Metabolism Enzymes AP Biology Day 1 Flow of
Metabolism & Enzymes AP Biology

Day 1 - Flow of energy through life § Life is built on chemical reactions u transforming energy from one form to organic molecules another ATP & organic molecules sun solar energy AP Biology ATP & organic molecules

Reactions in a closed system u Eventually reach equilibrium ∆G < 0 Figure 8. 7 A AP Biology ∆G = 0 (a) A closed hydroelectric system. Water flowing downhill turns a turbine that drives a generator providing electricity to a light bulb, but only until the system reaches equilibrium.

Reactions in an open system § Cells in our body u Experience a constant flow of materials in and out, preventing metabolic pathways from reaching equilibrium ∆G < 0 Figure 8. 7 AP Biology (c) A multi-step open hydroelectric system. Cellular respiration is analogous to this system: Glucose is broken down in a series of exergonic reactions that power the work of the cell. The product of each reaction becomes the reactant for the next, so no reaction reaches equilibrium.

Metabolism A cell is a miniature factory where thousands of reactions occur u Metabolism is the totality of an organism’s chemical reactions § Arises from interactions between molecules § Transforms matter and energy, subject to the laws of thermodynamics AP Biology

1 st Law of Thermodynamics Chemical energy (a) AP Biology Figure 8. 3 First law of thermodynamics: Energy can be transferred or transformed but Neither created nor destroyed. For example, the chemical (potential) energy in food will be converted to the kinetic energy of the cheetah’s movement in (b).

2 nd Law of Thermodynamics Heat co 2 + H 2 O (b) Figure 8. 3 AP Biology Second law of thermodynamics: Every energy transfer or transformation increases the disorder (entropy) of the universe. For example, disorder is added to the cheetah’s surroundings in the form of heat and the small molecules that are the by-products of metabolism.
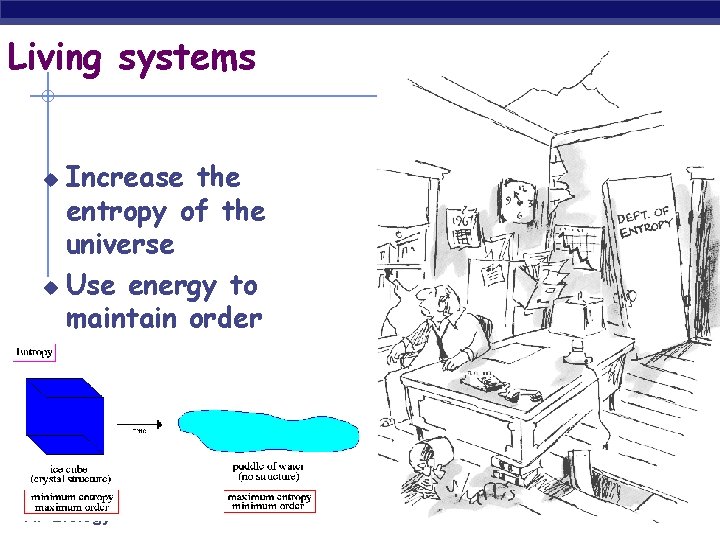
Living systems Increase the entropy of the universe u Use energy to maintain order u AP Biology

Metabolism § Chemical reactions of life u forming bonds between molecules § dehydration synthesis § anabolic reactions u breaking bonds between molecules § hydrolysis § digestion § catabolic reactions AP Biology
AP Biology
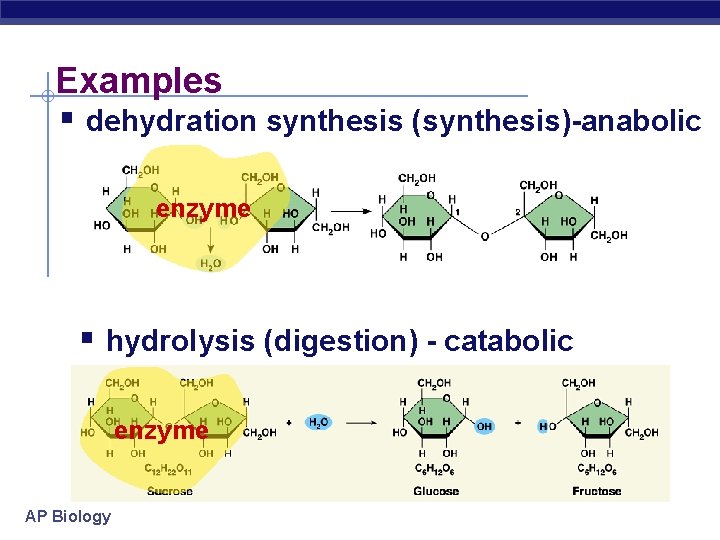
Examples § dehydration synthesis (synthesis)-anabolic enzyme § hydrolysis (digestion) - catabolic enzyme AP Biology

