METABOLISM and UTILIZATION OF AMINO ACIDS Amino nitrogen

METABOLISM and UTILIZATION OF AMINO ACIDS
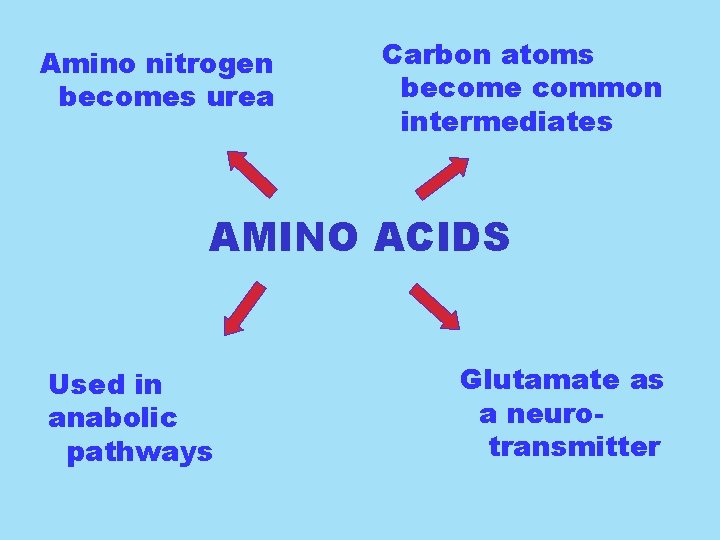
Amino nitrogen becomes urea Carbon atoms become common intermediates AMINO ACIDS Used in anabolic pathways Glutamate as a neurotransmitter

I. Amino-acid Nitrogen Removal by Transamination and its Utilization for Urea Transamination requires: 1) An amino acid an oxo- (keto-) acid 2) Vitamin pyridoxol 3) An aminotransferase. NH 2 O | || O O || || R 1 CH– C OH + R 2 C– C– OH OH O O || || R 1– C – C– O H + NH 2 O | || R 2 –CH – C – OH

The oxoacid can be either pyruvate, which produces alanine, oxaloacetate, which produces aspartate 2 -oxoglutarate, which produces glutamate

Transamination with 2 -oxoglutarate is important because this reaction allows an amino acid nitrogen to become the nitrogen of ammonia. NH 2 O | | | R– CH– C– O H + 2 -OG O || NH 2 O | || HO– C – CH 2 –CH – C– OH NH 3
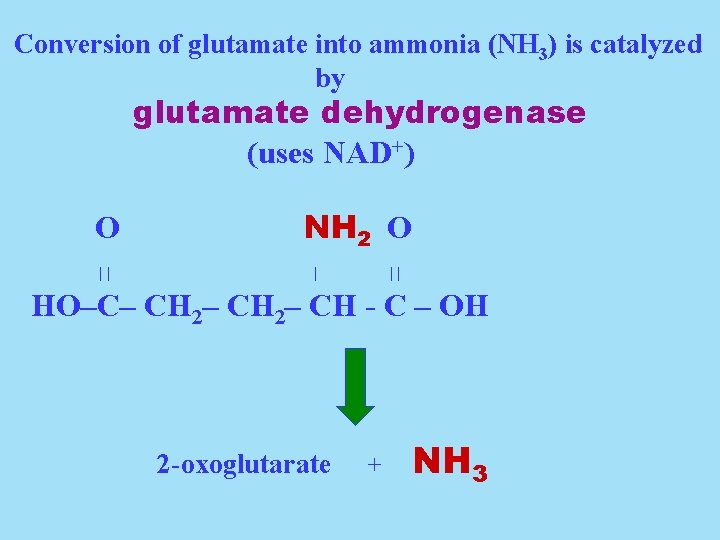
Conversion of glutamate into ammonia (NH 3) is catalyzed by glutamate dehydrogenase (uses NAD+) O || NH 2 O | || HO–C– CH 2– CH - C – OH 2 -oxoglutarate + NH 3
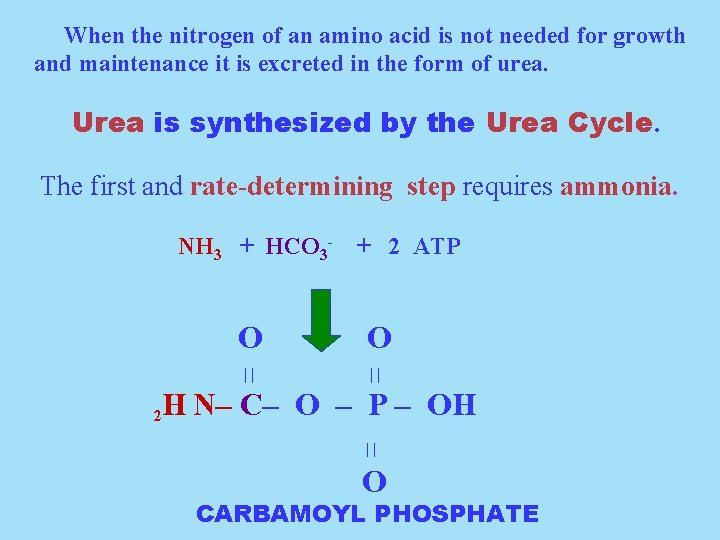
When the nitrogen of an amino acid is not needed for growth and maintenance it is excreted in the form of urea. Urea is synthesized by the Urea Cycle. The first and rate-determining step requires ammonia. NH 3 + HCO 3 - + 2 ATP O O || || H N– C– O – P – OH 2 || O CARBAMOYL PHOSPHATE

First and rate determining step (RDS) of the urea cycle is catalyzed by carbamoylphosphate synthetase. There are, in fact, two enzymes with this name. One is involved in pyrimidine synthesis (in humans). The other is the urea cycle enzyme that is designated carbamoylphosphate synthetase I or CPS I.
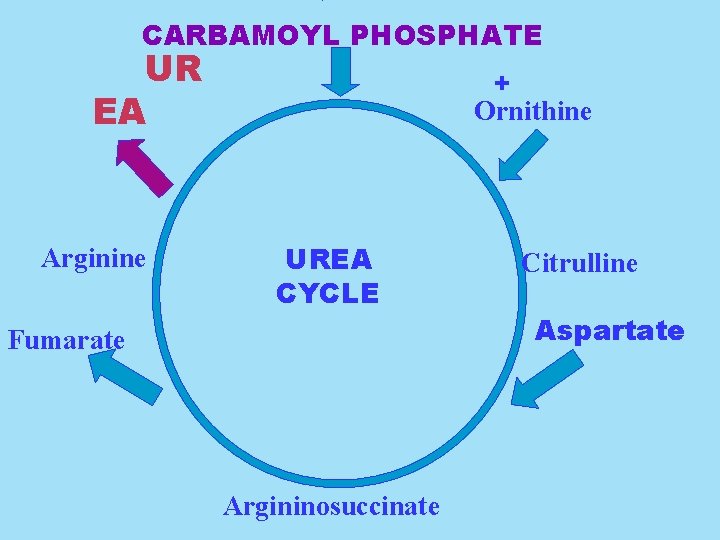
CARBAMOYL PHOSPHATE UR EA Arginine + Ornithine UREA CYCLE Fumarate Argininosuccinate Citrulline Aspartate

