Metabolism of Lipids I HP Kedilaya Biological importance

Metabolism of Lipids - I HP Kedilaya

Biological importance • Lipids are unique hydrophobic nature. • partly obtained from foods and partly derived from carbohydrates. • Fats are stored in adipocytes as highly concentrated sources of energy and oxidized for energy production.

Biological importance • The layer of fat as adipose tissue under the skin acts as shock absorbing cushions and also thermoinsulation against heat loss

Biological importance • Glycerophospholipids, sphingolipids and cholesterol are indispensible for cell structure and function. • Vitamin D, bile salts and steroid hormones are synthesized from cholesterol. • Prostaglandins, leukotrienes, thromboxanes and related compounds, synthesized from C 20 fatty acids possess many biological actions.
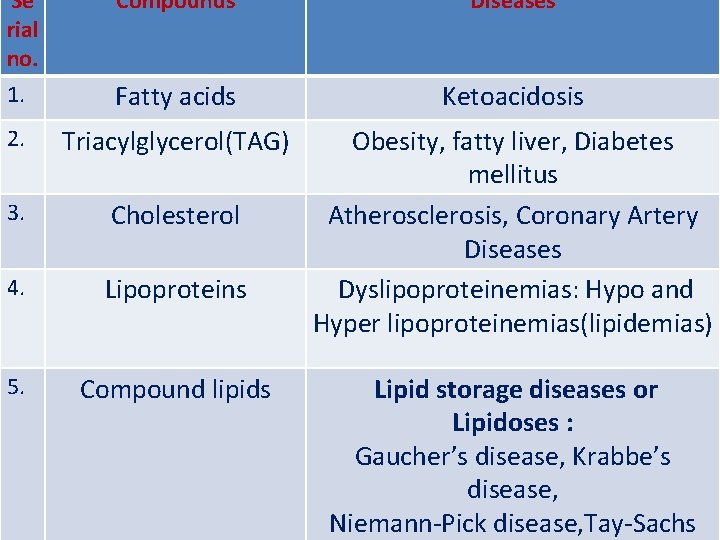
Se Compounds rial no. 1. Fatty acids 2. Triacylglycerol(TAG) 3. Cholesterol 4. Lipoproteins 5. Compound lipids Diseases Ketoacidosis Obesity, fatty liver, Diabetes mellitus Atherosclerosis, Coronary Artery Diseases Dyslipoproteinemias: Hypo and Hyper lipoproteinemias(lipidemias) Lipid storage diseases or Lipidoses : Gaucher’s disease, Krabbe’s disease, Niemann-Pick disease, Tay-Sachs

Contents: • Metabolism of fatty acids Fatty acid oxidation and Fatty acid synthesis • Metabolism of Ketone bodies Ketogenesis and Ketolysis • Metabolism of Triacylglycerol • Metabolism of Compound lipids • Metabolism of Cholesterol • Metabolism of lipoproteins • Fatty liver & lipotropic factors
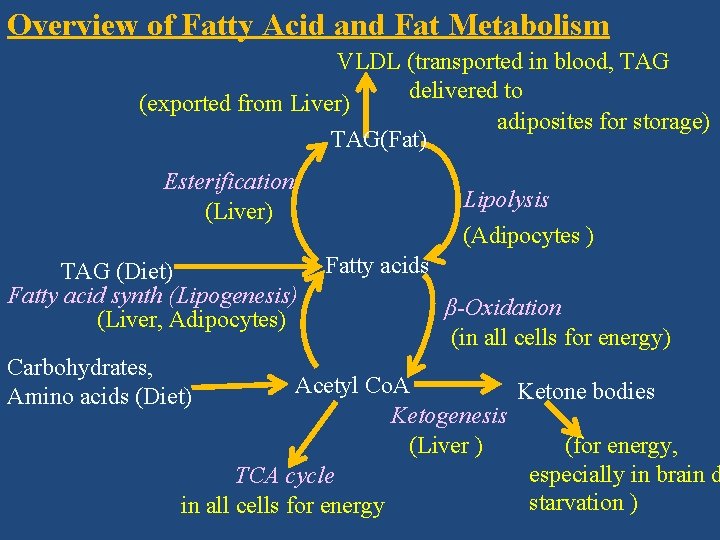
Overview of Fatty Acid and Fat Metabolism VLDL (transported in blood, TAG delivered to (exported from Liver) adiposites for storage) TAG(Fat) Esterification (Liver) TAG (Diet) Fatty acid synth (Lipogenesis) (Liver, Adipocytes) Carbohydrates, Amino acids (Diet) Lipolysis (Adipocytes ) Fatty acids β-Oxidation (in all cells for energy) Acetyl Co. A Ketone bodies Ketogenesis (Liver ) (for energy, especially in brain d TCA cycle starvation ) in all cells for energy

Metabolism of Fatty Acids- includes – • Fatty Acid Oxidation (breakdown for energy) • Fatty Acid Synthesis (Lipogenesis) – De Novo synthesis of fatty acids. (‘De Novo synthesis’ refers to synthesis of complex/large molecules from simple, small molecules. ) and • Ket one body metabolism

Fatty acid oxidation • Fatty acids are oxidized in the body for energy mostly by β-oxidation. • Other forms of oxidation, of minor importance, are • α-oxidation and • ω-oxidation.

