Metabolism of tryptophan An aromatic amino acids Indole

Metabolism of tryptophan • An aromatic amino acids • Indole ring containing amino acid • α-amino β- indole propionic acid • Neutral amino acid • Essential amino acids • Both glucogenic and ketogenic

Biologically important compound derived from tryptophan

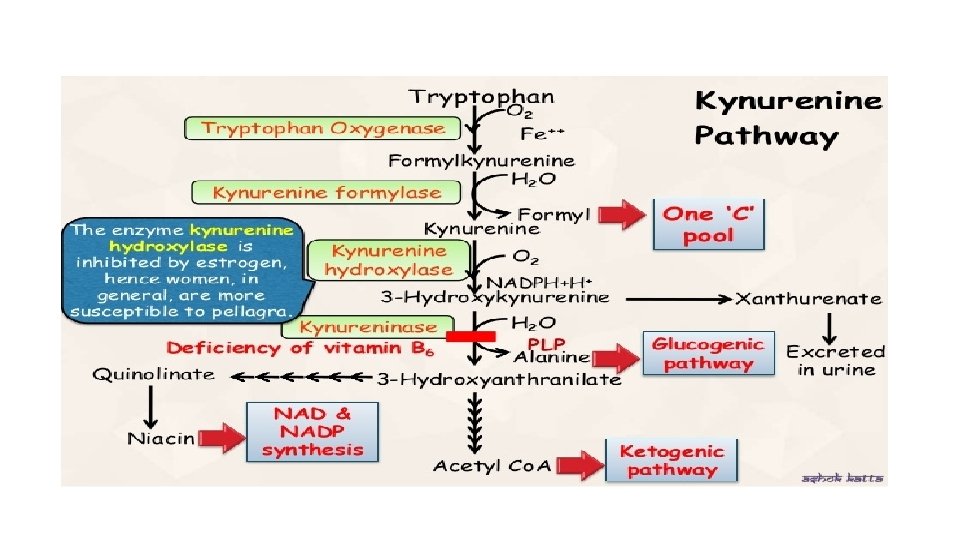
Metabolism of Tryptophan Metabolism is divided into • Kynurenine pathway • Serotonin pathway

Kynurenine pathway • Mainly takes place in liver • Through this pathway tryptophan is oxidized into : Alanine Acetyl Co. A Niacin Co-enzyme (NAD+, NADP+)

Serotonin pathway • Serotonin or 5 -hydroxytryptamine (5 HT) • It is a neurotransmitter • Normally about 1% of tryptophan is converted into serotonin and 99% are metabolized to form alanine , acetyl Co. A and niacin • Serotonin is widely distributed in the body • In mammals largest amount is synthesized in the intestinal cells
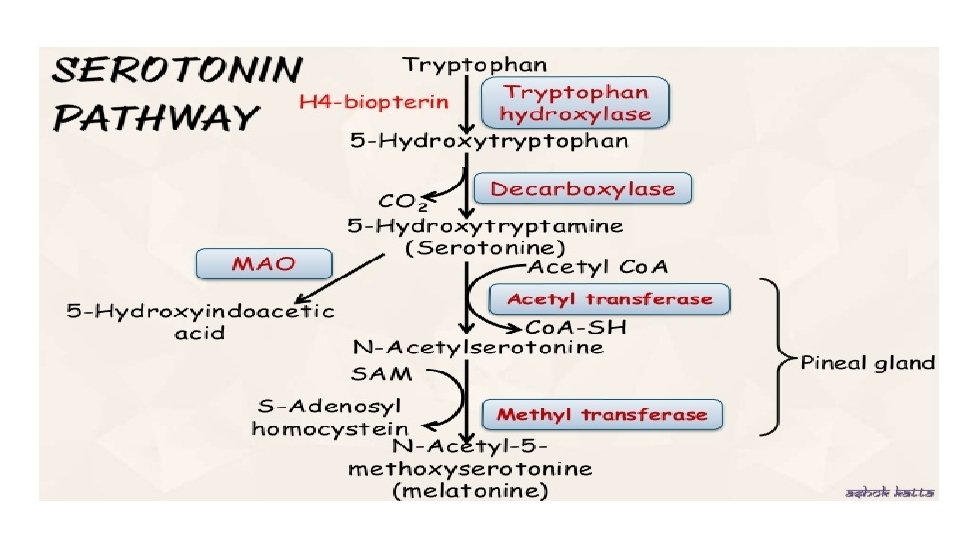


