kcal/gram n Each ATP holds energy & Kcal measures energy, so the more ATP generated the more Kcal collected. ¨ One glucose molecule yield 36 -38 ATP when oxidized completely. ¨ One 16 carbon FA yield 129 ATP when oxidized completely.

Energy Balance - Surplus n When energy intake exceeds energy output, there is a gain in weight. n Fat cells enlarge & multiply regardless of whether the excess comes from protein, CHO or fat. n Most direct & efficient pathway to body fat is dietary fat.

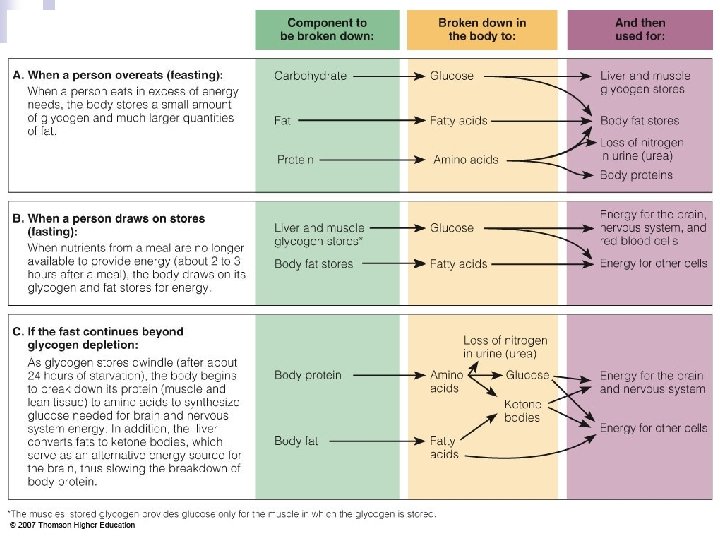
Energy Balance Body needs energy all the time n Relies on energy stores between meals n ¨ glycogen ¨ fatty acids ¨ lean mass tissue. . least preferred

Energy Balance – Fasting/Starvation n Fasting—Inadequate Energy ¨ Glucose needed for the brain ¨ Protein meets glucose needs ¨ The shift to ketosis n Ketones are produces when glucose is not available. ¨ Ketosis causes a suppression of the appetite. ¨ Slowing of metabolism

Energy Balance – Fasting/Starvation n Fasting—Inadequate Energy ¨ Symptoms of starvation n Muscle wasting n Decreased heart rate, respiratory rate, metabolic rate, and body temperature n Impaired vision n Organ failure n Decreased immunity n Depression, anxiety, and food-related dreams

Alcohol & Nutrition

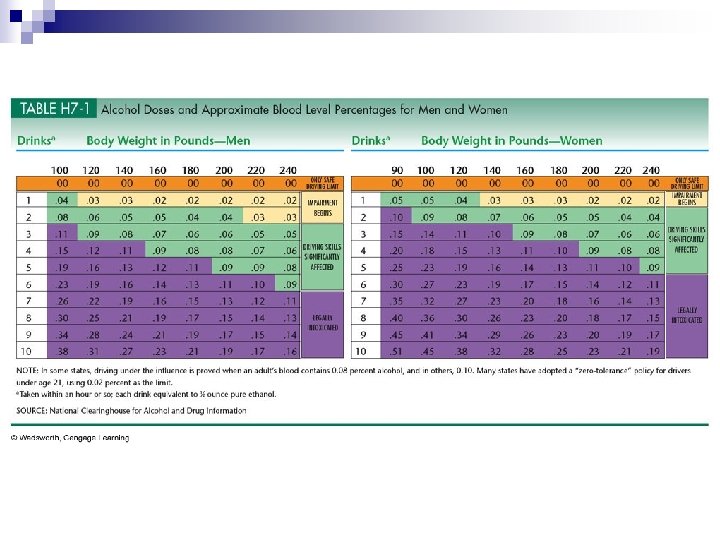
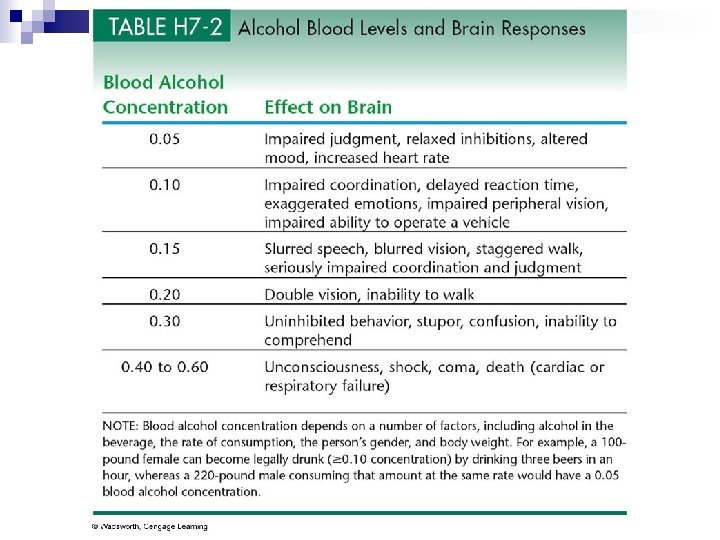
Alcohol n For most adults, moderate consumption of alcohol is no more than 1 drink a day & no more than 7 drinks a week. n Pregnant women are advised to avoid alcohol.

