Metabolism Transformations and Interactions Chapter 7 Introduction Energy

Metabolism: Transformations and Interactions Chapter 7

Introduction § Energy § Heat for temperature maintenance § Mechanical to move muscles § Electrical for nerve impulses § Chemical- how energy is stored in food and body (ATP) § Metabolism § Release of energy, water, and carbon dioxide

Chemical Reactions in the Body § Energy metabolism after absorption § How body obtains & uses energy from food § Where does a lot of metabolism happen? § In Cells, liver cells especially § Anabolism – condensation rxn’s § Requires energy to build body’s compounds § Catabolism – hydrolysis rxn’s § Releases energy when compounds are broken down


A Typical Cell

Inside the cell membrane lies the cytoplasm, a lattice-type structure that supports and controls the movement of the cell’s structures. A protein-rich jelly-like fluid called cytosol fills the spaces within the lattice. The cytosol contains the enzymes involved in glycolysis. a This network of membranes is known as smooth endoplasmic reticulum—the site of lipid synthesis. A membrane encloses each cell’s contents and regulates the passage of molecules in and out of the cell. A separate inner membrane encloses the cell’s nucleus. Inside the nucleus are The chromosomes, Which contain the genetic material DNA. Known as the “powerhouses” of the cells, the mitochondria are intricately folded membranes that house all the enzymes involved in the conversion of pyruvate to acetyl Co. A, fatty acid oxidation, the TCA cycle, and the electron transport chain. Rough endoplasmic reticulum is dotted with ribosomes—the site of protein synthesis.

Chemical Reactions in the Body
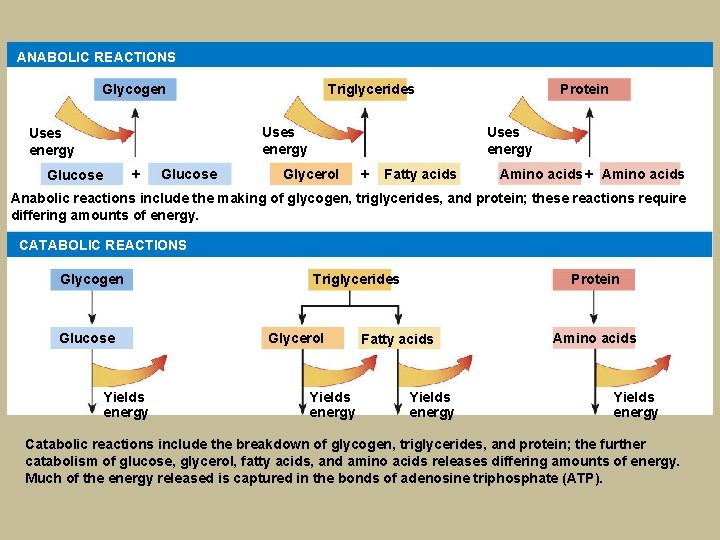
ANABOLIC REACTIONS Glycogen Triglycerides Uses energy + Glucose Protein Uses energy Glycerol + Fatty acids Amino acids + Amino acids Anabolic reactions include the making of glycogen, triglycerides, and protein; these reactions require differing amounts of energy. CATABOLIC REACTIONS Glycogen Glucose Yields energy Triglycerides Glycerol Yields energy Protein Fatty acids Yields energy Amino acids Yields energy Catabolic reactions include the breakdown of glycogen, triglycerides, and protein; the further catabolism of glucose, glycerol, fatty acids, and amino acids releases differing amounts of energy. Much of the energy released is captured in the bonds of adenosine triphosphate (ATP).

Metabolism in the Body § Transfer of energy in reactions – ATP § Released during breakdown of glucose, fatty acids, and amino acids § Form of phosphate groups § Negative charge – vulnerable to hydrolysis § Provides energy for all cell activities § Coupled reactions § Efficiency § Heat loss
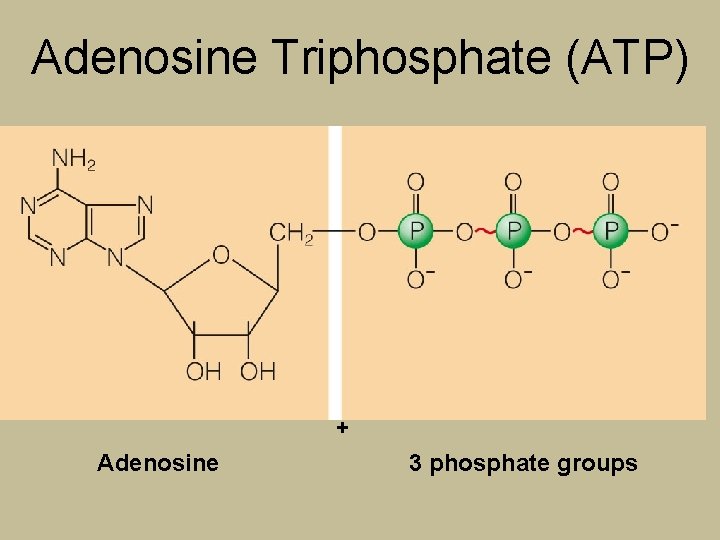
Adenosine Triphosphate (ATP) + Adenosine 3 phosphate groups
Capture/Release of Energy by ATP
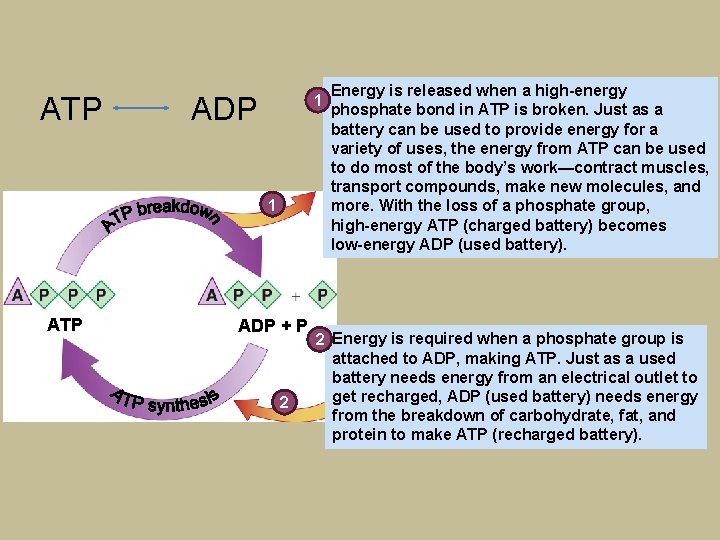
ATP 1 ADP 1 ATP ADP + P 2 Energy is released when a high-energy phosphate bond in ATP is broken. Just as a battery can be used to provide energy for a variety of uses, the energy from ATP can be used to do most of the body’s work—contract muscles, transport compounds, make new molecules, and more. With the loss of a phosphate group, high-energy ATP (charged battery) becomes low-energy ADP (used battery). 2 Energy is required when a phosphate group is attached to ADP, making ATP. Just as a used battery needs energy from an electrical outlet to get recharged, ADP (used battery) needs energy from the breakdown of carbohydrate, fat, and protein to make ATP (recharged battery).

Helpers in Metabolic Rxn’s § Enzymes § Facilitators of metabolic reactions § Coenzymes § Organic § Associate with enzymes § Without coenzyme, an enzyme cannot function

II. Break Down Nutri. for Energy § Digestion § Carbohydrates into glucose & other monosaccharides § Fats (triglycerides) into glycerol and fatty acids § Proteins into amino acids § Digestion Products: molecules of glucose, glycerol, amino acids, and fatty acids § Catabolism § Carbon, nitrogen, oxygen, hydrogen

Nutrient Breakdown for Energy § Two energy-releasing compounds headed for TCA cycle and electron transport chain § Pyruvate § 3 -carbon structure § Can be used to make glucose § Acetyl Co. A § 2 -carbon structure § Cannot be used to make glucose
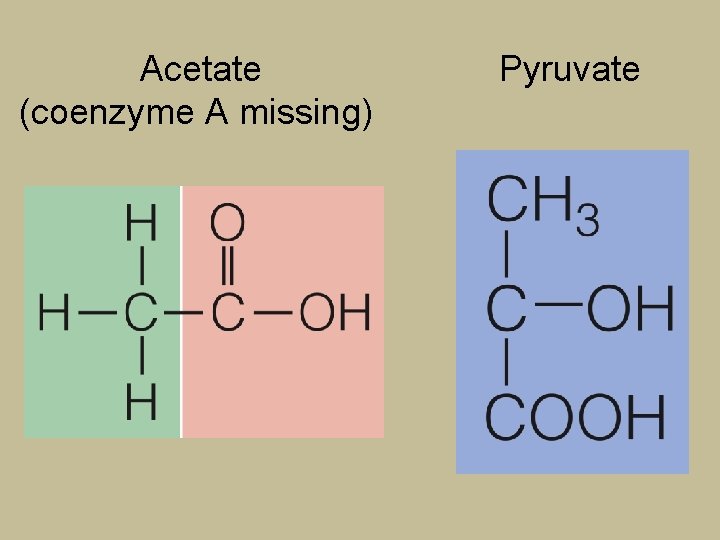
Acetate (coenzyme A missing) Pyruvate