β-Oxidation of Fatty Acids • Definition β-oxidation of fatty acids is the oxidation on the β-carbon atom of the fatty acid molecule and it occurs for the energy requirement of the cell during the fasting state. • Tissues: Most of the tissues except brain, RBC and adrenal medulla • Starting substrate: Fatty acid (Palmitic acid as example) • Pathway/Reactions of β-Oxidation

Pathway/Reactions of β-Oxidation consists of the following three stages 1. I. Activation of fatty acids (in cytosol) II. Transport of fatty acids into mitochondria III. β-Oxidation proper (in the mitochondrial matrix)
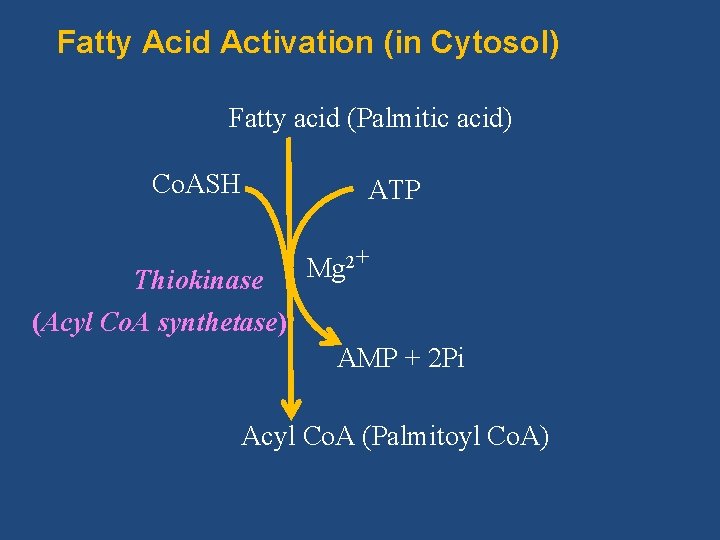
Fatty Acid Activation (in Cytosol) Fatty acid (Palmitic acid) Co. ASH ATP Thiokinase (Acyl Co. A synthetase) Mg 2+ AMP + 2 Pi Acyl Co. A (Palmitoyl Co. A)

Transport of Fatty Acyl Co. A Into Mitochondria (Role of Carnitine in Oxidation of Fatty Acids) • Long chain fatty acids cannot pass through the inner mitochondrial membrane. Hence, this needs a transport mechanism involving, • carnitine, Carnitine is β-hydroxy γ-trimethyl ammonium butyrate. (CH 3)3 N+–CH 2–CH(OH)–CH 2–COO– and three protein molecules present in the mitochondrial membranes – • translocase, • carnitine acyl transferase (CAT I) and • carnitine acyl transferase (CAT II).
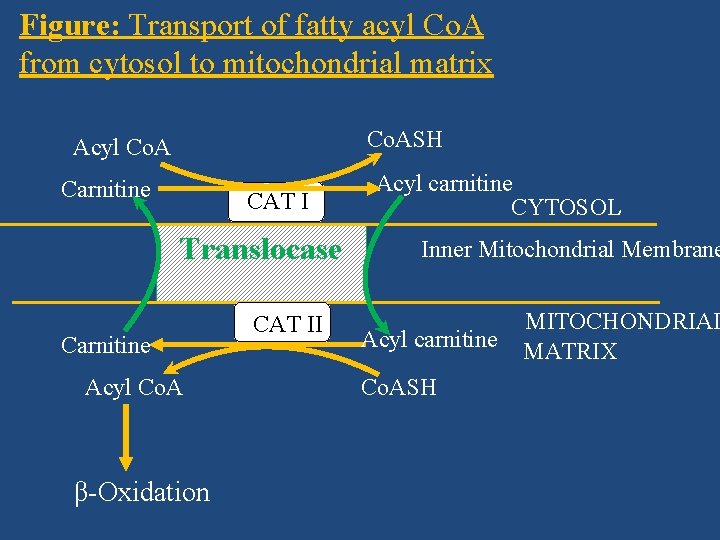
Figure: Transport of fatty acyl Co. A from cytosol to mitochondrial matrix Co. ASH Acyl Co. A Carnitine CAT I Translocase Carnitine Acyl Co. A β-Oxidation CAT II Acyl carnitine CYTOSOL Inner Mitochondrial Membrane Acyl carnitine Co. ASH MITOCHONDRIAL MATRIX

III. β-Oxidation Proper (Palmitic acid/Palmitoyl Co. A as example) Subcellular site: Mitochondrial matrix Starting substrate: Acyl Co. A/Palmitoyl Co. A During β-oxidation, fatty acids undergo oxidative removal of successive two-carbon units as acetyl Co. A, starting from carboxyl end of fatty acyl chain. •
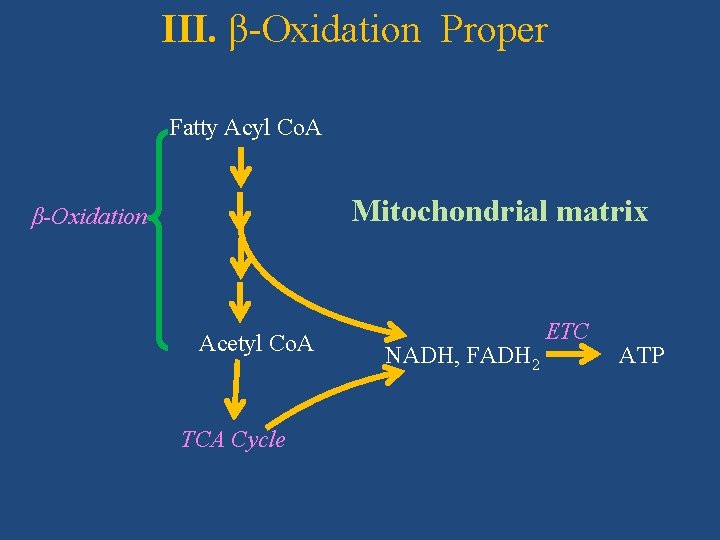
III. β-Oxidation Proper Fatty Acyl Co. A Mitochondrial matrix β-Oxidation Acetyl Co. A TCA Cycle NADH, FADH 2 ETC ATP

III. β-Oxidation Proper Each cycle of β-oxidation, accompanied by removal of a two-carbon unit as acetyl Co. A, has four steps/reactions in the following order. • Dehydrogenation • Hydration • Dehydrogenation • Thiolytic cleavage

