Metabolism Basic Concepts and Design Metabolism n n

Metabolism Basic Concepts and Design

Metabolism n n What is metabolism? u all the chemical reactions that take place in cells u series of energy transformations Why do living organism require energy? u to synthesize large molecules from small ones u to move substances in and out of cells u muscle contraction and cell movement

Metabolism n n What is the difference between phototrophs and chemotrophs? u phototrophs obtain energy from sunlight u chemotrophs obtain energy from breaking chemical bonds What is the difference between anabolic and catabolic reactions? u catabolic – fuels degraded to release useful energy u anabolic – energy used to synthesize biomolecules

Metabolism n Why are reactions often coupled in metabolic pathways? u to insure that the overall free energy change is negative t a reaction that requires energy may be paired with one that releases energy
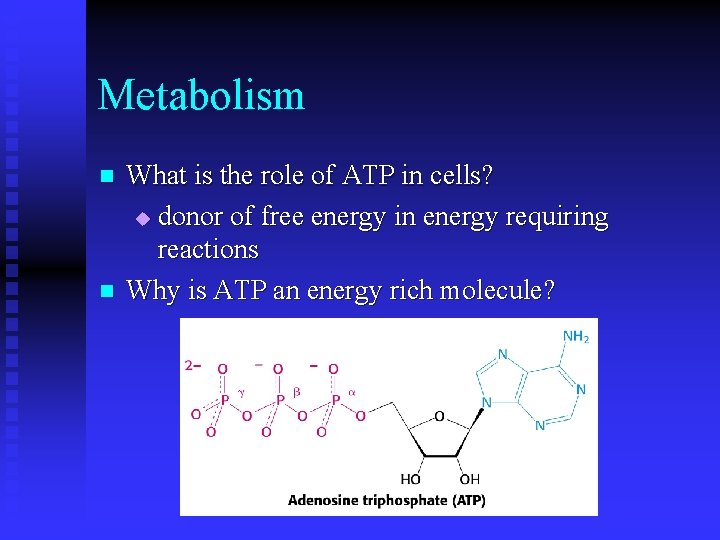
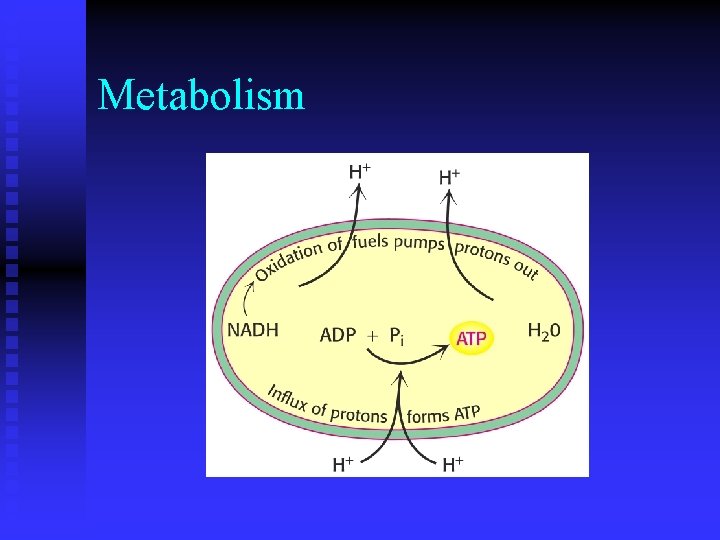
Metabolism n n What is the role of ATP in cells? u donor of free energy in energy requiring reactions Why is ATP an energy rich molecule?