∆G is Gibbs Free Energy Calculation to tell us if a reaction will proceed to the left or to the right…. . § The sign of ∆ G tells us in what direction the reaction has to shift to reach equilibrium. § Reactions go towards a NEGATIVE ∆ G § The magnitude of ∆ G tells us how far the reaction is from equilibrium at that moment. AP Biology
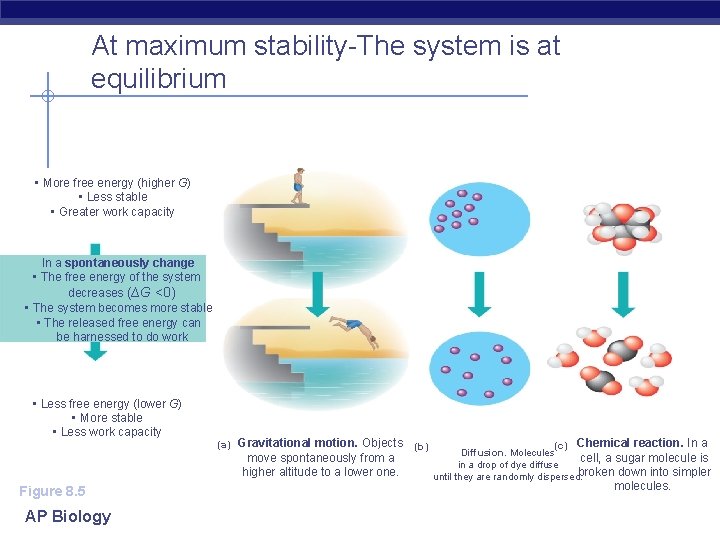
At maximum stability-The system is at equilibrium • More free energy (higher G) • Less stable • Greater work capacity In a spontaneously change • The free energy of the system decreases (∆G <0) • The system becomes more stable • The released free energy can be harnessed to do work. • Less free energy (lower G) • More stable • Less work capacity Figure 8. 5 AP Biology (a) Gravitational motion. Objects move spontaneously from a higher altitude to a lower one. (b) (c) Chemical reaction. In a Diffusion. Molecules cell, a sugar molecule is in a drop of dye diffuse broken down into simpler until they are randomly dispersed. molecules.
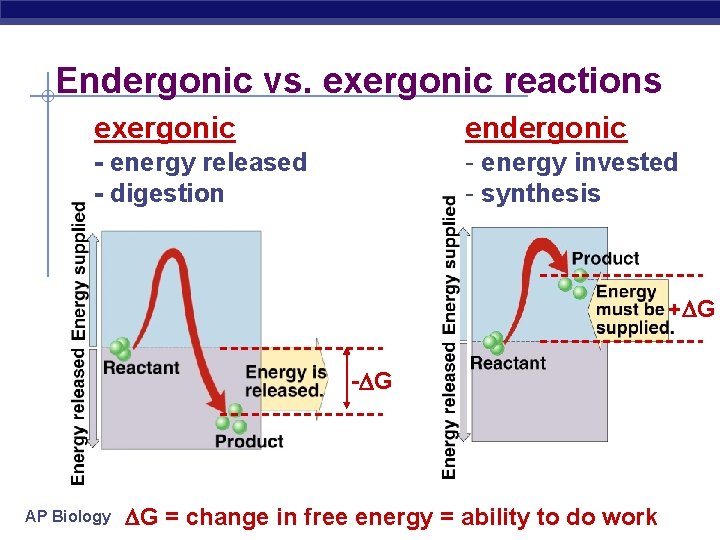
Endergonic vs. exergonic reactions exergonic endergonic - energy released - digestion - energy invested - synthesis + G - G AP Biology G = change in free energy = ability to do work
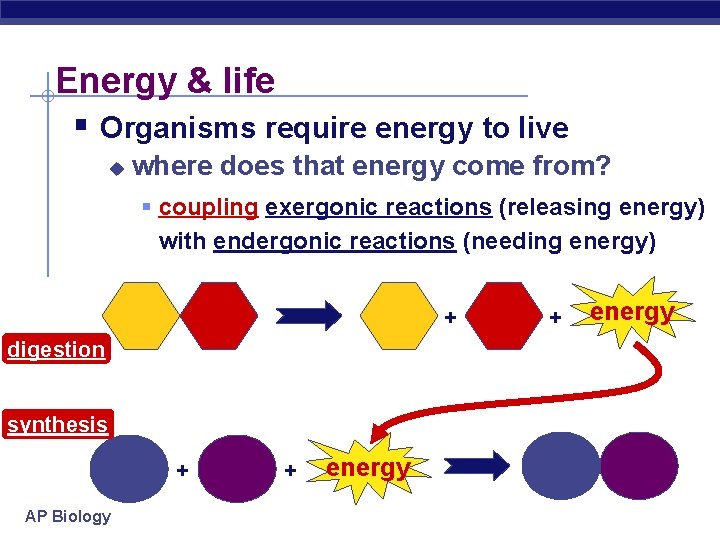
Energy & life § Organisms require energy to live u where does that energy come from? § coupling exergonic reactions (releasing energy) with endergonic reactions (needing energy) + digestion synthesis + AP Biology + energy