REGULATION OF THE UREA CYCLE Acute: N-acetylglutamate, an allosteric effector, up regulates carbamoylphosphate synthetase At the level of gene expression: Hepatic nuclear factors (HNFs) provide tissue specificity

Excessive ammonia is toxic to the central nervous system! Treatment Induce the formation of a compound that contains the ammonia nitrogen, that is then excreted. Sodium benzoate induces production of hippuric acid. Sodium phenylacetylglutamate induces production of phenylacetylglutamine.
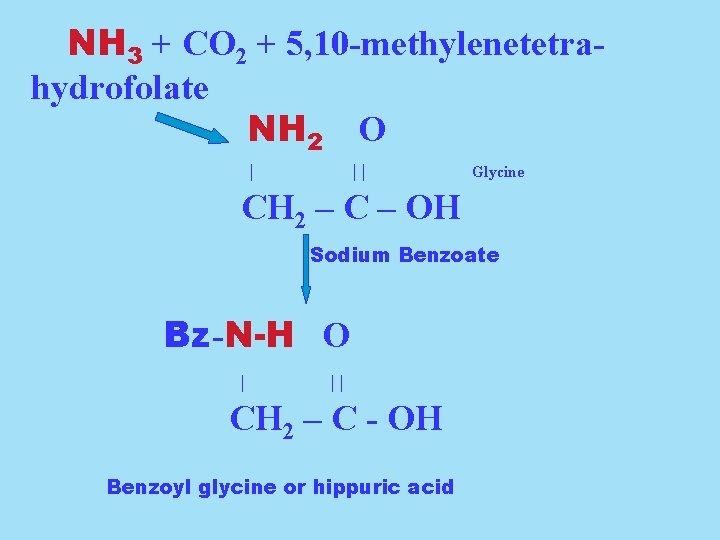
NH 3 + CO 2 + 5, 10 -methylenetetrahydrofolate NH 2 O | || Glycine CH 2 – C – OH Sodium Benzoate Bz-N-H O | || CH 2 – C - OH Benzoyl glycine or hippuric acid
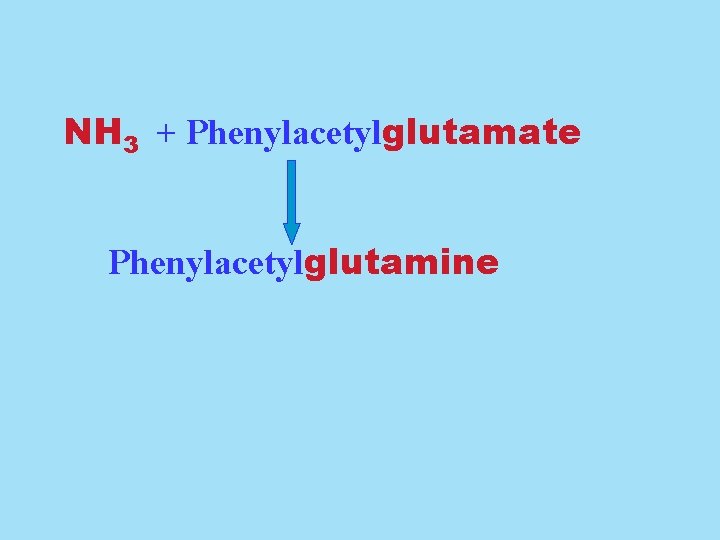
NH 3 + Phenylacetylglutamate Phenylacetylglutamine

In the presence of excessive ammonia, glutamine is synthesized in the brain’s astrocytes by the glutamine synthetase reaction, i. e. , NH 3 + Glutamate + ATP → Glutamine + ADP + Pi. Water then enters the cell, producing an “intracranial mass” that is manifested as edema. The astrocytes become dysfunctional which has ramifications with respect to the regulation of extracellular potassium concentrations, among other processes.

II. Catabolic Fates of Amino-acid Carbon Atoms Carbons derived from amino acid can become those of five very common biochemical intermediates. These are: pyruvate acetyl-Co. A acetoacetate succinyl-Co. A 2 -oxoglutarate

A. Amino Acid Degradation Provides Carbon Atoms of Pyruvate Examples of carbon atoms of amino acids becoming those of pyruvate are found in glycine and serine. Glycine can be converted into serine and then the carbon atoms of serine can become those of pyruvate.
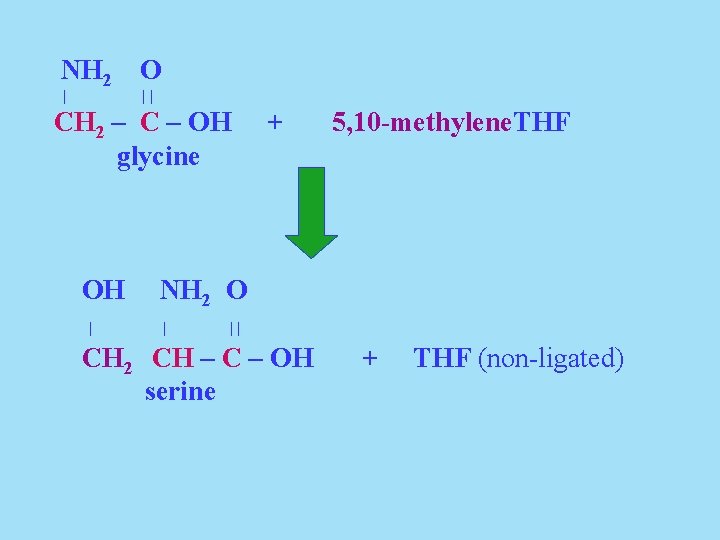
NH 2 O | || CH 2 – C – OH glycine OH | + 5, 10 -methylene. THF NH 2 O | || CH 2 CH – C – OH serine + THF (non-ligated)
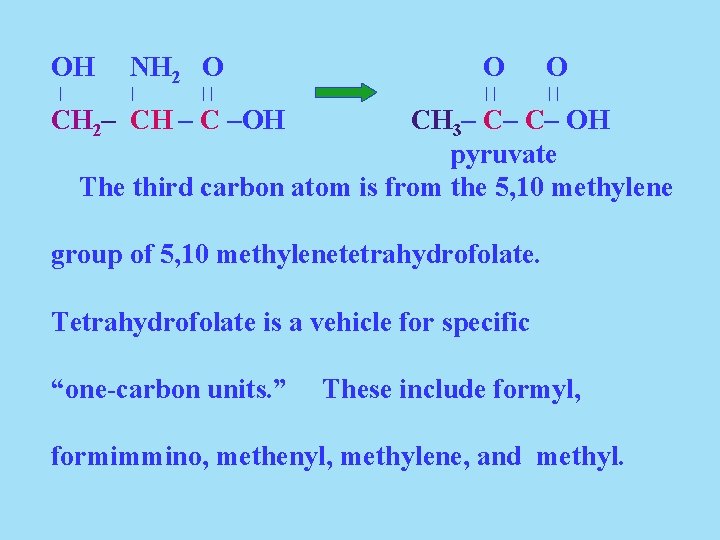
OH | NH 2 O | || O O || || CH 2– CH – C –OH CH 3– C– C– OH pyruvate The third carbon atom is from the 5, 10 methylene group of 5, 10 methylenetetrahydrofolate. Tetrahydrofolate is a vehicle for specific “one-carbon units. ” These include formyl, formimmino, methenyl, methylene, and methyl.

