Metabolism of Red Blood Cells RBCs DR SADIA

Metabolism of Red Blood Cells (RBCs) DR. SADIA HAROON LECTURE 1 BLOOD MODULE

BIOCHEMISTRY OF RBC METABOLISM

OBJECTIVES OF THE LECTURE 1 - Understanding the general structural & functional features of red blood cells (RBCs). 2 - Recognizing the main metabolic pathways occurring in RBCs with reference to their relations to functions of RBCs. 3 - Identifying some of the main & common diseases of RBCs as implication of defects of RBCs metabolism. 4 - Recognizing changes occurring in aging of RBCs.

INTRODUCTION TO THE RED BLOOD CELLS (RBCS) The red blood cells (RBCs) are not true cells. RBCs contain no nucleus or nucleic acids, and thus, can not reproduce. RBCs contain no cell organelles (as mitochondria, Golgi, ER or lysosomes) and thus possess no synthetic activities (no protein biosynthesis, no lipid synthesis & no carbohydrate synthesis). RBCs must be able to squeeze through some tight spots in microcirculation. For that RBCs must be easily & reversibly deformable

Importance of bi-concave shape are: 1. It can easily squeeze when it pass through capillary pores. 2. It allows considerable alteration of the cell volume without increasing the tension of the cell wall. 3. It can accommodate much fluid in hypotonic environment. 4. Hb remains distributed in a very thin layer. This facilitates quick saturation or desaturation with the gases.

Mature red cells do not have a nucleus, mitochondria, or endoplasmic reticulum, They have cytoplasmic enzymes that are capable of metabolizing glucose and forming small amounts of adenosine triphosphate. These enzymes also (1)maintain pliability of the cell membrane, (2)maintain membrane transport of ions, (3)keep the iron of the cell’s hemoglobin in the ferrous form rather than ferric form (4) prevent oxidation of the proteins in the red cells.

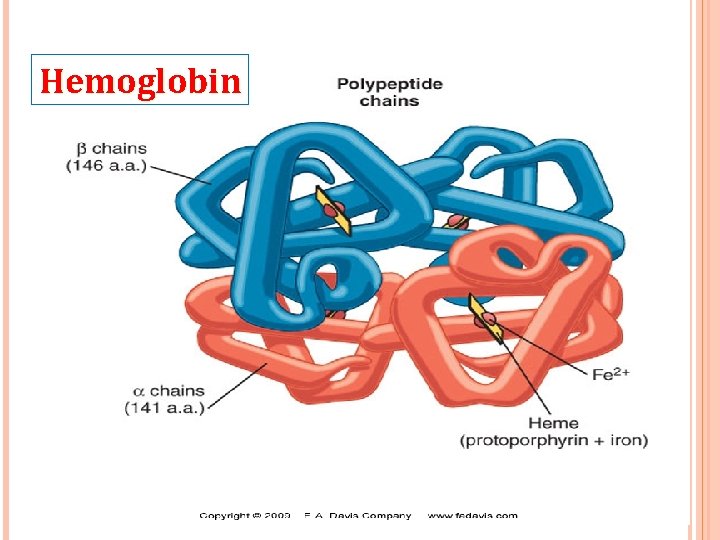
BIOCHEMICAL COMPOSITION OF THE RBCS Red cells contain 35 % solids. Hemoglobin, Hemoglobin the chief protein of the red cells. Other proteins are present in combination with lipids and oligosaccharide chains, forming the stroma and cell membrane. Potassium, Potassium magnesium, magnesium and zinc concentrations in red cells are much higher than in the plasma.
Hemoglobin

MAJOR FUNCTIONS OF THE RBC are small cell (6 -8μm in diameter) • Transport O 2 from lungs to the peripheral tissues. • Disposal of CO 2 and [H+]protons formed during tissue metabolism. • Carry CO 2, to lungs for elimination by exhalation.