Breaking Down Nutrients for Energy
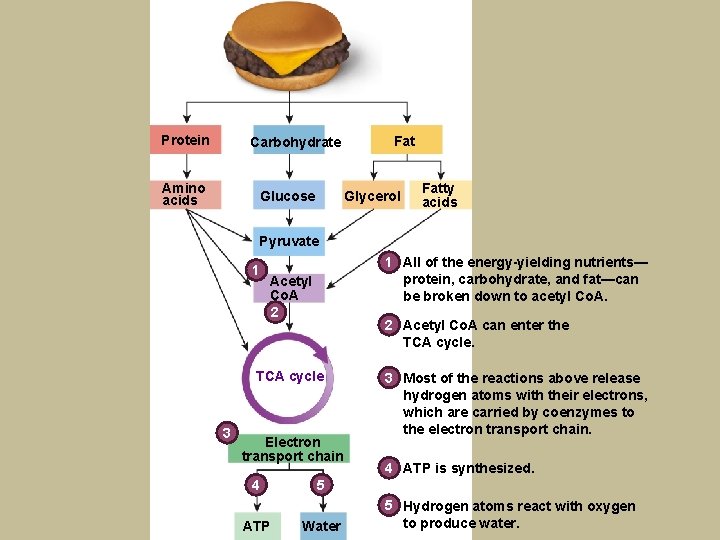
Protein Carbohydrate Amino acids Glucose Fat Glycerol Fatty acids Pyruvate 1 1 All of the energy-yielding nutrients— protein, carbohydrate, and fat—can be broken down to acetyl Co. A. Acetyl Co. A 2 2 Acetyl Co. A can enter the TCA cycle 3 Electron transport chain 44 ATP 3 Most of the reactions above release hydrogen atoms with their electrons, which are carried by coenzymes to the electron transport chain. 4 ATP is synthesized. 55 Water 5 Hydrogen atoms react with oxygen to produce water.
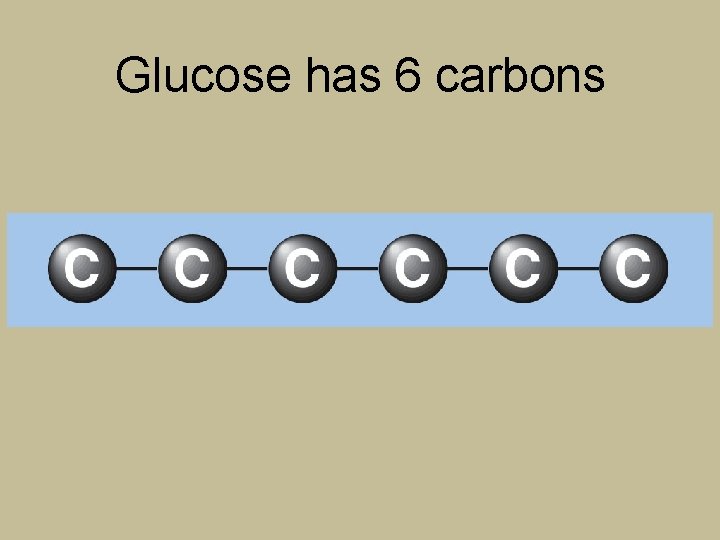
Glucose has 6 carbons
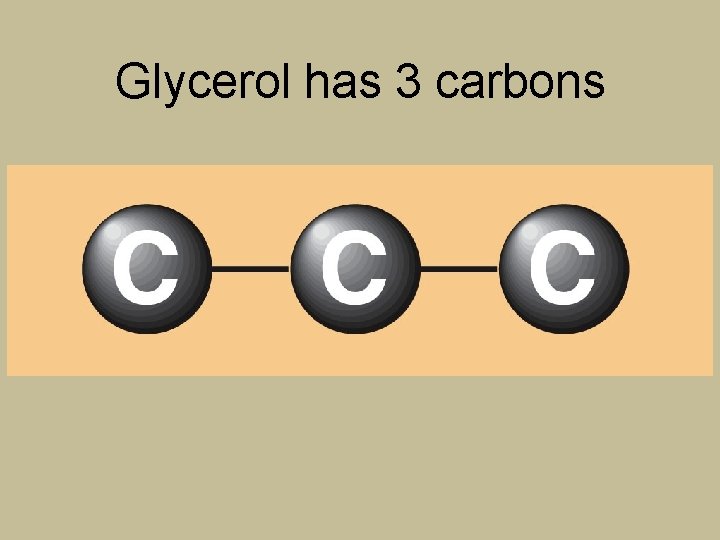
Glycerol has 3 carbons
Amino acids have varying no. ’s of carbons

Breaking Down Nutrients for Energy – Glucose § Glucose to pyruvate § Glycolysis § For short energy bursts and TCA cycle prep § 1 glucose yields 2 pyruvate § Hydrogen atoms carried to electron transport chain § Pyruvate can be converted back to glucose § Liver cells and kidneys (to some extent)

Glucose to Pyruvate Glycolysis § Fructose and galactose enter same pathway glucose is on § Needs ATP for jump start § file: ///E: /Media/Animations/chapter 7/0 705. html
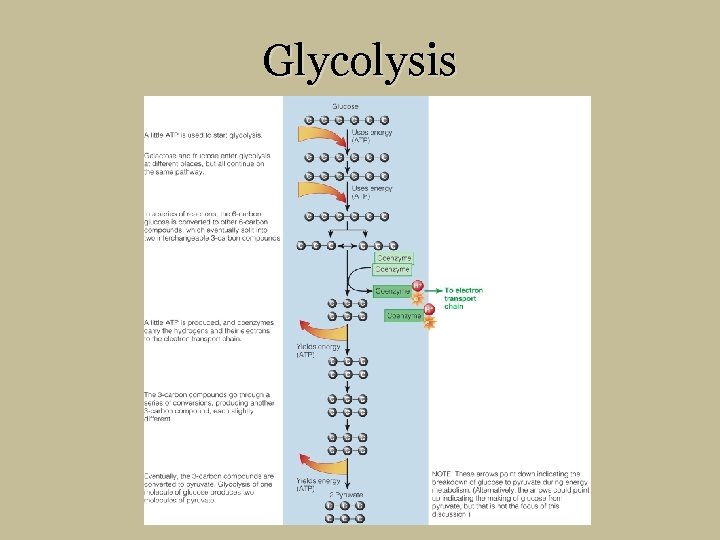
Glycolysis
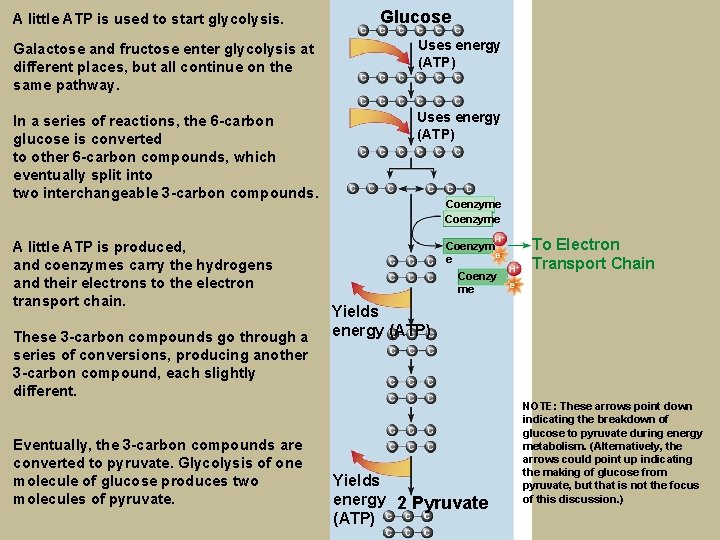
A little ATP is used to start glycolysis. Glucose Galactose and fructose enter glycolysis at different places, but all continue on the same pathway. Uses energy (ATP) In a series of reactions, the 6 -carbon glucose is converted to other 6 -carbon compounds, which eventually split into two interchangeable 3 -carbon compounds. Uses energy (ATP) A little ATP is produced, and coenzymes carry the hydrogens and their electrons to the electron transport chain. These 3 -carbon compounds go through a series of conversions, producing another 3 -carbon compound, each slightly different. Eventually, the 3 -carbon compounds are converted to pyruvate. Glycolysis of one molecule of glucose produces two molecules of pyruvate. Coenzyme Coenzym e Coenzy me To Electron Transport Chain Yields energy (ATP) Yields energy 2 Pyruvate (ATP) NOTE: These arrows point down indicating the breakdown of glucose to pyruvate during energy metabolism. (Alternatively, the arrows could point up indicating the making of glucose from pyruvate, but that is not the focus of this discussion. )

