Metabolism alcohol and toxicity Best of Digital ILC

Metabolism, alcohol and toxicity Best of Digital ILC 2020

About these slides • These slides provide highlights of new data presented at Digital ILC 2020 • Please feel free to use, adapt, and share these slides for your own personal use; however, please acknowledge EASL as the source • Definitions of all abbreviations shown in these slides, and a full copy of the original abstract text, are provided within the slide notes • Submitted abstracts are included in the slide notes, but data may not be identical to the final presented data shown on the slides • When you see a symbol like this: , you can click on it to return to the contents page These slides are intended for use as an educational resource and should not be used to make patient management decisions. All information included should be verified before treating patients or using any therapies described in these materials

Contents 1. NAFLD: target identification and drug pipeline GS 11 Macrophage scavenger receptor 1 mediates lipid-induced inflammation in human obesity-related non-alcoholic fatty liver disease AS 017 A polygenic risk score for progressive non-alcoholic fatty liver disease risk stratification AS 018 Carbohydrate restriction reverses NAFLD by altering hepatic mitochondrial fluxes in humans Click here to skip to this section 2. NAFLD: pharmacological therapy LBO 01 AS 076 Positive topline results from a 24 -week, randomized, double-blind, placebo-controlled, multicentre, phase 2 study of the FGF 19 analogue aldafermin (NGM 282) in patients with nonalcoholic steatohepatitis Effects of cotadutide on biomarkers of non-alcoholic steatohepatitis in overweight or obese subjects with type 2 diabetes mellitus: a 54 -week analysis of a randomized phase 2 b study Click here to skip to this section 3. Gut liver axis AS 110 MCJ deficiency results in gut barrier dysfunction and macrophage-elicited inflammation following ethanol consumption, facilitating alcoholic endotoxemia Click here to skip to this section
Contents 4. NAFLD: experimental AS 133 Myosteatosis is associated with early NASH in the context of obesity and metabolic syndrome Click here to skip to this section 5. NAFLD: clinical aspects except therapy THU 001 THU 053 Adipose tissue insulin resistance and inflammation, but not reduced hepatic fat oxidation, are associated to active NASH and severe fibrosis Europe's largest meta-analysis on the prevalence of non-alcoholic fatty liver disease, non-alcoholic steatohepatitis and advanced fibrosis (F 3−F 4) Click here to skip to this section 6. NAFLD: diagnostics and non-invasive assessment FRI 003 FRI 008 Machine learning models identify novel histologic features predictive of clinical disease progression in patients with advanced fibrosis due to non-alcoholic steatohepatitis Predictive modelling to identify and characterize fast progressors among patients with non-alcoholic steatohepatitis Click here to skip to this section
Contents 7. NAFLD: experimental and pathophysiology LBO 05 Genome-wide association study for alcohol-related cirrhosis identifies new risk loci in MARC 1 and HNRNPUL 1 SAT 065 Infiltrative monocyte derived adenosine deaminase 2 promotes liver fibrosis via signalling through extracellular inosine Click here to skip to this section