FUNCTIONS OF RBCS RBCs have relatively simple functions as they have much simpler structure than most human cells. The major functions of RBCs are delivering oxygen to the tissues & disposal of carbon dioxide & protons formed by tissue metabolism. This function is carried out by hemoglobin

11

METABOLISM OF RBCS Introduction: Introduction RBCs contain no mitochondria, so there is no respiratory chain, no citric acid cycle, and no oxidation of fatty acids or ketone bodies. RBCs contain no cell organelles (as mitochondria, Golgi, ER or lysosomes) and thus possess no synthetic activities (no protein biosynthesis, no lipid synthesis & no carbohydrate synthesis). RBCs must be able to squeeze through some tight spots in micro-circulation. For that RBCs must be easily & reversibly deformable The RBC is highly dependent upon glucose as its energy source. Energy in the form of ATP is obtained ONLY from the glycolytic breakdown of glucose with the production of lactate (anaerobic glycolysis).

ATP produced being used for keeping the biconcave shape of RBCs & in the regulation of transport of ions & water in and out of RBCs. (Na+ –K+-ATPase and the anion exchange protein Energy in the form of ATP is obtained ONLY from the glycolytic breakdown of glucose with the production of lactate (anaerobic glycolysis). ATP produced being used for keeping the biconcave shape of RBCs & in the regulation of transport of ions & water in and out of RBCs. 13

METABOLISM OF RBCS (CONT. ) 1 - Glucose transport through RBC membrane: membrane Glucose is transported through RBC membrane glucose by a facilitated diffusion by glucose transporters (GLUT-1). GLUT-1 Glucose transporters (GLUT-1) are independent on insulin i. e. insulin does not promote glucose transport to RBCS It functions by generating a gated pore in the membrane to permit passage of glucose; Hemoglobin, the chief protein of the red cells. Other proteins are present in combination with lipids and oligosaccharide chains, forming the stroma and cell membrane.

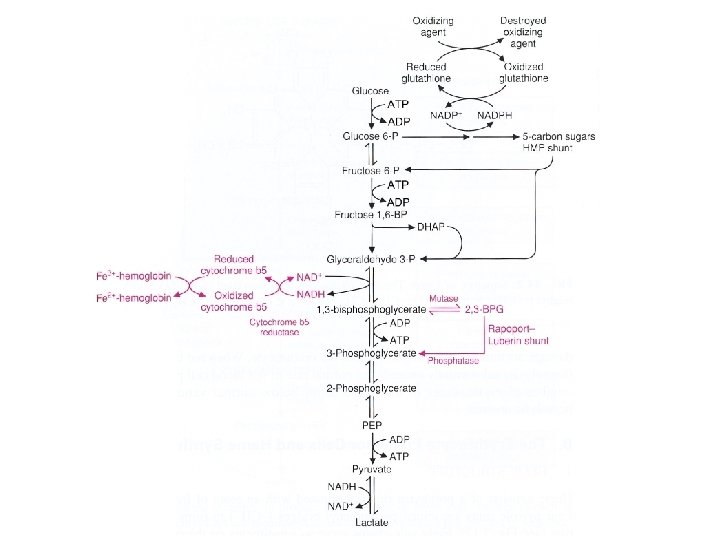
Erythrocyte metabolism Glucose as a source of energy Glycolysis generates ATP and 2, 3 -bisphoglycerate The pentose phosphate pathway produces NADPH Glutathione synthesis- the antioxidant defence system

Erythrocyte exceptions They lack organelles no ATP production in oxidative phosphorylation no ability to replace damaged lipids and proteins (low metabolic activities, with no ability to synthesize new proteins or lipids) Free radicals exposure haemoglobin autoxidation (O 2 • - release) a cell membrane rich in polyunsaturated fatty acids (susceptible to lipid peroxidation) deformation in tiny capillaries; catalytic ions leakage (cause of lipid peroxidation)

Hemoglobin autooxidation • • O 2 binds Fe 2+ - an intermediate structure - an electron is delocalized between the iron ion and the O 2 the side effect - every so often a molecule of oxyhaemoglobin undergoes decomposition and release superoxide Hem - Fe 2+- O 2 • Hem - Fe 3+ - O 2 • - 3% of the haemoglobin undergoes oxidation every day Methemoglobin (Fe 3+) is unable to bind O 2 (methaemoglobin reductase)