Breaking Down Glucose for Energy § Pyruvate’s options § Quick energy needs – anaerobic §Pyruvate-to-lactate or back to glucose § Slower energy needs – aerobic §Pyruvate-to-acetyl Co. A (irreversible to glucose)

Breaking Down Glucose for Anaerobic Energy § Pyruvate conversion to lactate § Pyruvate accepts hydrogens § Occurs during high-intensity exercise, has limited minutes § Produces ATP quickly when too few mitochondria or low oxygen § Accumulation of lactate in muscles from rapid glycolysis § Liver’s Cori cycle- lactate back to glucose
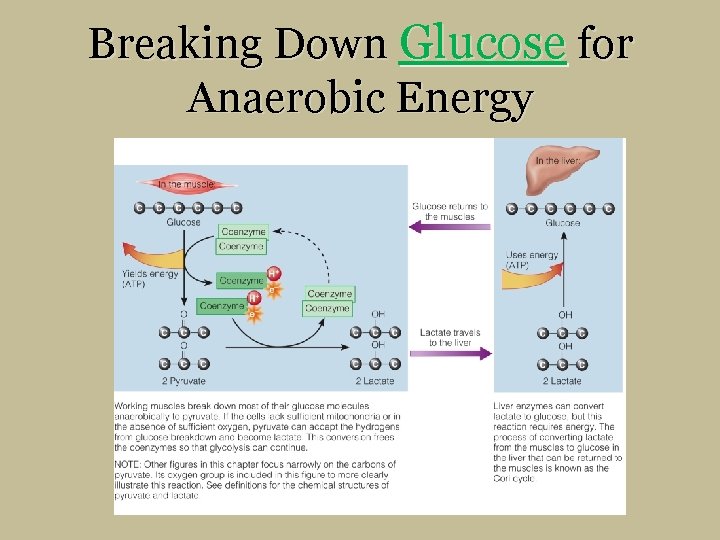
Breaking Down Glucose for Anaerobic Energy
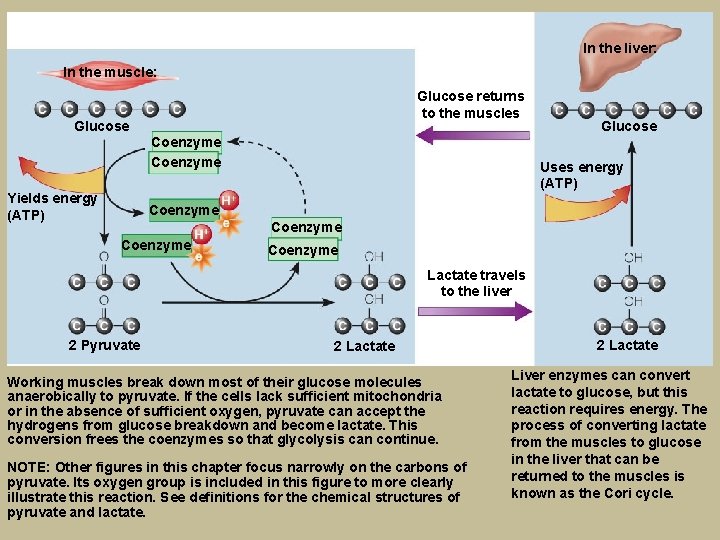
In the liver: In the muscle: Glucose Yields energy (ATP) Glucose returns to the muscles Coenzyme Glucose Uses energy (ATP) Coenzyme Lactate travels to the liver 2 Pyruvate 2 Lactate Working muscles break down most of their glucose molecules anaerobically to pyruvate. If the cells lack sufficient mitochondria or in the absence of sufficient oxygen, pyruvate can accept the hydrogens from glucose breakdown and become lactate. This conversion frees the coenzymes so that glycolysis can continue. NOTE: Other figures in this chapter focus narrowly on the carbons of pyruvate. Its oxygen group is included in this figure to more clearly illustrate this reaction. See definitions for the chemical structures of pyruvate and lactate. 2 Lactate Liver enzymes can convert lactate to glucose, but this reaction requires energy. The process of converting lactate from the muscles to glucose in the liver that can be returned to the muscles is known as the Cori cycle.
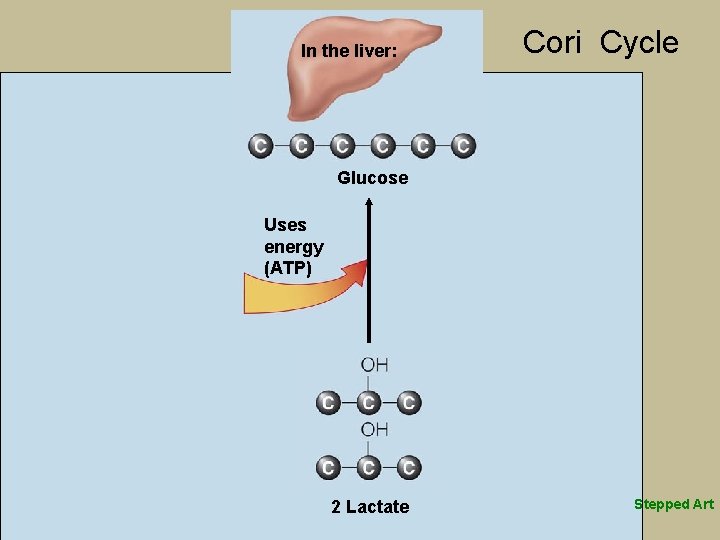
In the liver: Cori Cycle Glucose Uses energy (ATP) 2 Lactate Stepped Art

Breaking Down Glucose for AEROBIC Energy § Pyruvate-to-Acetyl Co. A § Pyruvate enters mitochondria of cell § Carbon removed – becomes carbon dioxide § 2 -carbon compound joins with Co. A becoming acetyl Co. A – irreversible

Breaking Down Glucose for AEROBIC Energy
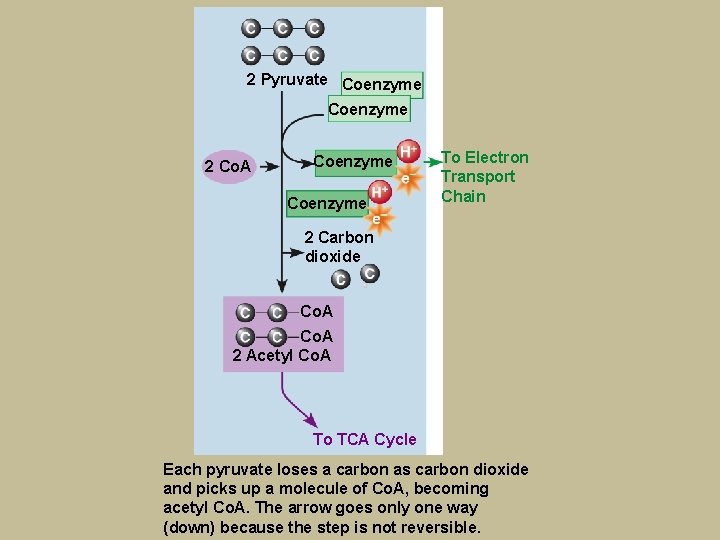
2 Pyruvate Coenzyme 2 Co. A Coenzyme To Electron Transport Chain 2 Carbon dioxide Co. A 2 Acetyl Co. A To TCA Cycle Each pyruvate loses a carbon as carbon dioxide and picks up a molecule of Co. A, becoming acetyl Co. A. The arrow goes only one way (down) because the step is not reversible.