III. β-Oxidation Proper • Palmitic acid (C 16) requires 7 cycles of β-oxidation, each cycle producing one molecule each of acetyl Co. A, NADH and FADH 2.
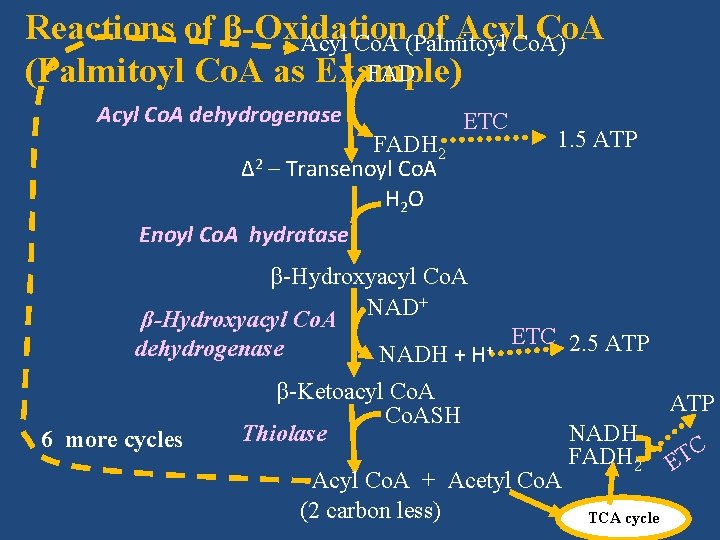
Reactions of β-Oxidation of Acyl. Co. A) Co. A Acyl Co. A (Palmitoyl FAD (Palmitoyl Co. A as Example) Acyl Co. A dehydrogenase FADH 2 Δ 2 – Transenoyl Co. A H 2 O Enoyl Co. A hydratase ETC β-Hydroxyacyl Co. A + NAD β-Hydroxyacyl Co. A dehydrogenase 6 more cycles NADH + H+ 1. 5 ATP ETC 2. 5 ATP β-Ketoacyl Co. ASH Thiolase Acyl Co. A + Acetyl Co. A (2 carbon less) ATP NADH FADH 2 TCA cycle C T E

III. β-Oxidation Proper • Overall reaction of β-oxidation of palmitoyl Co. A • Palmitoyl Co. A+7 Co. ASH+ 7 FAD+7 NAD++7 H 2 O • 8 Acetyl Co. A+ 7 FADH 2 +7 NADH + 7 H+

Energetics of β-Oxidation of Palmitic acid • Mechanism ATP Yield • I. β-Oxidation proper (7 cycles): • 7 NADH (by oxidative phosphorylation, • each NADH gives 2. 5 ATP) 17. 5 • 7 FADH 2 (by oxidative phosphorylation, • each FADH 2 gives 1. 5 ATP) 10. 5 • II. From 8 acetyl Co. A oxidized by TCA Cycle, • Each acetyl Co. A provides 10 ATP 80 • Total energy from one mole of palmitoyl Co. A 108 • Energy utilized for activation (formation of palmitoyl Co. A) – 2 Net ATP yield of β-oxidation of one molecule of palmitate = 106 •

Regulation of β-Oxidation • by availability of the substrate, namely fatty acids. • During fasting state rate of β-oxidation is increased due to high level of plasma free fatty acid, which is released from adipose tissue because of low insulin/glucagon ratio found during fasting. (See chapter ‘Lipolysis in Adipose Tissue’)

Regulation of β Oxidation • by availability of the substrate, namely fatty acids. • During fed state rate of β-oxidation is decreased as there is an increased level of malonyl Co. A, a substrate of fatty acid synthesis. CAT I is inhibited by malonyl Co. A decreasing the entry of fatty acids into mitochondrial matrix.

Regulation of β Oxidation fasting state ↓insulin/glucagon ratio ↑Lipolysis adipose tissue ↑plasma free fatty acid ↑β-Oxidation
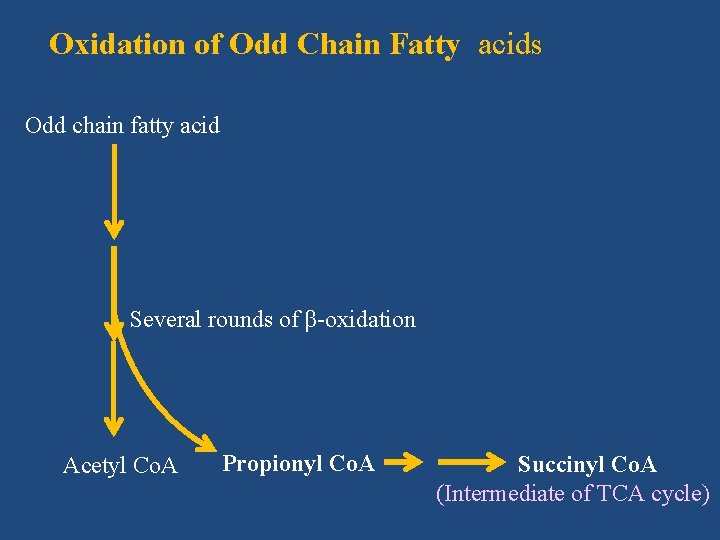
Oxidation of Odd Chain Fatty acids Odd chain fatty acid Several rounds of β-oxidation Acetyl Co. A Propionyl Co. A Succinyl Co. A (Intermediate of TCA cycle)

Fatty Acid Biosynthesis (De Novo Synthesis of Fatty Acid) • Synthesis is not the reversal of β-oxidation. • dietary carbohydrates and amino acids, when consumed in excess, converted to fatty acids and stored as triacylglycerols.

