Metabolism and Energy AP Biology Metabolic reactions energy
Metabolism and Energy AP Biology

Metabolic reactions & energy § Some chemical reactions release energy exergonic u breaking polymers u hydrolysis = catabolism u digesting molecules= less organization= lower energy state § Some chemical reactions require input of energy building molecules= more organization= higher energy state endergonic u building polymers u dehydration synthesis = anabolism u AP Biology
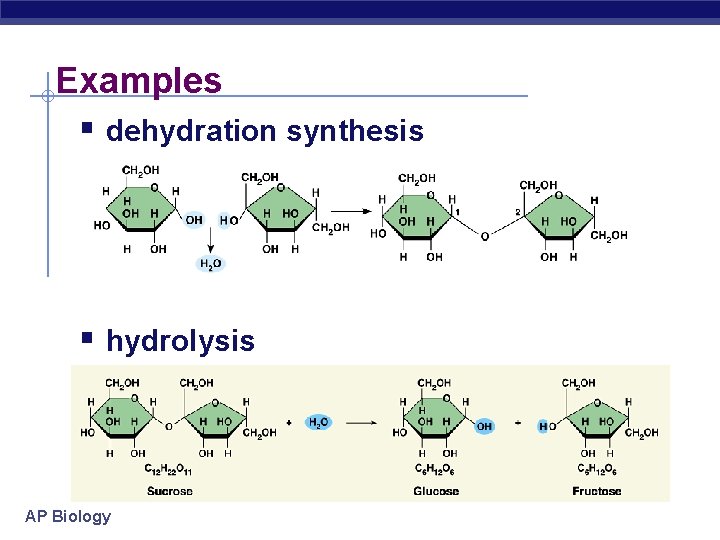
Examples § dehydration synthesis § hydrolysis AP Biology
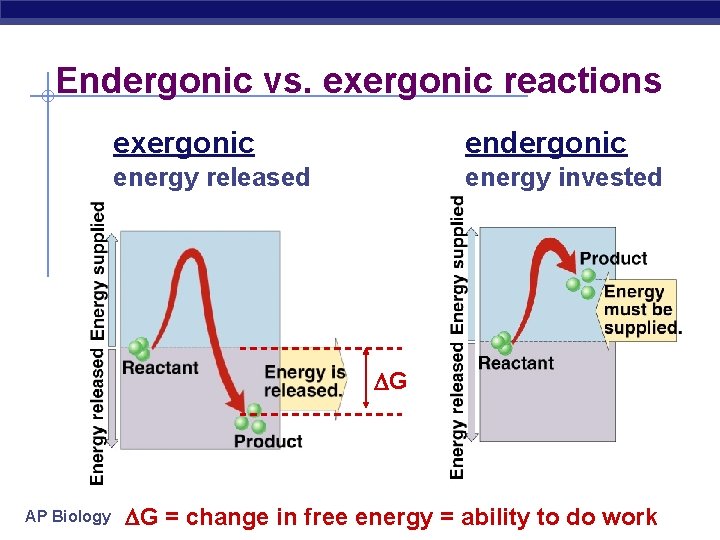
Endergonic vs. exergonic reactions exergonic endergonic energy released energy invested G AP Biology G = change in free energy = ability to do 2005 -2006 work
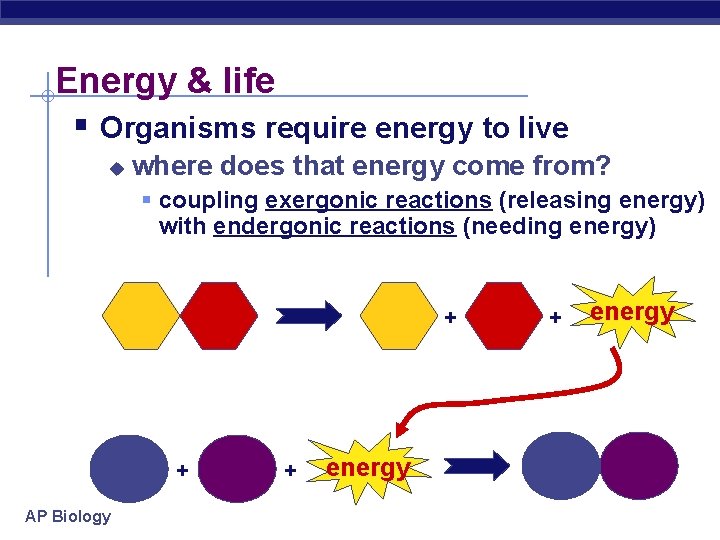
Energy & life § Organisms require energy to live u where does that energy come from? § coupling exergonic reactions (releasing energy) with endergonic reactions (needing energy) + + AP Biology + energy