ATP – Adenosine Tri-phosphate § ATP powers cellular work by coupling § exergonic reactions to endergonic reactions A cell does three main kinds of work Mechanical u Transport u Chemical u AP Biology
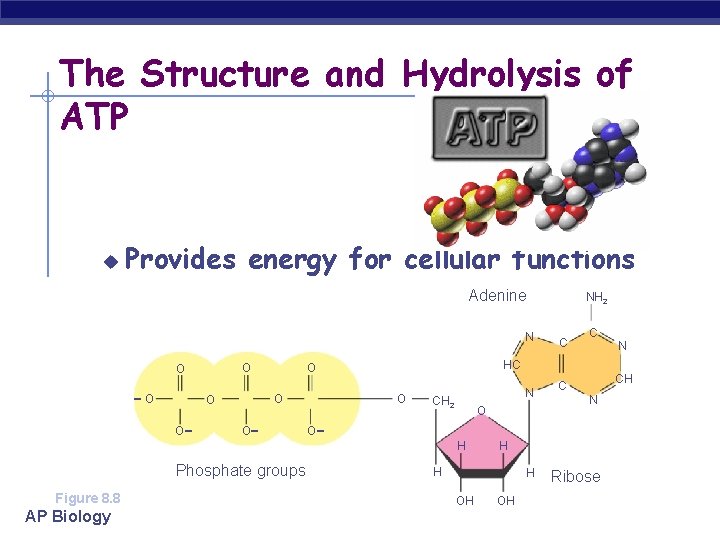
The Structure and Hydrolysis of ATP u Provides energy for cellular functions Adenine N O O - O - Phosphate groups Figure 8. 8 AP Biology O O C C N HC O O O NH 2 N CH 2 - O H N H H H OH CH C OH Ribose
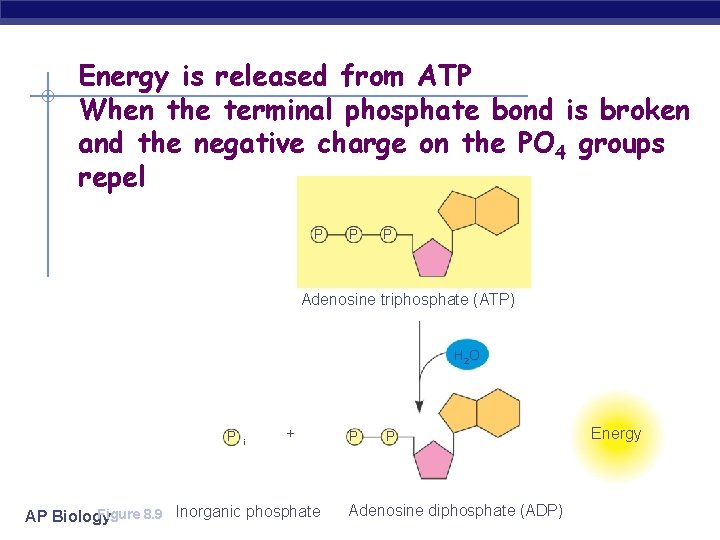
Energy is released from ATP When the terminal phosphate bond is broken and the negative charge on the PO 4 groups repel P P P Adenosine triphosphate (ATP) H 2 O P i + Figure 8. 9 Inorganic phosphate AP Biology P P Adenosine diphosphate (ADP) Energy

ATP hydrolysis can be coupled to other reactions Endergonic reaction: ∆G is positive, reaction is not spontaneous NH 2 Glu + Glutamic acid ATP hydrolysis AP Biology Glu Ammonia Glutamine ∆G = +3. 4 kcal/mol Exergonic reaction: ∆ G is negative, reaction is spontaneous ATP Figure 8. 10 NH 3 + H 2 O ADP + P Coupled reactions: Overall ∆G is negative; together, reactions are spontaneous ∆G = - 7. 3 kcal/mol ∆G = – 3. 9 kcal/mol
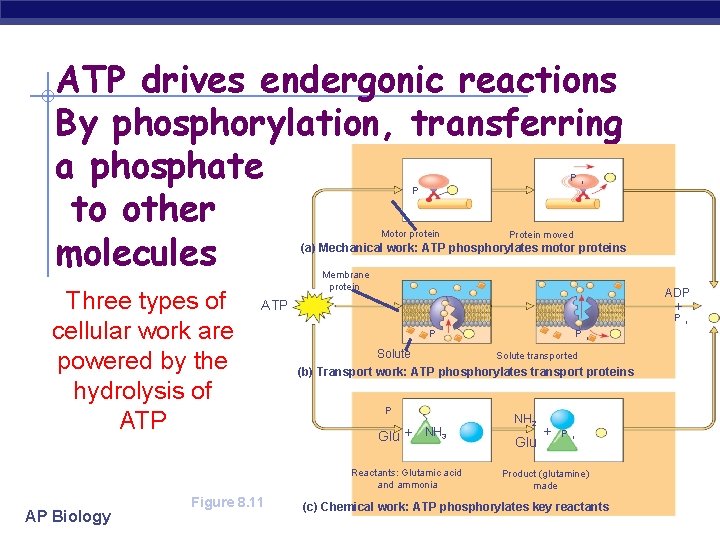
ATP drives endergonic reactions By phosphorylation, transferring a phosphate to other molecules P P Motor protein i Protein moved (a) Mechanical work: ATP phosphorylates motor proteins Three types of cellular work are powered by the hydrolysis of ATP Membrane protein ADP ATP + P P AP Biology i Solute transported (b) Transport work: ATP phosphorylates transport proteins P Glu + NH 3 Reactants: Glutamic acid and ammonia Figure 8. 11 P NH 2 Glu + P i Product (glutamine) made (c) Chemical work: ATP phosphorylates key reactants i
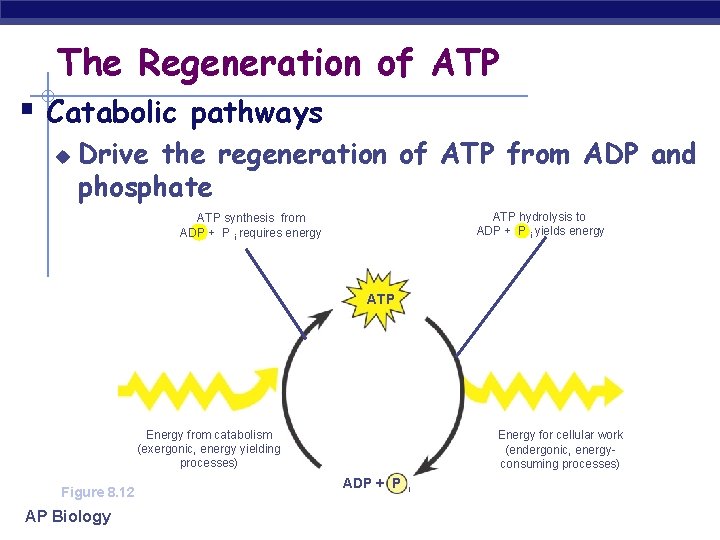
The Regeneration of ATP § Catabolic pathways u Drive the regeneration of ATP from ADP and phosphate ATP hydrolysis to ADP + P i yields energy ATP synthesis from ADP + P i requires energy ATP Energy from catabolism (exergonic, energy yielding processes) Figure 8. 12 AP Biology Energy for cellular work (endergonic, energyconsuming processes) ADP + P i
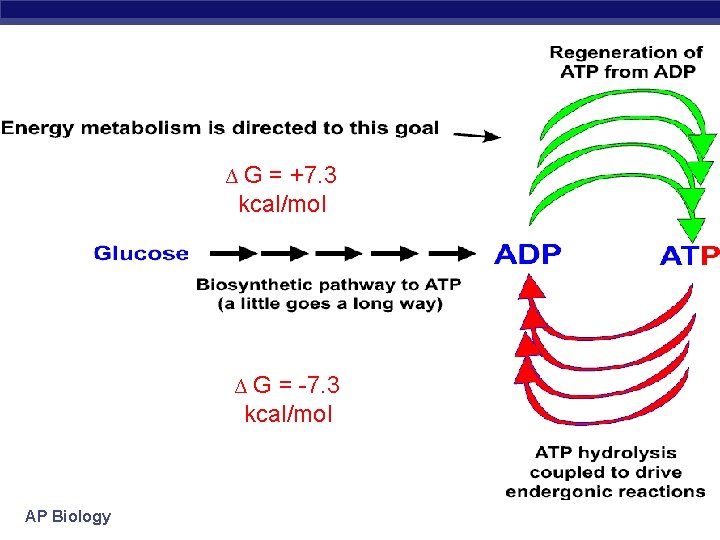
∆G = +7. 3 kcal/mol ∆G = -7. 3 kcal/mol AP Biology

