Metabolism Campbell and Reece Chapter 8 Metabolism total

Metabolism Campbell and Reece Chapter 8

Metabolism • total sum of all chemical reactions in an organism

Metabolic Pathways • begin with specific molecule which is altered in series of defined steps, resulting in certain product(s) • each step has own specific enzyme • mechanisms that regulate enzymes balance metabolic supply & demand
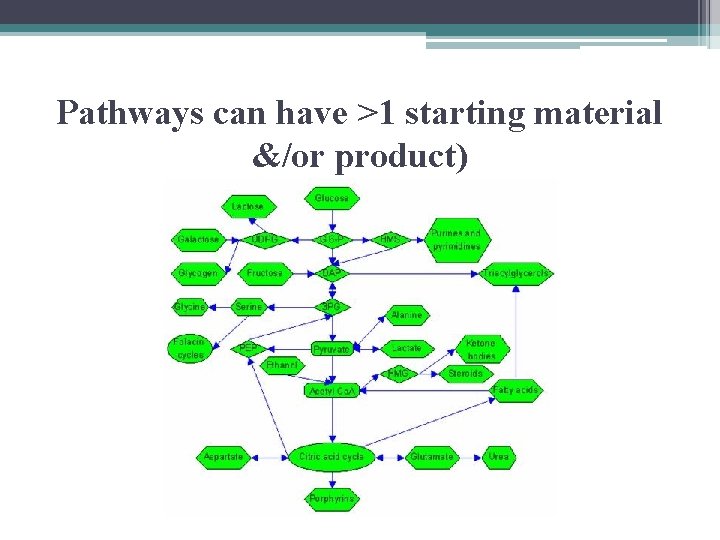
Pathways can have >1 starting material &/or product)
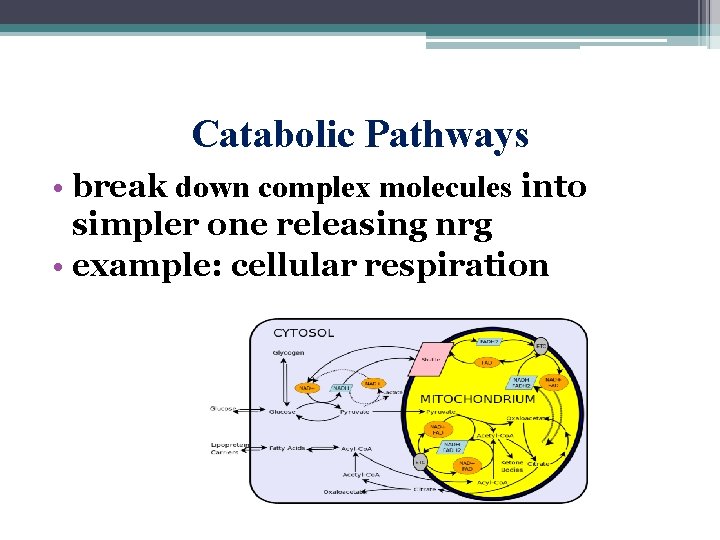
Catabolic Pathways • break down complex molecules into simpler one releasing nrg • example: cellular respiration

Anabolic Pathways • build complex molecules out of simpler ones • require nrg • example: protein synthesis • nrg released from catabolic pathways used to fuel anabolic pathways
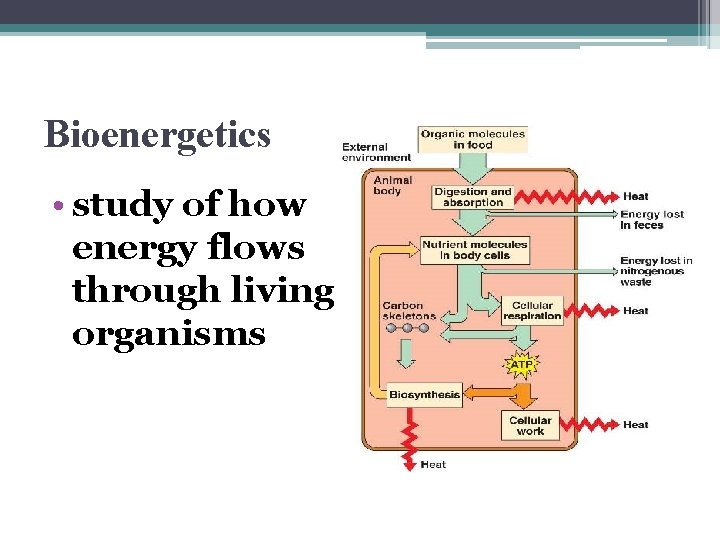
Bioenergetics • study of how energy flows through living organisms

Forms of Energy (capacity to do work) 1. KE 2. Thermal Energy 3. PE 4. Chemical Energy

KE (energy of motion)

Thermal Energy (heat) • KE ass’c with random movement of atoms or molecules

Light Energy

PE (energy matter possesses due to its position)
Chemical Energy (type of PE) • in catabolic pathways

Laws of Energy Transfer • thermodynamics: study of energy transfer that occurs in a system • system: matter being studied • surroundings: everything else in the universe • isolated or closed system: has no interaction with surroundings • open system: energy & matter can be transferred between system & surroundings

1 st Law of Thermodynamics (Law of Conservation of Energy) • amt of energy in universe is constant • Energy can neither be created or destroyed

1 st Law of Thermodynamics

2 nd Law of Thermodynamics (Entropy) • in most nrg transfers, some nrg is lost to the system, usually in form of heat nrg • in case of digesting food, most of chemical nrg is lost as heat • logical consequence of losing nrg with each transfer: universe is becoming more disordered

2 nd Law of Thermodynamics • entropy : a measure of the disorder or randomness of the universe • 2 nd Law: every nrg transfer or transformation increases the entropy of the universe • (there is an unstoppable trend toward randomization of the universe)

2 nd Law of Thermodynamics • spontaneous process: a process that can occur w/out input of energy ▫ will always increase entropy of the universe



• Biologists use the Laws of Thermodynamics to predict which chemical reactions will happen spontaneously & which one require an input of nrg

J. Willard Gibbs • Yale professor, 1878

Gibbs Free-Energy (G) • is the part of a system’s nrg that can perform work when the T & P are uniform thru out the system (living cell fits this description)

Gibbs Free Energy • system is the chemical reaction • enthalpy = total energy (in biologic systems) • ΔH = change in system’s enthalpy • ΔS = change in system’s entropy • absolute temperature (K) = ºC + 273