Breaking Down Glucose for AEROBIC Energy… now or later § Acetyl Co. A’s options – 2 functions 1. Synthesize fats when ATP is abundant § Any molecule that can make acetyl Co. A can make fat (glucose, glycerol, fatty/amino acids) § Acetyl Co. A itself can only make fatty acids 2. Generate more ATP through TCA cycle than glycolysis § Hydrogens – electron transport chain
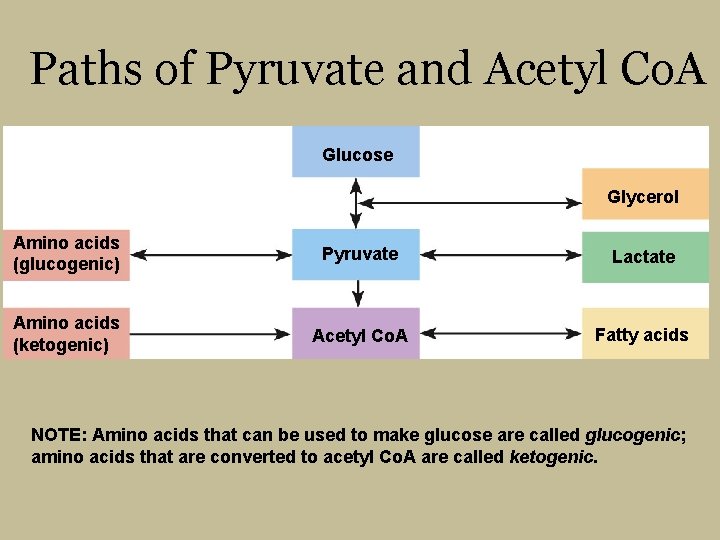
Paths of Pyruvate and Acetyl Co. A Glucose Glycerol Amino acids (glucogenic) Pyruvate Lactate Amino acids (ketogenic) Acetyl Co. A Fatty acids NOTE: Amino acids that can be used to make glucose are called glucogenic; amino acids that are converted to acetyl Co. A are called ketogenic.
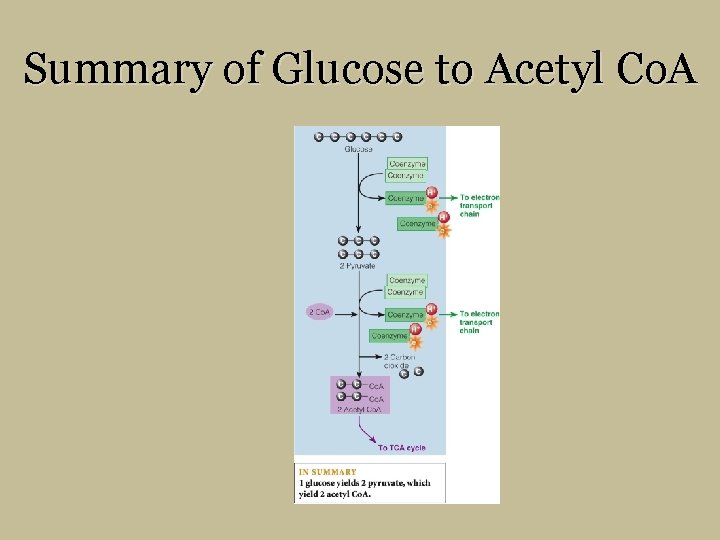
Summary of Glucose to Acetyl Co. A
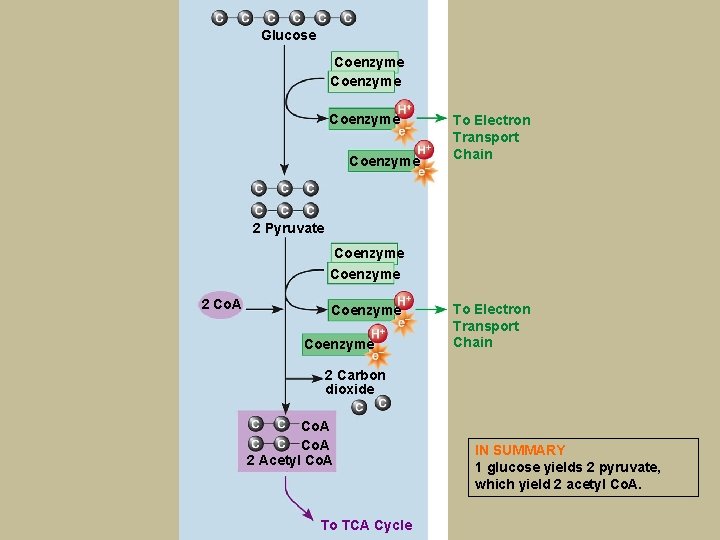
Glucose Coenzyme To Electron Transport Chain 2 Pyruvate Coenzyme 2 Co. A Coenzyme To Electron Transport Chain 2 Carbon dioxide Co. A 2 Acetyl Co. A To TCA Cycle IN SUMMARY 1 glucose yields 2 pyruvate, which yield 2 acetyl Co. A.

Breaking Down Glycerol and Fatty Acids from TG for Energy § Glycerol into pyruvate § Glycerol can be converted to § Glucose § Pyruvate § Fatty acids into Acetyl Co. A § Fatty acid oxidation § 2 -carbon units at a time join with Co. A § Hydrogens and electrons carried to electron transport chain

Glycerol Pyruvate

Breaking Down Glycerol and Fatty Acids from TG for Energy § Fatty Acid to Acetyl Co. A § 2 fatty acids are snapped off at a time to combine with Co. A to make Acetyl Co. A (oxidation rxn) § file: ///E: /Media/Animations/chapter 7/0710. ht ml § 16 -carbon fatty acid yields 8 Acetyl Co. A

Breaking Down Glycerol and Fatty Acids for Energy
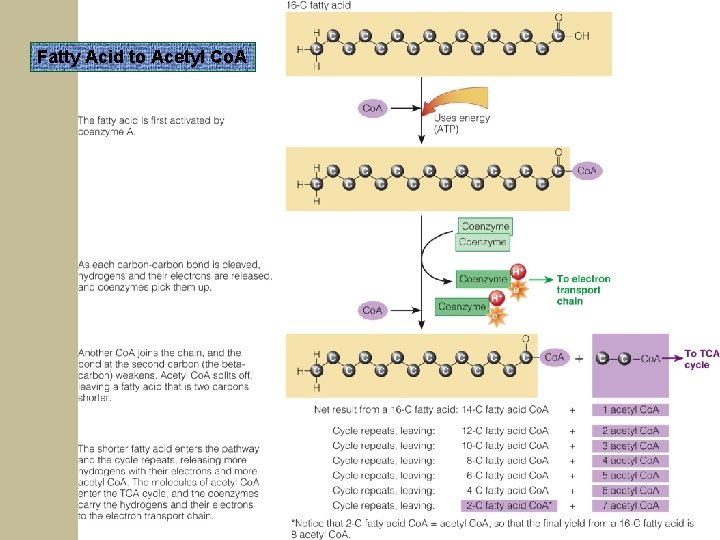
Fatty Acid to Acetyl Co. A
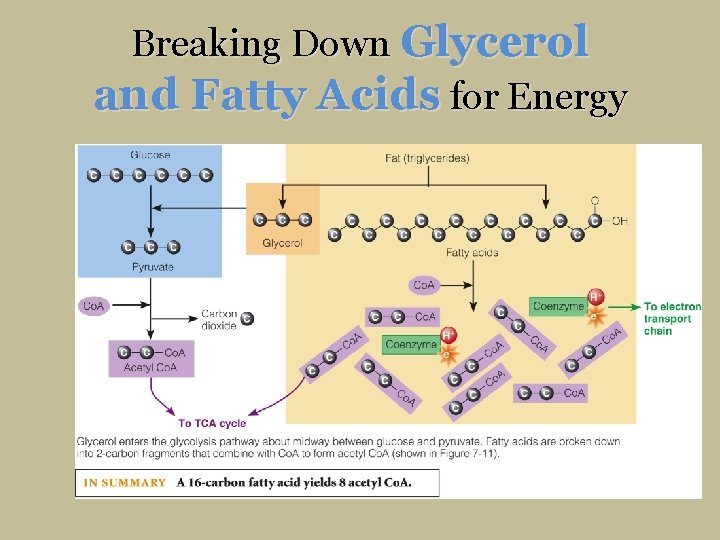
Breaking Down Glycerol and Fatty Acids for Energy

Breaking Down Glycerol and Fatty Acids for Energy Glycerol Fatty acids 18 C 3 C 54 C A typical triglyceride contains only one small molecule of glycerol (3 C) but has three fatty acids (each commonly 16 C or 18 C, or about 48 C to 54 C in total). Only the glycerol portion of a triglyceride can yield glucose.