1. NAFLD: target identification and drug pipeline

Macrophage scavenger receptor 1 mediates lipid-induced inflammation in human obesity-related NAFLD BACKGROUND & AIMS • Low-grade, chronic hepatic inflammation (steatohepatitis) plays a key role in NAFLD pathogenesis – Inter-patient variation is significant – Mechanisms underpinning obesityassociated inflammation, and thus disease progression, remain unclear • Pro-inflammatory activation of macrophages by saturated fatty acids is independent of TLR 4 – True receptor remains unknown • AIM: to investigate the role of MSR 1* in NAFLD by combining human transcriptomic and genetic data with Tg mouse models and in vitro assays *A phagocytic receptor also known as SR-A or CD 204; †From multiple centres in Europe Govaere O, et al. DILC 2020; GS 11 METHODS In vivo study • MSR 1 -/- and WT mice were fed on high-fat and highcholesterol diet • Tissue-specific macrophages isolated using magnetic beads/FACS • BMDMs co-cultured with adipocyte-like or hepatoma cells in the presence of lipids • Read-out based on histological and biochemical results, Seahorse and MS nano. String® • 214 liver and adipose tissue samples • Caucasian patients Histopathological and transcriptomics study GWAS • 1, 483 Caucasian patients with biopsy -proven NAFLD† • 17, 781 European general-population controls SNP analysis UK Biobank cohort • 430, 101 patients Quantitative trait analysis
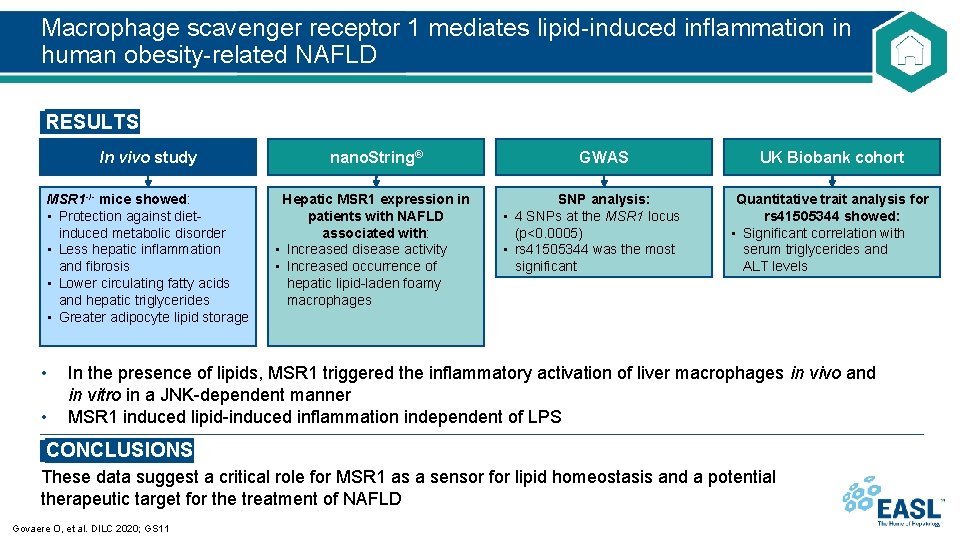
Macrophage scavenger receptor 1 mediates lipid-induced inflammation in human obesity-related NAFLD RESULTS • • In vivo study nano. String® MSR 1 -/- mice showed: • Protection against dietinduced metabolic disorder • Less hepatic inflammation and fibrosis • Lower circulating fatty acids and hepatic triglycerides • Greater adipocyte lipid storage Hepatic MSR 1 expression in patients with NAFLD associated with: • Increased disease activity • Increased occurrence of hepatic lipid-laden foamy macrophages GWAS SNP analysis: • 4 SNPs at the MSR 1 locus (p<0. 0005) • rs 41505344 was the most significant UK Biobank cohort Quantitative trait analysis for rs 41505344 showed: • Significant correlation with serum triglycerides and ALT levels In the presence of lipids, MSR 1 triggered the inflammatory activation of liver macrophages in vivo and in vitro in a JNK-dependent manner. MSR 1 induced lipid-induced inflammation independent of LPS CONCLUSIONS These data suggest a critical role for MSR 1 as a sensor for lipid homeostasis and a potential therapeutic target for the treatment of NAFLD Govaere O, et al. DILC 2020; GS 11
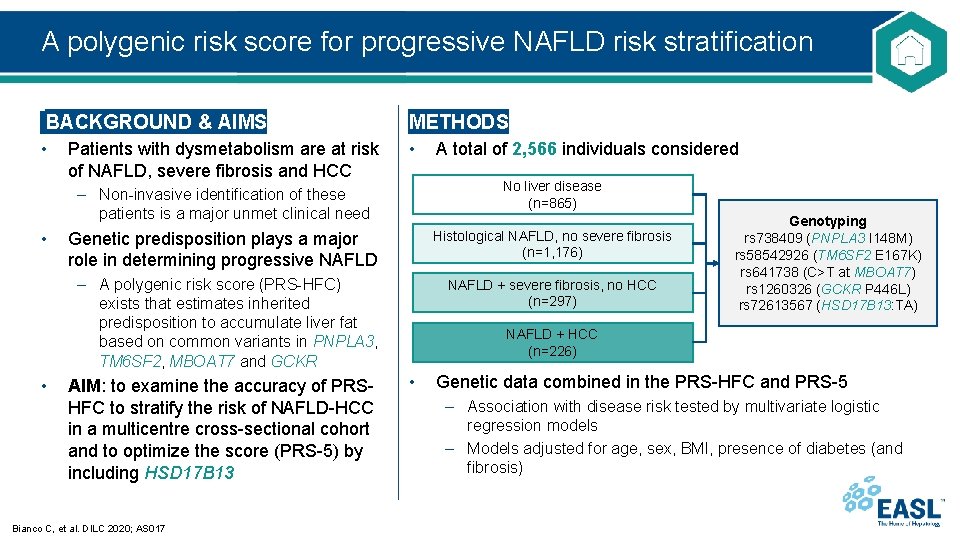
A polygenic risk score for progressive NAFLD risk stratification BACKGROUND & AIMS • Patients with dysmetabolism are at risk of NAFLD, severe fibrosis and HCC • METHODS • – Non-invasive identification of these patients is a major unmet clinical need No liver disease (n=865) Genetic predisposition plays a major role in determining progressive NAFLD Histological NAFLD, no severe fibrosis (n=1, 176) – A polygenic risk score (PRS-HFC) exists that estimates inherited predisposition to accumulate liver fat based on common variants in PNPLA 3, TM 6 SF 2, MBOAT 7 and GCKR • A total of 2, 566 individuals considered AIM: to examine the accuracy of PRSHFC to stratify the risk of NAFLD-HCC in a multicentre cross-sectional cohort and to optimize the score (PRS-5) by including HSD 17 B 13 Bianco C, et al. DILC 2020; AS 017 NAFLD + severe fibrosis, no HCC (n=297) Genotyping rs 738409 (PNPLA 3 I 148 M) rs 58542926 (TM 6 SF 2 E 167 K) rs 641738 (C>T at MBOAT 7) rs 1260326 (GCKR P 446 L) rs 72613567 (HSD 17 B 13: TA) NAFLD + HCC (n=226) • Genetic data combined in the PRS-HFC and PRS-5 – Association with disease risk tested by multivariate logistic regression models – Models adjusted for age, sex, BMI, presence of diabetes (and fibrosis)

A polygenic risk score for progressive NAFLD risk stratification RESULTS CONCLUSION • PRS was more robust than single variants in predicting NAFLD-HCC • Polygenic PRS improved detection accuracy for NAFLD-HCC compared with single variants • Overall cohort • Genetically determined hepatic fat accumulation causes liver fibrosis. Although results do not exclude an independent contribution due to pleiotropic effects of specific variants, predisposition to HCC development seemed mainly dependent on the induction of fibrosis • Studies evaluating PRS to improve diagnostic accuracy of classical risk factors for NAFLD, severe fibrosis, and HCC are ongoing – AUROC was 0. 64 for PRS-HFC – OR for PRS-HFC ≥ 0. 532: 3. 0 (95% CI, 2. 2– 3. 9, p<0. 01) • Patients with dysmetabolism – Prediction in patients with diabetes* • OR for PRS-HFC ≥ 0. 532: 1. 6 (95% CI, 1. 1– 2. 5, p=0. 02) – Independent of fibrosis severity – Prediction in obese patients* • OR for PRS-HFC ≥ 0. 532: 2. 7 (95% CI, 1. 8– 4. 0; p<0. 001) • Patients without severe fibrosis – OR for PRS-HFC ≥ 0. 532: 2. 0 (95% CI, 1. 1– 2. 8; p=0. 03) *Independently of confounders Bianco C, et al. DILC 2020; AS 017

Carbohydrate restriction reverses NAFLD by altering hepatic mitochondrial fluxes in humans BACKGROUND & AIMS • CR has emerged as an effective dietary regimen in the management of NAFLD • – Intrahepatic mechanisms remain poorly understood • – Mean age: 58 2 years – Mean BMI: 32 2 kg/m 2 – Sex: 50% male AIM: to examine mechanisms by which CR improves liver metabolism in vivo in humans before and after a 6 -day CR diet using PINTA • Assessments – Compliance METHODS • Isotope infusions were performed in 10 overweight/obese participants before and after a 6 -day CR diet • Measuring plasma -hydroxybutyrate and acetoacetate concentrations • 3 -day food records ]-glucose, [13 C Infusion of [D 7 4]-hydroxybutyrate and [3 -13 C]-lactate were used to quantify: – Endogenous glucose production – Ketogenesis – Hepatic citrate synthase (VCS) and pyruvate carboxylase (VPC) fluxes by positional isotopomer NMR tracer analysis (PINTA) *Determined by ratio of plasma -hydroxybutyrate and acetoacetate Luukkonen P, et al. DILC 2020; AS 018 – Liver fat: magnetic resonance spectroscopy – Liver stiffness: magnetic resonance elastography • Potential regulators of VCS: hepatic mitochondrial redox state* and plasma concentrations of leptin and triiodothyronine were also measured