Free-Energy Change (ΔG) ΔG = ΔH - TΔS • 0 nce you know ΔG you can say whether a reaction will be spontaneous or require an input of nrg • *to be spontaneous ΔG must be (-) • for ΔG to be (-), either ΔH must be (-) (enthalpy decreases) or TΔS must be (+) (entropy increases)


Spontaneous Reactions • all spontaneous reactions have a –ΔG which decreases the system’s free nrg • reactions with a (+) G or G = 0 are never spontaneous

Another Way to Look at ΔG • ΔG = G (final state) + G(initial state) • because G(final state) has less free nrg it will be less likely to change thus the system will be more stable • think of free nrg as a measure of a system’s instability • unstable systems have higher G tend to change in such a way as to end with a more stable, lower G

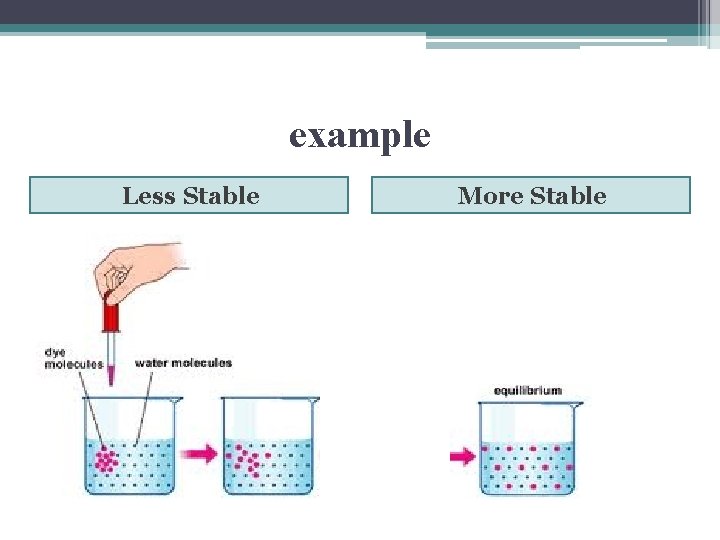
example Less Stable More Stable

Equilibrium • state of maximum stability • as a chemical reaction moves toward equilibrium the free nrg of reactants & products gradually decreases • free nrg increases if reactants & products somehow pulled away from equilibrium (removing products from system)

• for a system @ equilibrium, G is @ its lowest possible value in that system • any change from that equilibrium position will have a (+) ΔG & so will not be spontaneous

• *because a system @ equilibrium cannot spontaneously change, it cannot do work • A process is spontaneous & can do work only when it is moving toward equilibrium.

Free Energy & Metabolism • exergonic reaction: energy outward ▫ proceeds with net release of free nrg • endergonic reaction: energy inward ▫ proceeds only if absorption of free nrg ▫ reversible chemical reactions must be endergonic in one direction & exergonic in the other direction

Exergonic Reactions • reactions that occur spontaneously • ΔG is always (-) (reaction loses free nrg) • example: • 1 M C 6 H 12 O 6 + 6 O 2 6 CO 2 + 6 H 2 O + 686 kcal (2, 870 k. J)

• BREAKING BONDS OF REACTANTS DOES NOT RELEASE ENERGY… (IT REQUIRES ENERGY)

ENDERGONIC REACTIONS • absorbs free energy from surroundings • G increases so can think of it as reaction that stores free energy • ΔG is always (+) (amount of G tells you how much energy needed to drive reaction) • nonspontaneous reactions

Endergonic Reactions • example: ▫ if cellular respiration of glucose yielded 686 kcal of energy then…. ▫ plants had to add 686 kcal energy to make the glucose


Equilibrium & Metabolism • reactions that have reached equilibrium cannot do work • chemical reactions of metabolism would reach equilibrium if they occurred in isolation (like in a test tube)

• If a cell reaches equilibrium it is dead!

How do Cells Avoid Equilibrium? • products of reactions do not accumulate because they are used as reactants in the next reaction

How do Cells Avoid Equilibrium? • Sequence of reactions keeps going because there is a large free -energy difference between the original reactant(s) and final product(s)

Catabolic Pathways • energy released in “little packets” • if reaction simply started with original reactant(s ) final product(s) in a single step releasing all the energy at once would probably be catastrophic for the cell (and maybe the body)

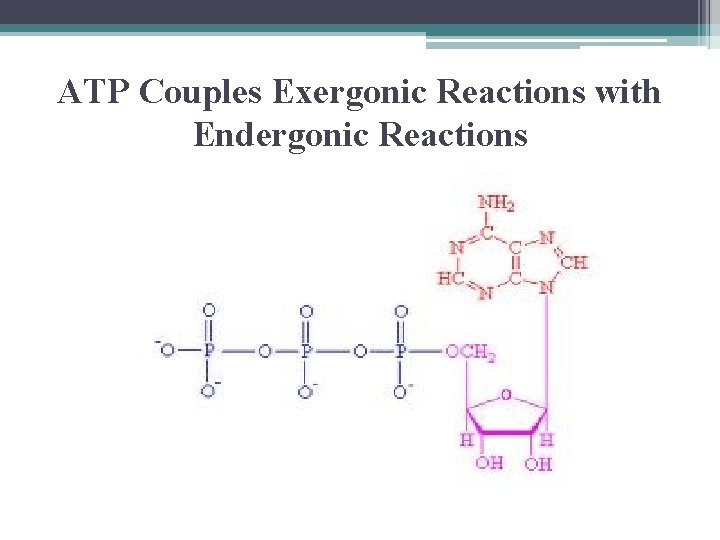
ATP Couples Exergonic Reactions with Endergonic Reactions

3 Kinds of Work Done by Cells 1. Chemical Work 2. Transport Work 3. Mechanical Work

Chemical Work • pushing endergonic reactions that do not occur spontaneously • example: monomers polymers

Transport Work • moving substances across membranes against their concentration gradients • example:

Mechanical Work • action of cells moves something • examples: cilia beating, muscle fibers contracting, chromosomes moving in anaphase • http: //biology. berkeley. edu/bio 1 a/topic/ Muscle_Motility/actin_myosin. html

