Metabolism Anabolism synthesis of materials n Catabolism breakdown

Metabolism Anabolism- synthesis of materials n Catabolism- breakdown of materials n Catabolism provides energy for life processes n Metabolic reactions require electron transfer n

Electron Transfer n Oxidation u Loss of electrons u Loss of Hydrogen u Addition of Oxygen n Reduction u Gain of electrons u Gain of Hydrogen u Loss of Oxygen

Electron Transfer Methane is fully reduced form of C n CO 2 is fully oxidized form of C n

Energy and C Sources Autotrophs- obtain C from CO 2 n Heterotrophs- obtain C from organic sources n Phototroph- energy from light n Chemotroph- energy from inorganic substances n

Photoautotrophs Use light for energy, CO 2 for carbon source n Includes green and purple sulfur bacteria, algae, green plants, and cyanobacteria n Green sulfur and purple sulfur B do not produce oxygen after photosynthesis n Use sulfur compounds (H 2 S) or hydrogen to reduce carbon dioxide n

Photoautotrophs Hydrogen sulfide ( S-2) is oxidized to sulfate (SO 4 -2) and Hydrogen to water n Photoheterotrophs- use light for energy, but does not convert carbon dioxide to sugar n Carbon comes from alcohols, fatty acids, and carbohydrates as sources for carbon n Green nonsulfur and purple nonsulfur B n

Chemoautotrophs Use electrons from inorganic sources as energy, carbon dioxide for carbon n Hydrogen sulfide, Sulfur, ammonia, nitrite (NO 2 -) are used n Energy is stored as ATP n Chemoheterotrophs- Organic sources used for both energy and carbon n

Factors Influencing Reaction Rate Temperature n p. H n Enzymes/Inhibitors n Concentration n State of Subdivision (size of particles) n

Inhibitors Competitive- fill the active site of the enzyme n Compete with normal substrate for the enzyme n Noncompetitive- do not compete with the substrate for the active site n Binds to another part of the enzymecalled “allosteric inhibition”- changes shape of the active site n
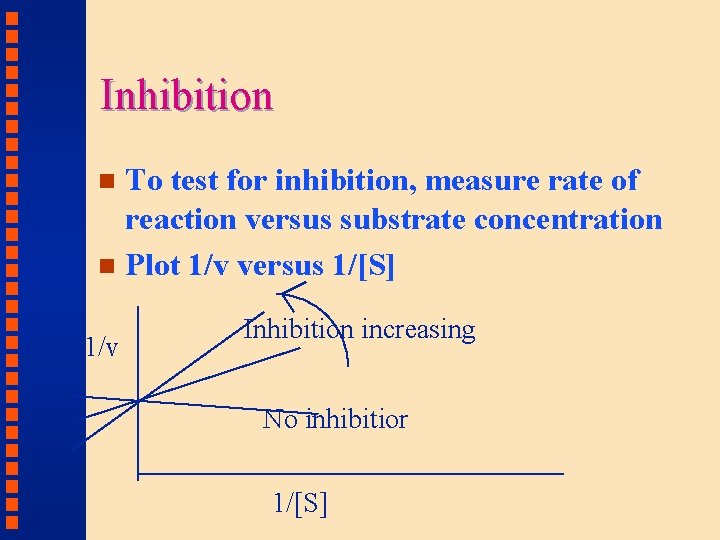
Inhibition To test for inhibition, measure rate of reaction versus substrate concentration n Plot 1/v versus 1/[S] n 1/v Inhibition increasing No inhibitior 1/[S]
![Inhibition n Noncompetitive inhibition 1/v 1/[S] Inhibition n Noncompetitive inhibition 1/v 1/[S]](http://slidetodoc.com/presentation_image_h2/3b588e106516540edb7d1f25365a6831/image-11.jpg)
Inhibition n Noncompetitive inhibition 1/v 1/[S]

Feedback Inhibition Product of a metabolic pathway inhibits the metabolic pathway n. A B C D E n E can inhibit reaction B under some conditions n

Respiration Complete oxidation of glucose to carbon dioxide n Consists of glycolysis, Krebs Cycle, and oxidative phosphorylation n

Pentose Phosphate Shunt Provides means to break down pentoses n Provides key intermediates that are precursors in synthesis of nucleic acids, glucose in photosynthesis, and some amino acids n Produces 12 NADPH/ glu, but only makes 1 ATP/ glu n B. Subtilis, E. Coli use this, in addition to glycolysis n

Entner- Doudoroff Pathway Substitutes for glycolysis n Produces 2 NADPH/ glu and 1 ATP/ glu n Found in some G - B, including Pseudomonas n Rarely found among G+ B n

Fermentation After pyruvic acid is formed in glycolysis, it can be fermented n Does not require O 2 - anaerobic n Does not use Krebs cycle, or ETC n Uses organic molecule as final electron acceptor n Produces little ATP n

Lactic Acid Fermentation Seen already on glycolysis chart n Pyruvic acid is reduced by NADH n Streptococcus does this- found in food spoilage, as well as yogurt, sauerkraut, and pickles n

Alcohol Fermentation Pyruvic acid loses CO 2 to form acetaldehyde (CH 3 COH) n Then reduced by NADH to form ethanol (CH 3 CH 2 OH) n Sacchromyces (yeast) uses the carbon dioxide to make dough rise n Can have homolactic (only lactic acid) or heterolactic (produce alcohol and lactic acid) n

Lipid Catabolism Lipases break down lipids into glycerol and fatty acids n Glycerol is converted into dihydroxyacetone phosphate n Then metabolized by glycolysis n Fatty acids are then broken down two carbons at a time and converted into acetyl groups, then acetyl co. A n

Protein Catabolism Proteases and peptidases break down proteins into amino acids n Deamination removes the amino group, converting it into ammonia n Remaining organic acid enters the Krebs Cycle n Decarboxylation also can occur n

Photosynthesis Cyclic- electrons return to chlorophyll n Noncyclic- electrons end up in NADPH n

Nutritional Patterns Phototrophs- get energy from light n Chemotrophs- get energy from inorganic or organic sources n Autotrophs- get Carbon from carbon dioxide n Heterotrophs- get carbon from organic sources n

Photoautotrophs Use light for energy, get carbon from carbon dioxide n Includes photosynthetic bacteria (green sulfur and purple sulfur and cyanobacteria), algae, and green plants n In most cases, oxygen is given offoxygenic n

Photoautotrophs Not all are oxygenic- can be anoxygenic n Green sulfur B use sulfur compounds H 2 S or H 2 to reduce carbon dioxide and produce sulfate (SO 4 -2) or water n Purple sulfur B do the same thing but have different chlorophyll, and thus a different color n

Photoheterotrophs Use light as source of energy n Cannot convert carbon dioxide to sugarinstead use organic compounds such as alcohols and fatty acids as source of carbon n Green nonsulfur bacteria (Chloroflexus) and purple nonsulfur B (Rhodopseudomonas) n

Chemoautotrophs Use electrons from reduced inorganic compounds as energy source n Use carbon dioxide as carbon source n Inorganic compounds include H 2 S, S, NH 3, H 2 n Nitrobacter uses nitrite (NO 2 -) n

Chemoheterotrophs Use organic compounds for both energy source and for carbon source n Most organisms are chemoheterotrophs n Saprophytes- live on dead organic matter n Parasites- live on living host n
- Slides: 27