Functions of serotonin • Excitatory neurotransmitter in brain • It is closely involved in the regulation of cerebral activity • Potent vasoconstrictor and causes smooth muscle contractions in bronchioles and arterioles • Causes platelet aggregation • Causes peristalsis, peptide hormone release in GI tract • Serotonin controls sleep, behavioural pattern, blood pressure and body temperature

Carcinoid syndrome • Serotonin is produced by Argentaffin cells of GI tract • These cells undergo uncontrolled growth and lead to carcinoid tumour • Flushing, sweating, Bronchoconstriction, Diarrhoea, heart failure • Tryptophan to niacin conversion is decreased– pallegra like symptoms(60% tryptophan is channeled towards formation) • Metabolite of serotonin 5 HIAA in urine is increased

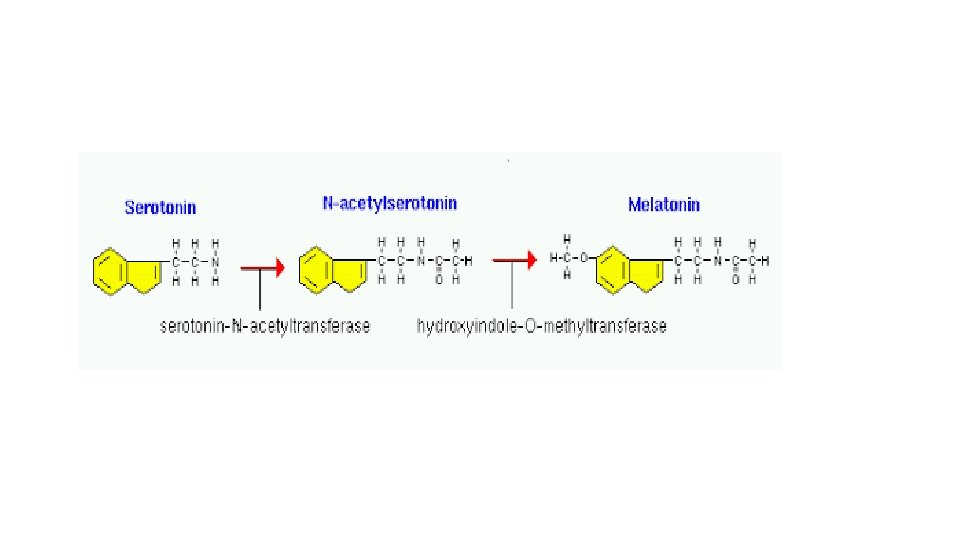
Melatonin • Produced by pineal gland • Synthesis is regulated by light dark cycle • Functions as neurotransmitter • Modulates circadian rhythm • Maintain sleep wake cycle • Regulates reproductive function

Hartnup’s disease • First described in family of Hartnup • Autosomal recessive disorder • Intestinal and renal transport of tryptophan and other neutral amino acids (Ala, Ser, Val, His) are decreased • The net outcome – low levels of these a. a. in blood and their excretion in urine is increased • Diagnosis –increased level in urine • Decreased plasma tryptophan level

Hartnup’s disease cont…… • Clinical symptoms include dermatitis, ataxia, mental retardation , neutral amino aciduria • Pellegra like and neurological symptoms due to tryptophan • Impairment in the synthesis of NAD+ and serotonin from tryptophan