Fats Enter the Energy Pathway Glucose Fat (triglycerides) Glycerol Pyruvate Fatty acids Co. A Carbon dioxide Co. A Acetyl Co. A Coenzyme To Electron Transport Chain To TCA Cycle Glycerol enters the glycolysis pathway about midway between glucose and pyruvate and can be converted to either. Fatty acids are broken down into 2 -carbon fragments that combine with Co. A to form acetyl Co. A (shown in Figure 7 -11). IN SUMMARY A 16 -carbon fatty acid yields 8 acetyl Co. A.

Fats Enter the Energy Pathway • Product of 16 -C fatty acid is 8 Acetyl Co. A for now or later file: ///E: /Media/Animations/chapter 7/0711. html

Breaking Down Amino Acids for Energy § § Amino acids into glucose, then energy § Several entry points in energy pathway § Converted to pyruvate (glucogenic) § Converted to acetyl Co. A (ketogenic) § Enter TCA cycle directly (glucogenic) § Amino acids-to-glucose
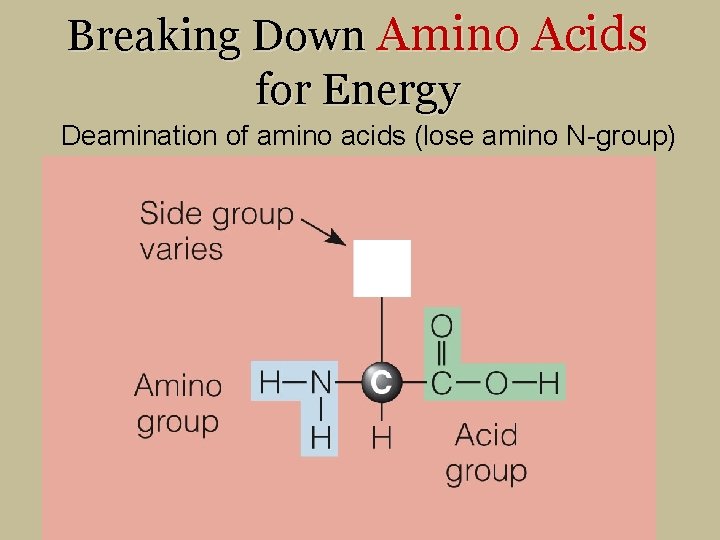
Breaking Down Amino Acids for Energy Deamination of amino acids (lose amino N-group)
Breaking Down Amino Acids for Energy
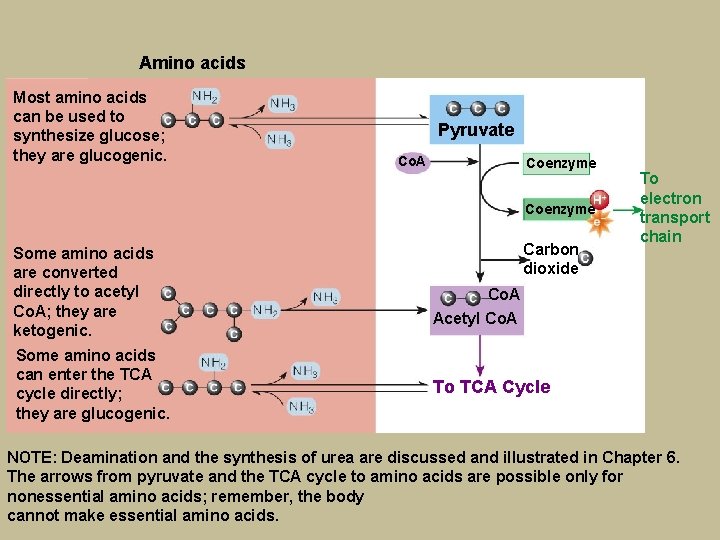
Amino acids Most amino acids can be used to synthesize glucose; they are glucogenic. Pyruvate Co. A Coenzyme Some amino acids are converted directly to acetyl Co. A; they are ketogenic. Some amino acids can enter the TCA cycle directly; they are glucogenic. Carbon dioxide To electron transport chain Co. A Acetyl Co. A To TCA Cycle NOTE: Deamination and the synthesis of urea are discussed and illustrated in Chapter 6. The arrows from pyruvate and the TCA cycle to amino acids are possible only for nonessential amino acids; remember, the body cannot make essential amino acids.

The TCA Cycle § Products of glucose, fat and amino acids now enter the TCA Cycle § Where? Inner compartment of mitochondria § Circular path because OAA is regenerated § Oxaloacetate – made primarily from pyruvate § Acetyl Co. A goes one direction § Carbon dioxide, H with electrons released § Coenzymes from niacin and riboflavin transfer H and electrons to Electron Transport Chain (ETC)

TCA Cycle § OAA starts the cycle § C released as CO 2; Co. A releases H and electrons § Each cycle releases 8 electrons total § file: ///E: /Media/Animations/chapter 7/0718. html
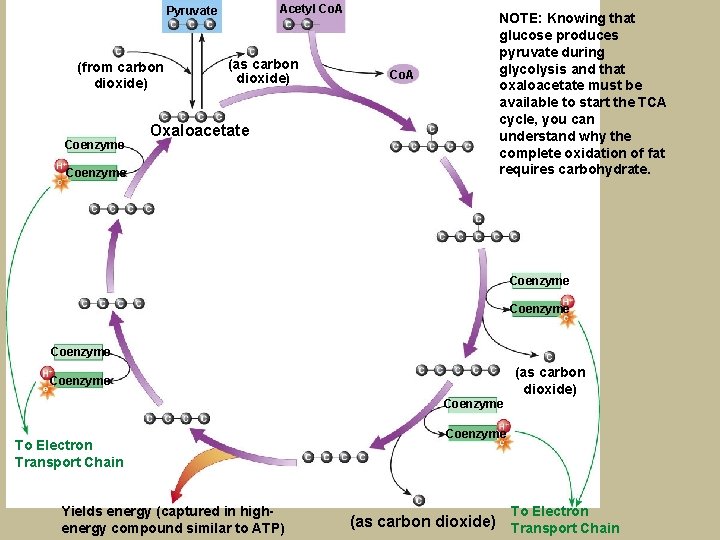
Acetyl Co. A Pyruvate (from carbon dioxide) Coenzyme (as carbon dioxide) Oxaloacetate Coenzyme Co. A NOTE: Knowing that glucose produces pyruvate during glycolysis and that oxaloacetate must be available to start the TCA cycle, you can understand why the complete oxidation of fat requires carbohydrate. Coenzyme (as carbon dioxide) Coenzyme To Electron Transport Chain Yields energy (captured in highenergy compound similar to ATP) Coenzyme To Electron (as carbon dioxide) Transport Chain

Electron Transport Chain § Energy captured in ATP bonds § The physical chain mounted on inner membrane of mitochondria (sl. 56) § Series of proteins acting as electron “carriers” § Electrons passed from carrier to carrier § End of chain –Oxygen accepts electrons, adds H to form water, water released § Rush of H ions into inner mitochondrion powers ATP synthesis
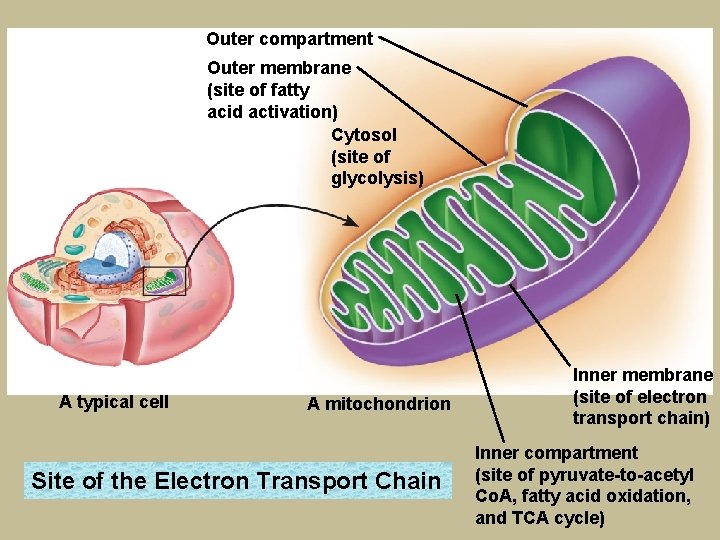
Outer compartment Outer membrane (site of fatty acid activation) Cytosol (site of glycolysis) A typical cell A mitochondrion Site of the Electron Transport Chain Inner membrane (site of electron transport chain) Inner compartment (site of pyruvate-to-acetyl Co. A, fatty acid oxidation, and TCA cycle)
file: ///E: /Media/Animations/chapter 7/ 0719. html