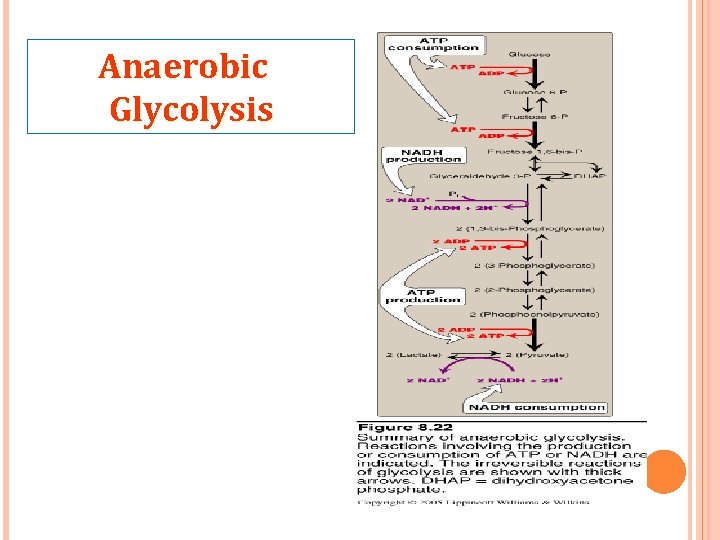
Metabolism of RBCs (cont. ) 2 - Glycolysis: Glucose is metabolized in RBCs through anaerobic glycolysis (that requires no mitochondria and no oxygen). One molecule of glucose yields 2 molecules of ATP by one anaerobic glycolytic pathway. In addition, 2 molecules of lactate are produced. Lactate is transported to blood & in the liver it is converted to glucose.
Anaerobic Glycolysis
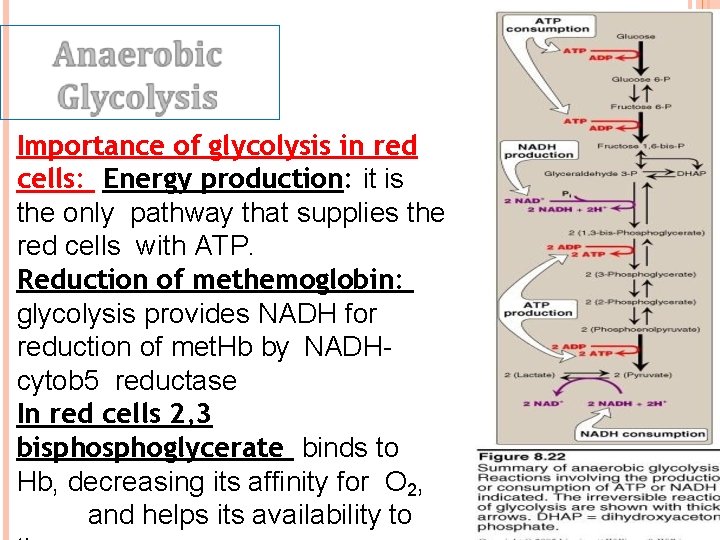
Importance of glycolysis in red cells: Energy production: it is the only pathway that supplies the red cells with ATP. Reduction of methemoglobin: glycolysis provides NADH for reduction of met. Hb by NADHcytob 5 reductase In red cells 2, 3 bisphoglycerate binds to Hb, decreasing its affinity for O 2, and helps its availability to
GLYCOLYSIS IN RED BLOOD CELLS 21
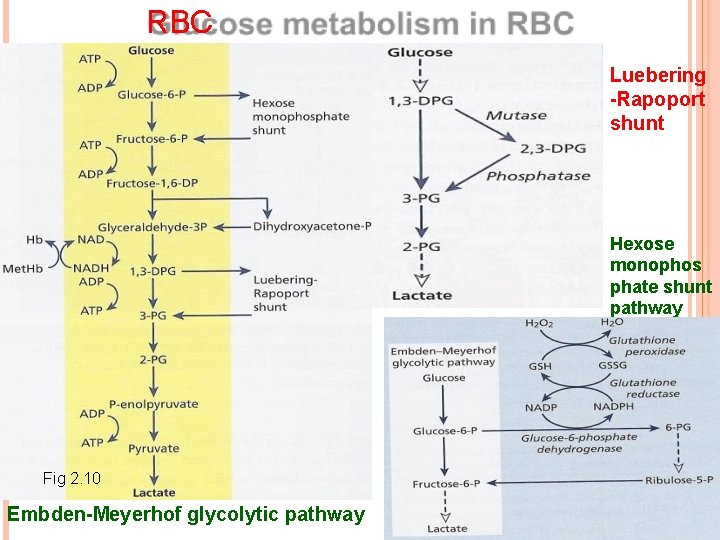
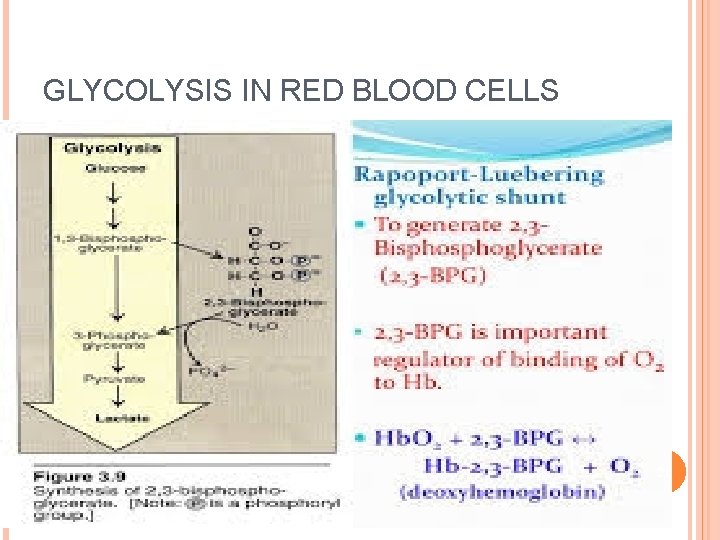
RBC Luebering -Rapoport shunt Hexose monophos phate shunt pathway Fig 2. 10 Embden-Meyerhof glycolytic pathway

