METABOLISM OF CARBOHYDRATES DIGESTION OF CARBOHYDRATES SYNTHESIS AND

METABOLISM OF CARBOHYDRATES: DIGESTION OF CARBOHYDRATES. SYNTHESIS AND DEGRADATION OF GLYCOGEN

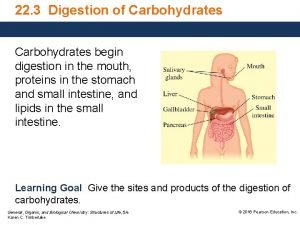
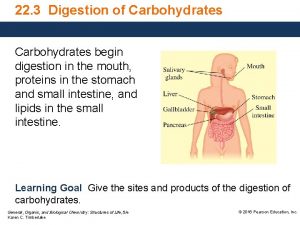
DIGESTION OF CARBOHYDRATES Glycogen, starch and disaccharides (sucrose, lactose and maltose) are hydrolyzed to monosaccharide units in the gastrointestinal tract. The process of digestion starts in the mouth by the salivary enzyme –amilase. The time for digestion in mouth is limited. Salivary -amilase is inhibited in stomach due to the action of hydrochloric acid. Another -amilase is produced in pancreas and is available in the intestine.

-amilase hydrolyzes the -1 -4 -glycosidic bonds randomly to produce smaller subunits like maltose, dextrines and unbranched oligosaccharides. -amilase
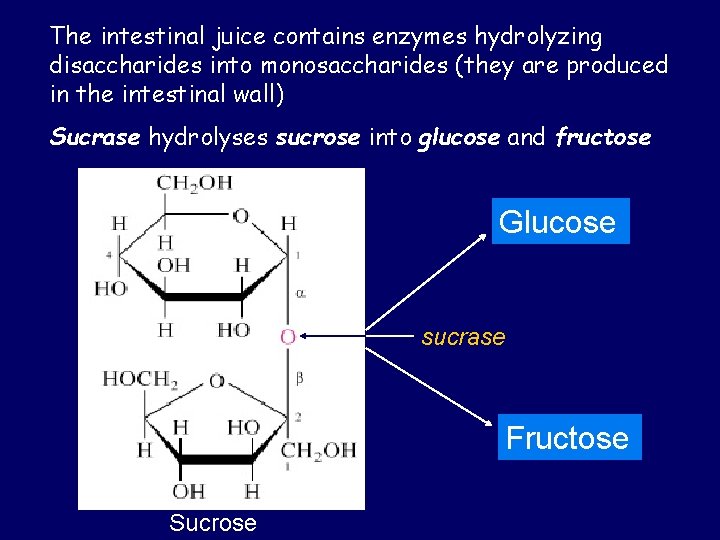
The intestinal juice contains enzymes hydrolyzing disaccharides into monosaccharides (they are produced in the intestinal wall) Sucrase hydrolyses sucrose into glucose and fructose Glucose sucrase Fructose Sucrose
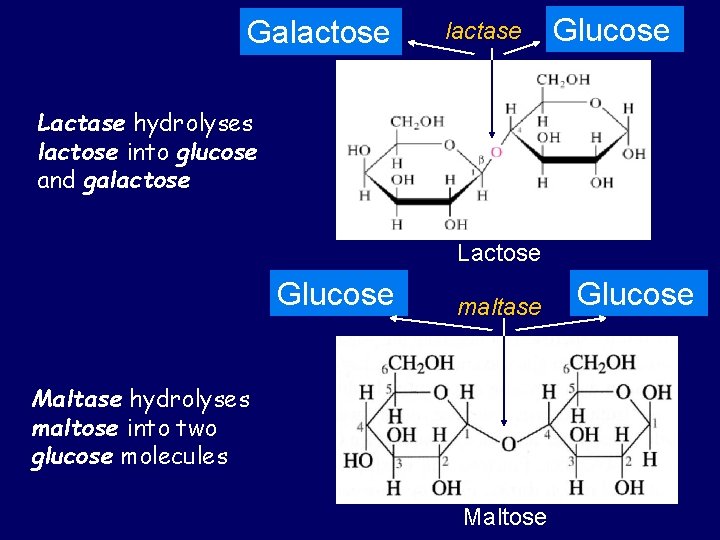
Galactose lactase Glucose Lactase hydrolyses lactose into glucose and galactose Lactose Glucose maltase Maltase hydrolyses maltose into two glucose molecules Maltose Glucose