Energy Coupling • * cells use an exergonic process to drive an endergonic one • example: ion pump:
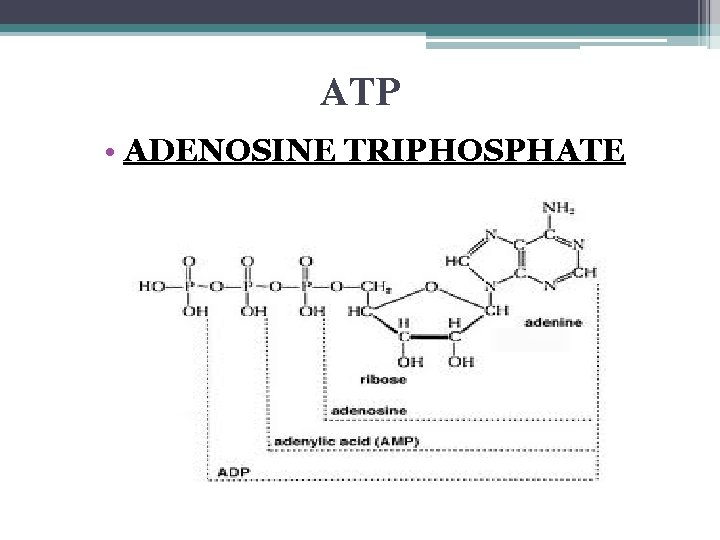
ATP • ADENOSINE TRIPHOSPHATE
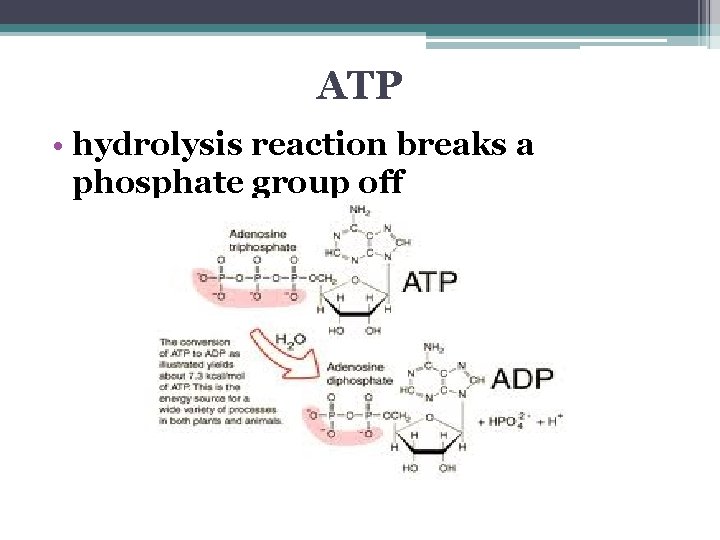
ATP • hydrolysis reaction breaks a phosphate group off

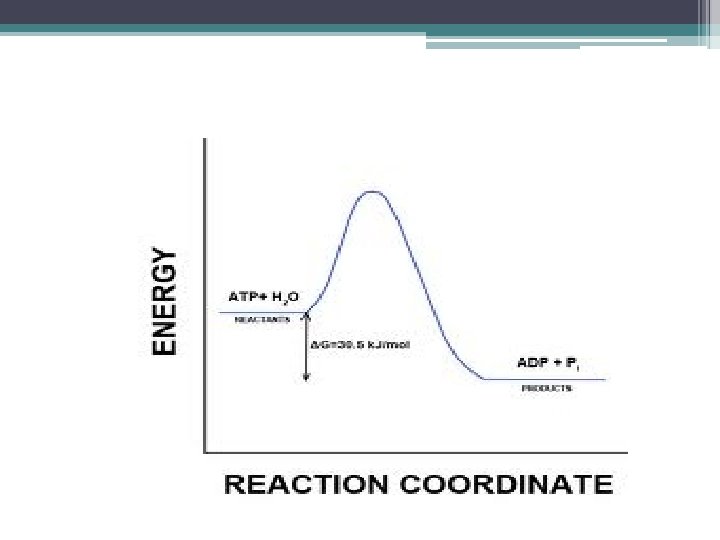
Hydrolysis of ATP • exergonic reaction • releases 7. 3 kcal / mole ATP so… the free energy change measured under standard conditions: • ΔG = -7. 3 kcal/mol

ATP Hydrolysis in Cells • not under standard conditions & not at concentrations of 1 M • under cellular conditions: ▫ ΔG = - 13 kcal / mol

Phosphate Bonds in ATP • are called “high-energy bonds” but that does not mean they are strong bonds • release of nrg comes from chemical change to a state of lower free energy (not from bonds themselves)
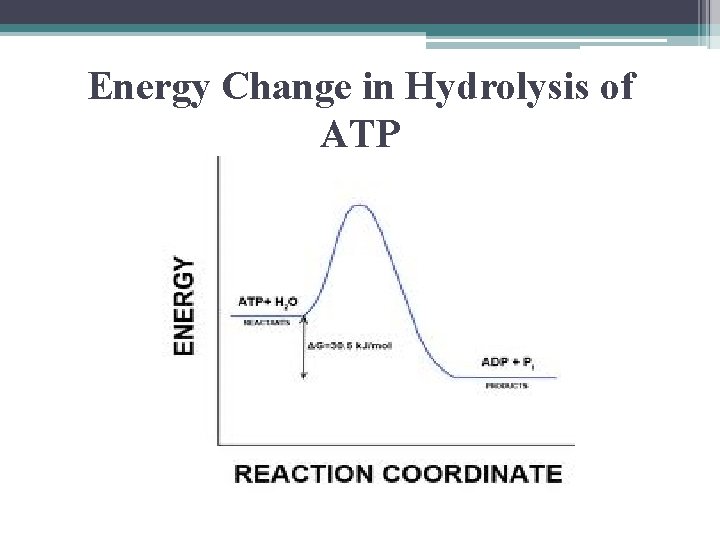
Energy Change in Hydrolysis of ATP

Why so much Energy in ATP? • 3 phosphate groups all (-) charged & close together so their mutual repulsion contributes to instability of this part of the ATP molecule • ATP w/all 3 phosphate groups


How Hydrolysis of ATP Performs Work • proteins harness the nrg in hydrolysis of ATP to perform the 3 types of cellular work: 1. Chemical 2. Transport 3. Mechanical