Amino-acid carbon atoms that become those of pyruvate can, in turn, become those of glucose (gluconeogenesis) and also those of palmitic acid (pyruvate acetyl-Co. A) cholesterol (pyruvate acetyl-Co. A) steroids (from cholesterol)

B. Amino Acid Degradation Provides Carbon Atoms of Acetyl-Co. A One example is found in the case of the branched chain amino acids leucine and isoleucine. Leucine is degraded to yield acetyl-Co. A and acetoacetate (another of the five key biochemical intermediates). Isoleucine yields acetyl-Co. A and propionyl-Co. A (precursor of another of the five).

The rate-determining reaction in the degradation of leucine or isoleucine is catalyzed by a branchedchain oxoacid dehydrogenase (BCOD) complex that is similar to the pyruvate dehydrogenase complex. The activity of the BCOD complex is tightly regulated by nutritional state and by hormones.

These factors exert their effects by controlling the activity and/or the expression of a specific protein kinase that modulates the activity of the BCOD complex. With respect to acute regulation, phosphorylation of the BCOD complex by its kinase (the BCOD kinase) down-regulates the activity of the complex.

Activity of the BCOD complex is also regulated by controlling the gene expression of the dehydrogenase subunit. This is appropriate because it is the activity of the dehydrogenase subunit that is rate-determining for the degradative process.

Starvation (>24 hours) leads to decreased BCOD kinase activity in the liver and, therefore, increased protein degradation. Diabetes, which can associate with increased protein degradation, also associates with decreased BCOD kinase activity. Since insulin promotes the activity of the BCOD kinase, it would follow that its lack, as in diabetes, would lead to (decreased activity of the kinase and) increased protein degradation.
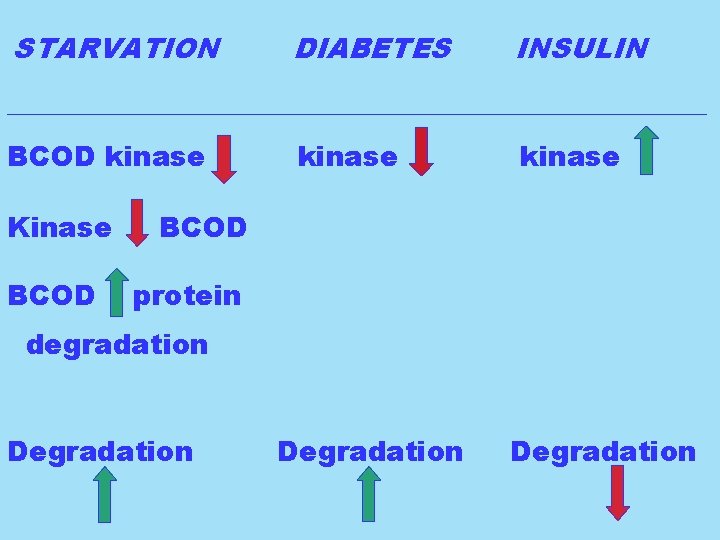
STARVATION DIABETES INSULIN _________________________ BCOD kinase Kinase BCOD kinase BCOD protein degradation Degradation

Amino-acid carbon atoms that become those of acetyl-Co. A can, in turn, become those of citrate (by entering the TCA cycle) palmitic acid (fatty acid synthesis) cholesterol (cholesterol synthesis uses 3) steroids (from cholesterol)

C. Amino Acid Degradation Provides Carbon Atoms of Acetoacetate. Examples are found in the case of phenylalanine and tyrosine. Phenylalanine can be converted into tyrosine and tyrosine can be further degraded (many steps) into acetoacetate and fumarate.

Acetoacetate is one of the “ketone bodies” and the precursor of the other two. ACETOACETATE -CO 2 ACETONE REDUCTION 3 -HYDROXYBUTYRATE

“Ketone bodies” are produced in the liver in order to provide substrates to be utilized by the brain in times of glucose deprivation. However, excessive “ketone body” production characterizes uncontrolled diabetes. In fact, acetoacetic acid and 3 -hydroxybutyric acid contribute to the metabolic acidosis that frequently accompanies diabetes.

Acetone and acetoacetate are each ketones and Contribute to the ketosis that frequently accompanies diabetes. Diabetes not only associates with increased protein degradation, as mentioned above, it also associates with increased circulating branched chain amino acids. The latter contribute to the production of “ketone bodies. ”

D. Amino Acid Degradation can Provide Carbon atoms of Succinyl-Co. A. Isoleucine, valine, threonine, and methionine (so-called “essential” amino acids) are degraded to yield propionyl-Co. A that is, in turn, converted into succinyl-Co. A by a vitamin B 12 -dependent reaction. Succinyl-Co. A can enter the TCA cycle.

E. Amino Acid Degradation can Provide Carbon Atoms of 2 -Oxoglutarate ( -ketoglutarate). These amino acids, likewise, provide carbon atoms that can be catabolized by the TCA cycle to produce energy.
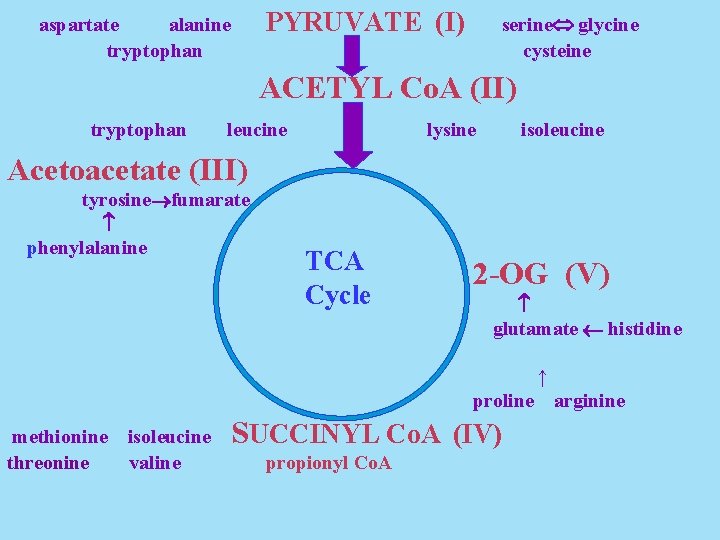
PYRUVATE (I) aspartate alanine tryptophan serine glycine cysteine ACETYL Co. A (II) tryptophan leucine lysine isoleucine Acetoacetate (III) tyrosine fumarate phenylalanine TCA Cycle 2 -OG (V) glutamate histidine ↑ proline methionine threonine isoleucine valine SUCCINYL Co. A (IV) propionyl Co. A arginine
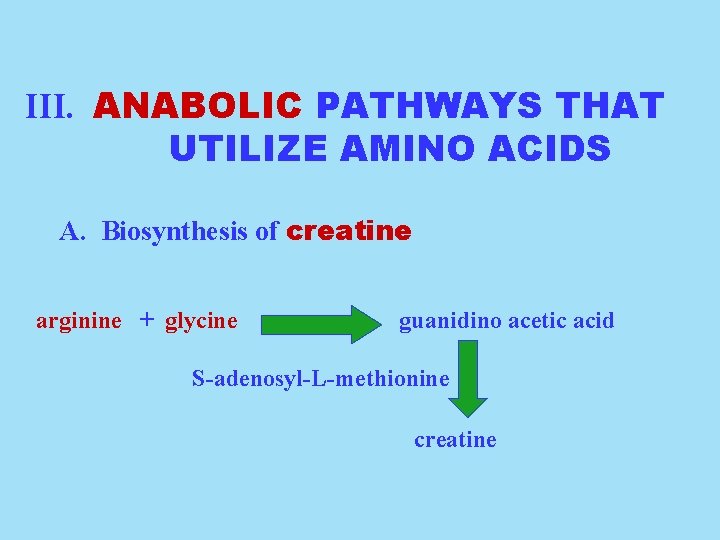
III. ANABOLIC PATHWAYS THAT UTILIZE AMINO ACIDS A. Biosynthesis of creatine arginine + glycine guanidino acetic acid S-adenosyl-L-methionine creatine