ABSORPTION OF CARBOHYDRATES Only monosaccharides are absorbed The rate of absorption: galactose > glucose > fructose Glucose and galactose from the intestine into endothelial cells are absorbed by secondary active transport Na+ Protein Glucose Protein

Carrier protein is specific for D-glucose or D-galactose. L-forms are not transported. There are competition between glucose and galactose for the same carrier molecule; thus glucose can inhibit absorption of galactose. Fructose is absorbed from intestine into intestinal cells by facilitated diffusion. Absorption of glucose from intestinal cells into bloodstream is by facilitated diffusion.

Transport of glucose from blood into cells of different organs is mainly by facilitated diffusion. The protein facilitating the glucose transport is called glucose transporter (Glu. T). Glu. T are of 5 types. Glu. T 2 is located mainly in hepatocytes membranes (it transport glucose into cells when blood sugar is high); Glu. T 1 is seen in erythrocytes and endothelial cells; Glu. T 3 is located in neuronal cells (has higher affinity to glucose); Glu. T 5 – in intestine and kidneys; Glu. T 4 - in muscles and fat cells.
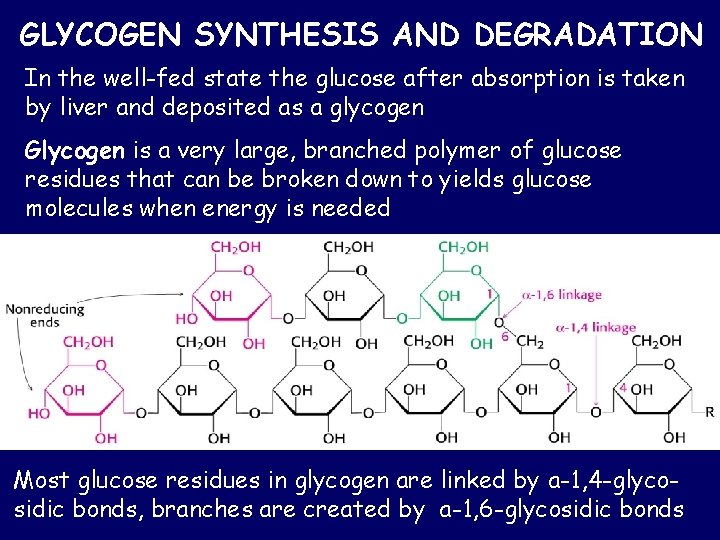
GLYCOGEN SYNTHESIS AND DEGRADATION In the well-fed state the glucose after absorption is taken by liver and deposited as a glycogen Glycogen is a very large, branched polymer of glucose residues that can be broken down to yields glucose molecules when energy is needed Most glucose residues in glycogen are linked by a-1, 4 -glycosidic bonds, branches are created by a-1, 6 -glycosidic bonds

Glycogen serves as a buffer to maintain blood-glucose level. Stable blood glucose level is especially important for brain where it is the only fuel. The glucose from glycogen is readily mobilized and is therefore a good source of energy for sudden, strenuous activity. Liver (10 % of weight) and skeletal muscles (2 %) – two major sites of glycogen storage Glycogen is stored in cytosolic granules in muscle and liver cells of vertebrates
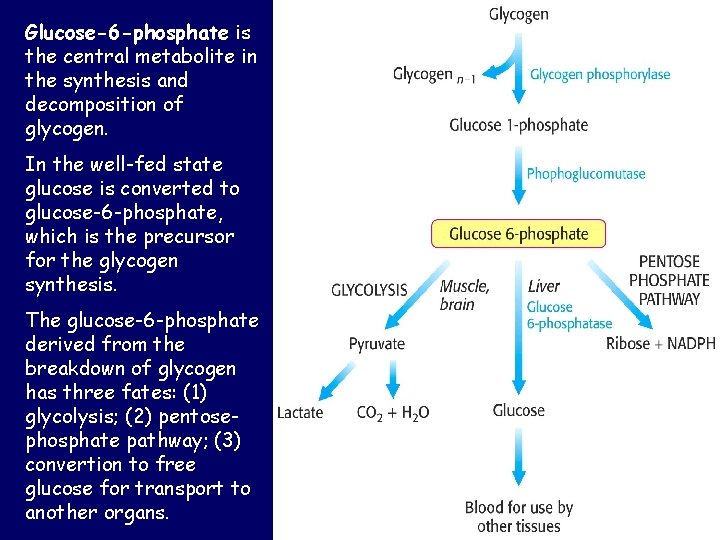
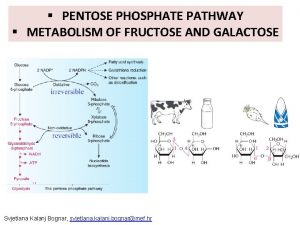
Glucose-6 -phosphate is the central metabolite in the synthesis and decomposition of glycogen. In the well-fed state glucose is converted to glucose-6 -phosphate, which is the precursor for the glycogen synthesis. The glucose-6 -phosphate derived from the breakdown of glycogen has three fates: (1) glycolysis; (2) pentosephosphate pathway; (3) convertion to free glucose for transport to another organs.