Fatty Acid Biosynthesis (De Novo Synthesis of Fatty Acid) • Tissues: Liver, adipose tissue, kidney, brain and mammary glands • Intracellular site: Cytosol • Substrates required: Acetyl Co. A, ATP and NADPH (provides reducing equivalents; main source: HMP shunt pathway)

Fatty Acid Biosynthesis (De Novo Synthesis of Fatty Acid) Steps of fatty acid synthesis include: • Transport of acetyl Co. A from mitochondrial matrix to cytosol • Formation of malonyl Co. A from acetyl Co. A • Synthesis of fatty acid involving fatty acid synthase complex
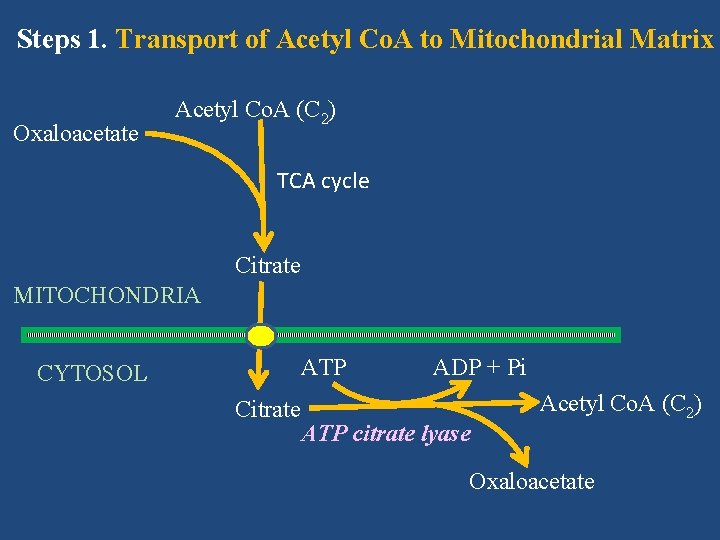
Steps 1. Transport of Acetyl Co. A to Mitochondrial Matrix Oxaloacetate Acetyl Co. A (C 2) TCA cycle Citrate MITOCHONDRIA ATP CYTOSOL Citrate ADP + Pi ATP citrate lyase Acetyl Co. A (C 2) Oxaloacetate
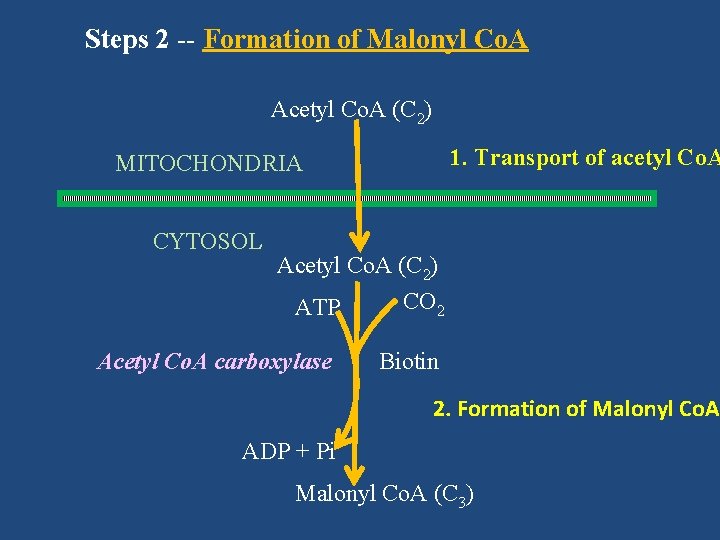
Steps 2 -- Formation of Malonyl Co. A Acetyl Co. A (C 2) 1. Transport of acetyl Co. A MITOCHONDRIA CYTOSOL Acetyl Co. A (C 2) CO 2 ATP Acetyl Co. A carboxylase Biotin 2. Formation of Malonyl Co. A ADP + Pi Malonyl Co. A (C 3)