Central Pathways of Energy Metabolism

Energy Balance – Feasting § Too much food? Metabolism favors fat formation § Regardless of excess from protein, fat, or carbohydrates § Dietary fat to body fat is most direct and efficient conversion § Carbohydrate and protein have other roles to fulfill before conversion to body fat § Fuel mix is ideal (balance)
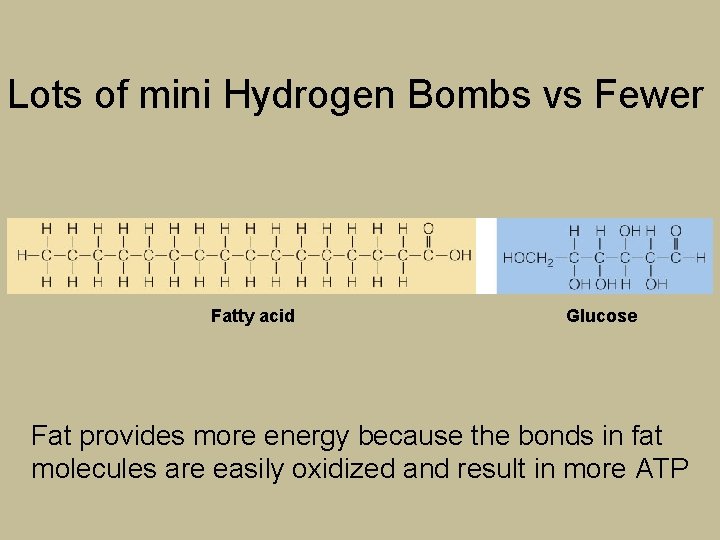
Lots of mini Hydrogen Bombs vs Fewer Fatty acid Glucose Fat provides more energy because the bonds in fat molecules are easily oxidized and result in more ATP

Feasting Reserves for Fasting § Glucose, glycerol, and fatty acids are used, then excess stored. Fasting state draws on these stores. § Glucose needed for Central Nervous Sys. § Glycogen and fat glycerol are released § Some amino acids can go to pyruvate § Ketones produced when no glucose § Basal metabolism slowed by hormones § Starvation: muscle wasting, organ slowdown and failure, impaired immunity and vision
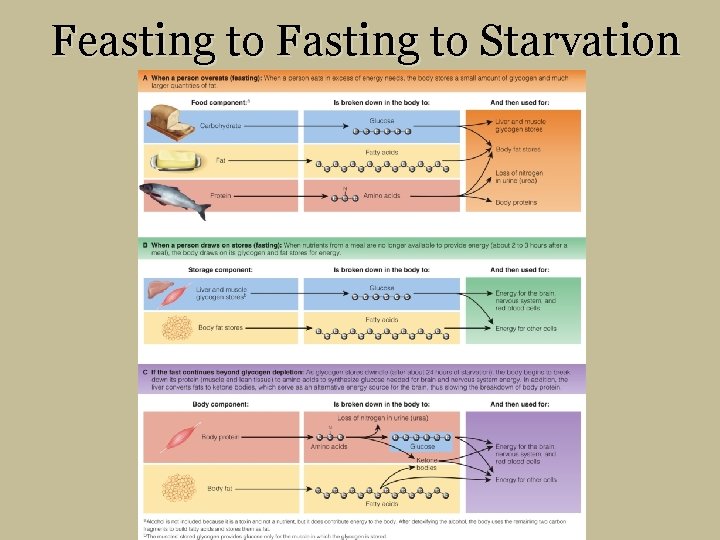
Feasting to Fasting to Starvation
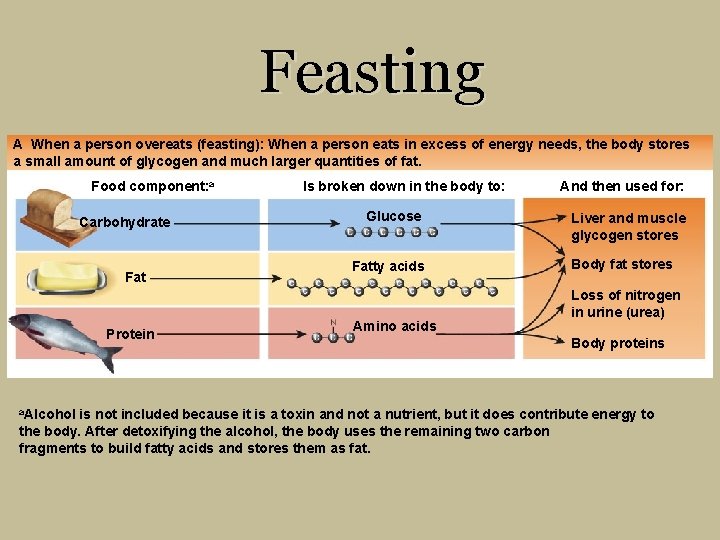
Feasting A When a person overeats (feasting): When a person eats in excess of energy needs, the body stores a small amount of glycogen and much larger quantities of fat. Food component: a Carbohydrate Fat Protein a. Alcohol Is broken down in the body to: Glucose Fatty acids Amino acids And then used for: Liver and muscle glycogen stores Body fat stores Loss of nitrogen in urine (urea) Body proteins is not included because it is a toxin and not a nutrient, but it does contribute energy to the body. After detoxifying the alcohol, the body uses the remaining two carbon fragments to build fatty acids and stores them as fat.
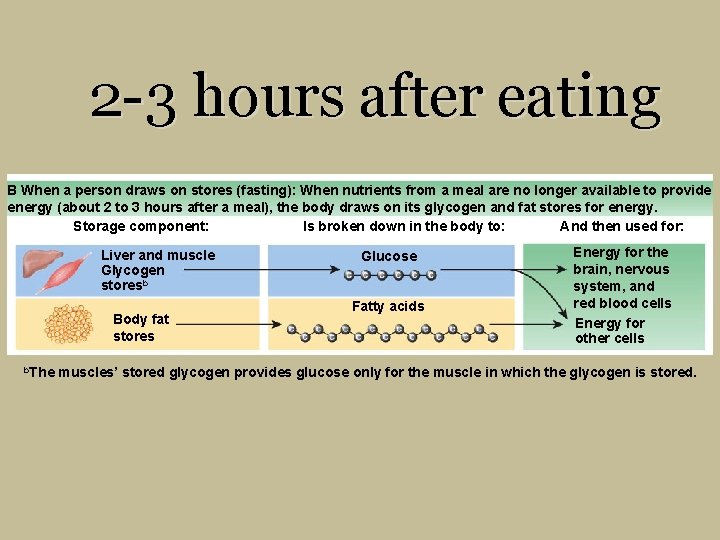
2 -3 hours after eating B When a person draws on stores (fasting): When nutrients from a meal are no longer available to provide energy (about 2 to 3 hours after a meal), the body draws on its glycogen and fat stores for energy. Storage component: Is broken down in the body to: And then used for: Liver and muscle Glycogen storesb Body fat stores b. The Glucose Fatty acids Energy for the brain, nervous system, and red blood cells Energy for other cells muscles’ stored glycogen provides glucose only for the muscle in which the glycogen is stored.

Fasting Body component: Body protein Is broken down in the body to: Loss of nitrogen in urine (urea) N Amino acids Glucose Ketone bodies And then used for : Energy for the brain, nervous system, and red blood cells Energy for other cells Body fat Fatty acids C If the fast continues beyond glycogen depletion: As glycogen stores dwindle (after about 24 hours of starvation), the body begins to break down its protein (muscle and lean tissue) to amino acids to synthesize glucose needed for brain and nervous system energy. In addition, the liver converts fats to ketone bodies, which serve as an alternative energy source for the brain, thus slowing the breakdown of body protein.