Metabolism n What are the factors that contribute to the large amount of energy released when ATP is hydrolzed? u resonance stabilization u electrostatic repulsion u ease of hydration
Metabolism
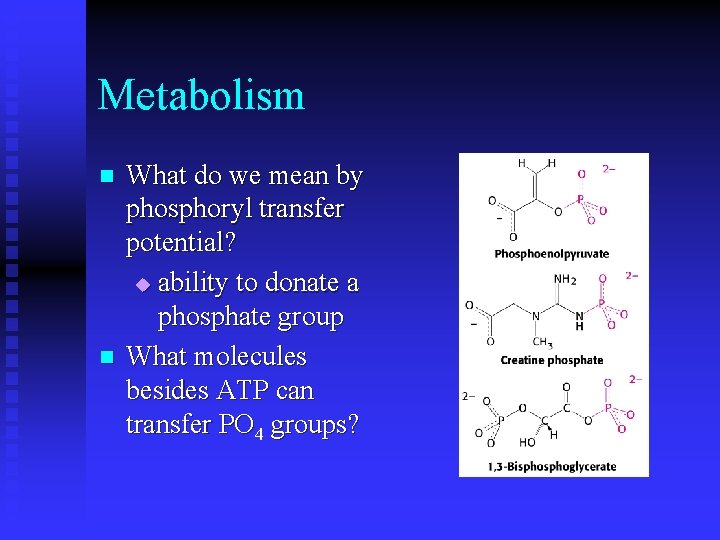
Metabolism n n What do we mean by phosphoryl transfer potential? u ability to donate a phosphate group What molecules besides ATP can transfer PO 4 groups?

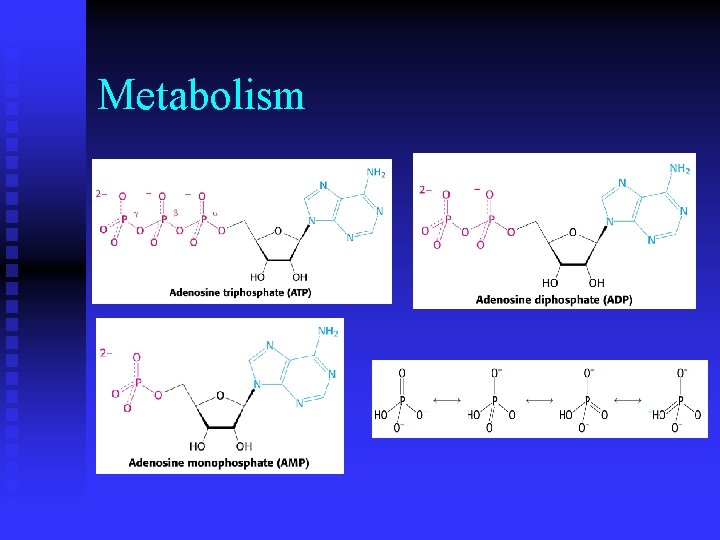
Metabolism n How is ATP regenerated in cells?
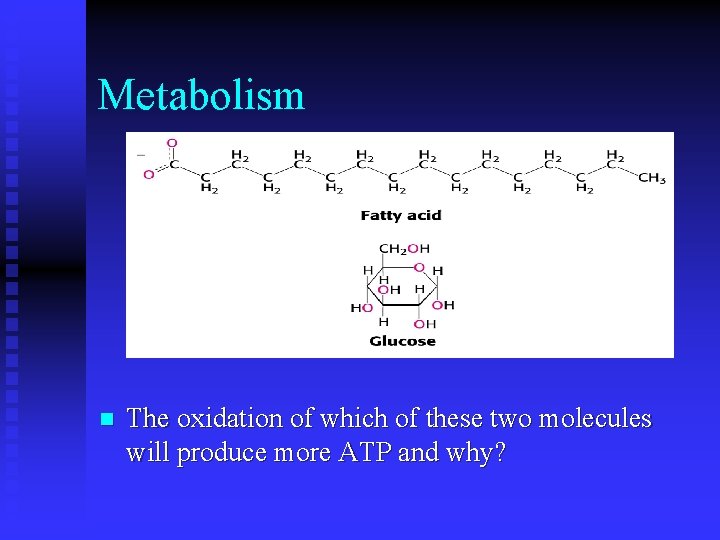
Metabolism n The oxidation of which of these two molecules will produce more ATP and why?