Step 3 – Synthesis of Fatty Acid Involving Fatty Acid Synthase Complex Fatty Acid Synthase (FAS) Complex 1 • FAS complex is a multienzyme complex and enzymes form a dimer with identical subunits. • The multienzyme complex facilitates easy interaction of substrates with the active sites of the enzymes.
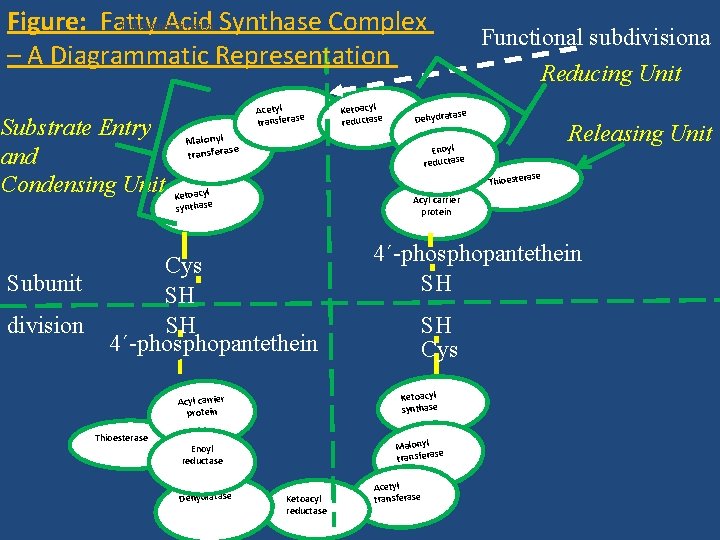
Functional division Figure: Fatty Acid Synthase Complex – A Diagrammatic Representation Substrate Entry and Condensing Unit Subunit division Acetyl se transfera se Dehydrata Malonyl se transfera se Thioestera Ketoacyl synthase Acyl carrier protein 4´-phosphopantethein SH SH Cys Acyl carrier protein Ketoacyl synthase Enoyl reductase Malonyl e transferas Dehydratase Releasing Unit Enoyl reductase Cys SH SH 4´-phosphopantethein Thioesterase Ketoacyl reductase Functional subdivisiona Reducing Unit Ketoacyl reductase Acetyl transferase
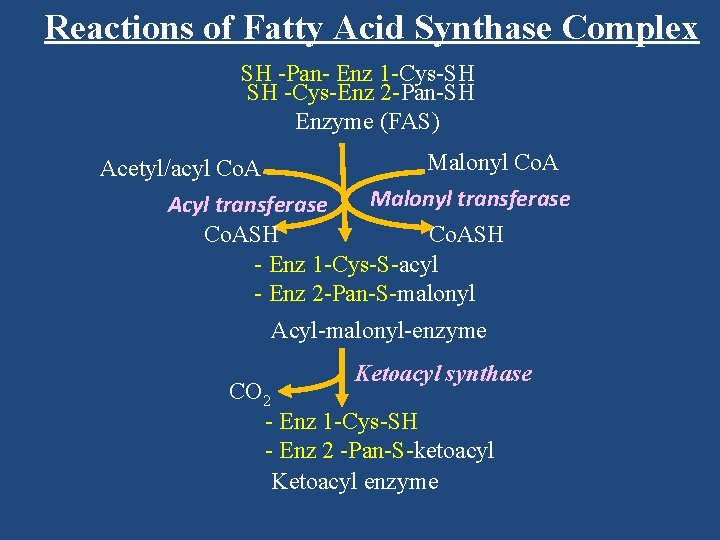
Reactions of Fatty Acid Synthase Complex SH -Pan- Enz 1 -Cys-SH SH -Cys-Enz 2 -Pan-SH Enzyme (FAS) Malonyl Co. A Acetyl/acyl Co. A Malonyl transferase Acyl transferase Co. ASH - Enz 1 -Cys-S-acyl - Enz 2 -Pan-S-malonyl Acyl-malonyl-enzyme Ketoacyl synthase CO 2 - Enz 1 -Cys-SH - Enz 2 -Pan-S-ketoacyl Ketoacyl enzyme
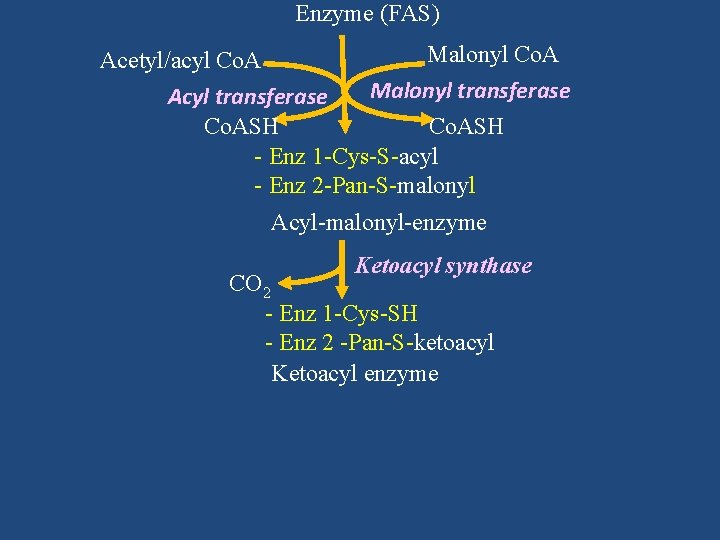
Enzyme (FAS) Malonyl Co. A Acetyl/acyl Co. A Malonyl transferase Acyl transferase Co. ASH - Enz 1 -Cys-S-acyl - Enz 2 -Pan-S-malonyl Acyl-malonyl-enzyme Ketoacyl synthase CO 2 - Enz 1 -Cys-SH - Enz 2 -Pan-S-ketoacyl Ketoacyl enzyme

Co. ASH - Enz 1 -Cys-S-acyl - Enz 2 -Pan-S-malonyl Acyl-malonyl-enzyme Ketoacyl synthase CO 2 - Enz 1 -Cys-SH - Enz 2 -Pan-S-ketoacyl Ketoacyl enzyme