Alcohol in body n Doesn’t need digestion & quickly absorbed. n About 20% is absorbed directly across walls of an empty stomach & can reach brain within minutes. n Stomach breaks down alcohol with its alcohol dehydrogenase enzyme. n Can reduce amount of alcohol entering blood by about 20%.

Alcohol in small intestine n Rapidly absorbed n From then on, it’s absorbed & metabolized before most nutrients which ensures a speedy disposal.

Alcohol in liver n Liver cells make alcohol dehydrogenase enzyme to oxidize alcohol. n It can process about ½ ounce of ethanol per hour ¨ Rate is set by amount of alcohol dehydrogenase available. n Extra alcohol circulates until liver enzymes are available to process it.
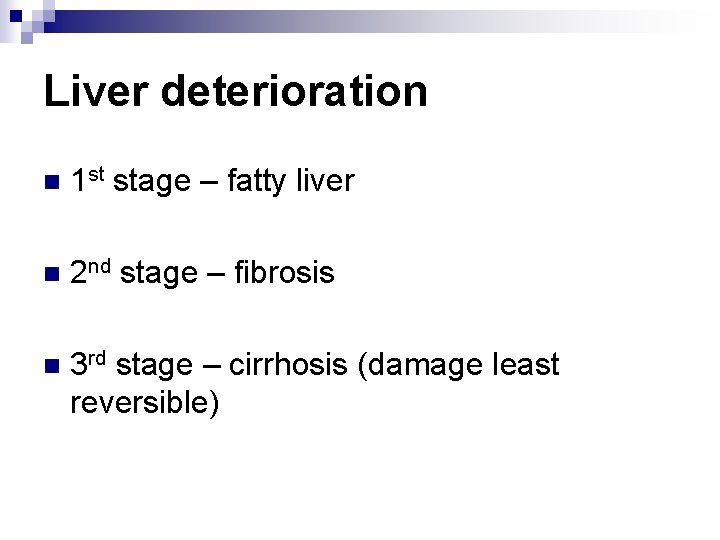
Liver deterioration n 1 st stage – fatty liver n 2 nd stage – fibrosis n 3 rd stage – cirrhosis (damage least reversible)
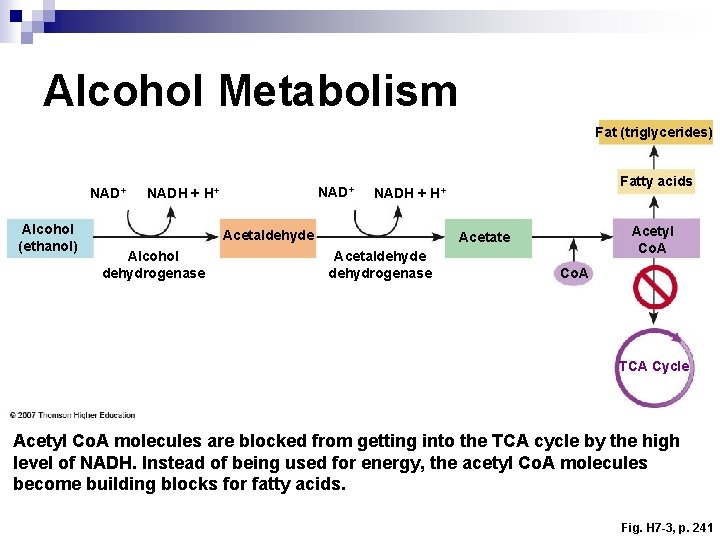
Alcohol Metabolism Fat (triglycerides) NAD+ Alcohol (ethanol) NAD+ NADH + H+ Acetaldehyde Alcohol dehydrogenase Fatty acids NADH + H+ Acetyl Co. A Acetate Acetaldehyde dehydrogenase Co. A TCA Cycle Acetyl Co. A molecules are blocked from getting into the TCA cycle by the high level of NADH. Instead of being used for energy, the acetyl Co. A molecules become building blocks for fatty acids. Fig. H 7 -3, p. 241