Day 2 Enzymes and Activation Energy AP Biology
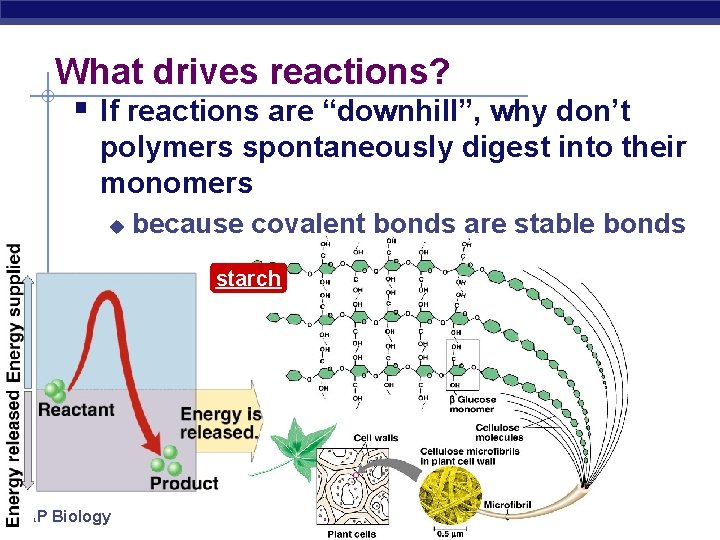
What drives reactions? § If reactions are “downhill”, why don’t polymers spontaneously digest into their monomers u because covalent bonds are stable bonds starch AP Biology

Activation energy § Breaking down large molecules requires an initial input of energy activation energy u large biomolecules are stable u must absorb energy to break bonds u AP Biology cellulose energy CO 2 + H 2 O + heat
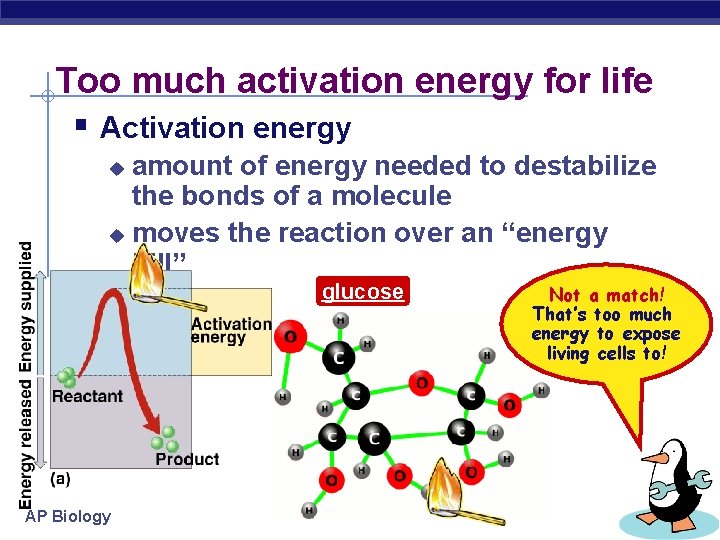
Too much activation energy for life § Activation energy amount of energy needed to destabilize the bonds of a molecule u moves the reaction over an “energy hill” u glucose AP Biology Not a match! That’s too much energy to expose living cells to!
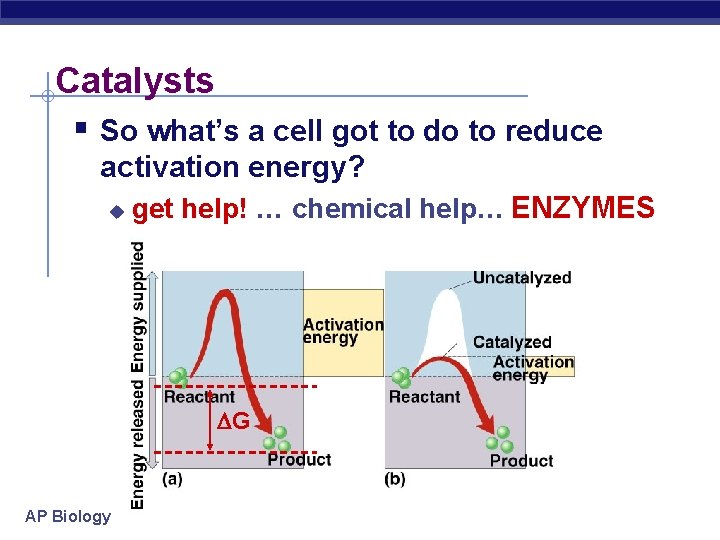
Catalysts § So what’s a cell got to do to reduce activation energy? u get help! … chemical help… ENZYMES G AP Biology