Carbohydrate restriction reverses NAFLD by altering hepatic mitochondrial fluxes in humans • RESULTS • • – Mobilization of fatty acids (+35%) – Ketogenesis (+232%) – VCS (-38%) Carbohydrate intake very low with CR diet* – Before CR diet: 183 ± 20 g/day – During CR diet: 23 ± 1 g/day p<0. 000001 Together with serum insulin limit hepatic lipogenesis p<0. 01 with CR diet p value Plasma -hydroxybutyrate 10 -fold <0. 001 Acetoacetate 6 -fold <0. 001 Body weight 3% <0. 001 Endogenous glucose production 22% <0. 001 Serum insulin concentrations 53% <0. 001 Liver fat content 31% <0. 001 Assessment *Intakes of other macronutrients were unchanged Luukkonen P, et al. DILC 2020; AS 018 p<0. 001 • Associated with mitochondrial redox state (+167%) Daily energy intake decreased with CR diet – Before CR diet: 2, 019 ± 177 kcal – During CR diet: 1, 444 kcal Reductions in liver fat content attributed to: CONCLUSION • CR reverses NAFLD in humans by altering hepatic mitochondrial fluxes to promote hepatic fatty acid disposal

2. NAFLD: pharmacological therapy

Positive topline results from a 24 -week, randomized, double-blind, placebocontrolled, multicenter, phase 2 study of aldafermin in patients with NASH BACKGROUND & AIMS • Aldafermin (NGM 282) is an engineered FGF 19 analogue – Significantly inhibited bile acid synthesis, reduced hepatic fibrosis, inflammation and steatosis in previous 12 -week NASH trials • AIM: to report the primary and key results from the 24 -week study with paired liver biopsy METHODS 78 subjects (from 9 US study sites) • • Inclusion criteria: biopsy-proven NASH* • • Primary endpoint: change in LFC from baseline to Week 24 Histological endpoints: – Improvement in liver fibrosis by ≥ 1 stage with no worsening of NASH – Resolution of NASH with no worsening of fibrosis† RESULTS • At Week 24, treatment with aldafermin resulted in statistically significant reductions in LFC PBO (n=25) R Absolute MRI-PDF, % Aldafermin (n=53) 1 mg SC QD Subjects underwent MRI-PDFF and liver biopsy at baseline and Week 24 % Subjects with 5% absolute Relative MRI-PDFF, % % Subjects with 30% relative Shown are LS mean (SE) or % subjects *NAS ≥ 4, stage 2− 3 fibrosis, absolute LFC ≥ 8%; †NASH CRN criteria; ‡One patient did not have any post-baseline measurements and was excluded from efficacy analysis as pre-specified in the statistical analysis plan Harrison SA, et al. DILC 2020; LBO 01 PBO (n=25) Aldafermin (n=52‡) p value -2. 7 (1. 3) -7. 7 (0. 8) 0. 002 24 68 <0. 001 -13. 1 -38. 8 0. 008 29 66 0. 004
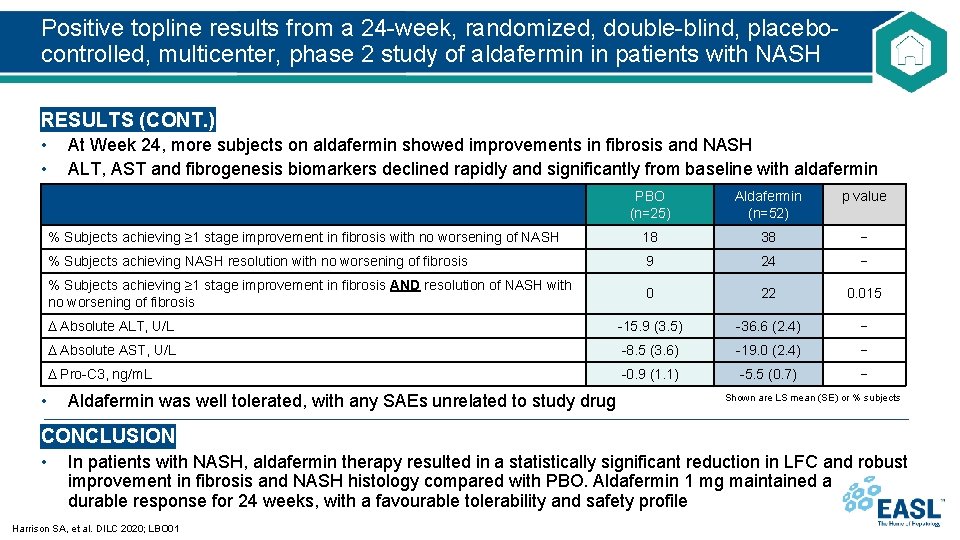
Positive topline results from a 24 -week, randomized, double-blind, placebocontrolled, multicenter, phase 2 study of aldafermin in patients with NASH RESULTS (CONT. ) • • • At Week 24, more subjects on aldafermin showed improvements in fibrosis and NASH ALT, AST and fibrogenesis biomarkers declined rapidly and significantly from baseline with aldafermin PBO (n=25) Aldafermin (n=52) p value % Subjects achieving ≥ 1 stage improvement in fibrosis with no worsening of NASH 18 38 − % Subjects achieving NASH resolution with no worsening of fibrosis 9 24 − % Subjects achieving ≥ 1 stage improvement in fibrosis AND resolution of NASH with no worsening of fibrosis 0 22 0. 015 Absolute ALT, U/L -15. 9 (3. 5) -36. 6 (2. 4) − Absolute AST, U/L -8. 5 (3. 6) -19. 0 (2. 4) − Pro-C 3, ng/m. L -0. 9 (1. 1) -5. 5 (0. 7) − Aldafermin was well tolerated, with any SAEs unrelated to study drug Shown are LS mean (SE) or % subjects CONCLUSION • In patients with NASH, aldafermin therapy resulted in a statistically significant reduction in LFC and robust improvement in fibrosis and NASH histology compared with PBO. Aldafermin 1 mg maintained a durable response for 24 weeks, with a favourable tolerability and safety profile Harrison SA, et al. DILC 2020; LBO 01