Metabolism • How do molecules with high phosphoryl transfer potential couple the oxidation of carbon to the synthesis of ATP? u glyceraldehyde-3 -PO 4 + NAD+ + HPO 4 1, 3 biphosphoglycerate + NADH + H+ u 1, 3 biphosphoglycerate + ADP 3 phosphoglyceric acid + ATP
Metabolism
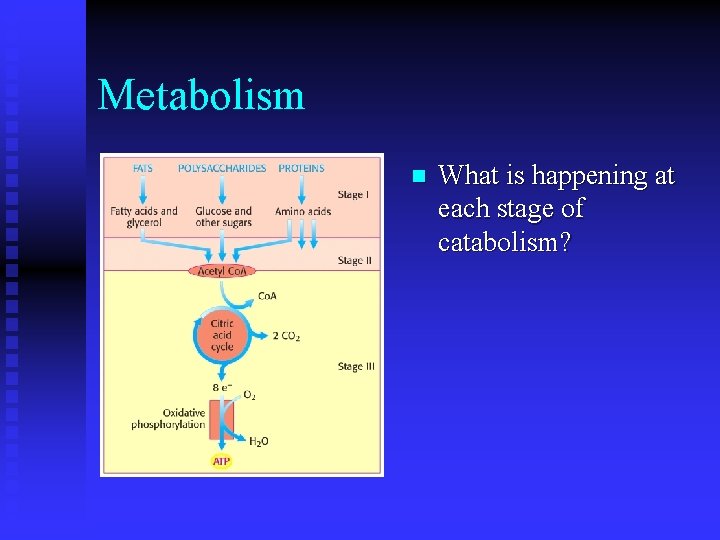
Metabolism n What is happening at each stage of catabolism?

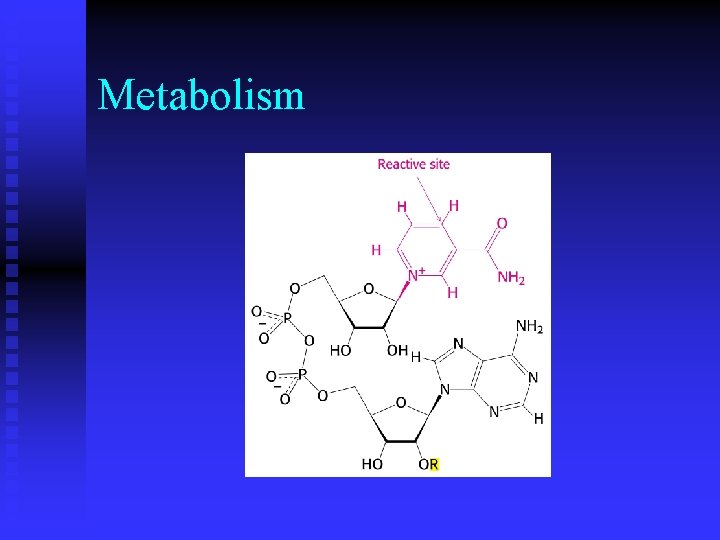
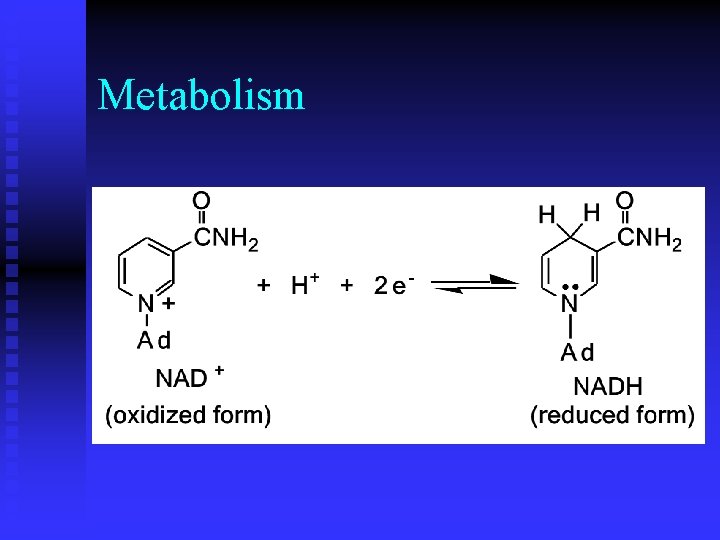