Reducing Activation energy § Enzymes are Biological Catalysts u reduce the amount of energy to start a reaction uncatalyzed reaction NEW activation energy reactant AP Biology product

Enzymes § are proteins (& RNA) u facilitate chemical reactions § increase rate of reaction without being consumed § reduce activation energy § don’t change free energy ( G) released or required u u required for most biological reactions highly specific § thousands of different enzymes in cells u AP Biology control reactions of life
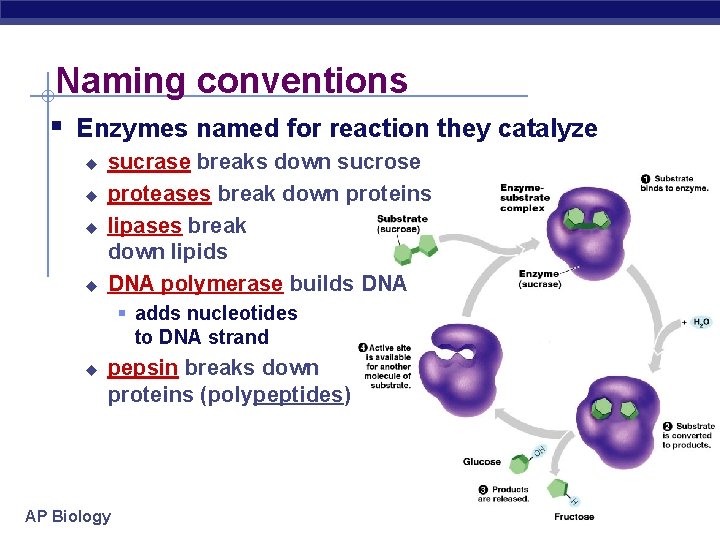
Naming conventions § Enzymes named for reaction they catalyze u u sucrase breaks down sucrose proteases break down proteins lipases break down lipids DNA polymerase builds DNA § adds nucleotides to DNA strand u pepsin breaks down proteins (polypeptides) AP Biology
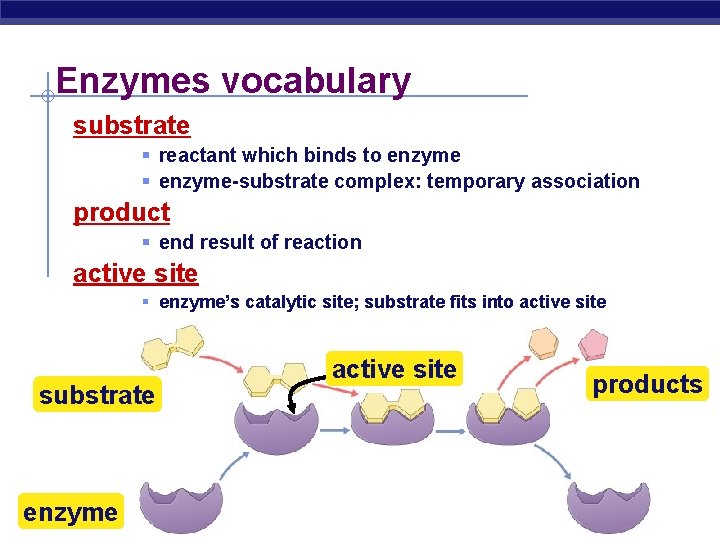
Enzymes vocabulary substrate § reactant which binds to enzyme § enzyme-substrate complex: temporary association product § end result of reaction active site § enzyme’s catalytic site; substrate fits into active site substrate enzyme AP Biology active site products

The active site can lower an EA barrier by: u Orienting substrates correctly § Synthesis: brings substrate closer together so they can bond to one another u Straining substrate bonds § Digestion: active site binds substrate and puts stress on bonds that must be broken, making it easier to separate molecules Providing a favorable microenvironment u Covalently bonding to the substrate u AP Biology