ONT. ) 3. Production of 2, 3 bisphoglycerate (2, 3 -BPG): In RBCs, some of glycolysis pathways are modified so that 2, 3 bisphoglycerate is formed (by bisphoglycerate mutase). 2, 3 bisphoglycerate decreases affinity of Hb for So, it helps oxyhemoglobin to unload oxygen. O 2. Storing blood results in decrease of 2, 3 -BPG leading to high oxygen affinity Hb. This leads to oxygen trap. 6 -24 hours are needed to restore the depleted 2, 3 BPG. Maximum storage time for RBCs is

ROLE OF 2, 3 -BPG IN HAEMOGLOBIN FUNCTION ü ü ü 2, 3 BPG acts as an allosteric regulator of hemoglobin that has the ability to decrease the affinity of O 2 to hemoglobin, . When a hemoglobin molecule is O 2 deficient, 2, 3 BPG inserts itself between the two beta chains, where it contains positively charged amino acids that form salt bridges with the negatively charged phosphate groups of 2, 3 -BPG. The lower affinity for O 2 means that the hemoglobin’s delivery of O 2 to the tissues is enhanced when needed.

For mothers to deliver oxygen to a fetus, it is necessary for the fetal hemoglobin to extract oxygen from the maternal oxygenated hemoglobin across the placenta. This requires the fetal hemoglobin to have a higher oxygen affinity than that of the maternal carrier. This is achieved by a fetal hemoglobin subunit γ (gamma), instead of the ß (beta) subunit. The γ subunit has fewer positive charges than the adult hemoglobin ß subunit. This means that 2, 3 -BPG is less electrostatically bound to fetal hemoglobin as compared to adult hemoglobin and therefore less effective in lowering the oxygen affinity of the fetal hemoglobin. This lowered affinity allows for adult hemoglobin (the maternal hemoglobin) to readily transfer its oxygen to the fetal hemoglobin.