Metabolism of sulphur containing amino acid • L-methionine –essential amino acid • L-Cysteine • L-cystine • Methionine , cysteine and cystine are the principal sources of Sulphur in the body • Methionine is essential a. a. • Cysteine can be synthesized in the body from methionine • Other sources of S in body is thiamine , lipoic acid and biotin
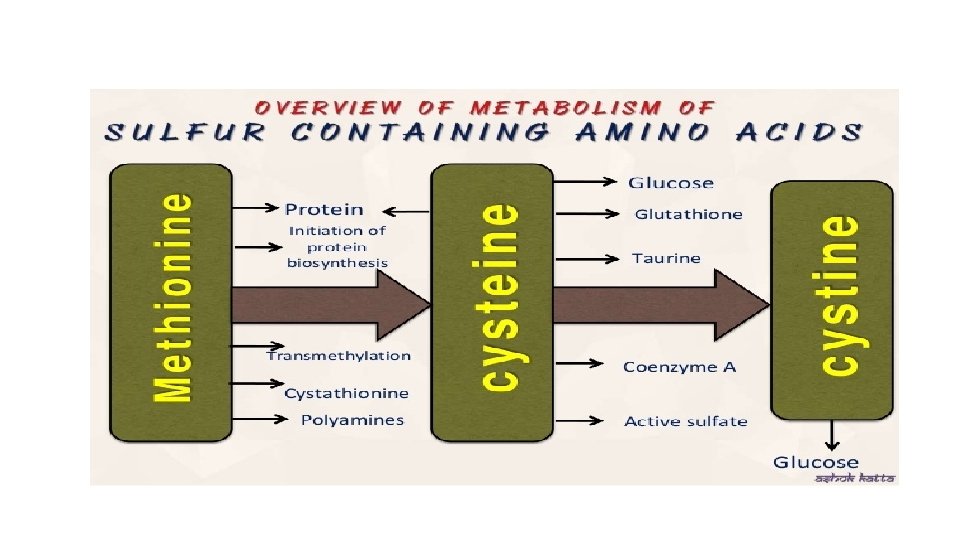

Compounds formed from methionine • Protein • Glucose • Cysteine • S-adenosyl methionine

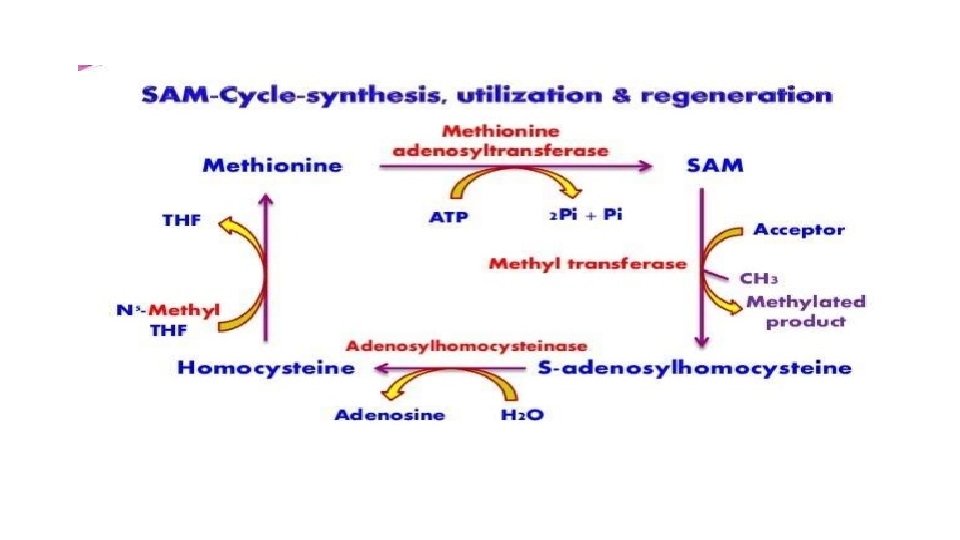
Fate of L-methionine • Stage 1 - Activation of methionine into S adenosyl methionine and its demethylation to form L-homocysteine • Stage 2 – Conversion of L-homocysteine to L-homoserine • Stage 3 – Degradation of homoserine to end products L-propionyl. Co. A and α-amino butyrate