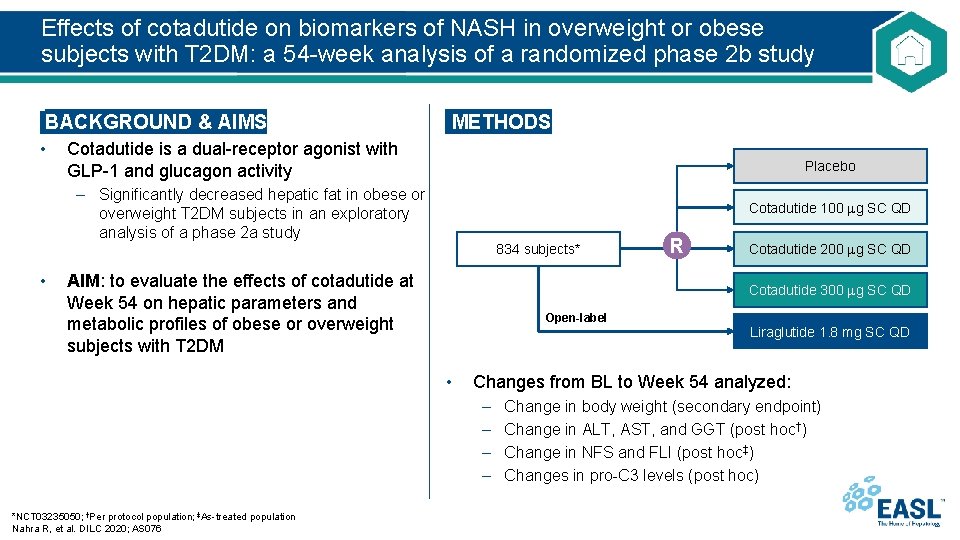
Effects of cotadutide on biomarkers of NASH in overweight or obese subjects with T 2 DM: a 54 -week analysis of a randomized phase 2 b study BACKGROUND & AIMS • Cotadutide is a dual-receptor agonist with GLP-1 and glucagon activity METHODS Placebo – Significantly decreased hepatic fat in obese or overweight T 2 DM subjects in an exploratory analysis of a phase 2 a study • Cotadutide 100 g SC QD 834 subjects* AIM: to evaluate the effects of cotadutide at Week 54 on hepatic parameters and metabolic profiles of obese or overweight subjects with T 2 DM Cotadutide 200 g SC QD Cotadutide 300 g SC QD Open-label • Liraglutide 1. 8 mg SC QD Changes from BL to Week 54 analyzed: – – *NCT 03235050; †Per protocol population; ‡As-treated population Nahra R, et al. DILC 2020; AS 076 R Change in body weight (secondary endpoint) Change in ALT, AST, and GGT (post hoc†) Change in NFS and FLI (post hoc‡) Changes in pro-C 3 levels (post hoc)
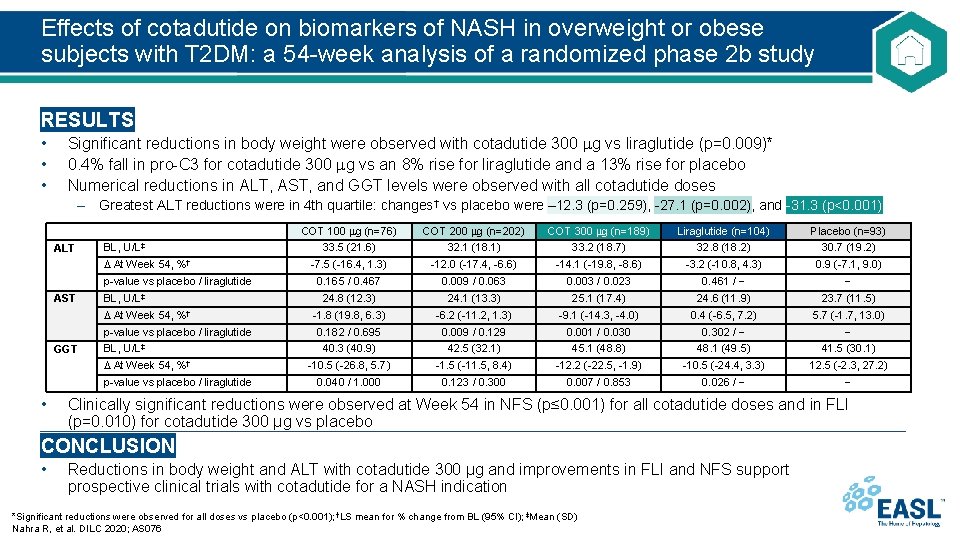
Effects of cotadutide on biomarkers of NASH in overweight or obese subjects with T 2 DM: a 54 -week analysis of a randomized phase 2 b study RESULTS • • • Significant reductions in body weight were observed with cotadutide 300 g vs liraglutide (p=0. 009)* 0. 4% fall in pro-C 3 for cotadutide 300 g vs an 8% rise for liraglutide and a 13% rise for placebo Numerical reductions in ALT, AST, and GGT levels were observed with all cotadutide doses – Greatest ALT reductions were in 4 th quartile: changes† vs placebo were – 12. 3 (p=0. 259), -27. 1 (p=0. 002), and -31. 3 (p<0. 001) ALT BL, U/L‡ At Week 54, %† p-value vs placebo / liraglutide AST BL, U/L‡ At Week 54, %† p-value vs placebo / liraglutide GGT BL, U/L‡ At Week 54, %† p-value vs placebo / liraglutide • COT 100 g (n=76) COT 200 g (n=202) COT 300 g (n=189) Liraglutide (n=104) Placebo (n=93) 33. 5 (21. 6) 32. 1 (18. 1) 33. 2 (18. 7) 32. 8 (18. 2) 30. 7 (19. 2) -7. 5 (-16. 4, 1. 3) -12. 0 (-17. 4, -6. 6) -14. 1 (-19. 8, -8. 6) -3. 2 (-10. 8, 4. 3) 0. 9 (-7. 1, 9. 0) 0. 165 / 0. 467 0. 009 / 0. 063 0. 003 / 0. 023 0. 461 / − − 24. 8 (12. 3) 24. 1 (13. 3) 25. 1 (17. 4) 24. 6 (11. 9) 23. 7 (11. 5) -1. 8 (19. 8, 6. 3) -6. 2 (-11. 2, 1. 3) -9. 1 (-14. 3, -4. 0) 0. 4 (-6. 5, 7. 2) 5. 7 (-1. 7, 13. 0) 0. 182 / 0. 695 0. 009 / 0. 129 0. 001 / 0. 030 0. 302 / − − 40. 3 (40. 9) 42. 5 (32. 1) 45. 1 (48. 8) 48. 1 (49. 5) 41. 5 (30. 1) -10. 5 (-26. 8, 5. 7) -1. 5 (-11. 5, 8. 4) -12. 2 (-22. 5, -1. 9) -10. 5 (-24. 4, 3. 3) 12. 5 (-2. 3, 27. 2) 0. 040 / 1. 000 0. 123 / 0. 300 0. 007 / 0. 853 0. 026 / − − Clinically significant reductions were observed at Week 54 in NFS (p≤ 0. 001) for all cotadutide doses and in FLI (p=0. 010) for cotadutide 300 µg vs placebo CONCLUSION • Reductions in body weight and ALT with cotadutide 300 µg and improvements in FLI and NFS support prospective clinical trials with cotadutide for a NASH indication *Significant reductions were observed for all doses vs placebo (p<0. 001); †LS mean for % change from BL (95% CI); ‡Mean (SD) Nahra R, et al. DILC 2020; AS 076