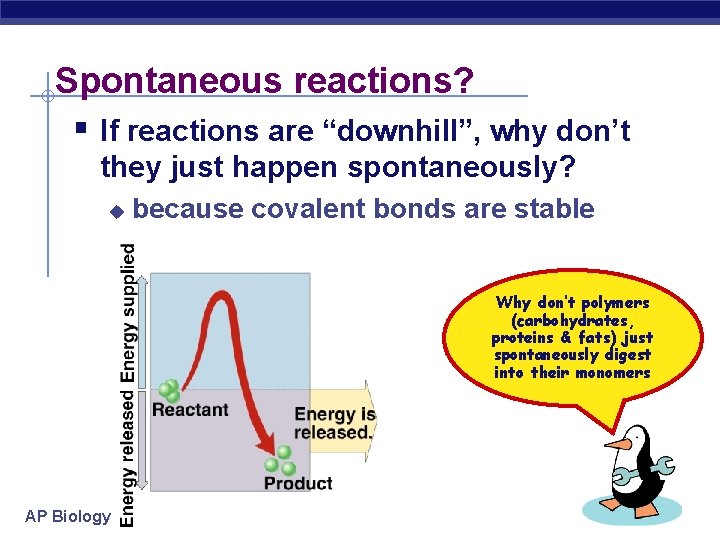
Spontaneous reactions? § If reactions are “downhill”, why don’t they just happen spontaneously? u because covalent bonds are stable Why don’t polymers (carbohydrates, proteins & fats) just spontaneously digest into their monomers AP Biology 2005 -2006

Activation energy § Breaking down large molecules requires an initial input of energy activation energy u large biomolecules are stable u must absorb energy to break bonds u AP Biology cellulose energy CO 2 + H 2 O + heat
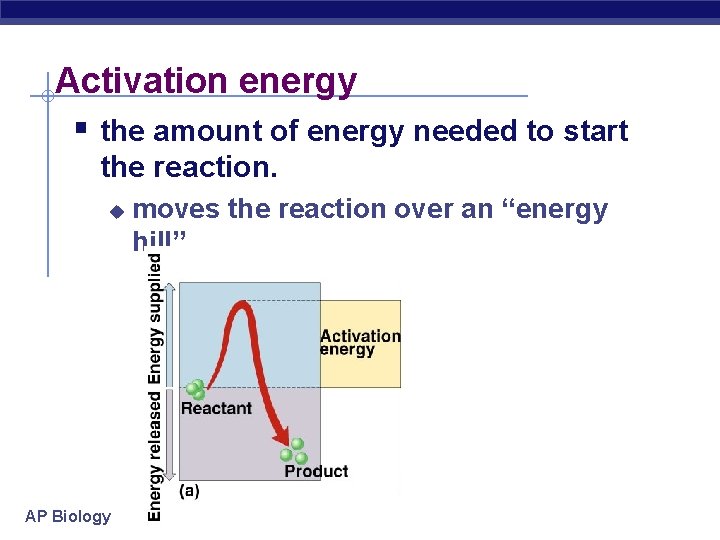
Activation energy § the amount of energy needed to start the reaction. u AP Biology moves the reaction over an “energy hill”
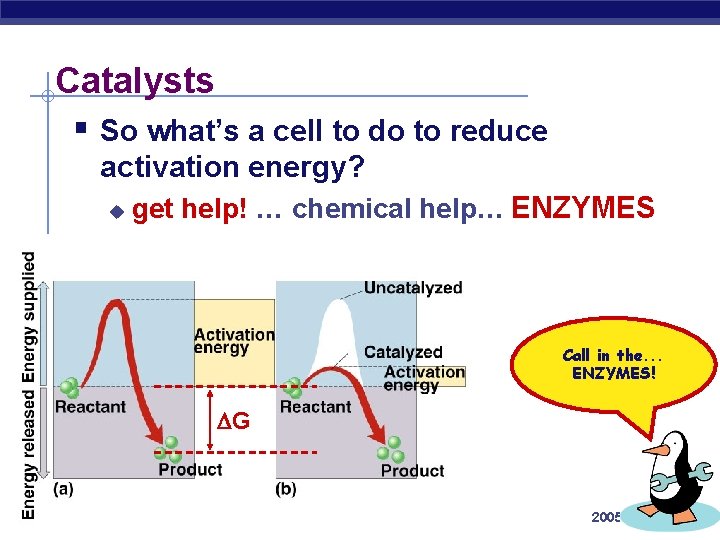
Catalysts § So what’s a cell to do to reduce activation energy? u get help! … chemical help… ENZYMES Call in the. . . ENZYMES! G AP Biology 2005 -2006