Active methionine/adenylation of methionine • Chemically called as S-adenosyl methionine • Activation of methionine is in presence of ATP (catalyzed by an enzyme , called L-methionine adenosyl transferase -MAT) • Requires presence of Mg ion and G-SH • ATP donates the entire adenosine moiety to methionine and looses 3 molecules of phosphate • In methionine , the thio-ether linkage (C-S-C) is very stable but in SAM CH 3 group forms a high energy bond with sulphur and methyl group becomes labile and easily transferred to the acceptor
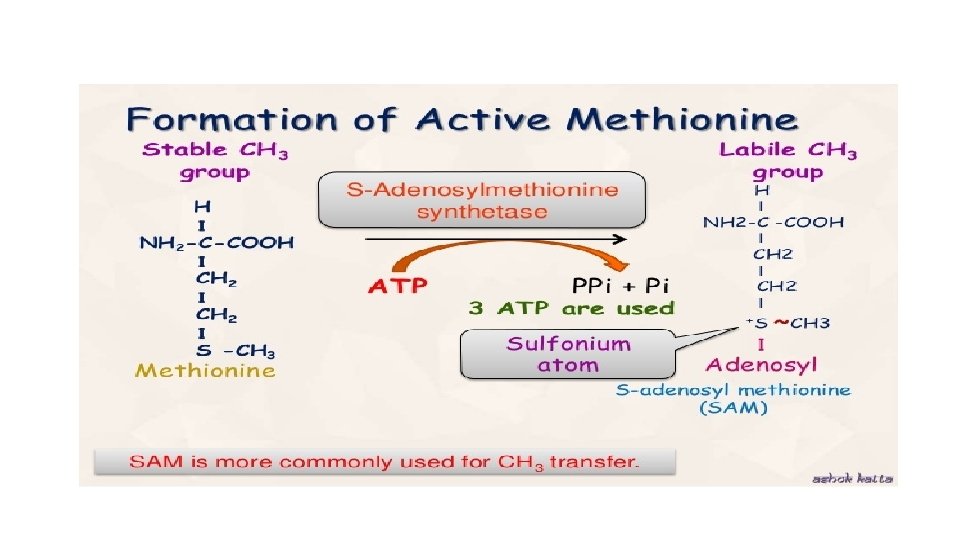

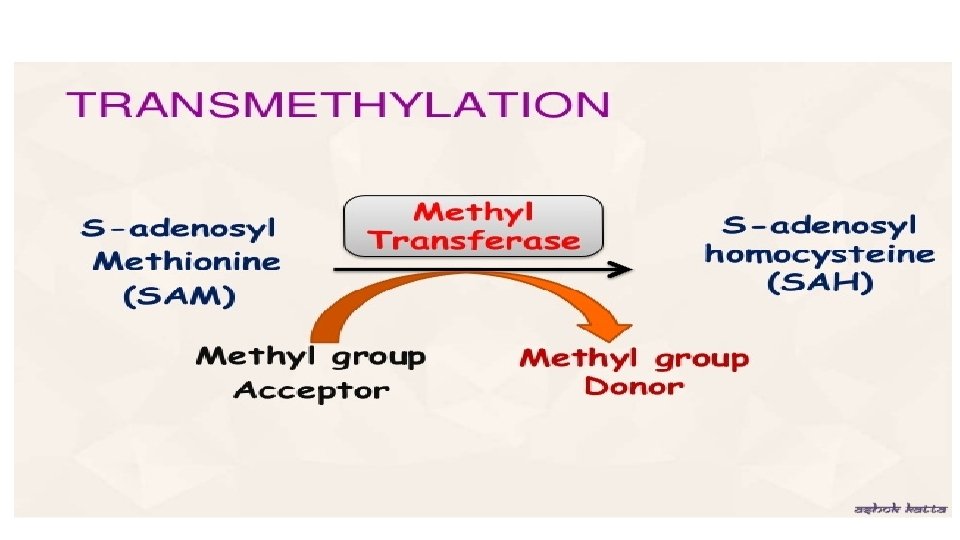
Transmethylation reaction Transfer of methyl group –CH 3 from active methionine to an acceptor is known as Transmethylation reaction In SAM due to presence of high energy bond , the methyl group is labile and may be transferred easily to other acceptors