Acyl-malonyl-enzyme Ketoacyl synthase CO 2 - Enz 1 -Cys-SH - Enz 2 -Pan-S-ketoacyl Ketoacyl enzyme
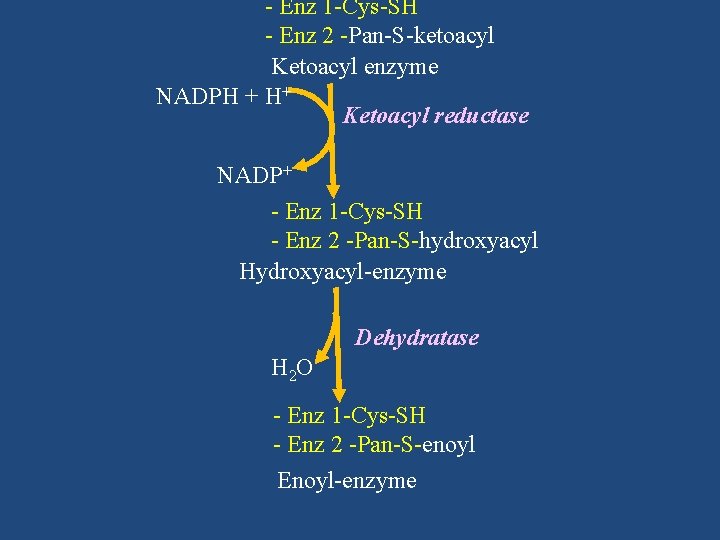
- Enz 1 -Cys-SH - Enz 2 -Pan-S-ketoacyl Ketoacyl enzyme NADPH + H+ Ketoacyl reductase NADP+ - Enz 1 -Cys-SH - Enz 2 -Pan-S-hydroxyacyl Hydroxyacyl-enzyme Dehydratase H 2 O - Enz 1 -Cys-SH - Enz 2 -Pan-S-enoyl Enoyl-enzyme
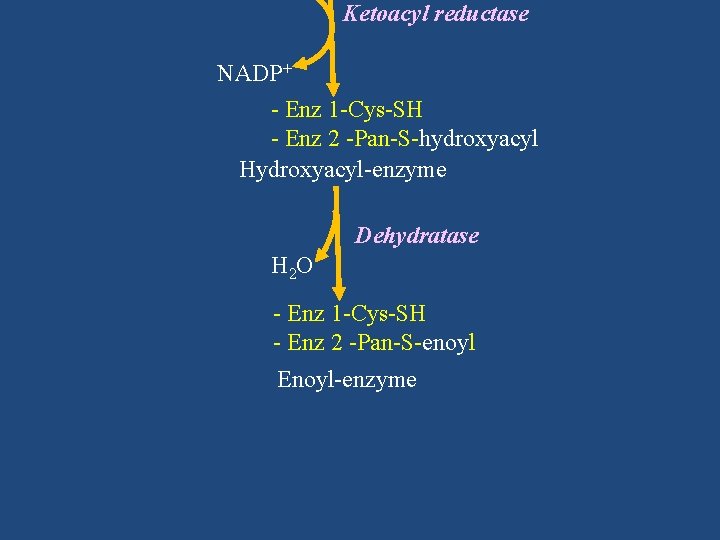
Ketoacyl reductase NADP+ - Enz 1 -Cys-SH - Enz 2 -Pan-S-hydroxyacyl Hydroxyacyl-enzyme Dehydratase H 2 O - Enz 1 -Cys-SH - Enz 2 -Pan-S-enoyl Enoyl-enzyme
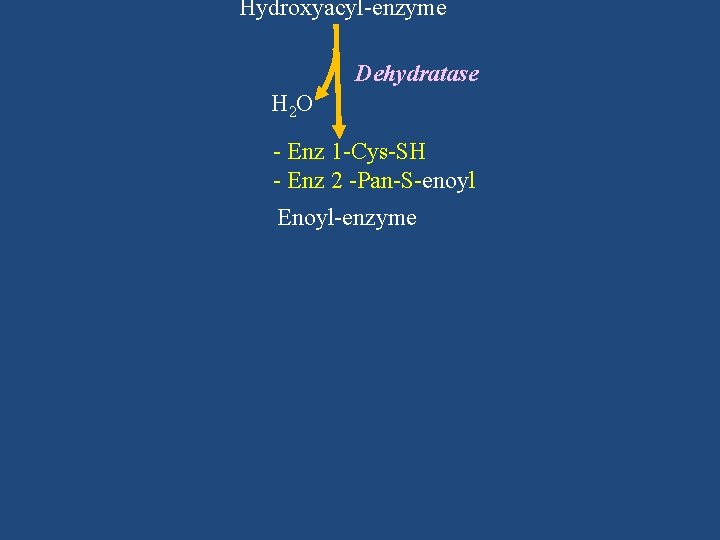
Hydroxyacyl-enzyme Dehydratase H 2 O - Enz 1 -Cys-SH - Enz 2 -Pan-S-enoyl Enoyl-enzyme
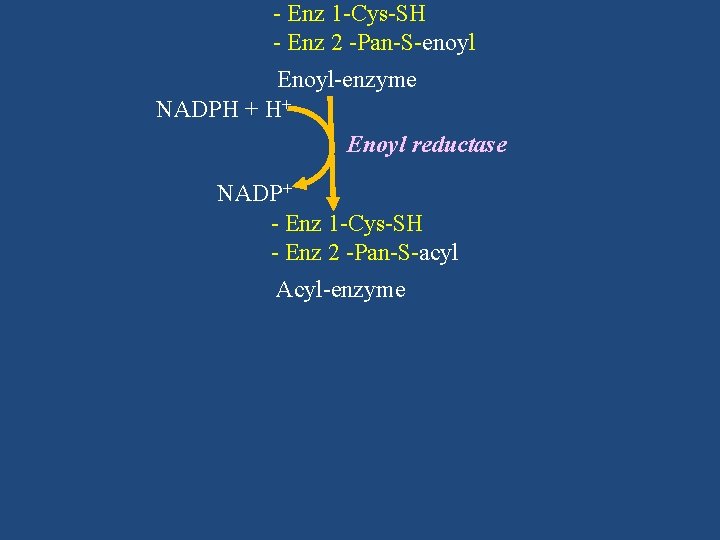
- Enz 1 -Cys-SH - Enz 2 -Pan-S-enoyl Enoyl-enzyme NADPH + H+ Enoyl reductase NADP+ - Enz 1 -Cys-SH - Enz 2 -Pan-S-acyl Acyl-enzyme
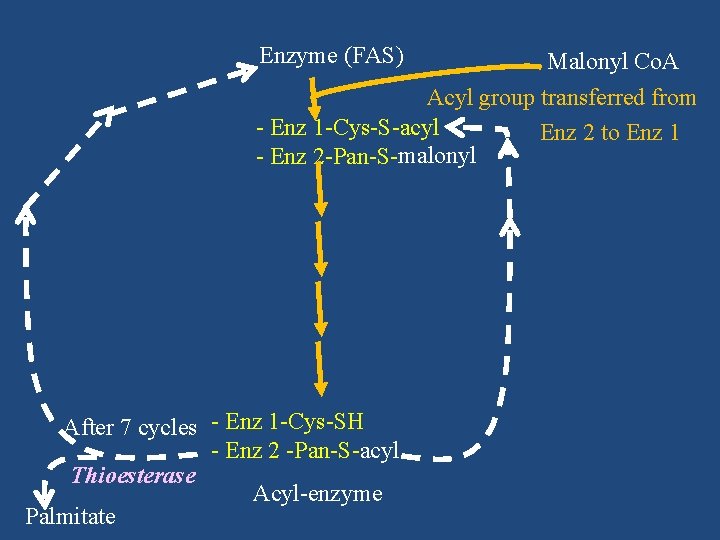
Enzyme (FAS) Malonyl Co. A Acyl group transferred from - Enz 1 -Cys-S-acyl Enz 2 to Enz 1 - Enz 2 -Pan-S-malonyl After 7 cycles - Enz 1 -Cys-SH - Enz 2 -Pan-S-acyl Thioesterase Acyl-enzyme Palmitate
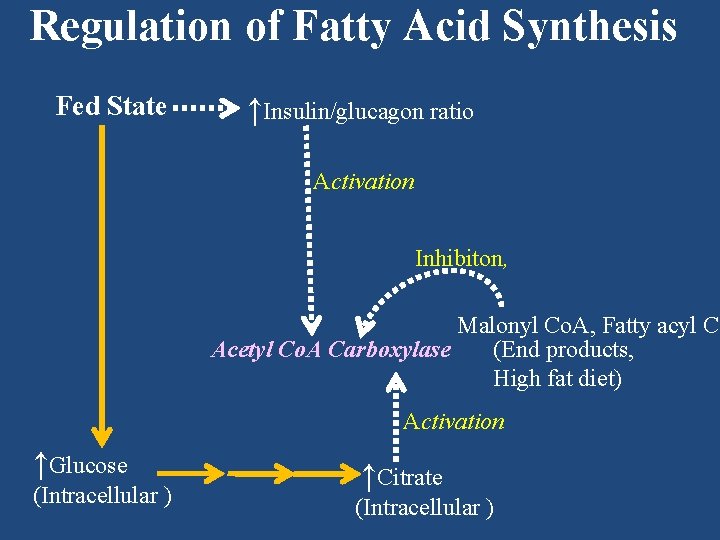
Regulation of Fatty Acid Synthesis Fed State ↑Insulin/glucagon ratio Activation Inhibiton, Malonyl Co. A, Fatty acyl Co Acetyl Co. A Carboxylase (End products, High fat diet) Activation ↑Glucose (Intracellular ) ↑Citrate (Intracellular )