Properties of enzymes § Reaction specific u each enzyme works with a specific substrate § chemical fit between active site & substrate w H bonds & ionic bonds § Not consumed in reaction u single enzyme molecule can catalyze thousands or more reactions per second § enzymes unaffected by the reaction § Affected by cellular conditions u any condition that affects protein structure § temperature, p. H, salinity AP Biology
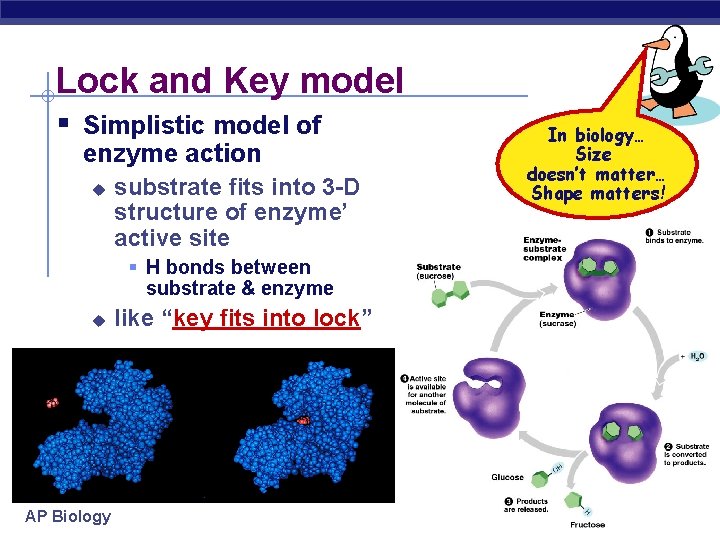
Lock and Key model § Simplistic model of enzyme action u substrate fits into 3 -D structure of enzyme’ active site § H bonds between substrate & enzyme u AP Biology like “key fits into lock” In biology… Size doesn’t matter… Shape matters!

Induced fit model § More accurate model of enzyme action 3 -D structure of enzyme fits substrate u substrate binding cause enzyme to change shape leading to a tighter fit u § “conformational change” § bring chemical groups in position to catalyze reaction AP Biology

Factors that Affect Enzymes AP Biology 2007 -2008
Factors Affecting Enzyme Function § Enzyme concentration § Substrate concentration § Temperature § p. H § Salinity § Activators § Inhibitors AP Biology catalase

Factors affecting enzyme function § Enzyme concentration u as enzyme = reaction rate § more enzymes = more frequently collide with substrate u reaction rate levels off reaction rate § substrate becomes limiting factor § not all enzyme molecules can find substrate AP Biology enzyme concentration

Factors affecting enzyme function § Substrate concentration u as substrate = reaction rate § more substrate = more frequently collide with enzyme u reaction rate levels off reaction rate § all enzymes have active site engaged § enzyme is saturated § maximum rate of reaction AP Biology substrate concentration

Factors affecting enzyme function § Temperature u Optimum T° § greatest number of molecular collisions § human enzymes = 35°- 40°C w body temp = 37°C u Heat: increase beyond optimum T° § increased energy level of molecules disrupts bonds in enzyme & between enzyme & substrate w H, ionic = weak bonds u § denaturation = lose 3 D shape (3° structure) Cold: decrease T° § molecules move slower § decrease collisions between enzyme & substrate AP Biology
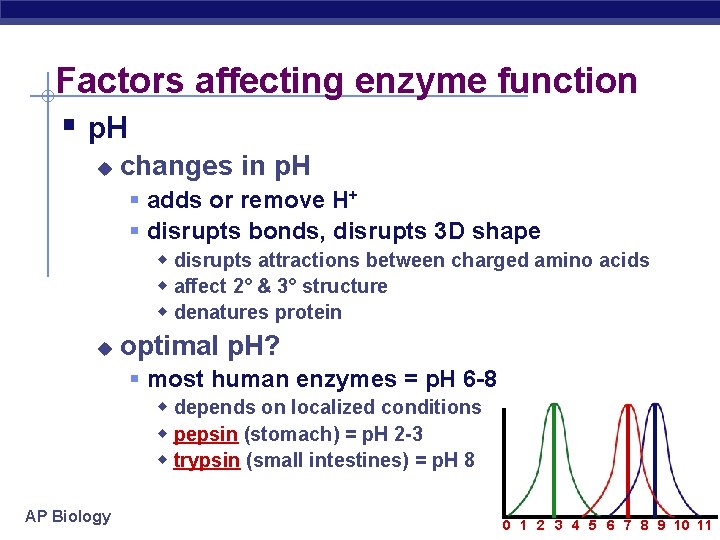
Factors affecting enzyme function § p. H u changes in p. H § adds or remove H+ § disrupts bonds, disrupts 3 D shape w disrupts attractions between charged amino acids w affect 2° & 3° structure w denatures protein u optimal p. H? § most human enzymes = p. H 6 -8 w depends on localized conditions w pepsin (stomach) = p. H 2 -3 w trypsin (small intestines) = p. H 8 AP Biology 0 1 2 3 4 5 6 7 8 9 10 11