Energy needs of life § Organisms are endergonic systems u What do we need energy for? § § § AP Biology synthesis (biomolecules) reproduction active transport movement temperature regulation 2005 -2006

Living economy § Fueling the economy u u u eat high energy organic molecules (food) break them down = catabolism (digest) capture energy in form cell can use § Need an energy currency u a way to pass energy around Whoa! Hot stuff! AP Biology ATP
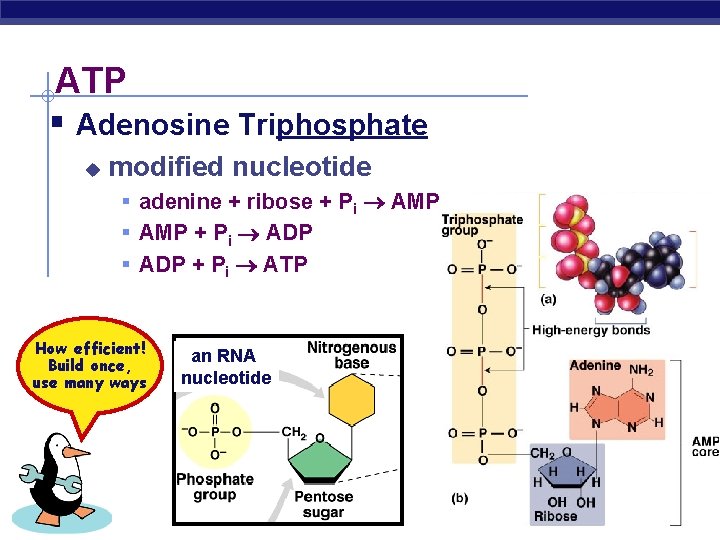
ATP § Adenosine Triphosphate u modified nucleotide § adenine + ribose + Pi AMP § AMP + Pi ADP § ADP + Pi ATP How efficient! Build once, use many ways AP Biology an RNA nucleotide 2005 -2006
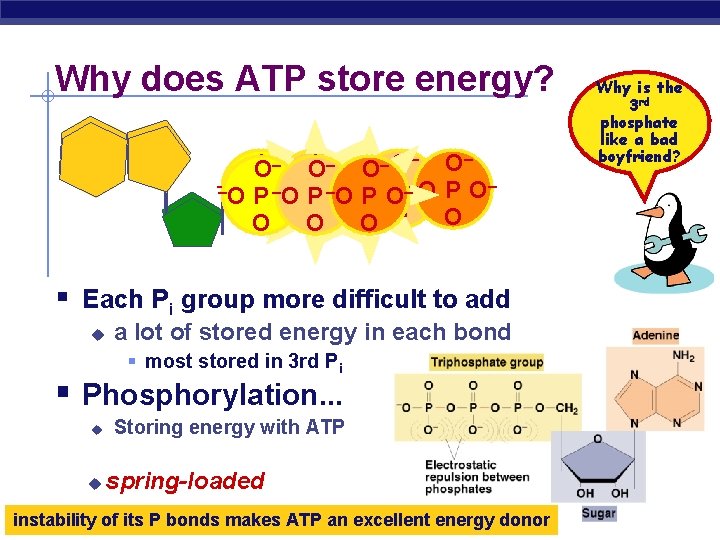
Why does ATP store energy? – – – – O O O O – –O– ––O P––O – – –O OP P –O O O– P O OPO O O O Why is the 3 rd phosphate like a bad boyfriend? § Each Pi group more difficult to add u a lot of stored energy in each bond § most stored in 3 rd Pi § Phosphorylation. . . u u Storing energy with ATP spring-loaded AP Biology instability of its P bonds makes ATP an excellent energy donor 2005 -2006