DEGRADATION OF GLYCOGEN Glycogenolysis - degradation of glycogen The reaction to release glucose from polysaccharide is not simple hydrolysis as with dietary polysaccharides but cleavage by inorganic phosphate – phosphorolytic cleavage Phosphorolytic cleavage or phosphorolysis is catalyzed by enzyme glycogen phosphorylase There are two ends on the molecules of starch or glycogen: a nonreducing end (the end glucose has free hydroxyl group on C 4) and a reducing end (the end glucose has an anomeric carbon center (free hydroxyl group on C 1)
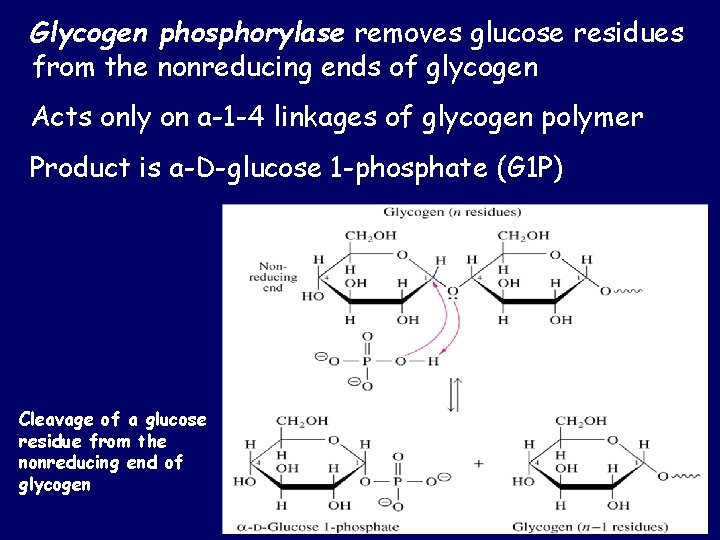
Glycogen phosphorylase removes glucose residues from the nonreducing ends of glycogen Acts only on a-1 -4 linkages of glycogen polymer Product is a-D-glucose 1 -phosphate (G 1 P) Cleavage of a glucose residue from the nonreducing end of glycogen