3. Gut–liver axis
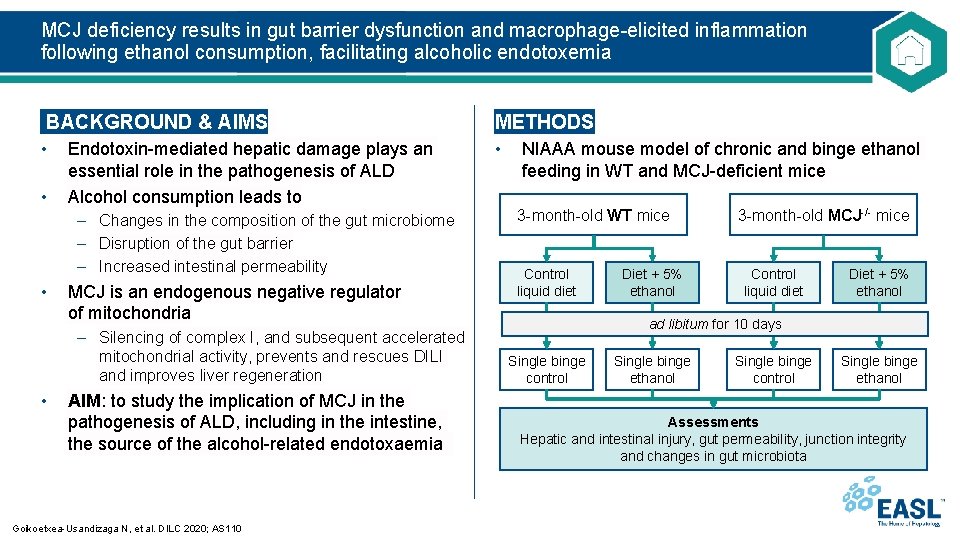
MCJ deficiency results in gut barrier dysfunction and macrophage-elicited inflammation following ethanol consumption, facilitating alcoholic endotoxemia BACKGROUND & AIMS METHODS • • • Endotoxin-mediated hepatic damage plays an essential role in the pathogenesis of ALD Alcohol consumption leads to – Changes in the composition of the gut microbiome – Disruption of the gut barrier – Increased intestinal permeability • MCJ is an endogenous negative regulator of mitochondria – Silencing of complex I, and subsequent accelerated mitochondrial activity, prevents and rescues DILI and improves liver regeneration • AIM: to study the implication of MCJ in the pathogenesis of ALD, including in the intestine, the source of the alcohol-related endotoxaemia Goikoetxea-Usandizaga N, et al. DILC 2020; AS 110 NIAAA mouse model of chronic and binge ethanol feeding in WT and MCJ-deficient mice 3 -month-old WT mice Control liquid diet Diet + 5% ethanol 3 -month-old MCJ-/- mice Control liquid diet Diet + 5% ethanol ad libitum for 10 days Single binge control Single binge ethanol Assessments Hepatic and intestinal injury, gut permeability, junction integrity and changes in gut microbiota

MCJ deficiency results in gut barrier dysfunction and macrophage-elicited inflammation following ethanol consumption, facilitating alcoholic endotoxemia RESULTS • Whole-body MCJ knockout was detrimental in endotoxin-mediated hepatic damage – 85% of ethanol-fed WT mice survived compared with 54% of ethanol-fed MCJ-/- mice • No differences were observed in both ethanol-fed WT and MCJ-/- livers – However, histological scores were higher in the intestine of ethanol-fed MCJ-/- mice • In ethanol-fed MCJ-/- compared with WT mice: – expression of TNF and IL-1ß detected by q. PCR – levels of F 4/80 in the gut on IHC • Suggests MCJ silencing causes infiltration of macrophages in the intestine, increasing the proinflammatory response Gut microbiota was altered in control-fed MCJ-/- compared with WT mice* – Worsened after ethanol diet, consistent with a dysbiosis event in ethanol-fed MCJ-/- mice Control-fed MCJ-/- mice Ethanol-fed MCJ-/- mice Ruminococcaceae, Bifidobacteriaceae Prevotella (degrades mucin leading to gut barrier disruption) Bifidobacteriaceae, Lactobacillaceae and Ruminococcaceae (maintain mucosal barrier integrity) • Intestinal permeability was greater† and junction integrity decreased‡ in ethanol-fed MCJ-/- mice vs controls – Facilitating bacterial and microbial product translocation CONCLUSION • Enhanced mitochondrial activity, due to MCJ silencing results in bacterial alterations, increased macrophage infiltration, and augmented inflammatory response after ethanol consumption, disrupting the gut barrier and increasing gut permeability to facilitate alcohol endotoxaemia and worsen ALD *Via sequencing V 3 -V 4 regions of 16 S r. DNA amplicon; †Evaluated with FITC-labelled dextran; ‡Reduced expression of tight junction proteins detected by q. PCR and IHC Goikoetxea-Usandizaga N, et al. DILC 2020; AS 110