2, 3 -bisphoglycerate Allosteric effector of haemoglobin: binds to deoxyhaemoglobin (a central cavity capable of binding 2, 3 -BPG) decreases haemoglobin‘s O 2 affinity Clinical aspects: In people with high-altitude adaptation or smokers the concentration of 2, 3 -BPG in the blood is increased (low oxygen supply) Fetal haemoglobin has low BPG affinity - the higher O 2 affinity - facilitates the transfer of O 2 to the fetus via the placenta

METABOLISM OF RBCS (CONT. ) 3 - Production of 2, 3 bisphoglycerate (2, 3 BPG): BPG) In RBCs, some of glycolysis pathways are modified so that 2, 3 bisphoglycerate is formed (by bisphoglycerate mutase). 2, 3 bisphoglycerate decreases affinity of HB for oxygen. So, it helps oxyhemoglobin to unload oxygen. Storing blood results in decrease of 2, 3 -BPG leading to high oxygen affinity Hb. This leads to oxygen trap. 6 -24 hours are needed to restore the depleted 2, 3 BPG Maximum storage time for RBCs is 21 -42 days
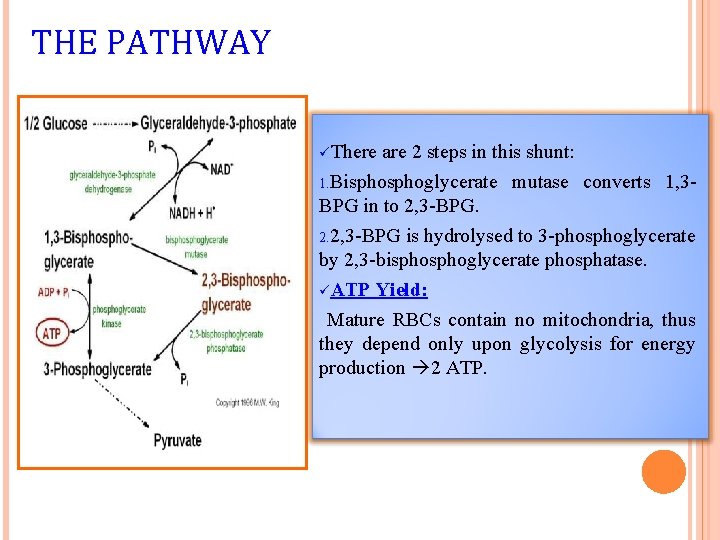
THE PATHWAY üThere are 2 steps in this shunt: 1. Bisphoglycerate mutase converts 1, 3 BPG in to 2, 3 -BPG. 2. 2, 3 -BPG is hydrolysed to 3 -phosphoglycerate by 2, 3 -bisphoglycerate phosphatase. üATP Yield: Mature RBCs contain no mitochondria, thus they depend only upon glycolysis for energy production 2 ATP.

METABOLISM OF RBCS (CONT. ) Genetic defects in enzymes of glycolysis: Genetic defects of one of the enzymes of glycolysis in RBCs results in a reduced rate of glycolysis in RBCs & by this way will deprive RBCs of the only means for producing energy. As a result, hemolytic anemia will be a consequence as RBCs will not be able to keep the biconcave flexible shape which allows it to squeeze through narrow capillaries with an end result of hemolysis (destruction of RBCs). 95% of cases of genetic defects in glycolytic enzymes is caused by pyruvate kinase deficiency 4% is caused by phosphoglucose isomerase deficiency

