Metabolism Enzymes Metabolism and Metabolic Pathways Enzymes n
Metabolism Enzymes Metabolism and Metabolic Pathways

Enzymes n n Proteins which function as biological catalysts Each biochemical transformation in a cell has a specific enzyme associated with it
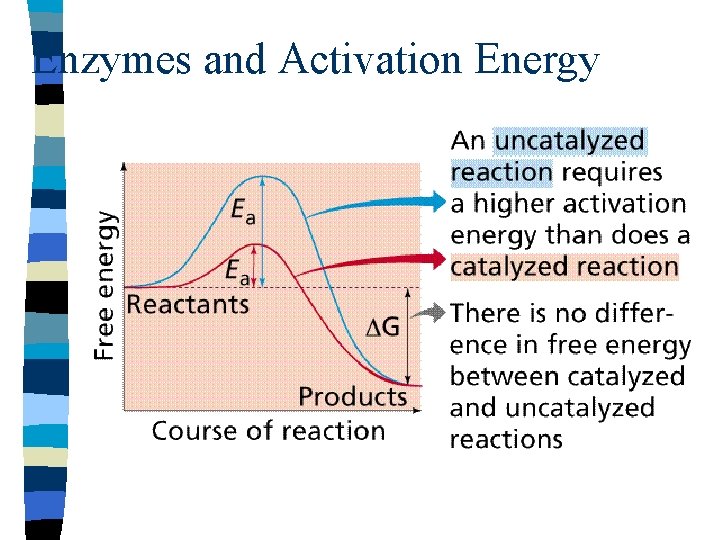
Enzymes and Activation Energy
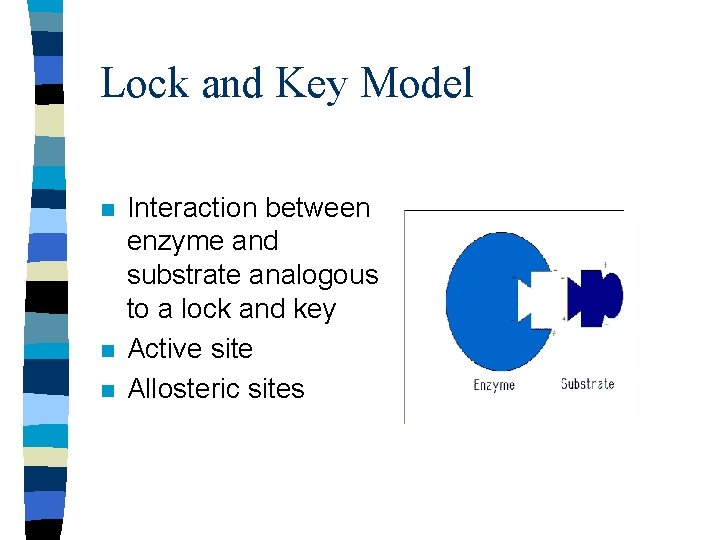
Lock and Key Model n n n Interaction between enzyme and substrate analogous to a lock and key Active site Allosteric sites