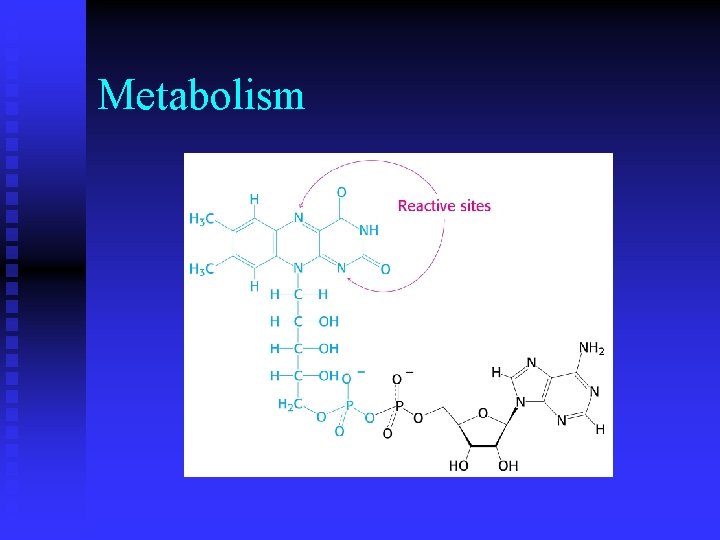
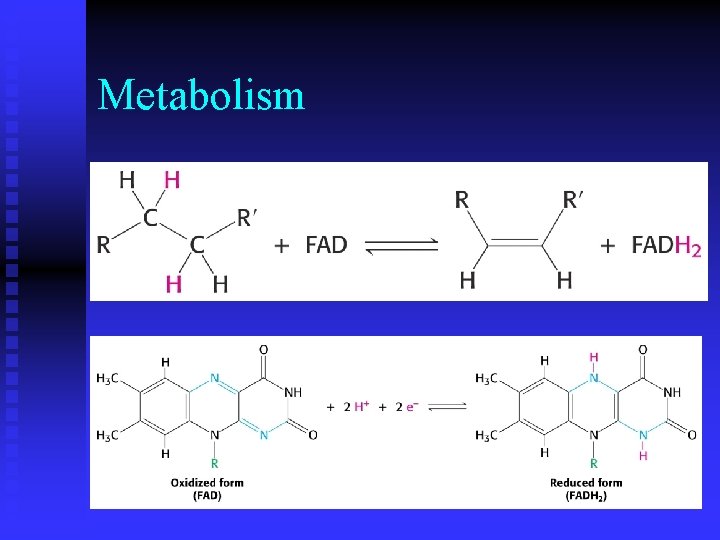
Metabolism n While ATP is an activated carrier of phosphoryl groups, what molecule(s) is (are) activated carrier(s) of electrons during oxidation? u nicotinamine adenine dinucleotide (NAD) u flavin adenine dinucleotide (FAD)
Metabolism
Metabolism
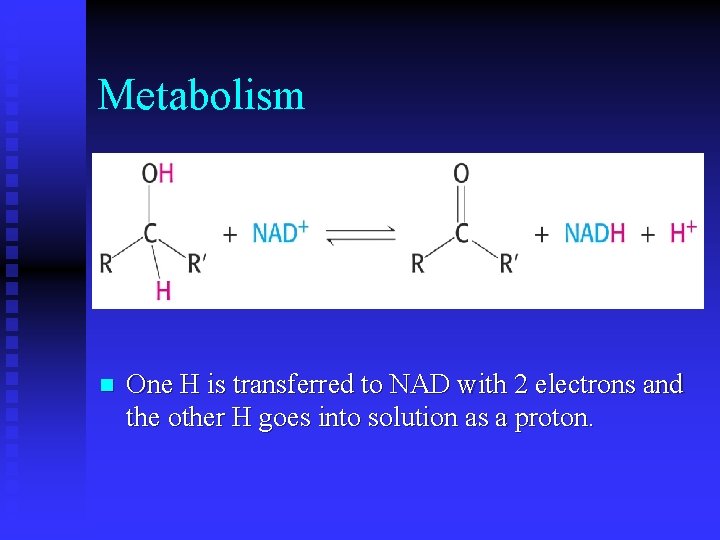
Metabolism n One H is transferred to NAD with 2 electrons and the other H goes into solution as a proton.