Coupling Reactions • if ΔG of endergonic reaction is < the amt of nrg released by ATP hydrolysis the 2 reactions can be coupled so…overall the reaction is exergonic
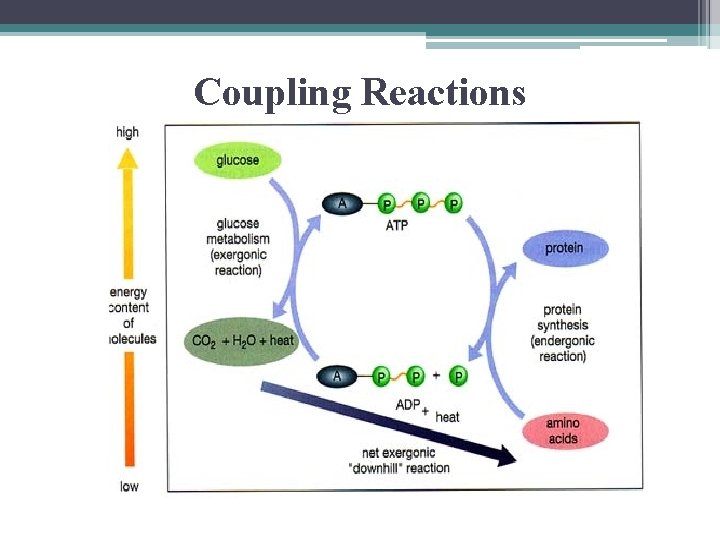
Coupling Reactions

Coupling Reactions • key involves formation of a phosphorylated intermediate which is more reactive (less stable) than the original unphosphorylated molecule
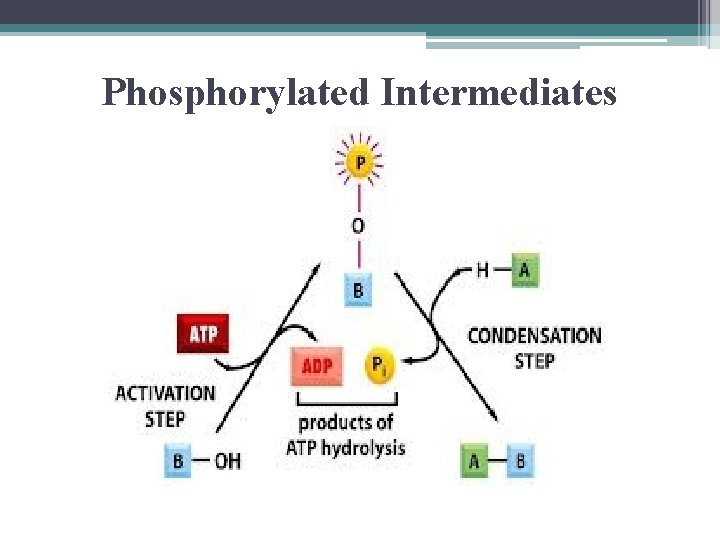
Phosphorylated Intermediates
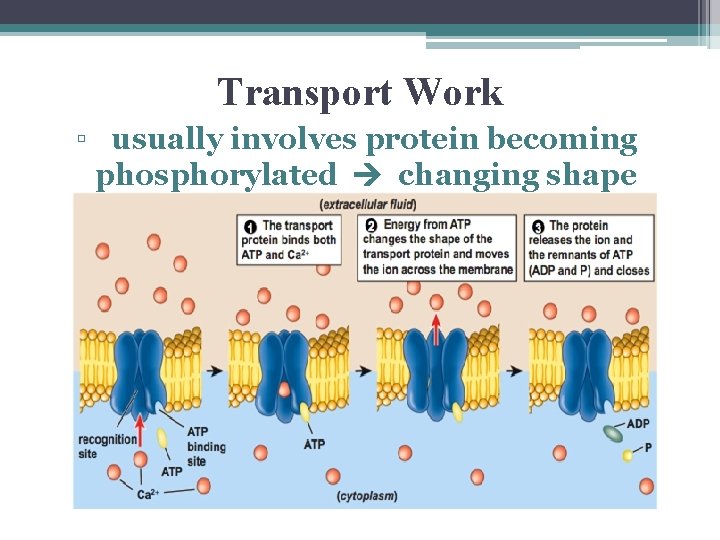
Transport Work ▫ usually involves protein becoming phosphorylated changing shape
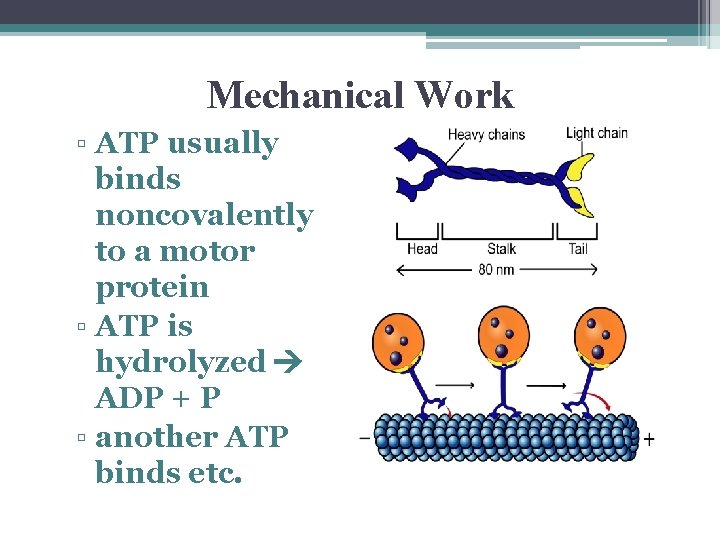
Mechanical Work ▫ ATP usually binds noncovalently to a motor protein ▫ ATP is hydrolyzed ADP + P ▫ another ATP binds etc.

Regeneration of ATP • free energy required to rephosphorylate ADP ATP comes from exergonic breakdown reactions (catabolic reactions)
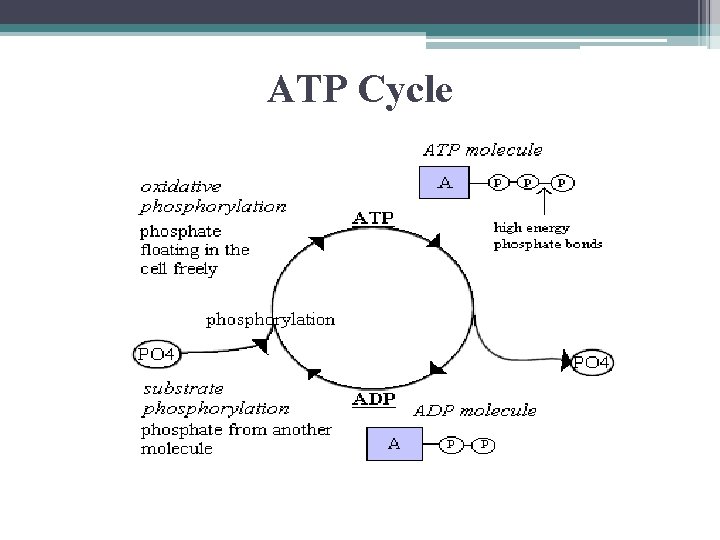
ATP Cycle