Factors affecting enzyme function § Salt concentration u changes in salinity § adds or removes cations (+) & anions (–) § disrupts bonds, disrupts 3 D shape w disrupts attractions between charged amino acids w affect 2° & 3° structure w denatures protein u enzymes intolerant of extreme salinity § Dead Sea is called dead for a reason! AP Biology
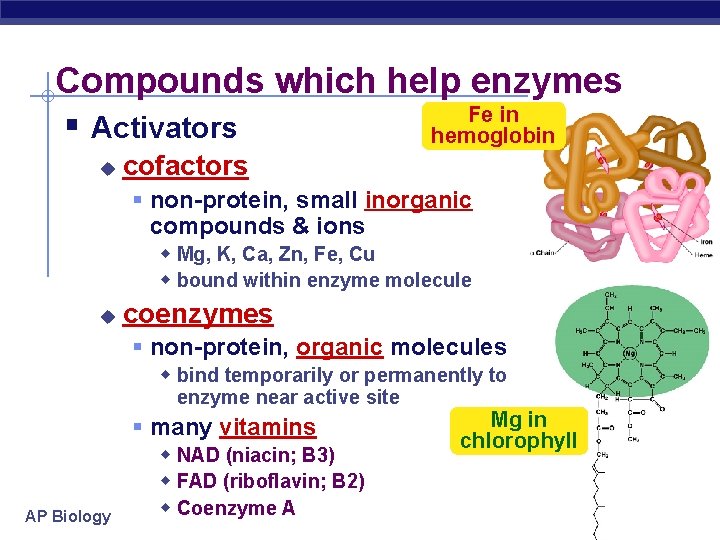
Compounds which help enzymes Fe in § Activators hemoglobin u cofactors § non-protein, small inorganic compounds & ions w Mg, K, Ca, Zn, Fe, Cu w bound within enzyme molecule u coenzymes § non-protein, organic molecules w bind temporarily or permanently to enzyme near active site AP Biology § many vitamins w NAD (niacin; B 3) w FAD (riboflavin; B 2) w Coenzyme A Mg in chlorophyll

Compounds which regulate enzymes § Inhibitors molecules that reduce enzyme activity u competitive inhibition u noncompetitive inhibition u irreversible inhibition u feedback inhibition u AP Biology

Competitive Inhibitor § Inhibitor & substrate “compete” for active site u u penicillin blocks enzyme bacteria use to build cell walls disulfiram (Antabuse) treats chronic alcoholism § blocks enzyme that breaks down alcohol § severe hangover & vomiting 5 -10 minutes after drinking § Overcome by increasing substrate concentration u AP Biology saturate solution with substrate so it out-competes inhibitor for active site on enzyme

Non-Competitive Inhibitor § Inhibitor binds to site other than active site u u u allosteric inhibitor binds to allosteric site causes enzyme to change shape § conformational change § active site is no longer functional binding site w keeps enzyme inactive some anti-cancer drugs inhibit enzymes involved in DNA synthesis § stop DNA production § stop division of more cancer cells u cyanide poisoning irreversible inhibitor of Cytochrome C, an enzyme in cellular respiration § stops production of ATP AP Biology

Irreversible inhibition § Inhibitor permanently binds to enzyme u competitor § permanently binds to active site u allosteric § permanently binds to allosteric site § permanently changes shape of enzyme § nerve gas, sarin, many insecticides (malathion, parathion…) w cholinesterase inhibitors n AP Biology doesn’t breakdown the neurotransmitter, acetylcholine

Allosteric regulation § Conformational changes by regulatory molecules u inhibitors § keeps enzyme in inactive form u activators § keeps enzyme in active form AP Biology. Conformational changes Allosteric regulation
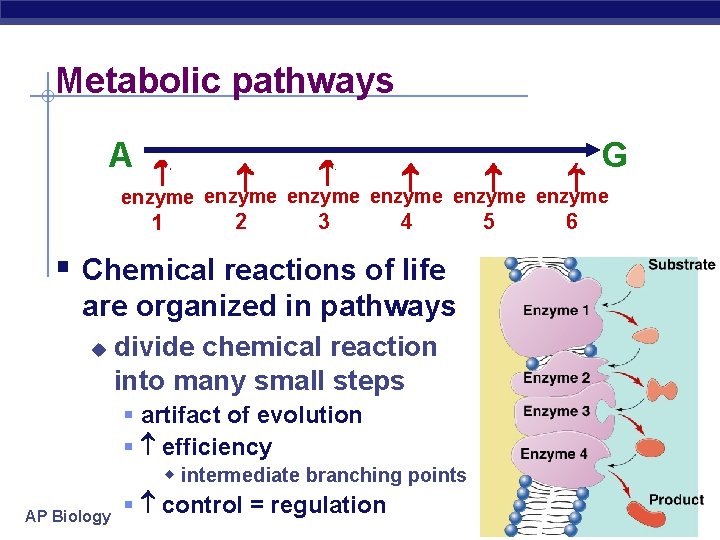
Metabolic pathways A B C D E F G 1 2 3 4 5 6 enzyme enzyme § Chemical reactions of life are organized in pathways u AP Biology divide chemical reaction into many small steps § artifact of evolution § efficiency w intermediate branching points § control = regulation
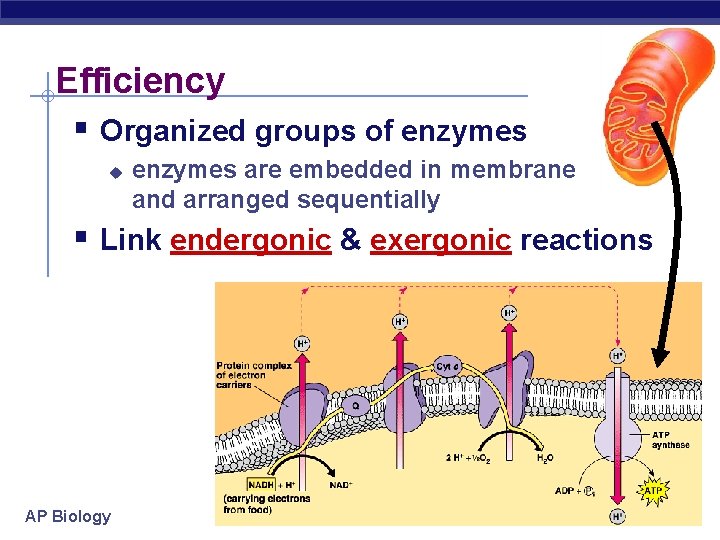
Efficiency § Organized groups of enzymes u enzymes are embedded in membrane and arranged sequentially § Link endergonic & exergonic reactions AP Biology