METABOLISM OF RBCS (CONT. ) 4 - Pentose phosphate pathway: RBCs contain an active pentose phosphate pathway (PPP) for glucose that supplies NADPH (PPP is the only source for NADPH in RBCs) NADPH is important in keeping glutathione in the reduced glutathione. Reduced glutathione plays a very important role in the survival of the red blood cells. (prevents oxidation of membrane) Glucose 6 - phosphate dehydrogenase deficiency (G 6 PD Deficiency): Deficiency Glucose 6 -phosphate dehydrogenase is the first enzyme of pentose phosphate pathway & its deficiency leads to reduced production of NADPH ending in acute hemolytic anemia.
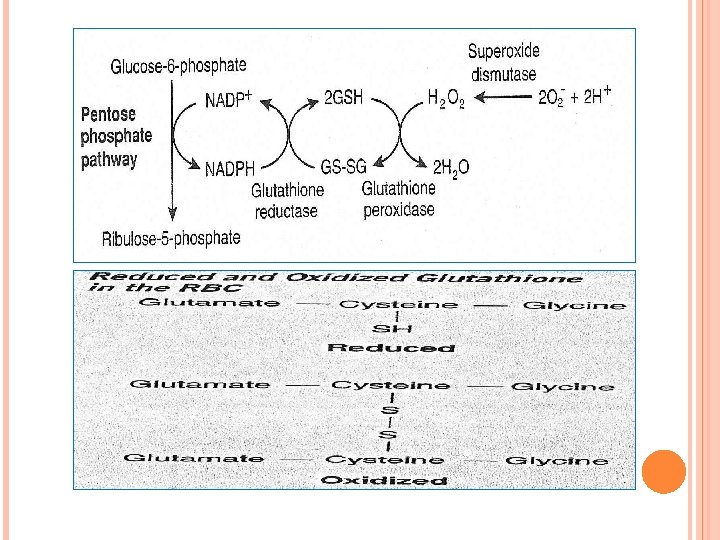
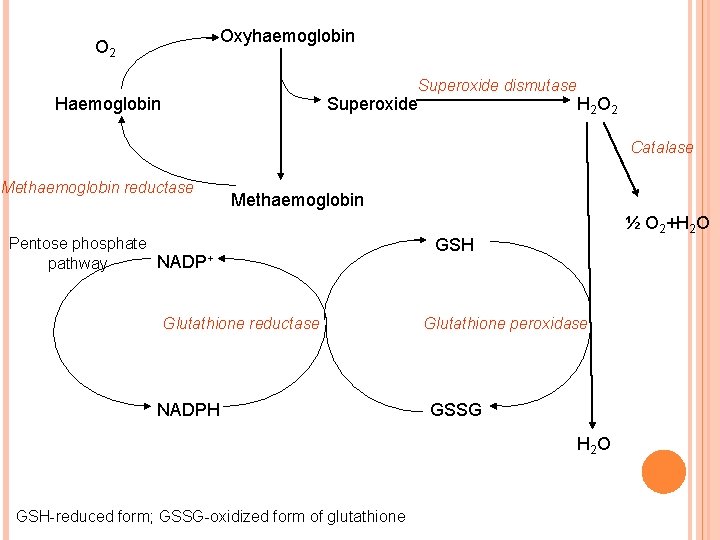
Oxyhaemoglobin O 2 Superoxide dismutase Haemoglobin Superoxide H 2 O 2 Catalase Methaemoglobin reductase Methaemoglobin Pentose phosphate NADP+ pathway Glutathione reductase NADPH ½ O 2+H 2 O GSH Glutathione peroxidase GSSG H 2 O GSH-reduced form; GSSG-oxidized form of glutathione

METABOLISM OF RBCS (CONT. ) ) • RBCs contain an active pentose phosphate pathway (PPP) for glucose that supplies • NADPH is important in keeping glutathione in the reduced glutathione. • Reduced glutathione plays a very important role in the survival of the red blood cells. (prevents oxidation of membrane)

The pentose phosphate pathway in erythrocytes Generates NADPH - reduction of glutathione (eliminates H 2 O 2 formed in erythrocytes) Clinical apect: Glucose-6 -phosphate dehydrogenase deficiency Causes hemolytic anemia (decreased production of NADPH - reduced protection against oxidative stress - haemoglobin oxidation and Heinz bodies formation, membrane lipid peroxidation and hemolysis) Hemolytic crises are evocated by drugs (primaquine, sulphonamide antibiotics) and foods (broad beans) The most common enzyme deficiency disease in the world (100 million people)