4. NAFLD: experimental
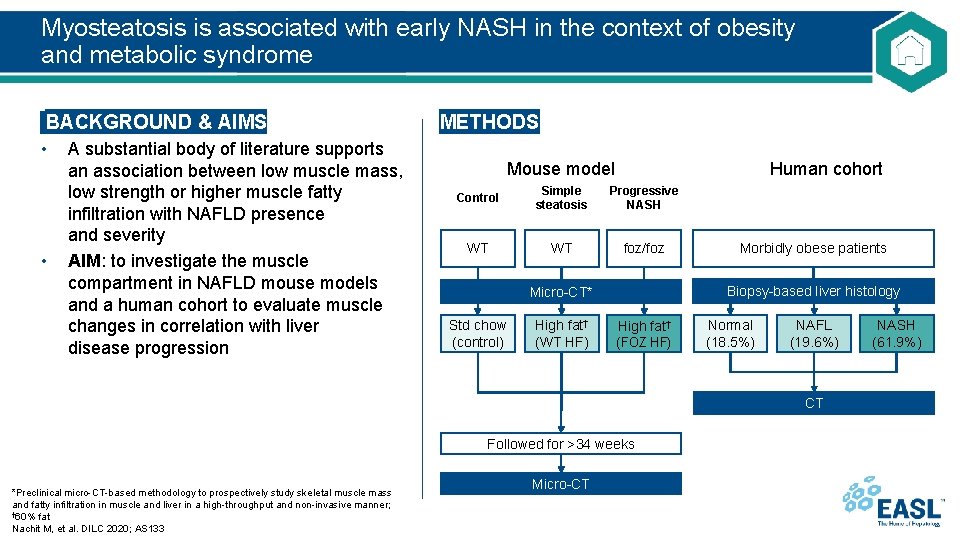
Myosteatosis is associated with early NASH in the context of obesity and metabolic syndrome BACKGROUND & AIMS • A substantial body of literature supports an association between low muscle mass, low strength or higher muscle fatty infiltration with NAFLD presence and severity • AIM: to investigate the muscle compartment in NAFLD mouse models and a human cohort to evaluate muscle changes in correlation with liver disease progression METHODS Mouse model Human cohort Control Simple steatosis Progressive NASH WT WT foz/foz Biopsy-based liver histology Micro-CT* Std chow (control) High fat† (WT HF) Morbidly obese patients High fat† (FOZ HF) Normal (18. 5%) NAFL (19. 6%) CT Followed for >34 weeks *Preclinical micro-CT-based methodology to prospectively study skeletal muscle mass and fatty infiltration in muscle and liver in a high-throughput and non-invasive manner; † 60% fat Nachit M, et al. DILC 2020; AS 133 Micro-CT NASH (61. 9%)
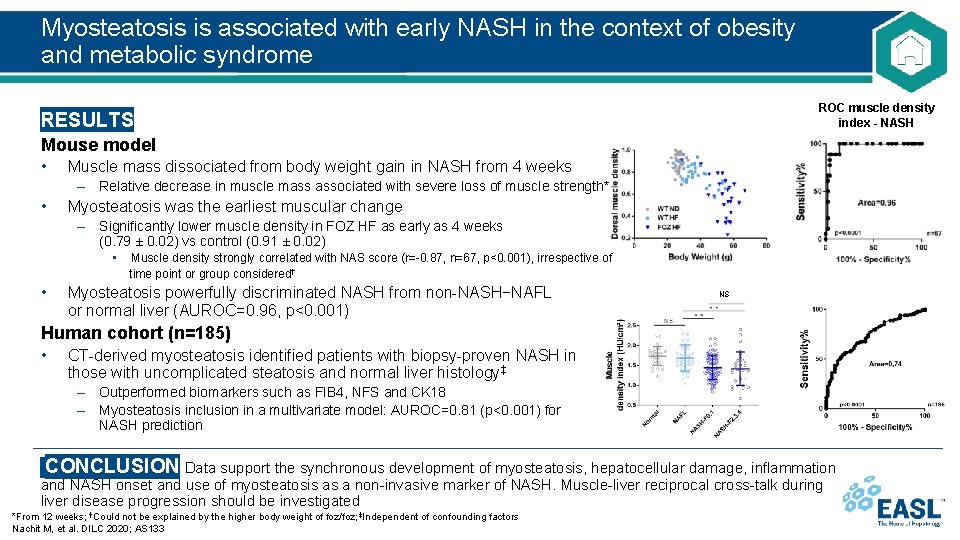
Myosteatosis is associated with early NASH in the context of obesity and metabolic syndrome ROC muscle density index - NASH RESULTS Mouse model • Muscle mass dissociated from body weight gain in NASH from 4 weeks – Relative decrease in muscle mass associated with severe loss of muscle strength* • Myosteatosis was the earliest muscular change – Significantly lower muscle density in FOZ HF as early as 4 weeks (0. 79 ± 0. 02) vs control (0. 91 ± 0. 02) • Muscle density strongly correlated with NAS score (r=-0. 87, n=67, p<0. 001), irrespective of time point or group considered† • Myosteatosis powerfully discriminated NASH from non-NASH−NAFL or normal liver (AUROC=0. 96, p<0. 001) NS Human cohort (n=185) • CT-derived myosteatosis identified patients with biopsy-proven NASH in those with uncomplicated steatosis and normal liver histology‡ – Outperformed biomarkers such as FIB 4, NFS and CK 18 – Myosteatosis inclusion in a multivariate model: AUROC=0. 81 (p<0. 001) for NASH prediction CONCLUSION Data support the synchronous development of myosteatosis, hepatocellular damage, inflammation and NASH onset and use of myosteatosis as a non-invasive marker of NASH. Muscle-liver reciprocal cross-talk during liver disease progression should be investigated *From 12 weeks; †Could not be explained by the higher body weight of foz/foz; ‡Independent of confounding factors Nachit M, et al. DILC 2020; AS 133

5. NAFLD: clinical aspects except therapy

Adipose tissue insulin resistance and inflammation, but not reduced hepatic fat oxidation, are associated with active NASH and severe fibrosis BACKGROUND & AIMS • Mechanisms that underpin the progression from NAFLD to NASH and to advanced liver disease are unclear – Reduced liver mitochondrial fat oxidation and VLDL secretion as consequences of increased fat overflow to the liver are implicated • AIM: to investigate a possible link between increased AT insulin resistance (Adipo-IR)*, reduced BOH†, and reduced Apo. B‡, and a more severe form of NAFLD/NASH METHODS EPo. S European NAFLD Registry 318 histologically characterized NAFLD subjects (188 with NASH) Circulating markers measured AT dysfunction FFA Adipo-IR=FFAx. Ins AT inflammation Leptin Adiponectin TNF-α MCP-1 Hepatic lipid oxidation BOH Data analysis by LMA to calculate OR (adjusted for age, gender and BMI) *Increased Adipo-IR reflects impaired lipolysis suppression; †Reduced BOH reflects hepatic mitochondrial fat oxidation; ‡Apo. B is a rate-limiting factor for VLDL secretion and thus implicated in the secretion of hepatic TG Gastaldelli A, et al. DILC 2020; THU 001 Hepatic lipid secretion TG Apo. B