Alcohol & Metabolism n Alcohol alters amino acid & protein metabolism n Synthesis of protein important to immune system slows down, weakening body’s defense against infection. n Protein deficiency can develop.
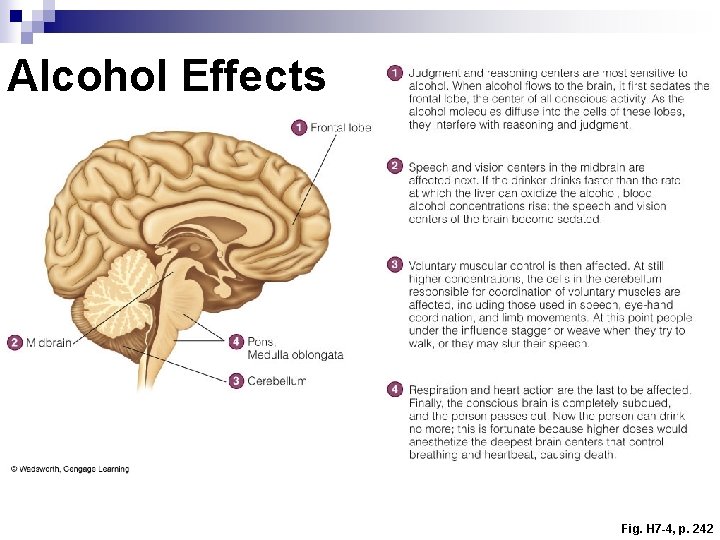
Alcohol Effects Fig. H 7 -4, p. 242

Alcohol Effects n Alcohol reduces production of Anti-diuretic hormone which retains H 2 O. n Drinking alcoholic beverages – ↑ water loss - leads to dehydration.

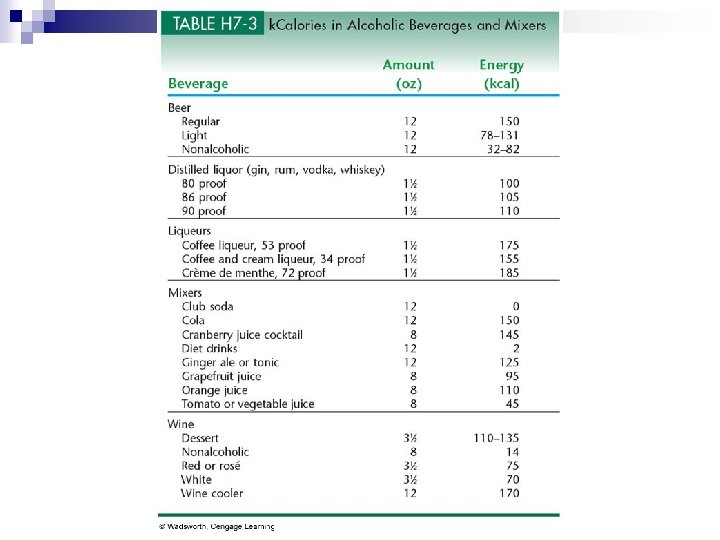
Alcohol Effects n Alcohol can contribute to body fat promote obesity. n High in calories n Most often associated with central obesity n More Kcal from alcohol - fewer Kcal from nutritious food

Alcohol Effects n Deficiency in folate & thiamin n Acetaldehyde (intermediate in alcohol metabolism) interferes with nutrient use.

Alcohol Effects n Alcohol causes stomach cells to over secrete gastric acid & histamine. n Beer stimulates gastric secretion, irritating stomach & esophagus linings making them vulnerable to ulcer formation.




Next Nutrients involved in fluid & electrolyte balance - Chapter 12
- Slides: 59