Feedback Inhibition § Regulation & coordination of production u u product is used by next step in pathway final product is inhibitor of earlier step § allosteric inhibitor of earlier enzyme § feedback inhibition u no unnecessary accumulation of product A B C D E F G 1 2 3 4 5 6 X enzyme enzyme AP Biology allosteric inhibitor of enzyme 1
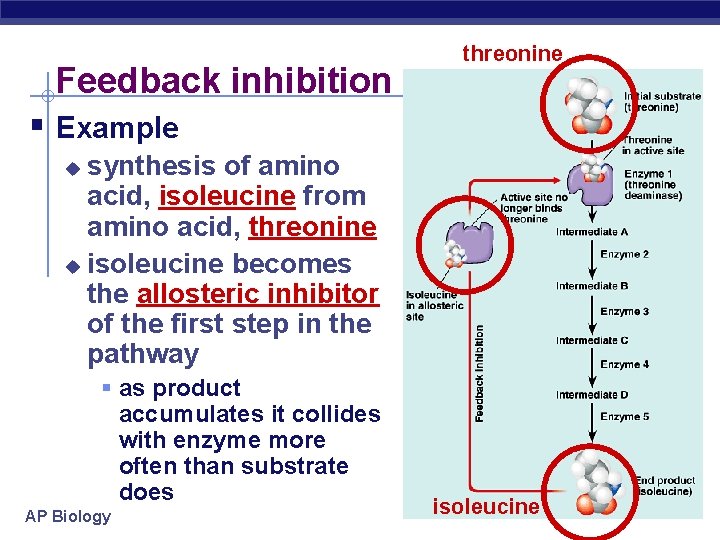
Feedback inhibition threonine § Example synthesis of amino acid, isoleucine from amino acid, threonine u isoleucine becomes the allosteric inhibitor of the first step in the pathway u § as product accumulates it collides with enzyme more often than substrate does AP Biology isoleucine

Review Feedback Loops § Learned about feedback loops at the start of school (August) Negative Feedback Loops u Positive Feedback Loops u § The END! AP Biology
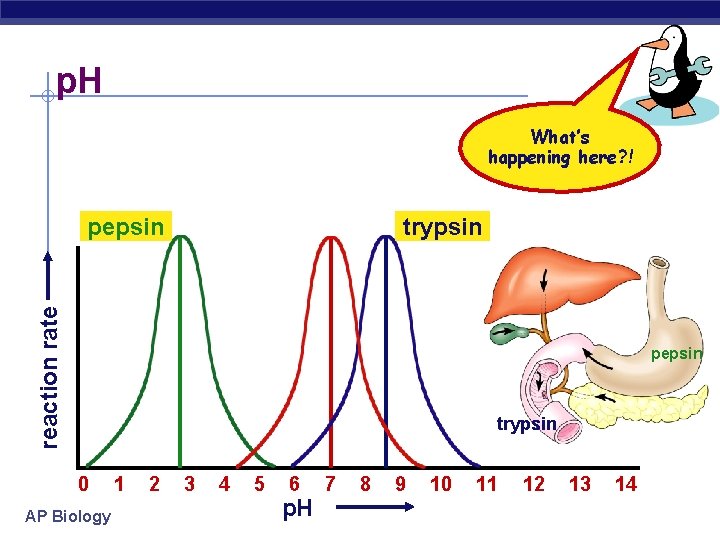
p. H What’s happening here? ! trypsin reaction rate pepsin trypsin 0 AP Biology 1 2 3 4 5 6 p. H 7 8 9 10 11 12 13 14
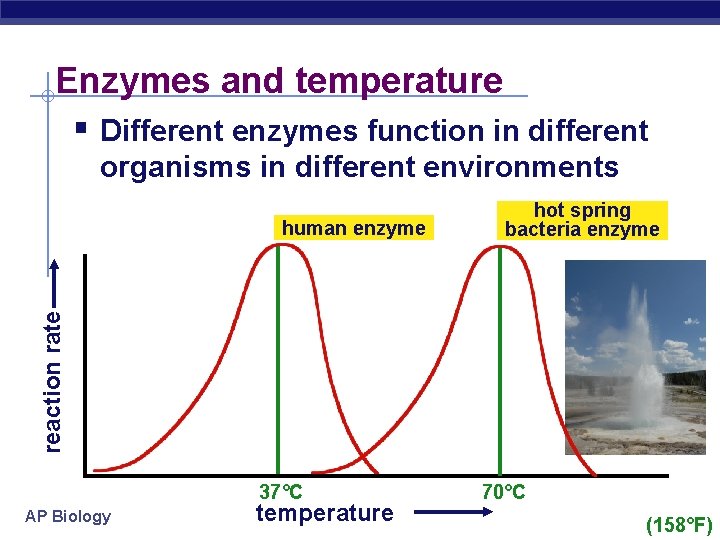
Enzymes and temperature § Different enzymes function in different organisms in different environments reaction rate human enzyme hot spring bacteria enzyme 37°C AP Biology temperature 70°C (158°F)
- Slides: 55