Adipose tissue insulin resistance and inflammation, but not reduced hepatic fat oxidation, are associated with active NASH and severe fibrosis RESULTS Marker (mean ± SEM) Subjects with NASH (n=188) Subjects with NAFL (n=130) p value BMI, kg/m 2 32. 9 ± 0. 4 31. 1 ± 0. 6 p<0. 001 Adipo-IR=FFAx. Ins 23. 2 ± 1. 5 16. 3 ± 1. 6 p<0. 001 TNF-α, pg/m. L 4. 9 ± 0. 2 3. 3 ± 0. 2 p<0. 001 MCP-1, pg/m. L 112 ± 3 92 ± 3 p<0. 001 Leptin, ng/m. L 13. 8 ± 1. 0 10. 4 ± 1. 0 p<0. 001 Adiponectin, μg/m. L 11. 0 ± 0. 6 13. 5 ± 0. 8 p<0. 001 TG, mg/d. L 177 ± 10 165 ± 12 ns Apo. B, mg/d. L 76 ± 2 78 ± 3 ns BOH, μmol/L 77 ± 8 76 ± 8 ns FFA, mmol/L 0. 59 ± 0. 02 0. 57 ± 0. 02 ns • Dysfunctional AT* independently associated with: – steatosis (OR=8. 3, p<0. 001) – Active NASH (OR=3. 1, p<0. 001) – Fibrosis F 3–F 4 (OR=2. 5, p<0. 001) in NASH • in NASH Surprisingly, BOH† increased in subjects with fibrosis F 3–F 4 – Associated with Adipo-IR (overflow of fat from AT to the liver [p<0. 0001]) • Adipo-IR showed a strong positive correlation with TG, leptin, TNF-α and MCP-1 levels (p<0. 0001) CONCLUSIONS NAFLD severity is strongly associated with markers of AT dysfunction and inflammation, but mitochondrial beta oxidation does not appear to be dysfunctional. AT plays a significant role in the progression of NAFL to NASH *Shown by increased Adipo-IR; †Which reflects hepatic mitochondrial fat oxidation Gastaldelli A, et al. DILC 2020; THU 001
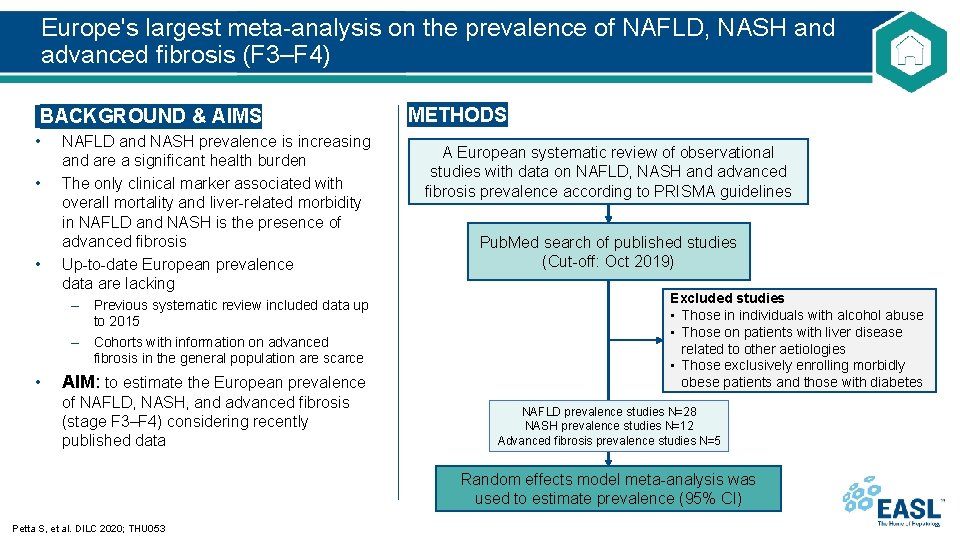
Europe's largest meta-analysis on the prevalence of NAFLD, NASH and advanced fibrosis (F 3–F 4) BACKGROUND & AIMS • • • NAFLD and NASH prevalence is increasing and are a significant health burden The only clinical marker associated with overall mortality and liver-related morbidity in NAFLD and NASH is the presence of advanced fibrosis Up-to-date European prevalence data are lacking – Previous systematic review included data up to 2015 – Cohorts with information on advanced fibrosis in the general population are scarce • AIM: to estimate the European prevalence of NAFLD, NASH, and advanced fibrosis (stage F 3–F 4) considering recently published data METHODS A European systematic review of observational studies with data on NAFLD, NASH and advanced fibrosis prevalence according to PRISMA guidelines Pub. Med search of published studies (Cut-off: Oct 2019) Excluded studies • Those in individuals with alcohol abuse • Those on patients with liver disease related to other aetiologies • Those exclusively enrolling morbidly obese patients and those with diabetes NAFLD prevalence studies N=28 NASH prevalence studies N=12 Advanced fibrosis prevalence studies N=5 Random effects model meta-analysis was used to estimate prevalence (95% CI) Petta S, et al. DILC 2020; THU 053
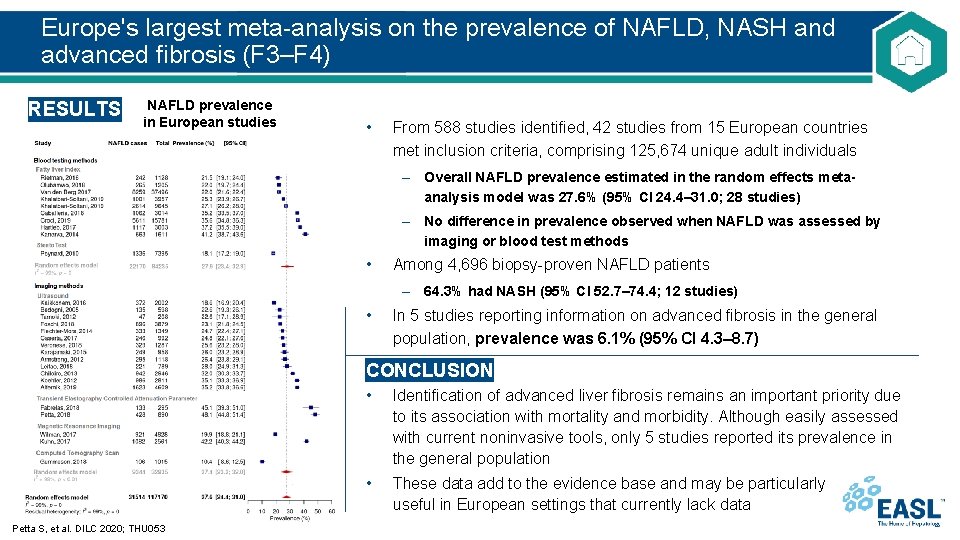
Europe's largest meta-analysis on the prevalence of NAFLD, NASH and advanced fibrosis (F 3–F 4) RESULTS NAFLD prevalence in European studies • From 588 studies identified, 42 studies from 15 European countries met inclusion criteria, comprising 125, 674 unique adult individuals – Overall NAFLD prevalence estimated in the random effects metaanalysis model was 27. 6% (95% CI 24. 4– 31. 0; 28 studies) – No difference in prevalence observed when NAFLD was assessed by imaging or blood test methods • Among 4, 696 biopsy-proven NAFLD patients – 64. 3% had NASH (95% CI 52. 7– 74. 4; 12 studies) • In 5 studies reporting information on advanced fibrosis in the general population, prevalence was 6. 1% (95% CI 4. 3– 8. 7) CONCLUSION Petta S, et al. DILC 2020; THU 053 • Identification of advanced liver fibrosis remains an important priority due to its association with mortality and morbidity. Although easily assessed with current noninvasive tools, only 5 studies reported its prevalence in the general population • These data add to the evidence base and may be particularly useful in European settings that currently lack data