B. Phenylalanine and tyrosine are precursors of the catecholamines, dopamine, norepinephrine, and epinephrine. 1) Clinical ramifications involving dopamine: Dopamine has a role in the pathogenic mechanisms that cause Parkinson’s disease is characterized by a loss of dopaminergic neurons that results in diminished levels of dopamine in the striatum.

It has recently been suggested that diminished uptake of dopamine into its storage vesicles is a causative factor in Parkinson’s. A protein called α-synuclein may have a role in this process since it promotes the formation of these vesicles. Dopamine that is not sequestered intracellularly can have deleterious effects upon the cell.
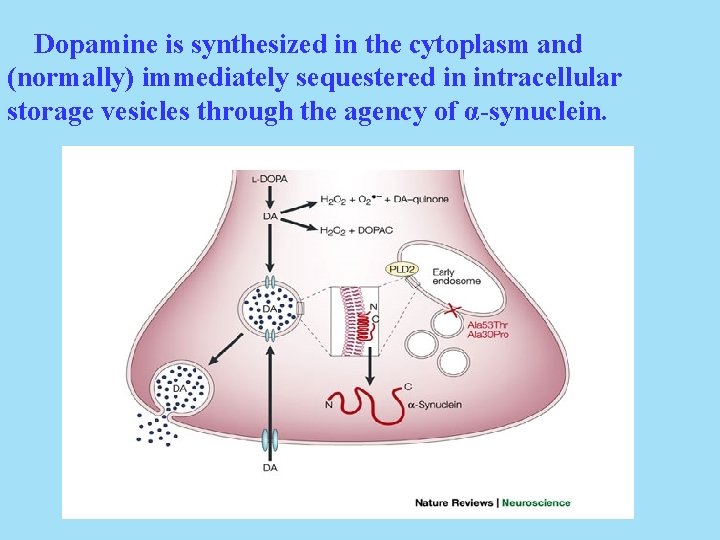
Dopamine is synthesized in the cytoplasm and (normally) immediately sequestered in intracellular storage vesicles through the agency of α-synuclein.
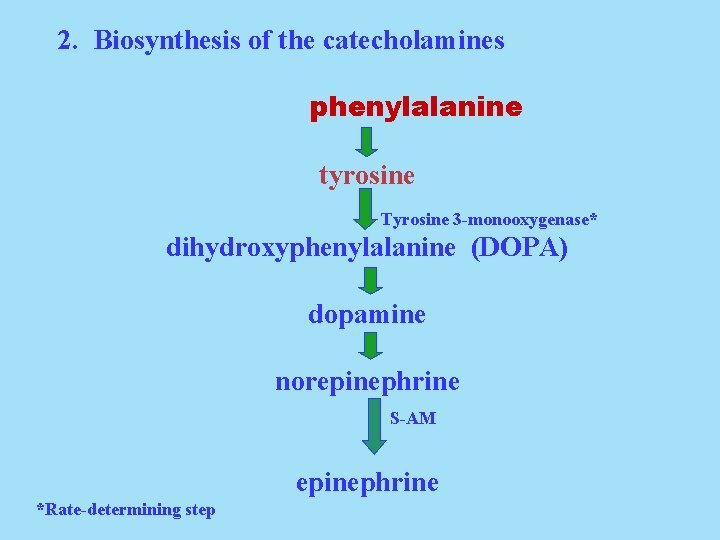
2. Biosynthesis of the catecholamines phenylalanine tyrosine Tyrosine 3 -monooxygenase* dihydroxyphenylalanine (DOPA) dopamine norepinephrine S-AM epinephrine *Rate-determining step

C. Tryptophan is the precursor of 5 -hydroxytryptamine (serotonin). 1. Clinical ramifications The broad actions of the neurotransmitter 5 hydroxytryptamine involve effects upon emotion, mood, and reward. Suicide and depression have been associated with reduced serotonergic transmission. One recent postmortem study examined the capacity to synthesize 5 -hydroxytryptamine and the capacity

to bind 5 -hydroxytryptamine. Their findings indicated that the brains of suicide victims had reduced serotonergic function. 2. Biosynthesis of 5 -hydroxytryptamine In the conversion of tryptophan into 5 -hydroxytryptamine, tryptophan undergoes hydroxylation to yield 5 -hydroxytryptophan that is then decarboxylated to yield 5 -hydroxytryptamine.
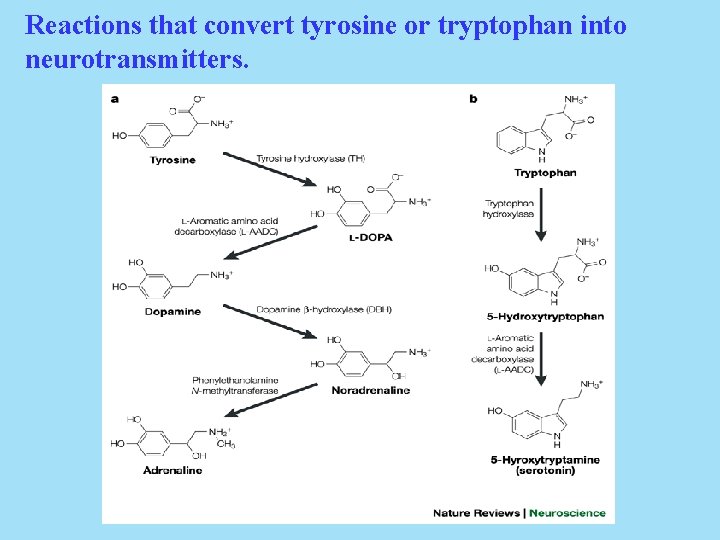
Reactions that convert tyrosine or tryptophan into neurotransmitters.