How Enzymes Work • Laws of Thermodynamics tell us which rxs will occur spontaneously but do not tell us how quickly • many rxs occur spontaneously but so slowly it is imperceptable ▫ example: sucrose + H 2 O glucose + fructose + 7 kcal/mol would have no perceptable change for years unless you add sucrase (then takes seconds)
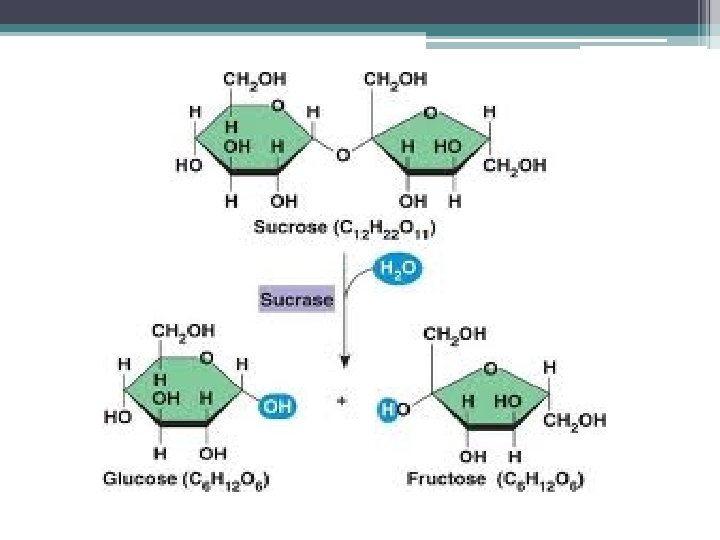

Enzymes • biologic catalysts that speed up chemical rxs w/out being consumed by the rx • macromolecules ▫ proteins ▫ ribozymes (RNA enzymes)

Activation Energy Barrier • to review: ▫ chemical rx involve breaking old bonds & making new one ▫ changing 1 molecule another usually involves contortion of reactant molecule into a highly unstable state b/4 reactants

Unstable State

Activation Energy Barrier • to reach the contorted state where bonds can change, reactants must absorb nrg from their surroundings • when new bonds formed in products nrg is released as heat & molecules return to stable shapes with lower nrg than they had in their contorted shape

Free Energy 0 f Activation Energy (EA ) • is nrg required to contort reactant molecules so bonds can break • often supplied in form of thermal nrg (absorbed by reactant from their environment)
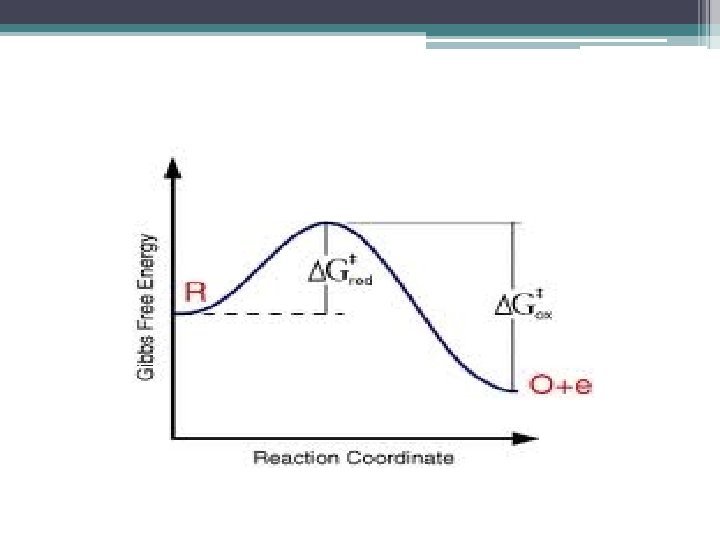

Activation Energy/ Unstable Transition State • as reactants absorb thermal nrg move faster collide more often & with more force • atoms w/in the reactant molecules more agitated so more likely bonds will break • both happen in the unstable transition state

Exergonic Reaction: occurs spontaneously (ΔG <0)

Activation Energy • in most cases EA is so high & transition state is reached so rarely that rx hardly ever product

How Enzymes Lower EA • many complex molecules (proteins, DNA) high in free nrg so you would think they have the potential to spontaneously decompose • doesn’t happen because temperatures typical for most cells do not provide enough energy to get over the EA hump

How Enzymes Lower EA • enzymes lower EA enabling reactants to absorb enough nrg to reach the transition state even at normal temperatures • *enzymes cannot change the ΔG for a reaction


Substrate Specificity of Enzymes • substrate: ▫ reactant an enzyme acts on • enzyme-substrate complex ▫ when enzyme is bound to subtrate ▫ enzymeenzyme + substrate + substrate complex product
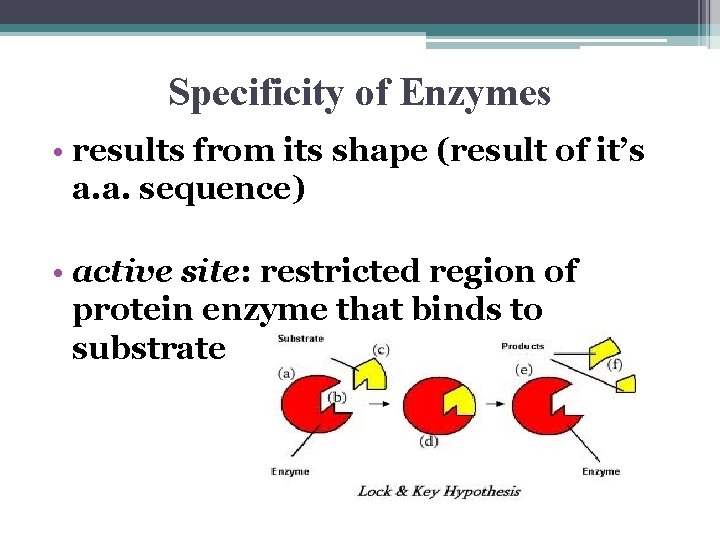
Specificity of Enzymes • results from its shape (result of it’s a. a. sequence) • active site: restricted region of protein enzyme that binds to substrate