Early Fasting Stage: 2 -3 hrs § Carbohydrate, fat, and protein are all eventually used for energy § Begin with release of glucose from glycogen and fatty acids from adipose § Low blood glucose levels signal § Fat breakdown § Release of amino acids from muscles

Continued Fasting § Protein meets glucose needs via breakdown of body proteins (amino acids yielding pyruvate) for § Nervous system § Red blood cells § Shift to ketosis § Acetyl Co. A makes ketone bodies to fuel brain § Slows the rate of body protein breakdown § Keto acid production rises, lowering body p. H § Acidic blood denatures proteins
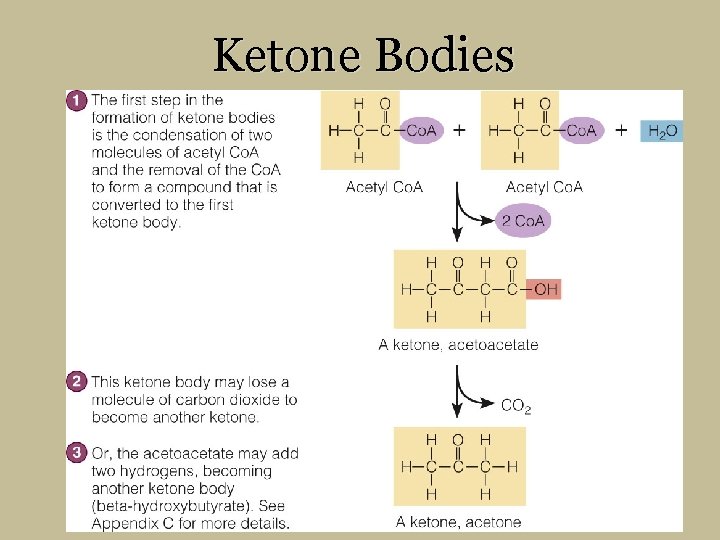
Ketone Bodies
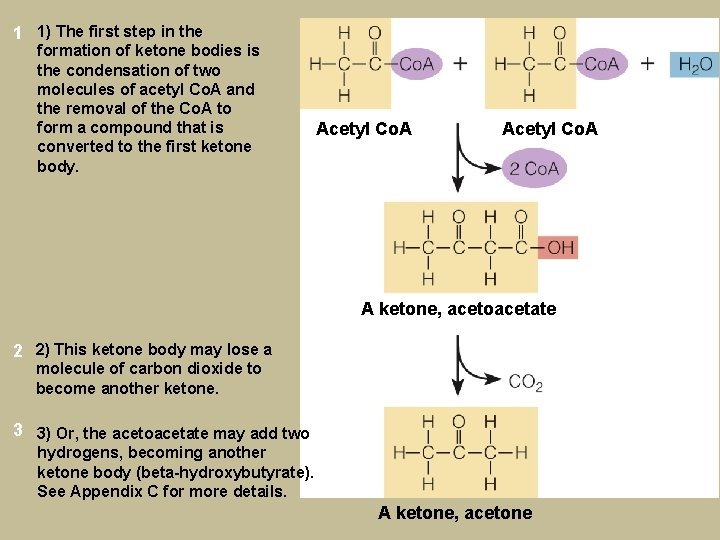
1 1) The first step in the formation of ketone bodies is the condensation of two molecules of acetyl Co. A and the removal of the Co. A to form a compound that is converted to the first ketone body. Acetyl Co. A A ketone, acetoacetate 2 2) This ketone body may lose a molecule of carbon dioxide to become another ketone. 3 3) Or, the acetoacetate may add two hydrogens, becoming another ketone body (beta-hydroxybutyrate). See Appendix C for more details. A ketone, acetone

Ketosis/ Very low-carb diets § Ketosis causes a loss of appetite § Slowing of metabolism § Hormones § Reduces energy output § Supports weight loss but not fat loss § Symptoms of starvation § Physical symptoms § Psychological symptoms

Low-Carbohydrate Diets § Metabolism similar to fasting § Uses glycogen and protein stores, body fluids + minerals first § Gluconeogenesis when glycogen is depleted § Body tissues used somewhat even when protein provided in diet § Ketogenic diet fat losses more quickly regained


Highlight 7 Alcohol and Nutrition
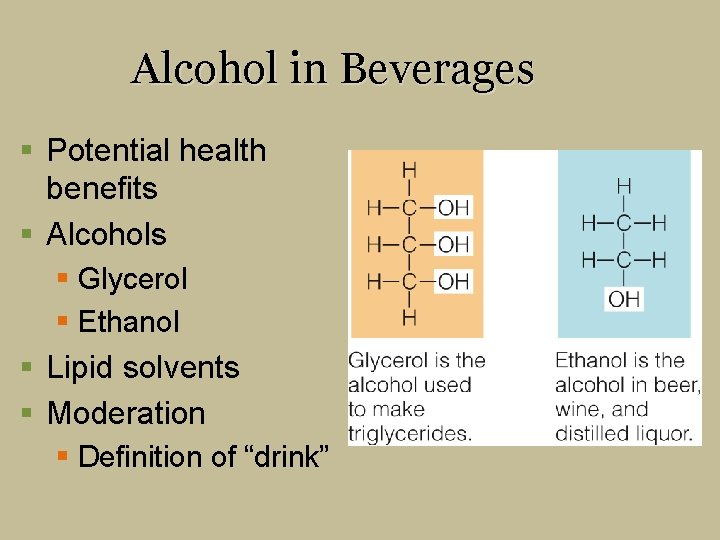
Alcohol in Beverages § Potential health benefits § Alcohols § Glycerol § Ethanol § Lipid solvents § Moderation § Definition of “drink”

Alcohol in the Body § Alcohol’s special privileges § No digestion § Quick absorption § Slowing absorption § Stomach § Alcohol dehydrogenase § Small intestine § Priority over nutrients

Alcohol Arrives in the Liver § Liver cells § First to receive alcohol-laden blood § Alcohol dehydrogenase § Disrupts liver activity § Can permanently change liver cell structure § Rate of alcohol metabolism § Acetaldehyde § Acetate
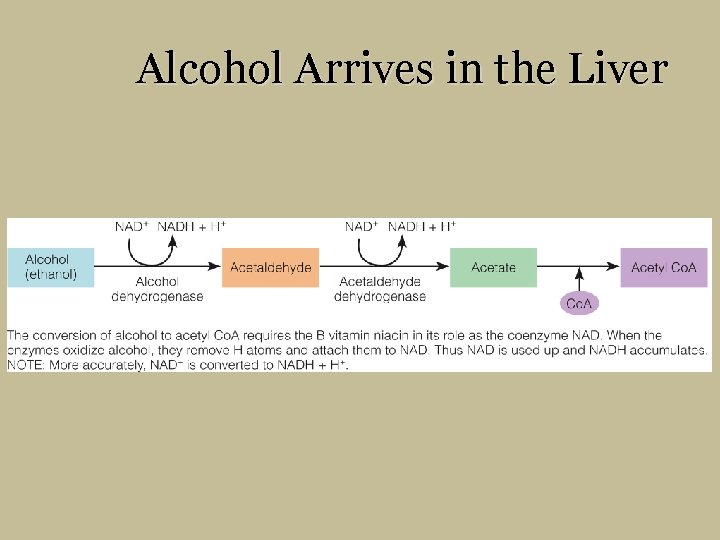
Alcohol Arrives in the Liver

Alcohol Disrupts the Liver § Nicotinamide adenine dinucleotide (NAD) § Glycolysis § TCA cycle § Electron transport chain § Development of fatty liver § Damage to central nervous system § Inflammation of joints § Amino acid and protein metabolism
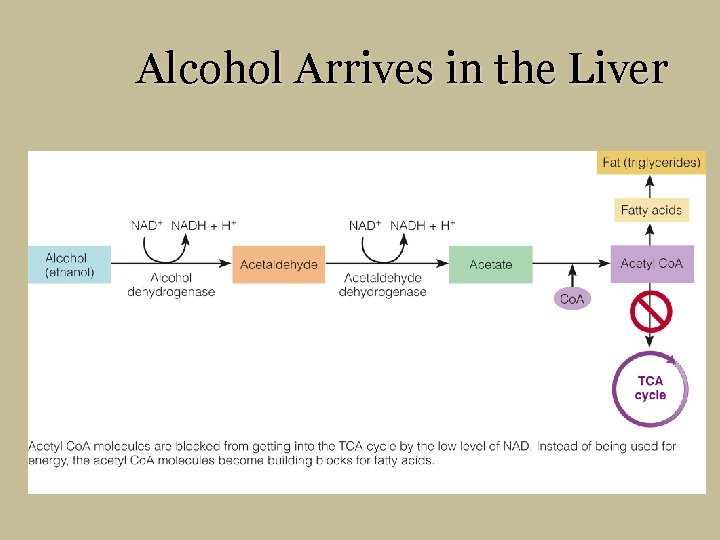
Alcohol Arrives in the Liver