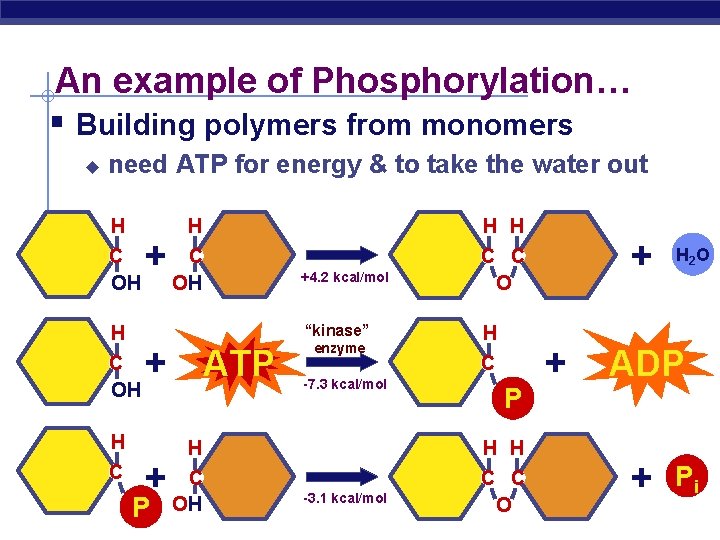
An example of Phosphorylation… § Building polymers from monomers u need ATP for energy & to take the water out H C OH + C OH C C O + ATP H AP Biology H H +4. 2 kcal/mol “kinase” H C H + P enzyme -7. 3 kcal/mol H C P H H H C OH C C O -3. 1 kcal/mol + + H 2 O ADP + 2005 -2006 Pi
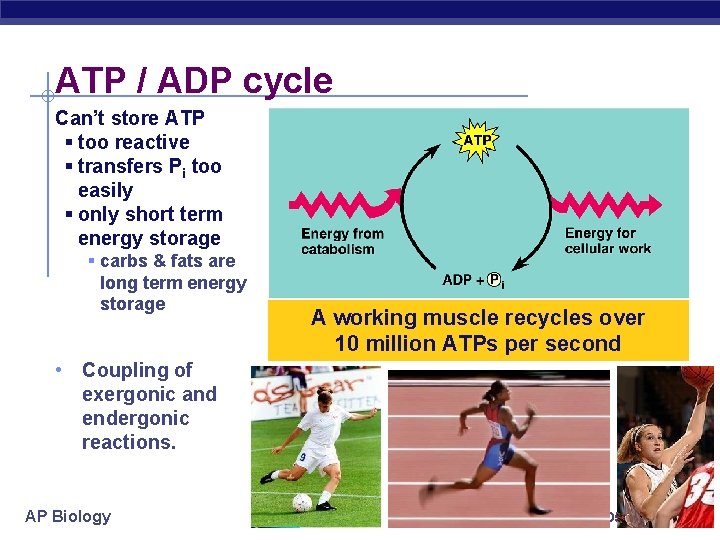
ATP / ADP cycle Can’t store ATP § too reactive § transfers Pi too easily § only short term energy storage § carbs & fats are long term energy storage A working muscle recycles over 10 million ATPs per second • Coupling of exergonic and endergonic reactions. AP Biology 2005 -2006

What’s the point? § Cells spend a lot of time making ATP! “The point is to make ATP!” For chemical, mechanical, and transport work Make ATP! That’s all I do all day. And no one even notices! AP Biology I didn’t HEAR you!
- Slides: 16