It has been shown that the intensity and duration of catecholamine signaling at the synapse is governed by the efficiency of the re-uptake of released neurotransmitter. Re-uptake occurs by means of highaffinity membrane transporters. The finding that a dopamine “re-uptake” transporter can serve as a cocaine “receptor” has provided new insight into mechanisms of addiction.
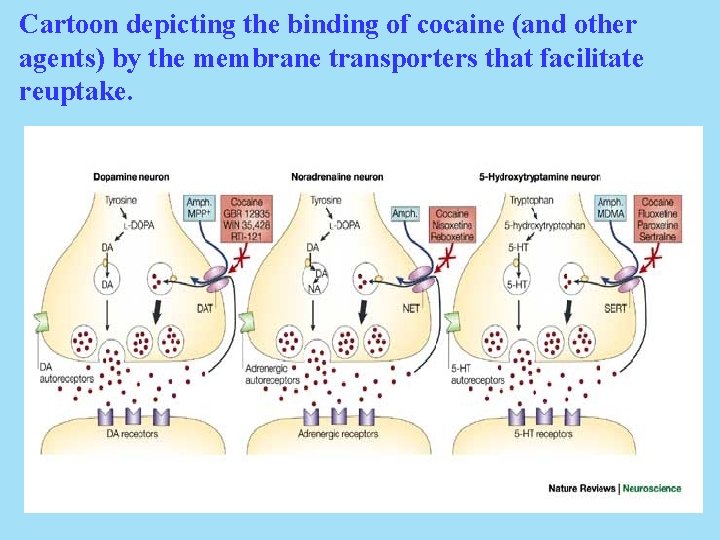
Cartoon depicting the binding of cocaine (and other agents) by the membrane transporters that facilitate reuptake.

D. Arginine is the precursor of the second messenger nitric oxide. This a second messenger that appears to have distinctly different functions in different cell types. For example, the nitric oxide synthesized by platelets is seen to inhibit platelet aggregation and adherence. In this way it contributes to the anti-thrombogenic properties of the endothelium.

In the kidney (in experimental animals), a diminution in nitric oxide was associated with glucocorticoid-induced hypertension. Such studies were carried out because it had been suggested that decreased nitric oxide contributes to the impaired endothelium-dependent vasodilatation seen in essential hypertension.

On the other hand, cortical cells from patients with Alzheimer’s disease showed increased levels of nitric oxide m. RNA and protein. Hence, nitric oxide has been associated with the progression of Alzheimer’s disease. From an overview however, nitric oxide is generally considered to be a vasodilator.

As a second messenger, nitric oxide up regulates the activity of the enzyme that synthesizes cyclic GMP, i. e. , guanylyl cyclase. The cyclic GMP then up regulates a cyclic GMP-dependent protein kinase. The protein kinase, in turn, phosphorylates specific substrates in order to bring about its effects.

For their pioneering work that lead to the discovery of nitric oxide (NO) and its signaling in humans, Dr. Robert Furchgott (SUNY, Brooklyn, NY) Dr. Louis J. Ignarro (UCLA, Los Angeles, CA) Dr. Ferid Murad (Univ. Texas Med. Sch. Houston, TX) received the 1998 Nobel Prize (Physiology or Medicine).

2. Biosynthesis of nitric oxide Nitric oxide is synthesized by the enzyme nitric oxide synthase (NOS). There are three types of enzymes, the neuronal nitric oxide synthase (n. NOS), the endothelial nitric oxide synthase (e. NOS), and the inducible nitric oxide synthase (i. NOS). They each catalyze the complex oxidation/reduction reaction that utilizes one of arginine’s nitrogen atoms in order to make the “free radical” nitric oxide.
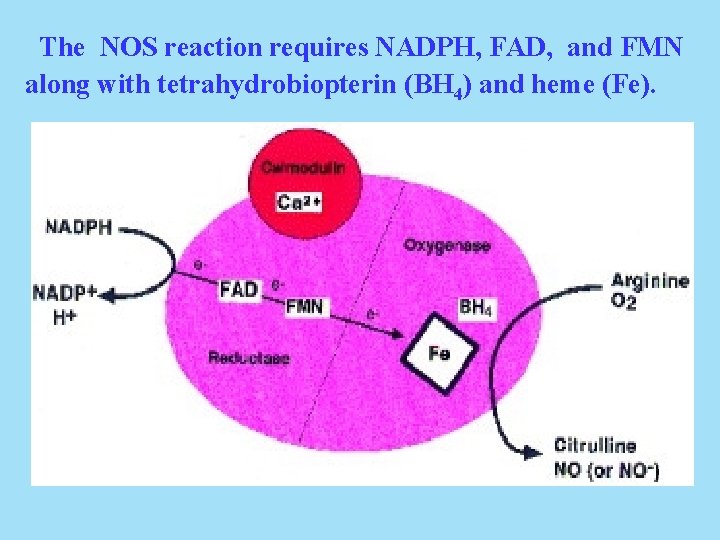
The NOS reaction requires NADPH, FAD, and FMN along with tetrahydrobiopterin (BH 4) and heme (Fe).
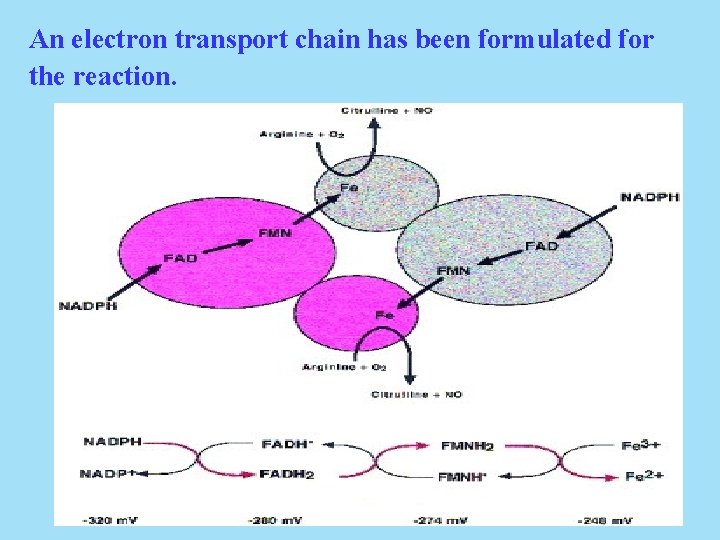
An electron transport chain has been formulated for the reaction.