Alcohol Arrives in the Liver § Immune system functioning § Alcohol interferes with drug metabolism § Microsomal ethanol-oxidizing system (MEOS)

Alcohol Arrives in the Brain § Sedates inhibitory nerves § Acts as central nervous system depressant § Blood alcohol levels and brain responses § Death of liver and brain cells § Depression of antidiuretic hormone (ADH) § Loss of body water § Loss of important minerals
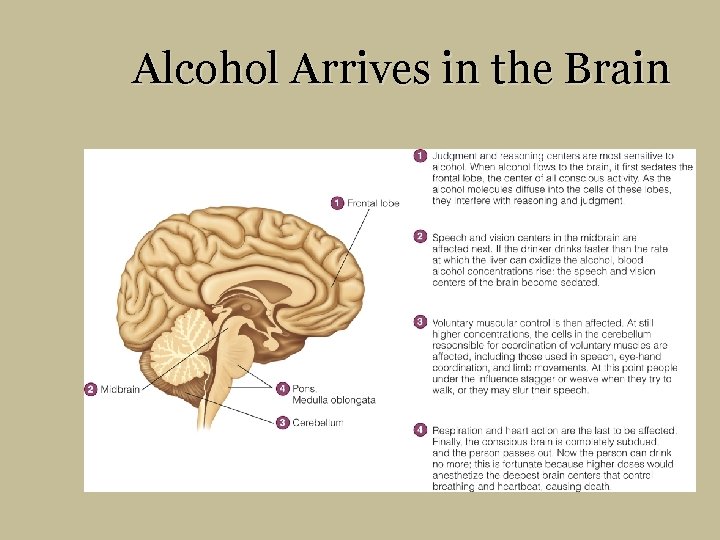
Alcohol Arrives in the Brain
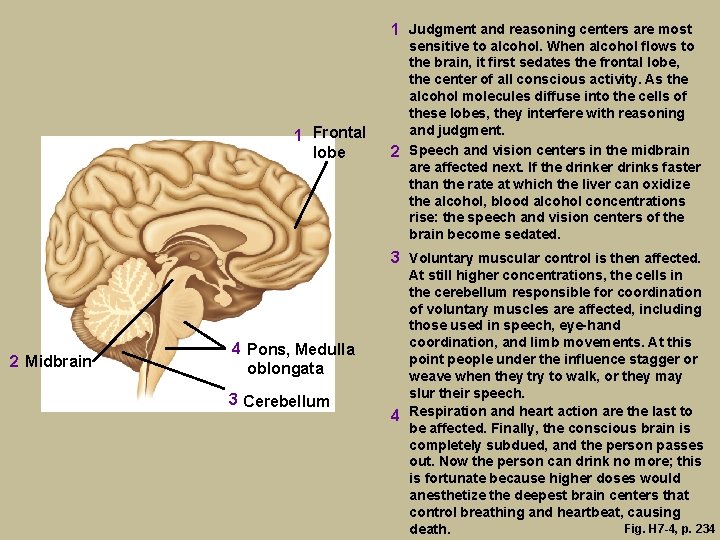
1 Judgment and reasoning centers are most 1 Frontal lobe sensitive to alcohol. When alcohol flows to the brain, it first sedates the frontal lobe, the center of all conscious activity. As the alcohol molecules diffuse into the cells of these lobes, they interfere with reasoning and judgment. 2 Speech and vision centers in the midbrain are affected next. If the drinker drinks faster than the rate at which the liver can oxidize the alcohol, blood alcohol concentrations rise: the speech and vision centers of the brain become sedated. 3 Voluntary muscular control is then affected. 2 Midbrain 4 Pons, Medulla oblongata 3 Cerebellum At still higher concentrations, the cells in the cerebellum responsible for coordination of voluntary muscles are affected, including those used in speech, eye-hand coordination, and limb movements. At this point people under the influence stagger or weave when they try to walk, or they may slur their speech. 4 Respiration and heart action are the last to be affected. Finally, the conscious brain is completely subdued, and the person passes out. Now the person can drink no more; this is fortunate because higher doses would anesthetize the deepest brain centers that control breathing and heartbeat, causing Fig. H 7 -4, p. 234 death.

Alcohol Arrives in the Brain

Alcohol and Malnutrition § Can contribute to body fat and weight gain § 1 ounce of alcohol represents 0. 5 ounce of fat § Central obesity § Substituted energy § 7 kcalories per gram § Nutrient displacement § B vitamins


Alcohol’s Effects § Short-term effects § Excessive drinking § Heavy drinking § Binge drinking § Long-term effects § Third leading preventable cause of death in U. S. § Sobering up
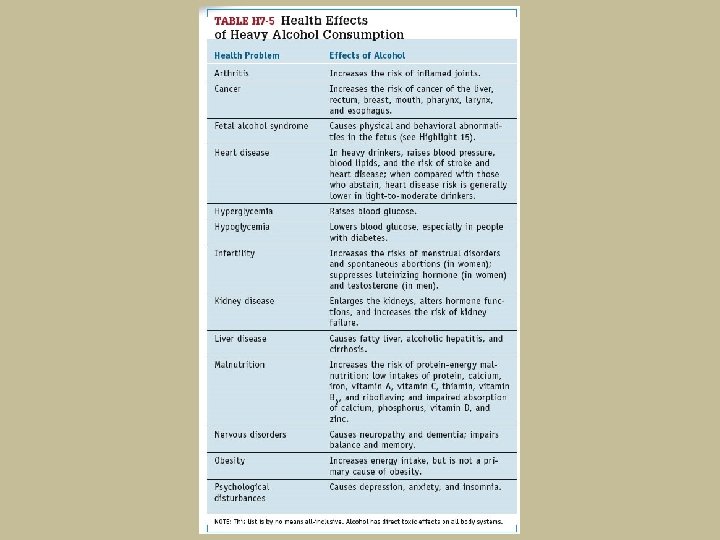
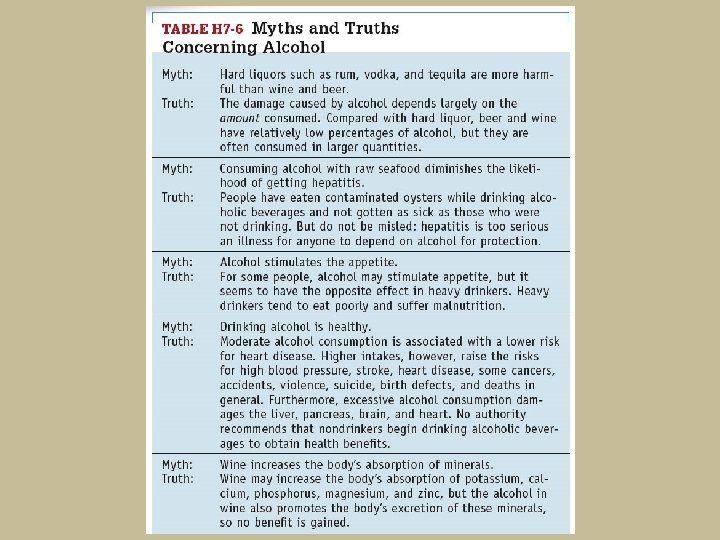
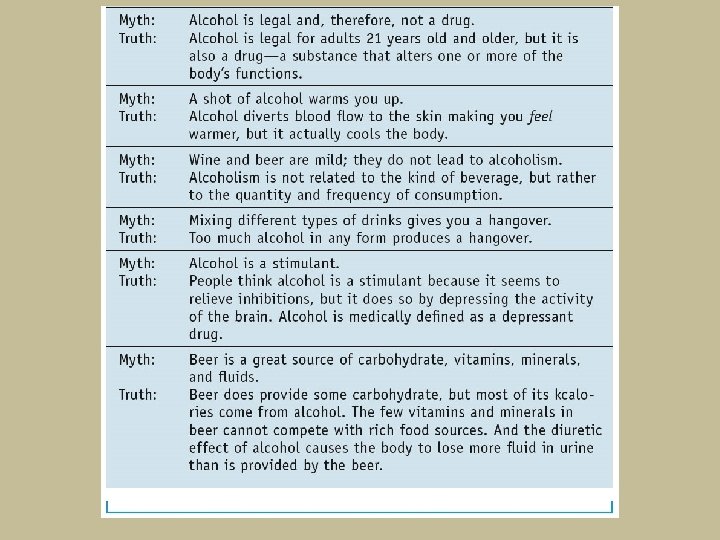
- Slides: 89