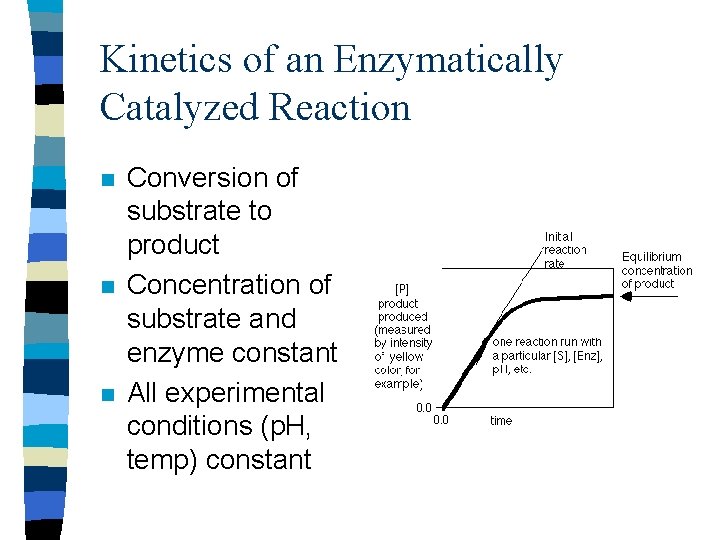
Kinetics of an Enzymatically Catalyzed Reaction n Conversion of substrate to product Concentration of substrate and enzyme constant All experimental conditions (p. H, temp) constant
![Effect of [S] on V 0 n n n Initial linear relationship between increasing Effect of [S] on V 0 n n n Initial linear relationship between increasing](http://slidetodoc.com/presentation_image/95907263635a9c27cefd315fd92ccc67/image-7.jpg)
Effect of [S] on V 0 n n n Initial linear relationship between increasing [S] and initial reaction velocity Saturation kinetics Overall a hyperbolic curve
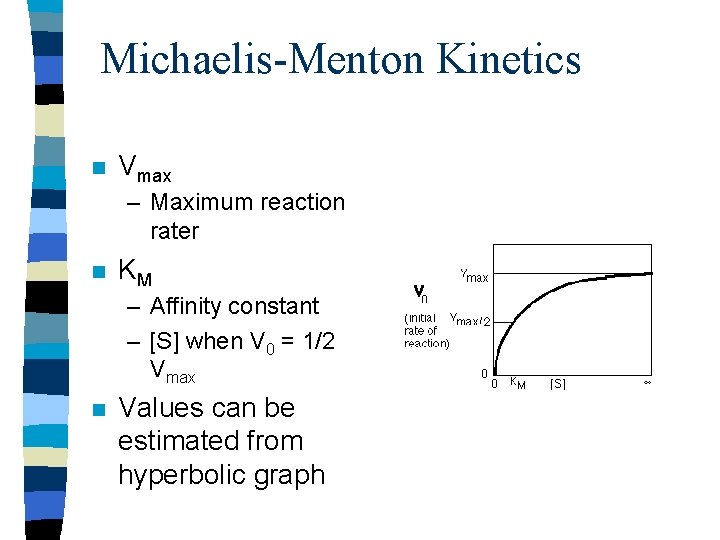
Michaelis-Menton Kinetics n Vmax – Maximum reaction rater n KM – Affinity constant – [S] when V 0 = 1/2 Vmax n Values can be estimated from hyperbolic graph
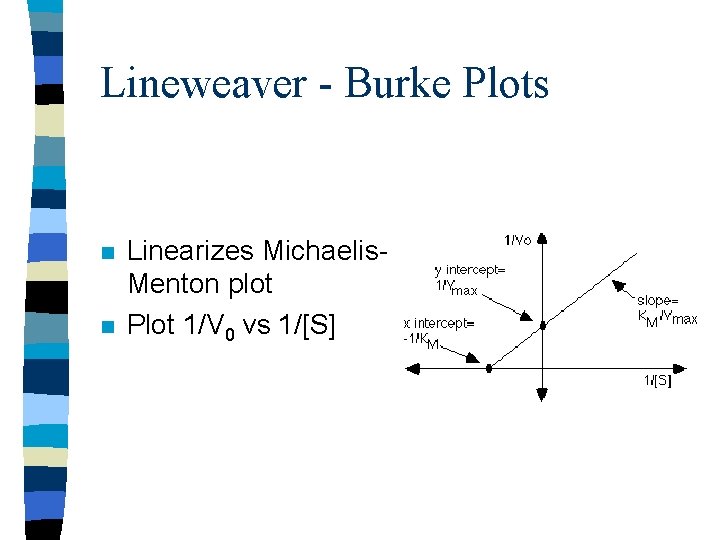
Lineweaver - Burke Plots n n Linearizes Michaelis. Menton plot Plot 1/V 0 vs 1/[S]

Monod Equation

Metabolism

Definitions n n Metabolism: The sum of the biochemical reactions which occur in a cell Pathway: – A series of connected reactions – A --> B--> C-->D-->E n Catabolism: – Breakdown complex substrates – Generally oxidations

– Energy yielding – Generate reduced electron carriers n Anabolism – Build up complex molecules from precursors – Generally reductions – Energy requiring – Oxidize electron carriers
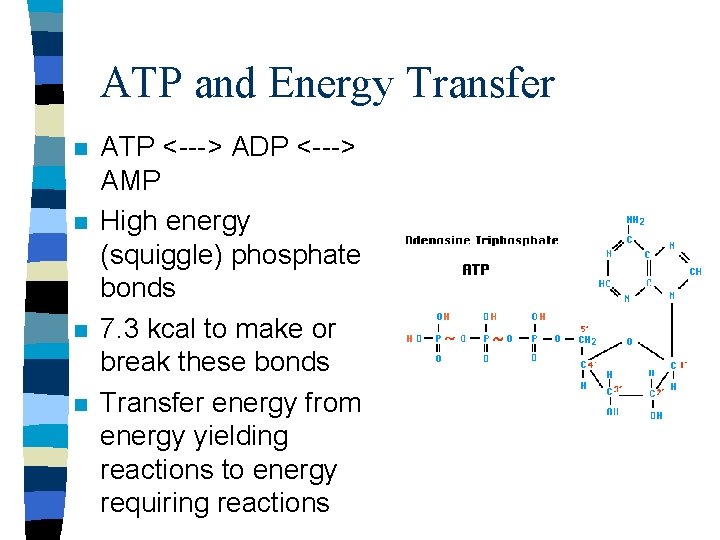
ATP and Energy Transfer n n ATP <---> ADP <---> AMP High energy (squiggle) phosphate bonds 7. 3 kcal to make or break these bonds Transfer energy from energy yielding reactions to energy requiring reactions