Metabolism of RBCs (cont. ) The erythrocytes contain carbonic anhydrase Carbon dioxide combines with water only after it enters the red cells where hemoglobin, the most important buffer for the resulting carbonic acid, is present. CO 2 + H 2 O HCO 3 - + H+ The red cell also contain rhodanese enzyme responsible for the detoxication of cyanides.
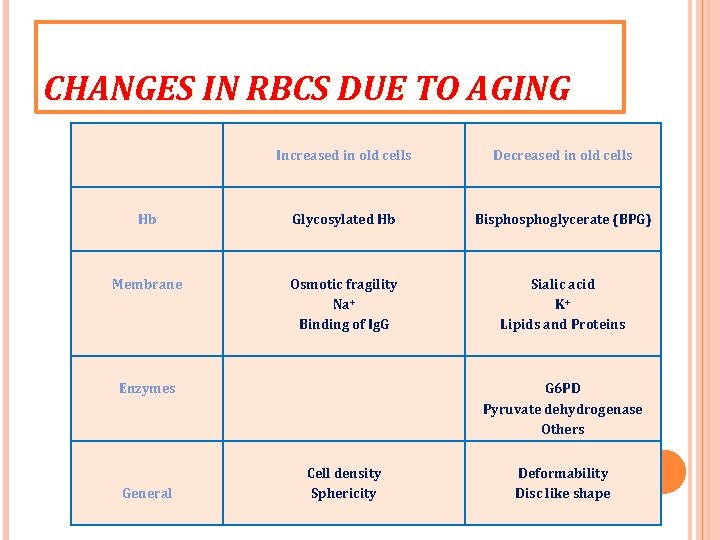
CHANGES IN RBCS DUE TO AGING Increased in old cells Decreased in old cells Hb Glycosylated Hb Bisphoglycerate (BPG) Membrane Osmotic fragility Na+ Binding of Ig. G Sialic acid K+ Lipids and Proteins Enzymes General G 6 PD Pyruvate dehydrogenase Others Cell density Sphericity Deformability Disc like shape

GLUCOSE 6 -P DEHYDROGENASE DEFICIENCY Diminished G 6 PD activity impairs the ability of the cell to form the NADPH that is essential for the maintenance of the reduced glutathione pool. This results in a decrease in the cellular detoxification of free radicals and peroxides formed within the cell
GLUCOSE 6 - PHOSPHATE DEHYDROGENASE DEFICIENCY (G 6 PD DEFICIENCY): • Glucose 6 -phosphate dehydrogenase is the first enzyme of pentose phosphate pathway & its deficiency leads to reduced production of NADPH ending in acute hemolytic anemia.

PRECIPITATING FACTORS IN G 6 PD DEFICIENCY Oxidant drugs (Antibiotics (for example, sulfamethoxazole and chloramphenicol), Antimalarials (for example, primaquine but not quinine), and Antipyretics (for example, acetanilid but not acetaminophen. ( Favism Infection Neonatal jaundice

Glutathione also helps maintain the reduced states of sulfhydryl groups in proteins, including hemoglobin. Oxidation of those sulfhydryl groups leads to the formation of denatured proteins that form insoluble masses (called Heinz bodies) that attach to the red cell membranes
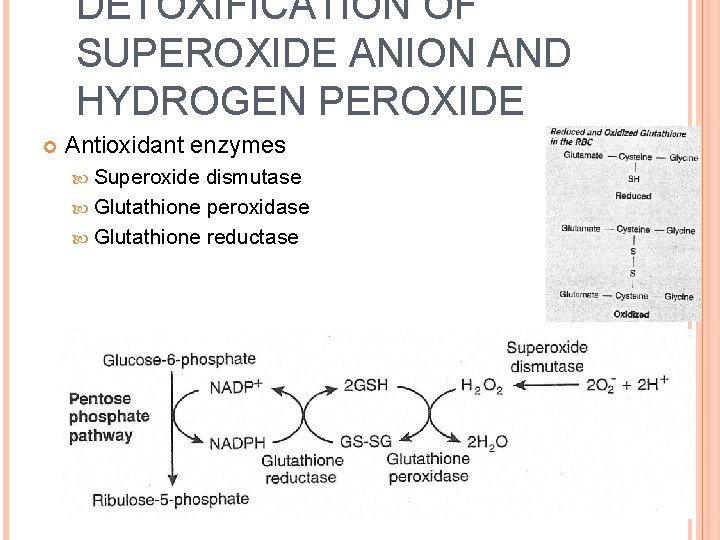
DETOXIFICATION OF SUPEROXIDE ANION AND HYDROGEN PEROXIDE Antioxidant enzymes Superoxide dismutase Glutathione peroxidase Glutathione reductase

FUNCTIONS OF REDUCED GLUTATHIONE 43

ASSIGNMENTS Pyruvate kinase deficiency Glucose , 6 phosphate dehydrogenase deficiency Role of glycolysis and pentose phosphate pathway in red blood cells
- Slides: 44