E. Methionine has multiple anabolic fates. 1. As a precursor of the polyamines, spermidine and spermine. While it is generally accepted that the polyamines, spermidine and spermine, are essential for cell proliferation in mammalian cells, the critical reaction(s) have not been identified. Inhibiting ornithine decarboxylase (ODC) (the rate-determining enzyme) has been shown to inhibit cell growth, however.
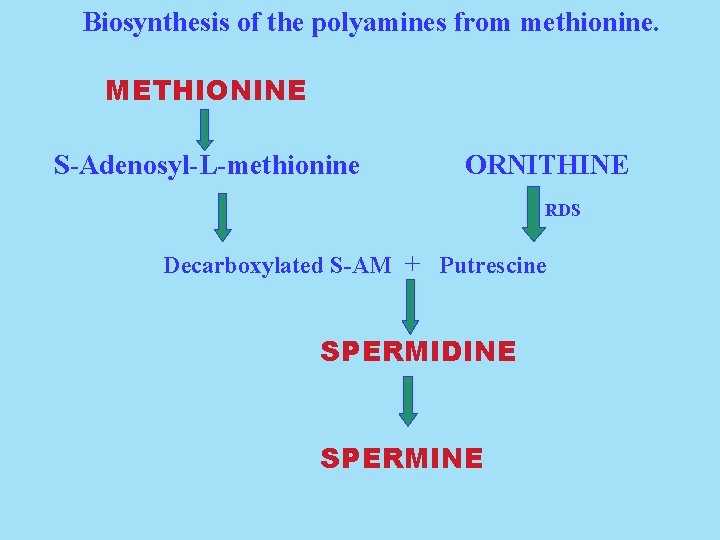
Biosynthesis of the polyamines from methionine. METHIONINE S-Adenosyl-L-methionine ORNITHINE RDS Decarboxylated S-AM + Putrescine SPERMIDINE SPERMINE
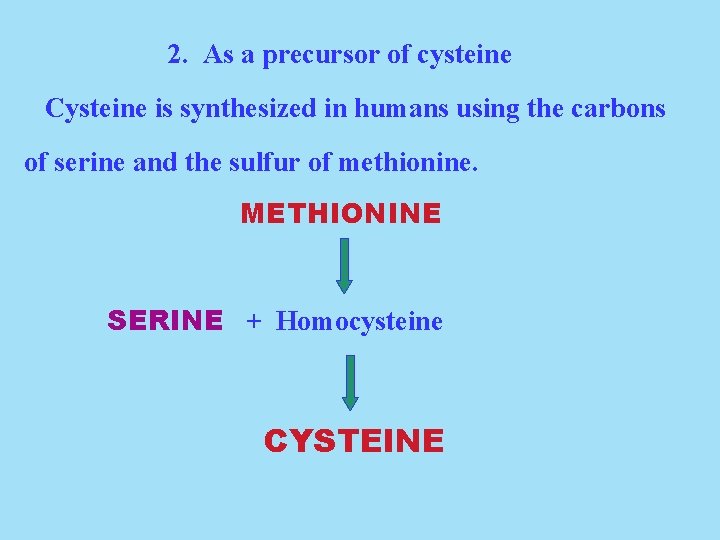
2. As a precursor of cysteine Cysteine is synthesized in humans using the carbons of serine and the sulfur of methionine. METHIONINE SERINE + Homocysteine CYSTEINE
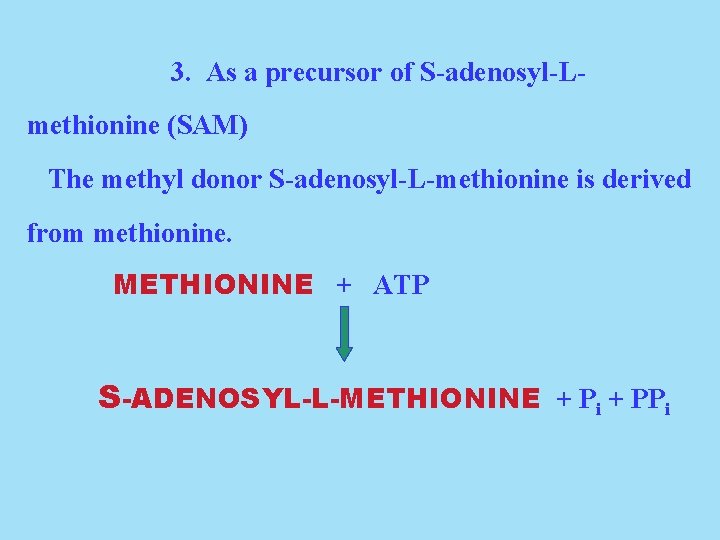
3. As a precursor of S-adenosyl-Lmethionine (SAM) The methyl donor S-adenosyl-L-methionine is derived from methionine. METHIONINE + ATP S-ADENOSYL-L-METHIONINE + Pi + PPi

4. As a necessary participant in the synthesis of deoxyribothymidylic acid and, hence, DNA The conversion of deoxyribouridylic acid monophosphate (d. UMP) into the corresponding thymidylic acid derivative (d. TMP) depends upon converting 5 -methyltetrahydrofolate into the unligated tetrahydrofolate. This is accomplished by the enzyme methionine synthase.

The synthesis of deoxyribothymidylic acid (d. TMP) requires both vitamin B 12 and folic acid. 5 -methyl THF methionine synthase homocysteine METHIONINE vitamin B 12 THF (non-ligated) serine hydroxymethyl transferase serine glycine 5, 10 -methylene THF thymidylate synthase d. UMP dihydrofolate d. TMP

In summary, methionine provides carbon and nitrogen atoms that become a part of spermidine and spermine, the sulfur atom that is required for the biosynthesis of cysteine, and the “active” methyl group of the methyl donor, S-adenosyl-L-methionine. H 3 C ─S NH O CH 2─CH──C─OH
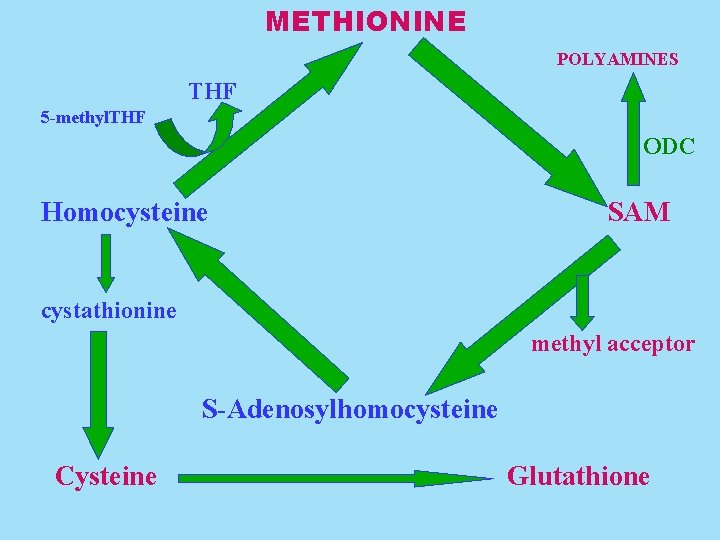
METHIONINE POLYAMINES THF 5 -methyl. THF ODC Homocysteine SAM cystathionine methyl acceptor S-Adenosylhomocysteine Cysteine Glutathione

IV. “INBORN ERRORS” OF AMINO ACID METABOLISM A. PHENYLKETONURIA 1. Clinical Ramifications: neurological, mental retardation 2. Critical Reaction: phenylalanine + tetrahydrobiopterin + O 2 tyrosine + dihydrobiopterin + H 2 O

B. ALBINISM 1. Clinical Ramifications: usually not severe, sensitivity to sun 2. Critical Reaction: Melanogenesis entails the oxidation of tyrosine by a tyrosinase. In vertebrates this enzyme is active only in specialized organelles including, primarily, the retinal pigment epithelium and melanocytes.