Structure of glycogen phosphorylase (GP) • GP is a dimer of identical subunits (97 k. D each) • Catalytic sites are in clefts between the two domains of each subunit • Binding sites for glycogen, allosteric effectors and a phosphorylation site • Two forms of GP Phosphorylase a (phosphorylated) active form Phosphorylase b (dephosphorylated) less active
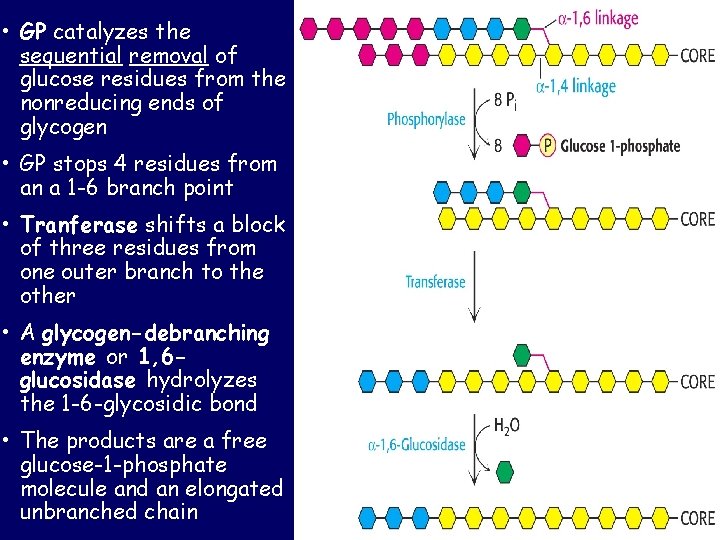
• GP catalyzes the sequential removal of glucose residues from the nonreducing ends of glycogen • GP stops 4 residues from an a 1 -6 branch point • Tranferase shifts a block of three residues from one outer branch to the other • A glycogen-debranching enzyme or 1, 6 glucosidase hydrolyzes the 1 -6 -glycosidic bond • The products are a free glucose-1 -phosphate molecule and an elongated unbranched chain
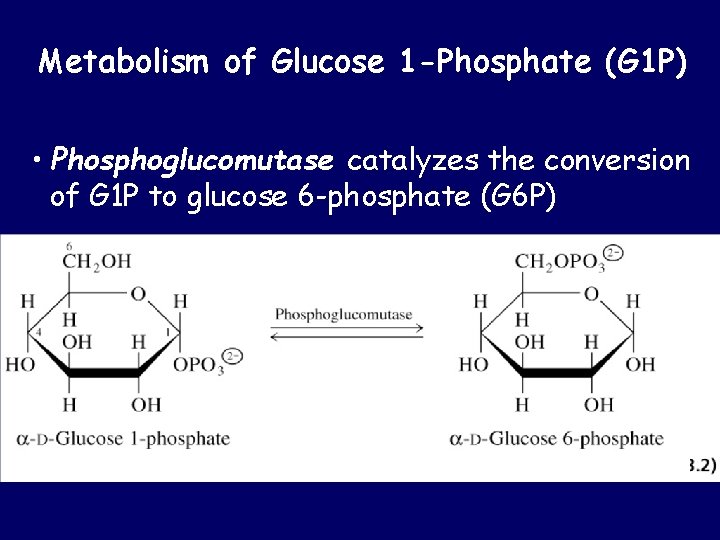
Metabolism of Glucose 1 -Phosphate (G 1 P) • Phosphoglucomutase catalyzes the conversion of G 1 P to glucose 6 -phosphate (G 6 P)

Glycogen Synthesis • Synthesis and degradation of glycogen require separate enzymatic steps • Cellular glucose converted to G 6 P by hexokinase • Three separate enzymatic steps are required to incorporate one G 6 P into glycogen • Glycogen synthase is the major regulatory step

Glucose 1 -Phosphate formation • Phosphoglucomutase catalyzes the conversion of glucose 6 -phosphate (G 6 P) to glucose 1 phosphate (G 1 P).
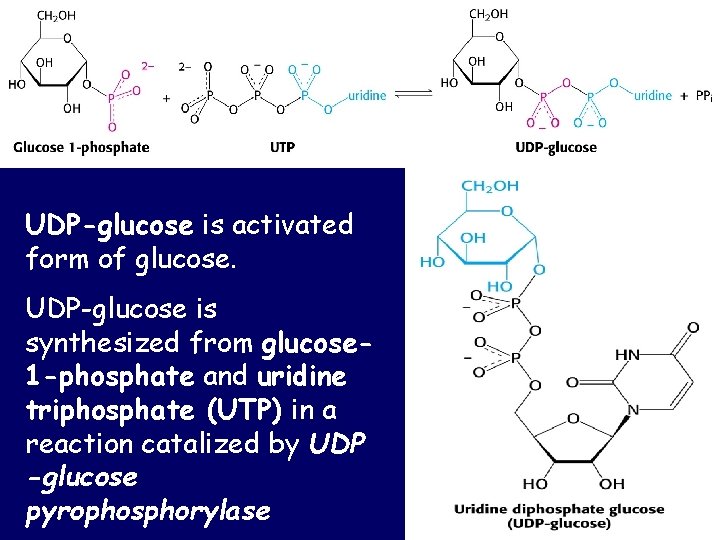
UDP-glucose is activated form of glucose. UDP-glucose is synthesized from glucose 1 -phosphate and uridine triphosphate (UTP) in a reaction catalized by UDP -glucose pyrophosphorylase