Active Sites • proteins alter between different shapes in a dynamic equilibrium with slight differences in free nrg in each “pose” • shape that best fits the substrate not always the one with lowest free nrg
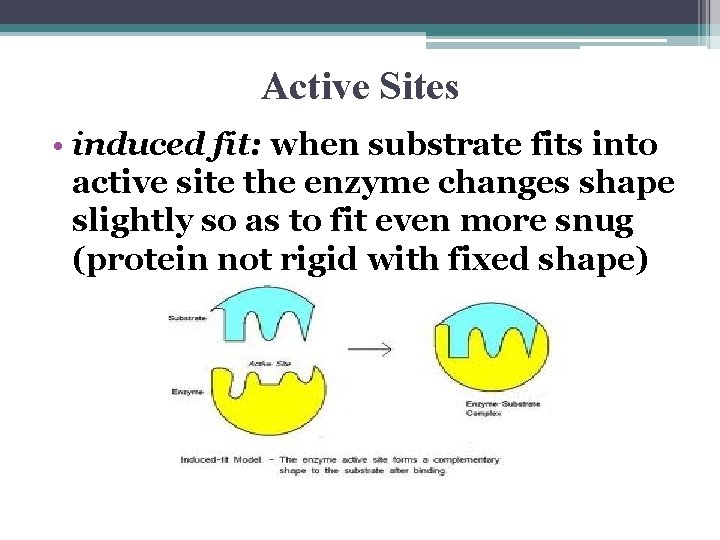
Active Sites • induced fit: when substrate fits into active site the enzyme changes shape slightly so as to fit even more snug (protein not rigid with fixed shape)

Catalysis in the Active Site • enzymes can catalyze either the forward or reverse reaction: depends on which direction has a ()G • which depends on the relative concentrations of reactants & products • net effect: reaction will move in direction that favors equilibrium

Mechanisms Used by Enzymes to Lower EA 1. if 2 or more reactants, active site positions them in proper orientation for rx to occur

Mechanisms Used by Enzymes to Lower EA 2. remember EA is proportional to the difficulty of breaking bonds, so having the active site stretch & bend the bonds toward their transitionstate form helps reduce the amt of free nrg that must be absorbed to achieve transition-state

Mechanisms Used by Enzymes to Lower EA 3. active site might provide a microenvironment more conducive to reaction than the solution would be w/out the enzyme if enzyme has acidic a. a. it would lower p. H of the solution in otherwise neutral cell

Mechanisms Used by Enzymes to Lower EA 4. direct participation of active site in chemical reaction (subsequent reactions restores active site to original state so active site same before & after)
![When Enzyme Becomes Saturated • can increase rate of reaction initially by increasing [substrate] When Enzyme Becomes Saturated • can increase rate of reaction initially by increasing [substrate]](http://slidetodoc.com/presentation_image_h2/885e304cdf8ad6b45af1a1b17c73f908/image-97.jpg)
When Enzyme Becomes Saturated • can increase rate of reaction initially by increasing [substrate] up to point where every enzyme has its active site occupied then increasing amt substrate will have no further effect • when that happens only way to increase rate of rx is to add more enzyme

Enzyme Animations • http: //highered. mcgrawhill. com/sites/0072495855/student_view 0/cha pter 2/animation__how_enzymes_work. html • http: //bcs. whfreeman. com/thelifewire/content/ chp 06/0602002. html

Effects of Local Conditions on Enzyme Activity 1. Temperature 2. p. H 3. cofactors 4. Enzyme Inhibitors

Effects of Temperature on Enzyme Activity • rate of enzyme activity increases with increasing temperature up to a point • as T increases so does KE of molecules so more collisions. . more likely substrate will find active site • when point reached speed of reaction drops sharply
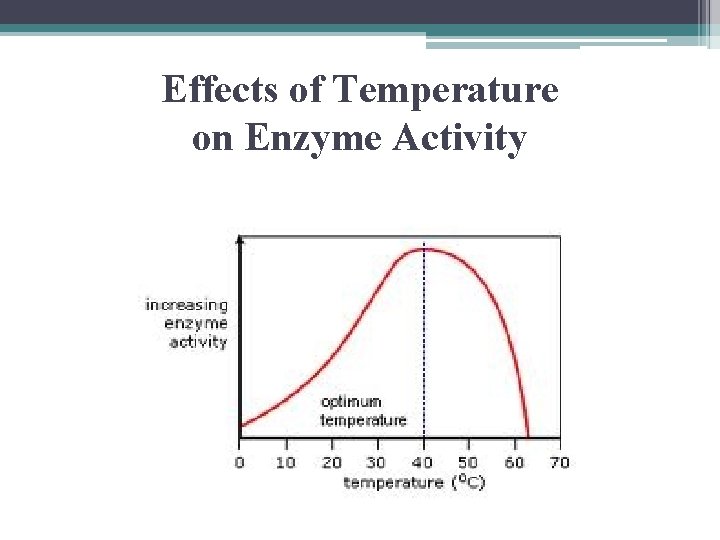
Effects of Temperature on Enzyme Activity

Effects of Temperature on Enzyme Activity • each enzyme has its optimal T @ which its rx rate is greatest • above some point the protein denatures • most human enzymes have optimal T @ 35 – 40 ºC • some thermophilic bacteria have enzymes with optimal activity @ 70 ºC

Effects of p. H on Enzyme Activity • each enzyme has optimal p. H • most enzymes have optimal p. H of 6 -8

Cofactors • nonprotein helpers for enzymes • might be 1. bound to the enzyme as permanent fixtures 2. bound loosely & reversibly like the substrate
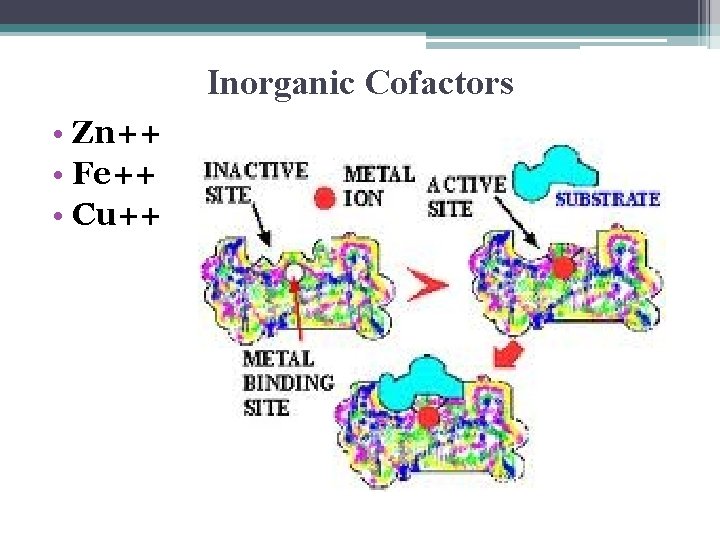
Inorganic Cofactors • Zn++ • Fe++ • Cu++