The oxidation of tyrosine yields dopaquinone that, subsequently, is converted into the polymer referred to as melanin. It has recently been shown that oculocutaneous albinism (OCA), type II, an autosomal recessive disorder, results from failure of appropriate intracellular transport of the tyrosinase.

Studies showed that in affected individuals, the melanocytes contained markedly diminished quantities of tyrosinase. The fault resided in a gene that encodes a protein that is critical for the processing and transport of tyrosinase to the melanocyte.
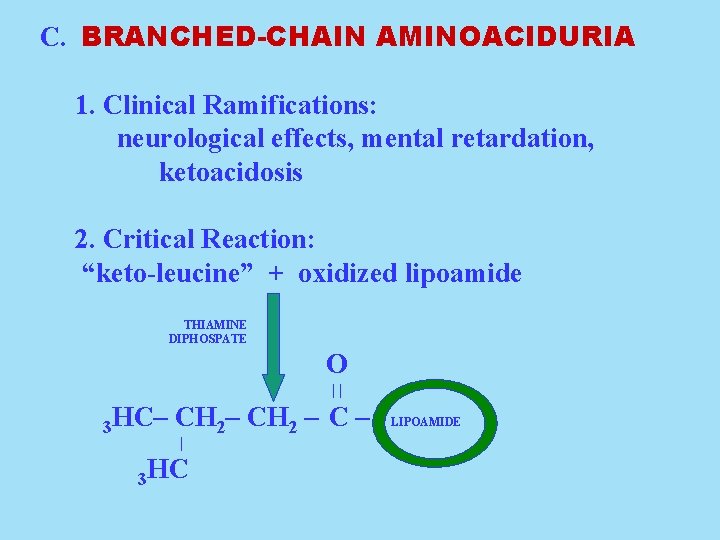
C. BRANCHED-CHAIN AMINOACIDURIA 1. Clinical Ramifications: neurological effects, mental retardation, ketoacidosis 2. Critical Reaction: “keto-leucine” + oxidized lipoamide THIAMINE DIPHOSPATE O || 3 HC– CH 2 – C – | 3 HC LIPOAMIDE

This reaction is catalyzed by the BCOD component of the multi-enzyme complex. (It is rate-determining for the process. ) Several mutations in this enzyme have been identified and characterized. Attempts are being made to correlate particular mutations with the severity of the disease.

D. HOMOCYSTEINURIA 1. Clinical ramifications: First described in 1962 in relation to mental retardation in children. Now associated primarily with vascular pathologies including atherosclerosis and premature thromboembolic disease. Also associated with certain CNS disorders and abnormal bone development.

Homocysteine can alter the surface properties of endothelial cells, causing them to be procoagulant as a result of inhibiting antithrombin III and inhibiting the synthesis of an anticoagulant (heparan sulfate). This has been offered as a rationale for the suggested role of homocysteine in atherosclerosis.

2. Critical reaction The most common cause for genetically determined homocysteinuria is a “loss of function” mutation that results in decreased activity of the enzyme cystathionine β-synthase. This is the enzyme that catalyzes the formation of cysteine using the carbon atoms of serine and a sulfur from methionine (as discussed above).

V. Glutamate and its Receptors and Signaling The amino acid glutamate is an endogenous excitatory neurotransmitter, utilized by neurons in the human central nervous system and the peripheral nervous system. Glutamate acts by binding glutamate receptors that allow it to play a pivotal role in synaptic mechanisms involved in learning and memory.

Learning and memory involve synaptic plasticity, the phenomenon in which the efficacy, or efficiency, of synaptic transmission varies in an activity-dependent manner. The effect can be transient (short-term) or persistent (long-term). Signal transduction involving glutamate receptors governs the reactions involved in these processes. .
Cartoon depicting signal transduction involving the glutamate receptor in the phenomenon of synaptic plasticity.

Glutamate binds two types of receptors, the ionotropic glutamate receptors and the metabotropic receptors. There are three of the former, the NMDA (N-methyl-D-aspartate), the AMPA (α-amino-3 hydroxy-5 -methyl-4 -isoxazolepropionic acid), and the kainate receptors. They are referred to as ionotropic because they associate to form cation channels that facilitate the entry of Ca 2+ (or Na+) into the cell.
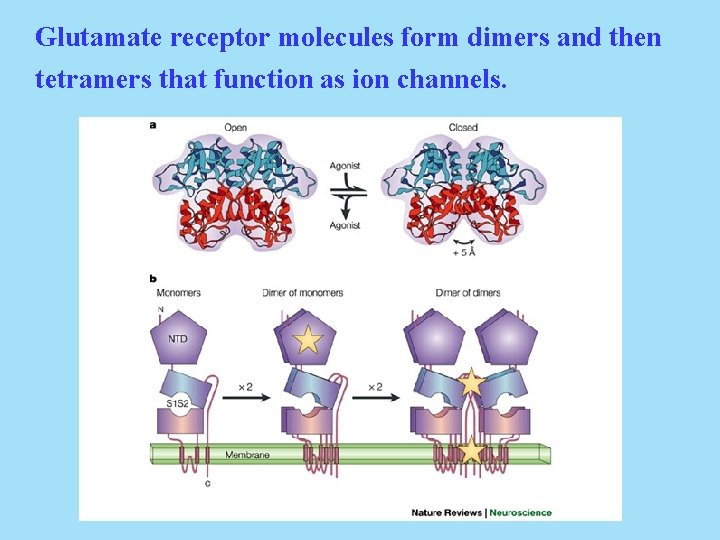
Glutamate receptor molecules form dimers and then tetramers that function as ion channels.

These ion channels are ligand-gated channels that assume an “open” configuration upon glutamate (or agonist) binding. They are locked in a “closed” configuration when an antagonist is bound. It should be remembered that when a protein binds a ligand, the protein generally undergoes a conformational change that can alter its properties and its activity.
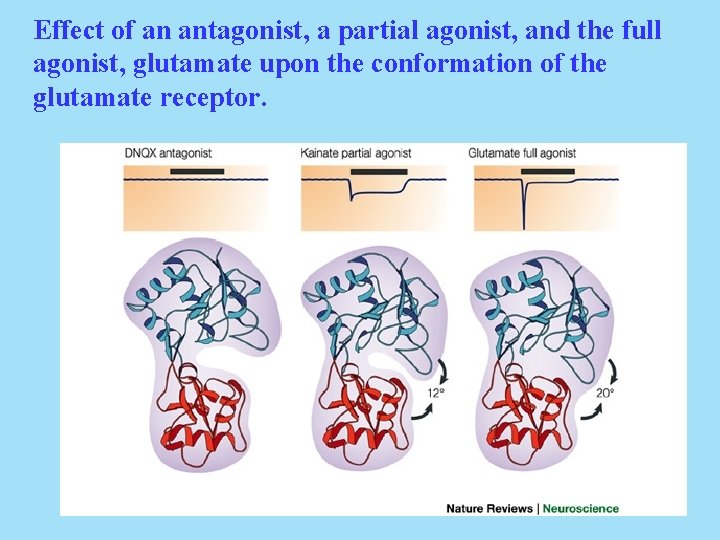
Effect of an antagonist, a partial agonist, and the full agonist, glutamate upon the conformation of the glutamate receptor.