Mechanisms of Energy Generation n Substrate level phosphorylation Oxidative phosphorylation Photophosphorylation

Electron Carriers n Coenzymes – NAD/NADH + H+ – FAD/FADH 2 n n Transfer electrons from oxidation to reduction reactions Need for initial electron donor and terminal electron acceptor
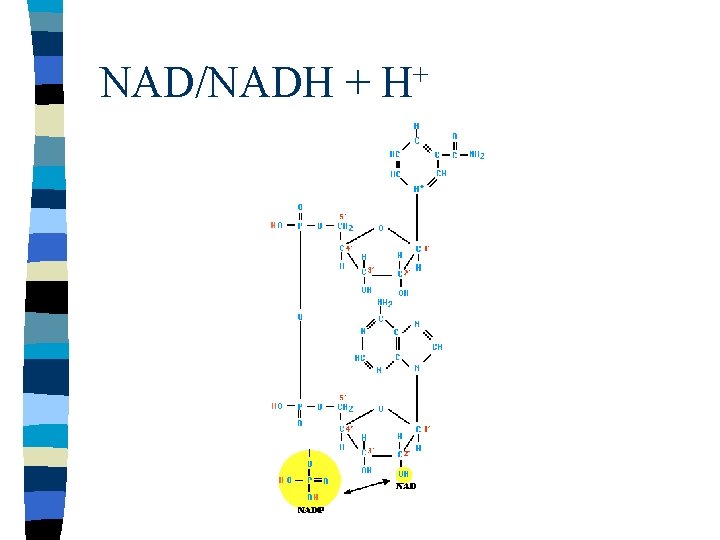
NAD/NADH + + H

Understanding Metabolic Pathways n Keep track of: – Elements – Energy (ATP/ADP) – Electrons n Why are there so many steps in the pathways – Energetic constraints – Generation of intermediates

Central Metabolic Pathways n n n Essential pathways Found in all organisms Include: – Glycolysis (EMP) – TCA (Kreb’s) cycle – ETS

Other Pathways n n Specific catabolic pathways not found in all organisms If an organism can convert a compound into an intermediate in Central metabolism, complete mineralization (catabolism to carbon dioxide and water) is possible.
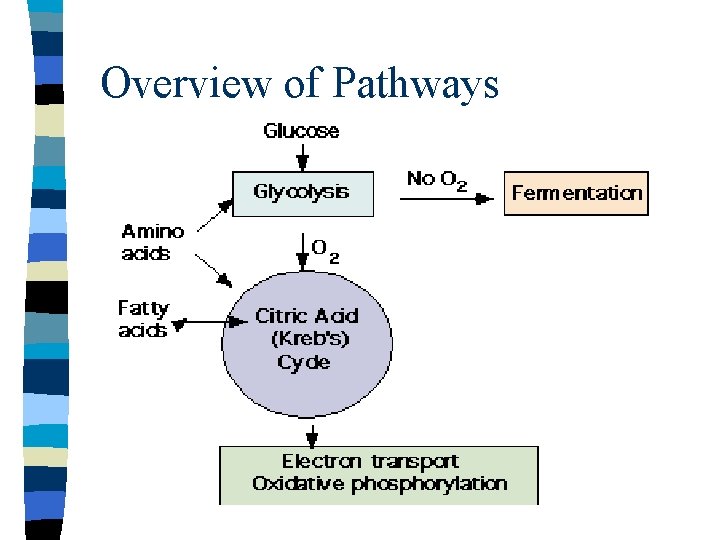
Overview of Pathways