Organic Cofactors • called: coenzymes • most vitamins are essential because they act as coenzymes or are the raw material that a coenzyme is made from

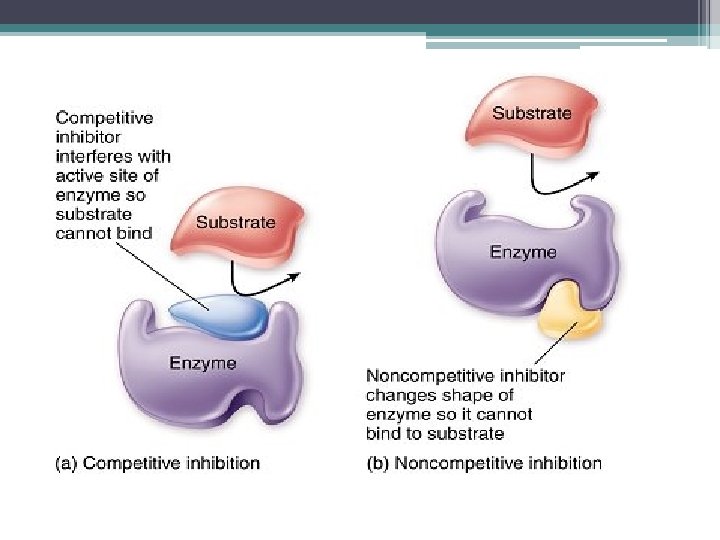
Enzyme Inhibitors certain chemicals that inhibit action of specific enzymes reversible: if inhibitor has weak, transient attachment irreversible: if inhibitor forms covalent bond with enzyme Types: 1. Competitive 2. Noncompetitive
![Competitive Inhibitors • inhibitor mimics substrate • can be overcome by increasing [substrate] Competitive Inhibitors • inhibitor mimics substrate • can be overcome by increasing [substrate]](http://slidetodoc.com/presentation_image_h2/885e304cdf8ad6b45af1a1b17c73f908/image-108.jpg)
Competitive Inhibitors • inhibitor mimics substrate • can be overcome by increasing [substrate]

Noncompetitive Inhibitor • do not compete with substrate @ active site • bind to another part of enzyme causes enzyme to change shape so active site becomes less effective

• http: //biologyanimations. blogspot. com/search/label/enzymes

Evolution of Enzymes • accepted Model: ▫ naturally occurring mutations that change a. a. in active site or some other crucial position ▫ then changes in environment so natural selection could tend to favor the mutation
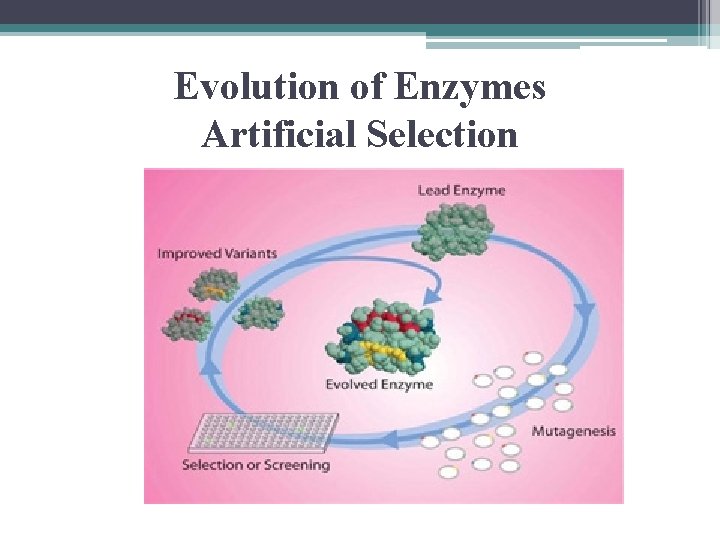
Evolution of Enzymes Artificial Selection
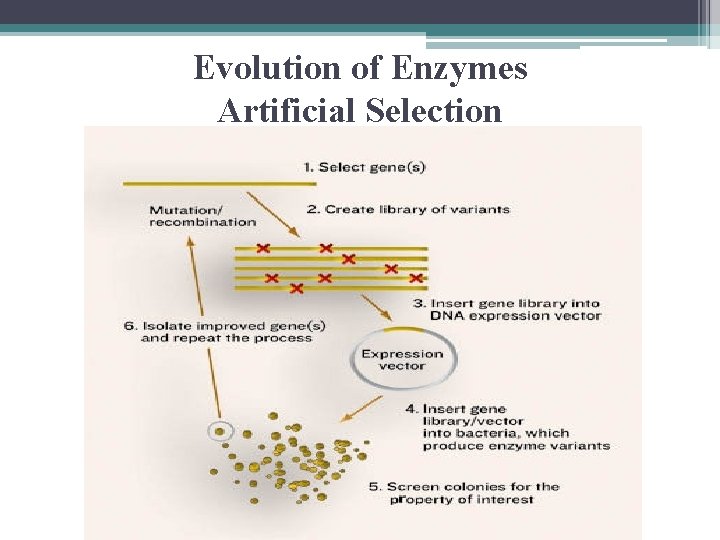
Evolution of Enzymes Artificial Selection


Regulation of Enzyme Activity • Cells must control which metabolic pathways are active when or there would be mass chaos. • control is achieved by controlling which enzymes are active when