Glycogen synthase adds glucose to the nonreducing end of glycogen
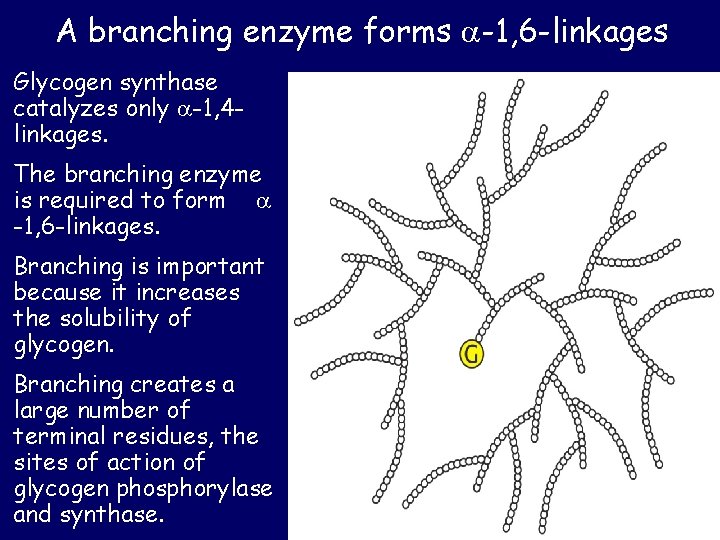
A branching enzyme forms -1, 6 -linkages Glycogen synthase catalyzes only -1, 4 linkages. The branching enzyme is required to form -1, 6 -linkages. Branching is important because it increases the solubility of glycogen. Branching creates a large number of terminal residues, the sites of action of glycogen phosphorylase and synthase.

Regulation of Glycogen Metabolism • Muscle glycogen is fuel for muscle contraction • Liver glycogen is mostly converted to glucose for bloodstream transport to other tissues • Both mobilization and synthesis of glycogen are regulated by hormones • Insulin, glucagon and epinephrine regulate mammalian glycogen metabolism

Hormones Regulate Glycogen Metabolism Insulin • Insulin is produced by b-cells of the pancreas (high levels are associated with the fed state) • Insulin increases rate of glucose transport into muscle, adipose tissue via Glu. T 4 transporter • Insulin stimulates glycogen synthesis in the liver via the second messenger phosphatidylinositol 3, 4, 5 -triphosphate (PIP 3)

Glucagon • Secreted by the a cells of the pancreas in response to low blood glucose (elevated glucagon is associated with the fasted state) • Stimulates glycogen degradation to restore blood glucose to steady-state levels • Only liver cells are rich in glucagon receptors and therefore respond to this hormone

Epinephrine (Adrenalin) • Released from the adrenal glands in response to sudden energy requirement (“fight or flight”) • Stimulates the breakdown of glycogen to G 1 P (which is converted to G 6 P) • Increased G 6 P levels increase both the rate of glycolysis in muscle and glucose release to the bloodstream from the liver and muscles • Both liver and muscle cells have receptors to epinephrine

Effects of hormones on glycogen metabolism

Reciprocal Regulation of Glycogen Phosphorylase and Glycogen Synthase • Glycogen phosphorylase (GP) and glycogen synthase (GS) control glycogen metabolism in liver and muscle cells • GP and GS are reciprocally regulated both covalently and allosterically (when one is active the other is inactive) • Covalent regulation by phosphorylation (-P) and dephosphorylation (-OH) • Allosteric regulation by glucose-6 -phosphate (G 6 P)

Reciprocal Regulation of GP and GS COVALENT REGULATION Active form “a” Inactive form “b” Glycogen phosphorylase -P -OH Glycogen synthase -OH -P ALLOSTERIC REGULATION by G 6 P GP a (active form) - inhibited by G 6 P GS b (inactive form) - activated by G 6 P

Activation of GP and inactivation of GS by Epinephrine and Glucagone

Activation of GS and inactivation of GP by Insulin
- Slides: 30