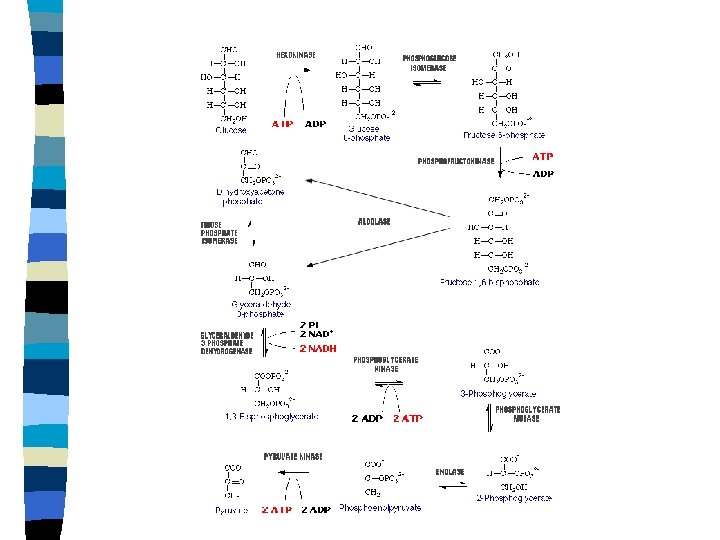
Glycolysis n n Conversion of glucose to 2 pyruvate Most common pathway for initial metabolism of glucose Anaerobic pathway Substrate level phosphorylation n n Low energy yield/incomplete oxidation Production of NADH + H+
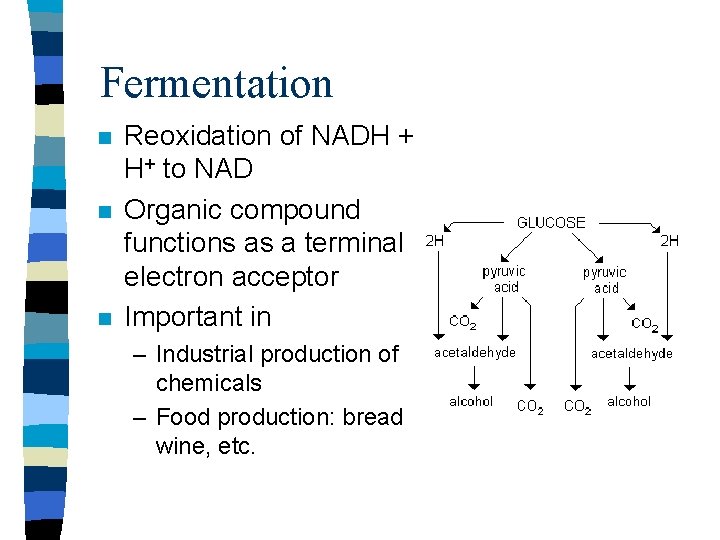
Fermentation n Reoxidation of NADH + H+ to NAD Organic compound functions as a terminal electron acceptor Important in – Industrial production of chemicals – Food production: bread, wine, etc.

TCA Cycle n n Oxidation of pyruvate to carbon dioxide Low direct energy yield Generates large amounts of reduced coenzymes Produces biosynthetic intermediates

Respiration n Transfer of electrons from electron acceptors to terminal electron acceptors – Aerobic organisms: oxygen – Anaerobic organisms: other inorganic compounds • • Nitrate Sulfate Iron Carbonate
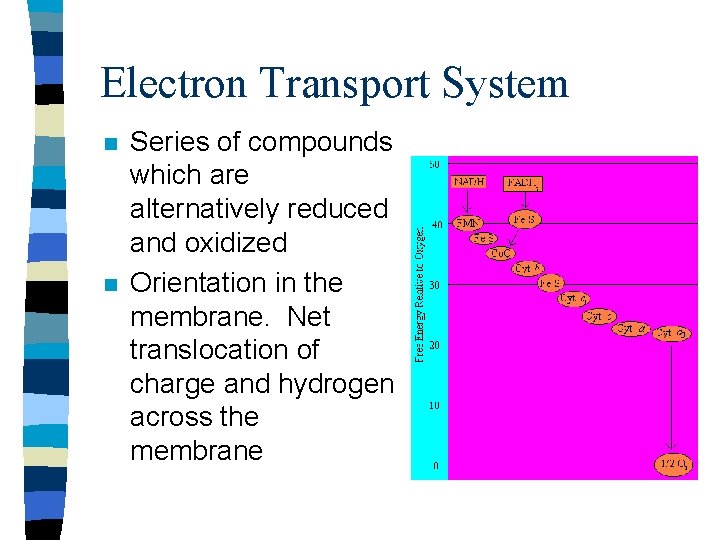
Electron Transport System n n Series of compounds which are alternatively reduced and oxidized Orientation in the membrane. Net translocation of charge and hydrogen across the membrane

Chemiosmosis n n n Couples electron transport with ATP generation Development of transmembrane potential by transfer of electrons and hydrogens Where H+ reenters the cell, ATP synthease is present



- Slides: 32