Allosteric Regulation of Enzymes • allosteric regulation describes any case in which a protein’s function @ 1 site is affected by the binding of a regulatory molecule to a separate site
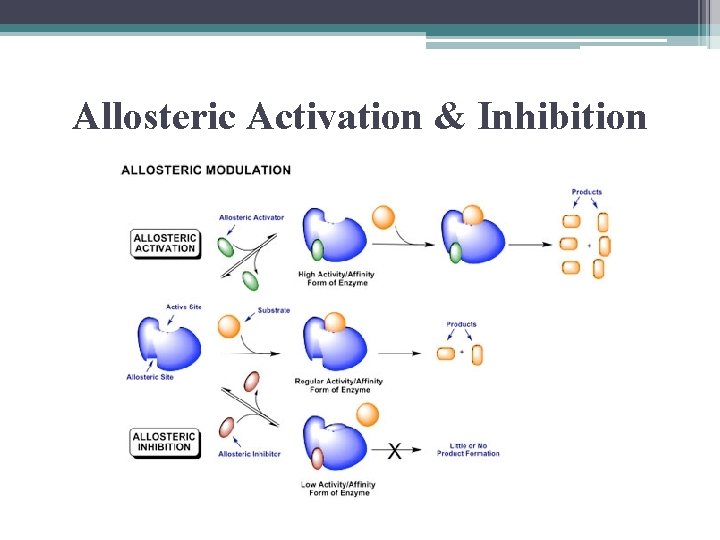
Allosteric Activation & Inhibition

Allosteric Regulators • few of many known metabolic enzymes have been shown to be regulated this way • pharmaceutical research uses this model to design medications to act as regulators (usually bind to enzyme longer than real regulators so are more effective)

Allosteric Regulators • ADP & P play role in balancing the flow of traffic between anabolic & catabolic pathways by their effects on key enzymes

• ATP binds to several catabolic enzymes allosterically, lowering their affinity for substrate & so inhibiting their activity • ADP & P activate same enzymes

• http: //programs. northlandcollege. edu/biology/ Biology 1111/animations/enzyme. html
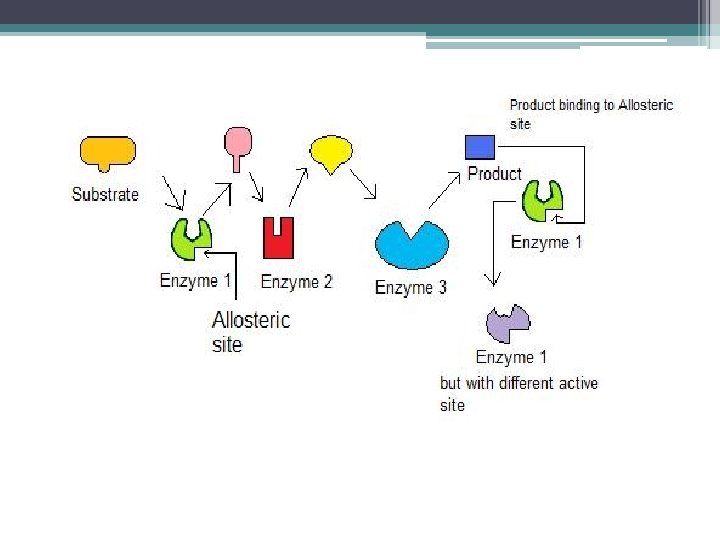

Cooperativity • substrate binds to 1 active site on multisubunit shape change in all subunits increases catalytic activity@ other sites • in effect, amplifies the response of enzymes to substrates
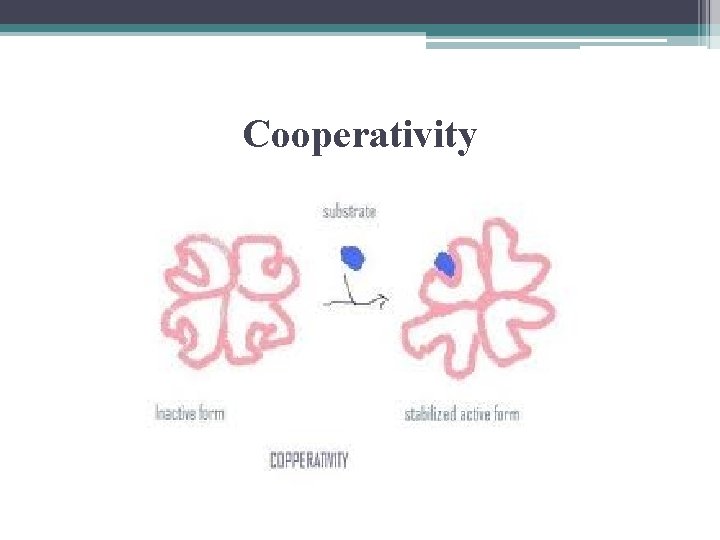
Cooperativity

Cooperativity also in Hgb • Hemoglobin (Hgb) made of 4 polypeptides (subunits) • each 1 has oxygen-binding site • O 2 binds to 1 site increases affinity for O 2 to other 3 sites

Hemoglobin • when Hgb in oxygen-poor capillary affinity of oxygen-binding sites decreases & O 2 released • as 1 O 2 leaves the affinity for others continues to decrease • http: //www. youtube. com/watch? v=WXO BJEXx. NEo

Feedback Inhibition • end product of metabolic pathway binds to enzyme early in pathway and switches off the pathway
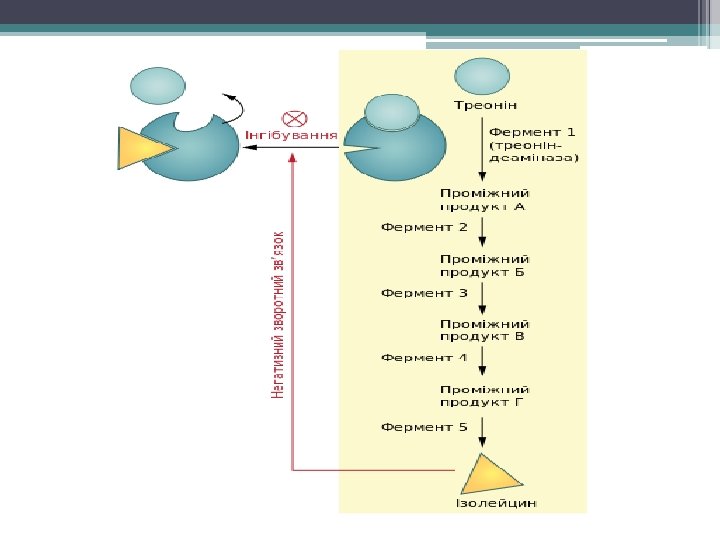

Cells are Compartmentalized • organelles bring order to metabolic pathways ▫ some have enzymes for several steps of pathway assembled in a multienzyme complex ▫ some have fixed locations in certain membranes ▫ some w/in membrane spaces

- Slides: 131