Metabolism
Metabolism
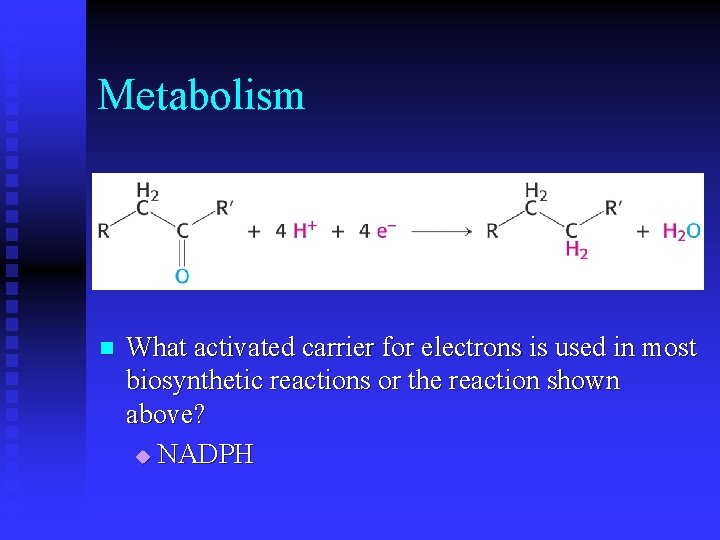
Metabolism n What activated carrier for electrons is used in most biosynthetic reactions or the reaction shown above? u NADPH
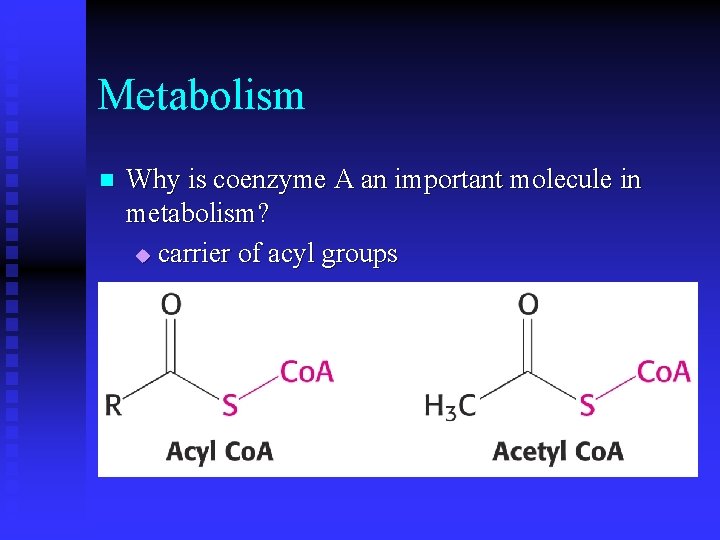
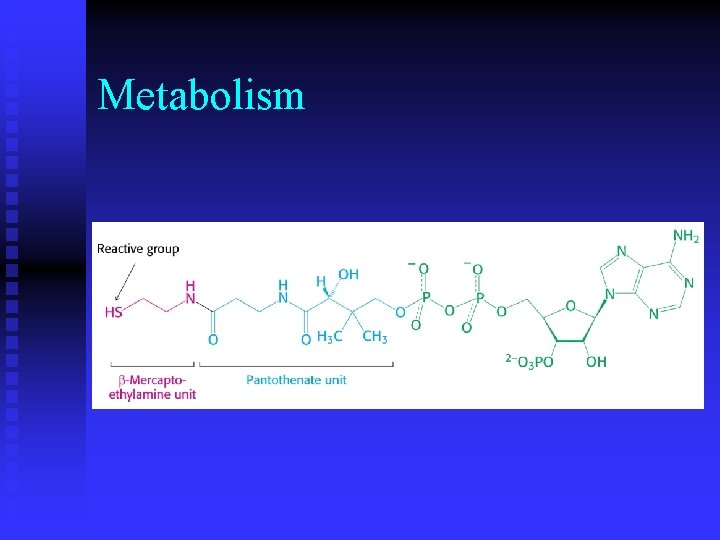
Metabolism n Why is coenzyme A an important molecule in metabolism? u carrier of acyl groups
Metabolism

Metabolism n While metabolism consists of thousands of reactions, each reaction falls into one of six categories.