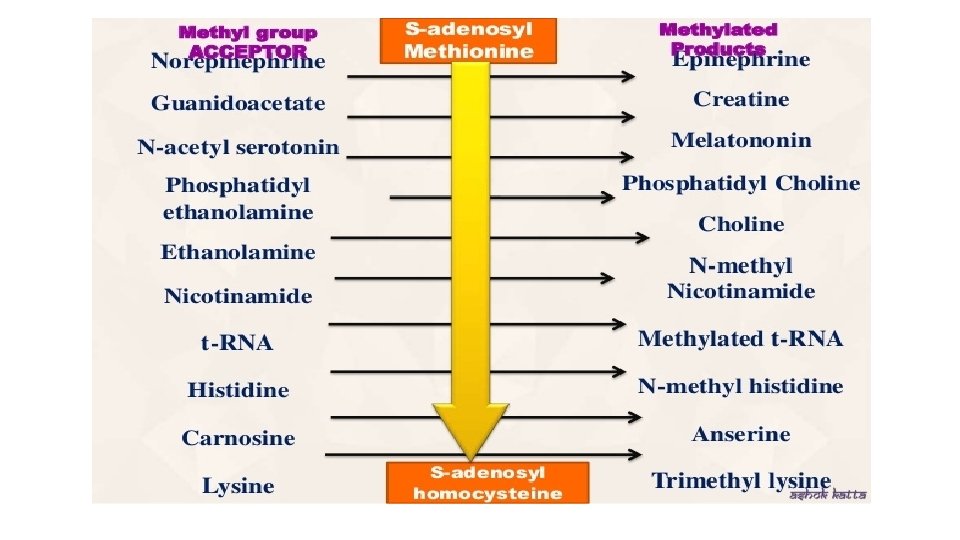
Importance of Transmethylation reaction • Many compounds in our body become functional or active only after methylation • Methylated protein is less degraded

Fate of homocysteine which is formed after transmethylation reaction • From S-adenosyl homocysteine , the adenosyl group is removed to form homocysteine (Homologous with cysteine ) • Homocysteine again converted into methionine by addition of methyl group donated from one carbon pool with the help of Vit B 12 • Homocysteine degradation – Homocysteine condenses with serine to form cystathionine (by enzyme cystathionine beta synthase in presence of Pyridoxal phosphate ) • Cystathionine is hydrolyzed to form cysteine and homoserine (enzyme –cystathionase ) • Homoserine is deaminated and then decarboxylated to propionyl. Co. A • Propionyl Co. A is changed into succinyl Co. A and enters TCA cycle
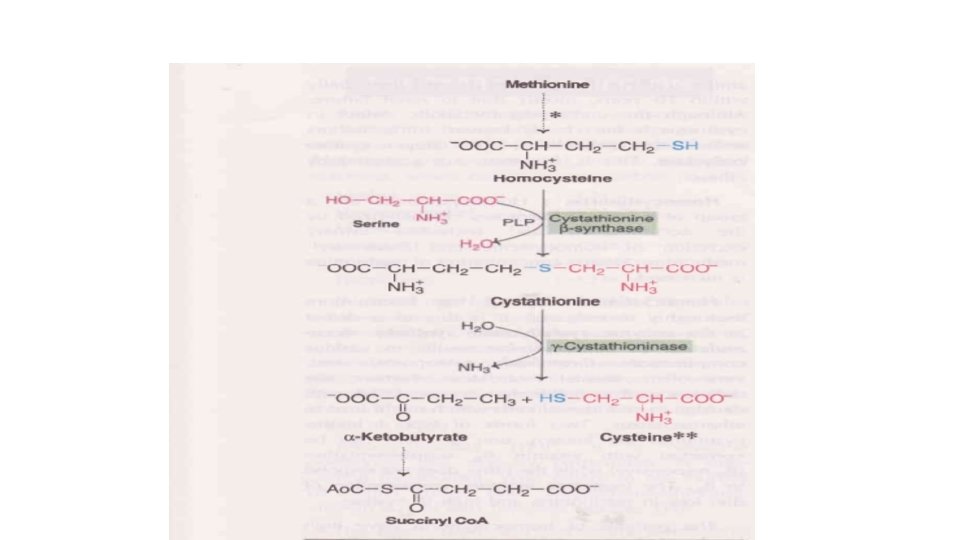

Inborn errors of metabolism of Sulfur containing amino acids • Cystinuria • Cystinosis • Homocystinuria type I • Homocystinuria III • Cystathionuria

- Slides: 29