Ketone Body Metabolism and Ketosis ketone bodies – A collective name for • Acetone, • Acetoacetate (acetoacetic acid) and • β-hydroxy butyrate (β-hydroxy butyric acid) • synthesized in the liver from fatty acids metabolic fuel molecules during prolonged fasting and Starvation especially for brain
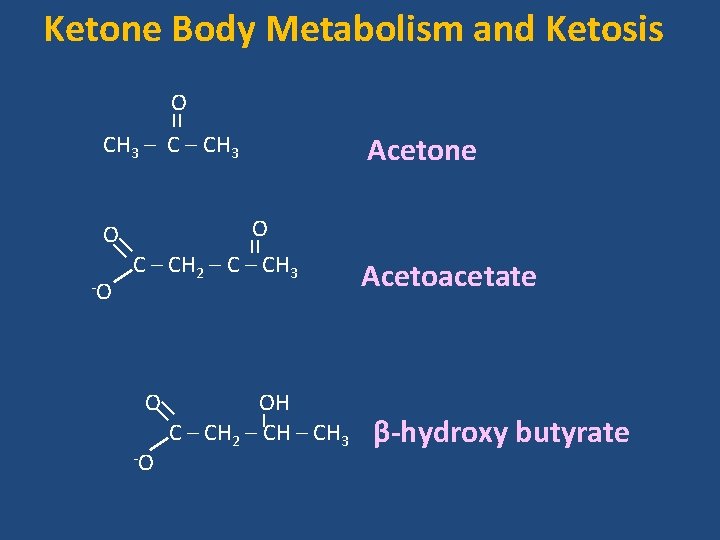
Ketone Body Metabolism and Ketosis O CH 3 – CH 3 O C – CH 2 – CH 3 -O O OH C – CH 2 – CH 3 -O Acetone Acetoacetate β-hydroxy butyrate

Ketone Body Metabolism and Ketosis • Ketosis, accumulation of ketone bodies in the body, a metabolic disorder seen in starvation and uncontrolled diabetes mellitus (Type 1)

Ketone Body Metabolism and Ketosis Ketone body metabolism includes – • 1) Ketogenesis (synthesis of ketone bodies), • 2) Ketolysis (breakdown of ketone bodies for energy) and

1) Ketogenesis (Synthesis of Ketone Bodies) • Acetyl Co. A immediate precursor However, since acetyl Co. A is formed from fatty acids by β-oxidation, fatty acids are the ultimate precursors • Site: Liver • Sub-cellular site: Mitochondria
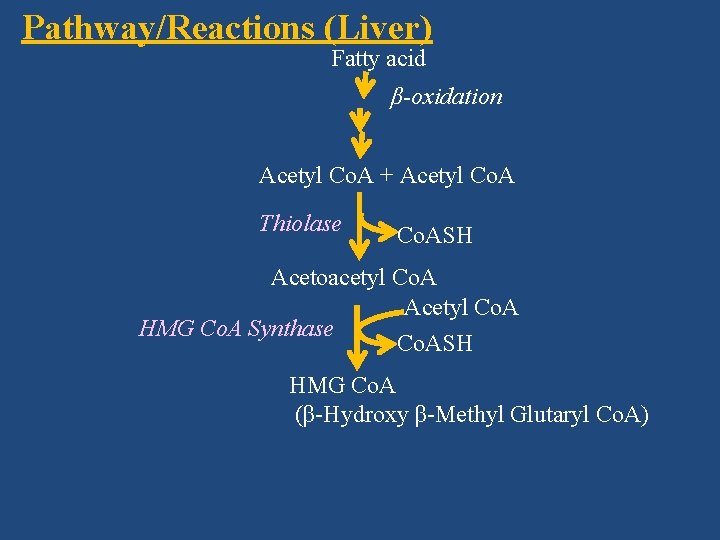
Pathway/Reactions (Liver) Fatty acid β-oxidation Acetyl Co. A + Acetyl Co. A Thiolase Co. ASH Acetoacetyl Co. A Acetyl Co. A HMG Co. A Synthase Co. ASH HMG Co. A (β-Hydroxy β-Methyl Glutaryl Co. A)