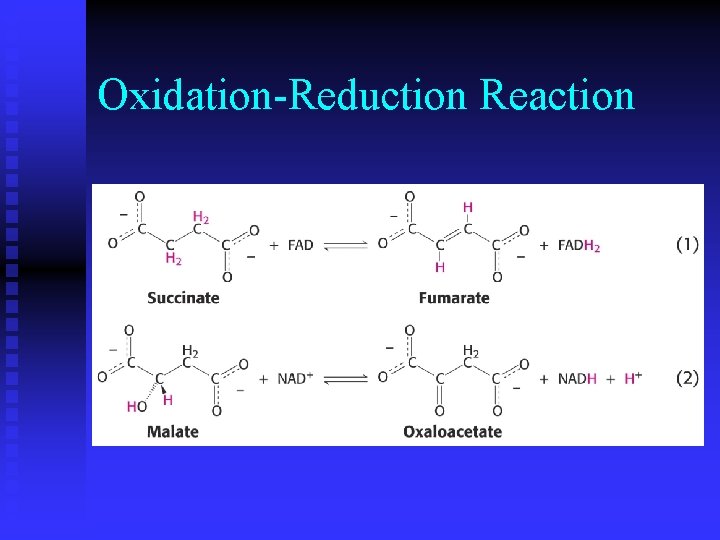
Oxidation-Reduction Reaction
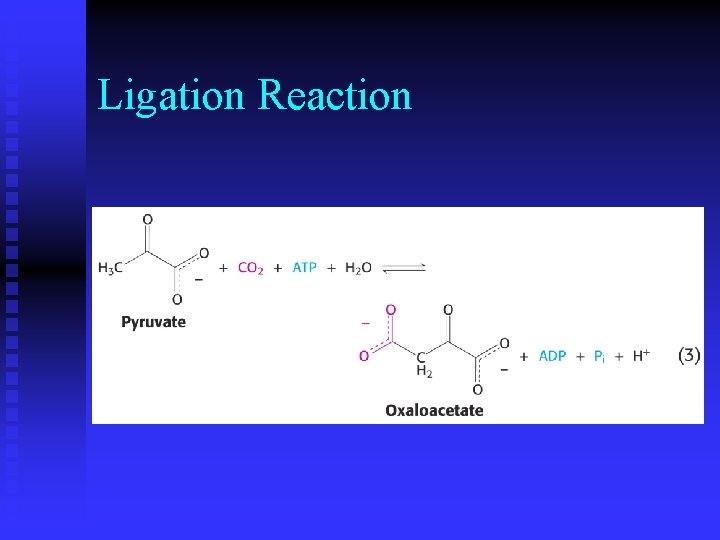
Ligation Reaction
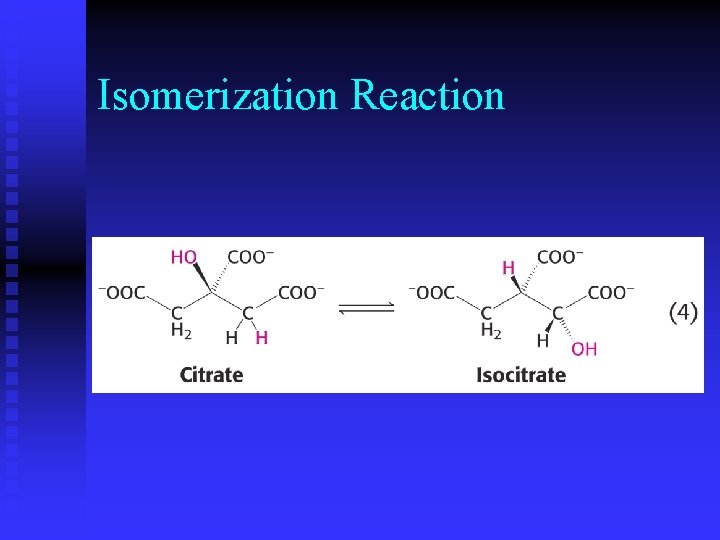
Isomerization Reaction
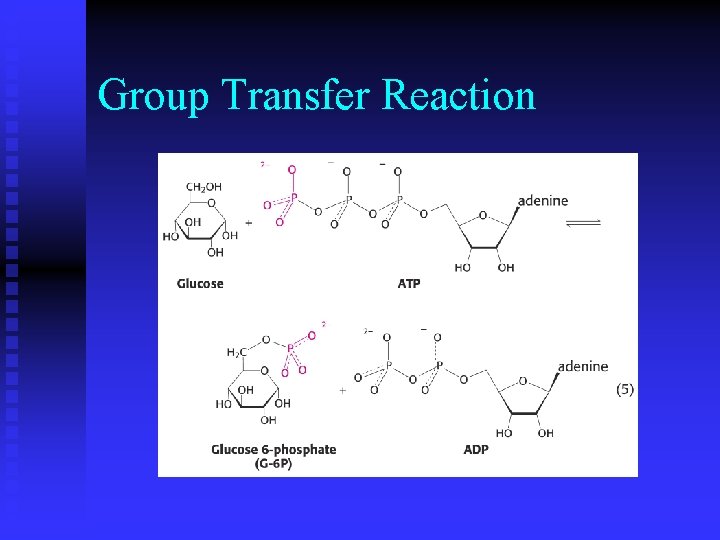
Group Transfer Reaction
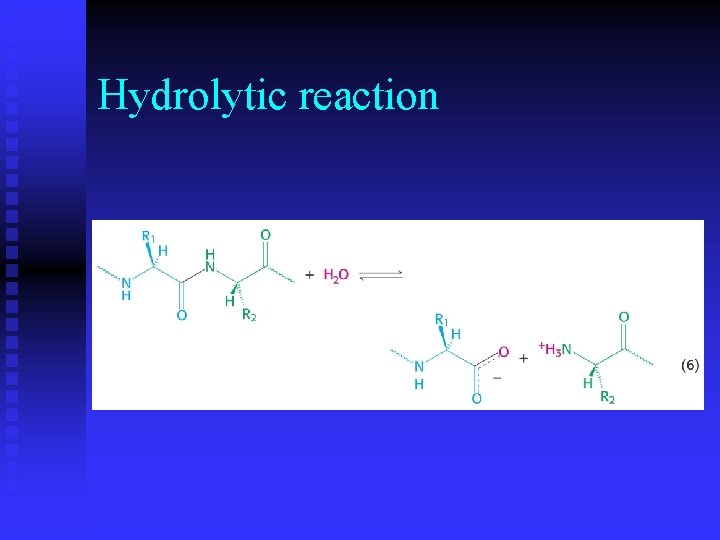
Hydrolytic reaction
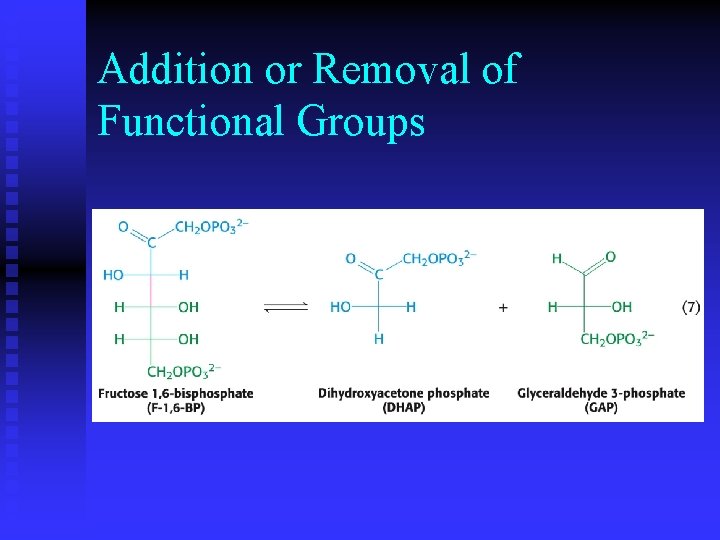
Addition or Removal of Functional Groups

Regulation of Metabolism n What are three basic ways in which metabolic reactions are regulated? u controlling amount of enzyme t influencing rate of transcription u altering catalytic activity t allosterically t covalent modification u controlling accessibility of substrates

Regulation of Metabolism How do hormones influence metabolism? u alteration of enzyme activity t glucagon and glycogen phosphorylase n What other factors influence metabolism? u compartmentalization u energy status of cell n

Metabolism n What do ATP, NADH, FAD and coenzyme A have in common and what does this indicate about these molecules? u adenosine diphosphate u evolved from early RNA catalysts
- Slides: 32