Excessive glutamate binding to glutamate receptors in the brain can bring about excitotoxic neuronal cell death, however. Excessively activated receptors lead to brain damage seen with cerebral ischemia, traumatic brain injury, and the neurodegeneration associated with Huntington’s chorea, epileptic disorders, and Alzheimer’s disease.

These excitotoxic effects are due, in part, to signaling downstream of certain glutamate receptors that leads to in increased intracellular Ca 2+. The ionotropic N-methyl-D-aspartate (NMDA) glutamate receptor is one of the principal ones that facilitates the entry of extracellular calcium (Ca 2+) into the neuronal cell.
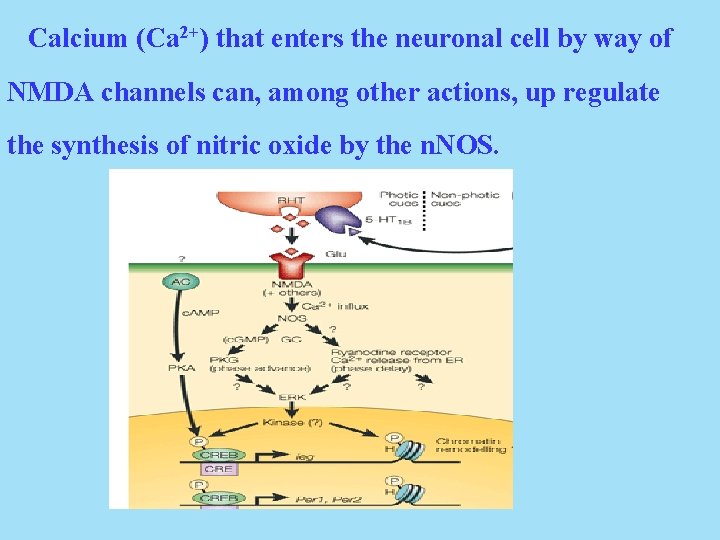
Calcium (Ca 2+) that enters the neuronal cell by way of NMDA channels can, among other actions, up regulate the synthesis of nitric oxide by the n. NOS.
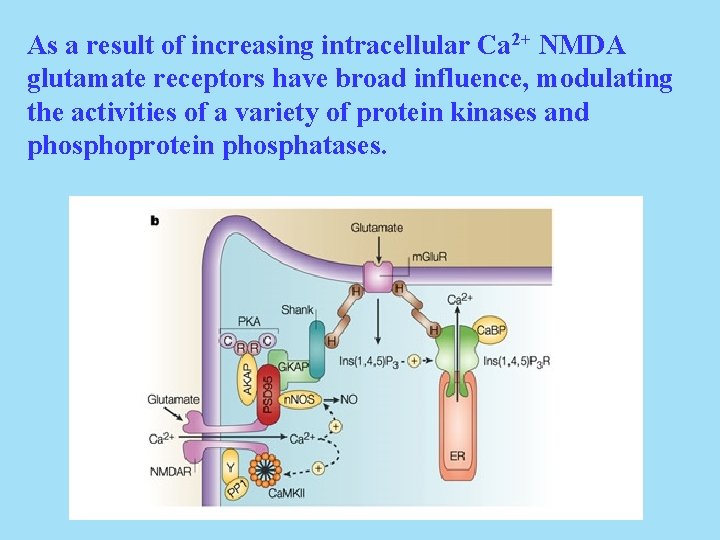
As a result of increasing intracellular Ca 2+ NMDA glutamate receptors have broad influence, modulating the activities of a variety of protein kinases and phosphoprotein phosphatases.
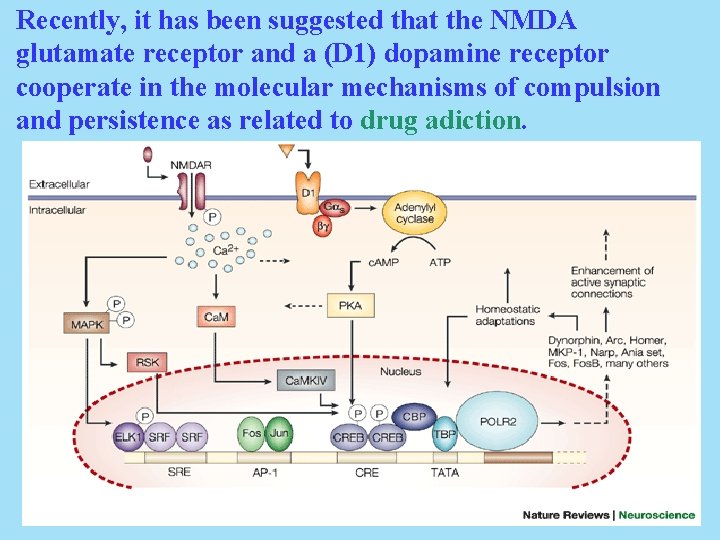
Recently, it has been suggested that the NMDA glutamate receptor and a (D 1) dopamine receptor cooperate in the molecular mechanisms of compulsion and persistence as related to drug adiction.
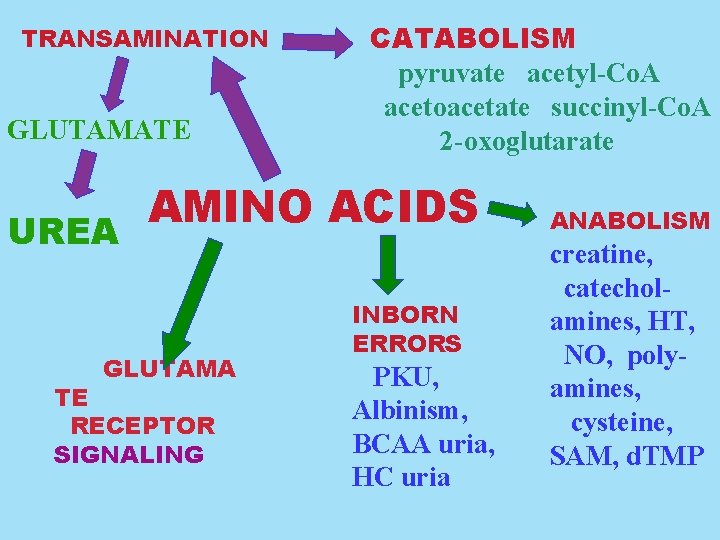
TRANSAMINATION GLUTAMATE UREA CATABOLISM pyruvate acetyl-Co. A acetoacetate succinyl-Co. A 2 -oxoglutarate AMINO ACIDS GLUTAMA TE RECEPTOR SIGNALING INBORN ERRORS PKU, Albinism, BCAA uria, HC uria ANABOLISM creatine, catecholamines, HT, NO, polyamines, cysteine, SAM, d. TMP
- Slides: 81