Synthase Acetyl Co. A + Acetyl Co. A Thiolase Co. ASH Acetoacetyl Co. A Acetyl Co. A HMG Co. A Synthase Co. ASH HMG Co. A (β-Hydroxy β-Methyl Glutaryl Co. A)
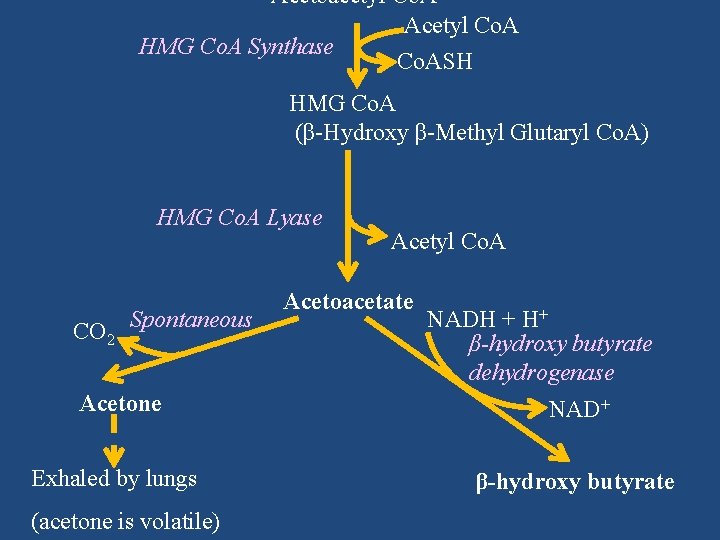
Acetoacetyl Co. A Acetyl Co. A HMG Co. A Synthase Co. ASH HMG Co. A (β-Hydroxy β-Methyl Glutaryl Co. A) HMG Co. A Lyase CO 2 Spontaneous Acetyl Co. A Acetoacetate NADH + H+ β-hydroxy butyrate dehydrogenase Acetone NAD+ Exhaled by lungs β-hydroxy butyrate (acetone is volatile)

2. Ketolysis (Breakdown of ketone bodies) • Ketone bodies formed in the liver utilized as fuels by extrahepatic tissues during prolonged fasting and Starvation especially for especially by brain heart muscle, skeletal muscle and renal cortex • They prefer ketone bodies to fatty acids and glucose
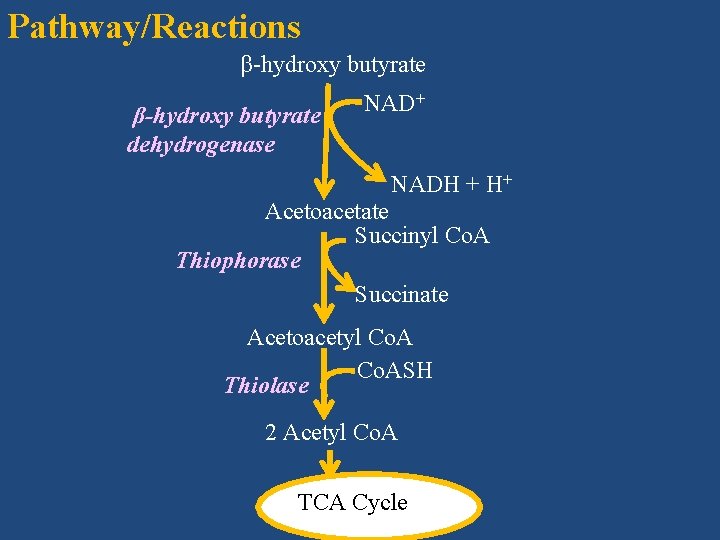
Pathway/Reactions β-hydroxy butyrate dehydrogenase NAD+ NADH + H+ Acetoacetate Succinyl Co. A Thiophorase Succinate Acetoacetyl Co. ASH Thiolase 2 Acetyl Co. A TCA Cycle

3. Ketosis/Ketoacidosis (Accumulation of ketone bodies in body) • Normal ketone body level in blood is less than 1 mg/dl. • Normally only traces of ketone bodies are found in urine. • Ketone bodies accumulate in body if rate of synthesis exceeds ability of extra hepatic tissues to utilize them. • Causes of Ketosis • 1) Starvation • 2) Uncontrolled type 1 diabetes mellitus (type 1 DM)
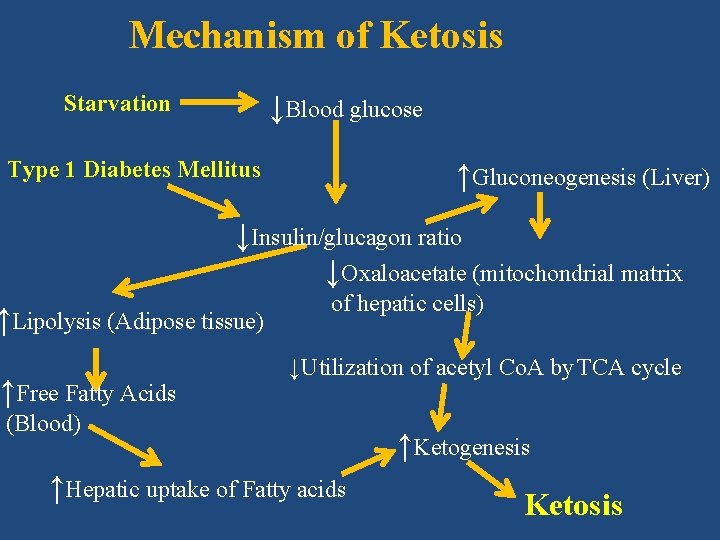
Mechanism of Ketosis ↓Blood glucose Starvation Type 1 Diabetes Mellitus ↑Gluconeogenesis (Liver) ↓Insulin/glucagon ratio ↓Oxaloacetate (mitochondrial matrix ↑Lipolysis (Adipose tissue) ↑Free Fatty Acids of hepatic cells) ↓Utilization of acetyl Co. A by TCA cycle (Blood) ↑Hepatic uptake of Fatty acids ↑Ketogenesis Ketosis

Biochemical Changes in Ketosis leads to: • Ketonemia (Excess ketone body in blood) • Ketonuria (Excretion of ketone body in urine) and • Acidosis (ketoacidosis) – acetoacetic acid and β-hydroxy butyric acid are acidic.

Ketosis • Clinical Manifestation • Deeper and faster rate of breathing (Kussmual breathing) • Acetone smelling breath • Laboratory Diagnosis 1. Blood - Estimation of p. H, HCO 3, CO 2 (see ‘Disorders of Acid Base Balance’) 2. Urine – Rothera’s test, for ketone bodies, will be is positive.

Thank You
- Slides: 57