6. NAFLD: diagnostics and non-invasive assessment
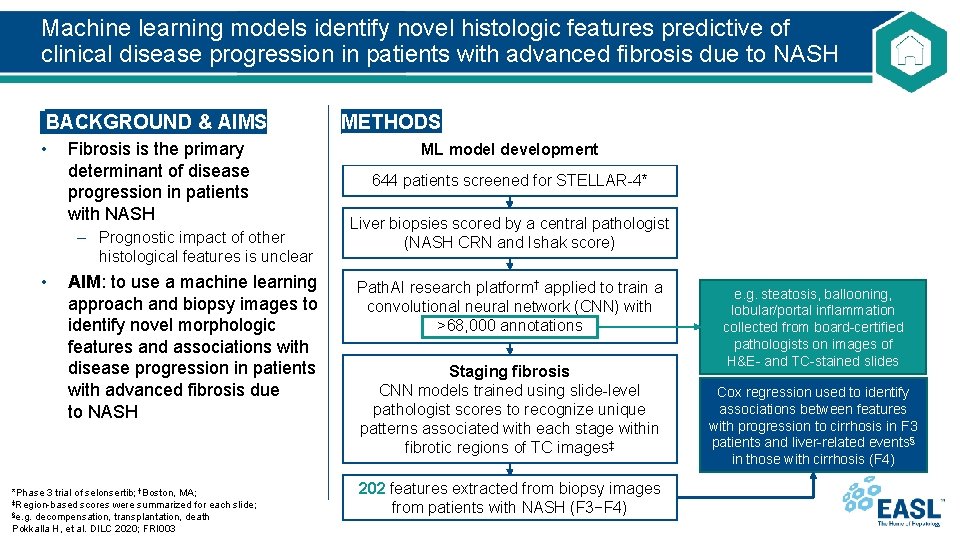
Machine learning models identify novel histologic features predictive of clinical disease progression in patients with advanced fibrosis due to NASH BACKGROUND & AIMS • Fibrosis is the primary determinant of disease progression in patients with NASH – Prognostic impact of other histological features is unclear • AIM: to use a machine learning approach and biopsy images to identify novel morphologic features and associations with disease progression in patients with advanced fibrosis due to NASH *Phase 3 trial of selonsertib; †Boston, MA; ‡Region-based scores were summarized for each slide; §e. g. decompensation, transplantation, death Pokkalla H, et al. DILC 2020; FRI 003 METHODS ML model development 644 patients screened for STELLAR-4* Liver biopsies scored by a central pathologist (NASH CRN and Ishak score) Path. AI research platform† applied to train a convolutional neural network (CNN) with >68, 000 annotations Staging fibrosis CNN models trained using slide-level pathologist scores to recognize unique patterns associated with each stage within fibrotic regions of TC images‡ 202 features extracted from biopsy images from patients with NASH (F 3−F 4) e. g. steatosis, ballooning, lobular/portal inflammation collected from board-certified pathologists on images of H&E- and TC-stained slides Cox regression used to identify associations between features with progression to cirrhosis in F 3 patients and liver-related events§ in those with cirrhosis (F 4)

Machine learning models identify novel histologic features predictive of clinical disease progression in patients with advanced fibrosis due to NASH RESULTS • Associations between machine learning-based morphological features with disease progression in patients with advanced fibrosis due to NASH 1, 593 NASH patients with F 3−F 4 fibrosis – Median age 59 years; 73% with diabetes; 52% F 4 • F 3: median follow-up of 16. 5 months – 15% (112/755) of F 3 patients cirrhosis • Associated with greater proportion of fibrosis area consistent with stage 6 Ishak score • Associated with greater proportion of tissue area consistent with portal inflammation – Reduced risk of progression associated with: • Greater proportion of fibrosis area consistent with stage 1 Ishak score • F 4: median follow-up of 15. 8 months – 3% (22/838) of F 4 patients had liver-related events • Associated with greater proportion of fibrosis area consistent with stage 6 Ishak score • Associated with hepatocellular ballooning, portal inflammation and bile duct areas CONCLUSION • – Reduced risk of progression associated with: • Greater proportion of fibrosis area consistent with stage 1 Ishak score • Greater proportion of total tissue area consistent with steatosis Pokkalla H, et al. DILC 2020; FRI 003 • Automated, machine learning-based evaluation of liver histology identified novel features associated with progression in patients with NASH and advanced fibrosis Data support the use of machine learning approaches to evaluate liver histology as endpoints in NASH clinical trials

Predictive modelling to identify and characterize fast progressors among patients with NASH BACKGROUND & AIMS • A subset of patients with NAFLD and NASH progress more rapidly to cirrhosis or HCC than typical patients • AIM: to develop a machine learning model to predict the progression risk to end-stage liver disease using data available from standard clinical care and electronic health care records METHODS Time to progression† Data source Optum Electronic Health records (2007− 2018, USA) Study cohort Patients with a diagnosis of cirrhosis or HCC* due to NAFLD/NASH (after exclusion of other causes) Fast progressors (<3 years) • • Intermediate progressors (3− 6 years) Fast and standard cohorts used to train model Standard progressors (>6− 10 years) Multiple machine learning classification techniques were tested and the best performing model selected Covariates used in model included lab tests and observations, comorbidities and their treatments, rate of change in lab tests and observations *ICD-9/ICD-10; †Established retrospectively from the date of cirrhosis or HCC to the first evidence of disease (index date), which was the first diagnosis of either NASH, NAFLD, or one of several comorbidities (e. g. type 2 diabetes, obesity) used as proxies Reinhart B, et al. DILC 2020; FRI 008

Predictive modelling to identify and characterize fast progressors among patients with NASH RESULTS • 951 fast and 992 standard progressors were used for modelling (fast; standard) – Age at index, mean (SD): 59. 5 (13. 6); 57. 2 (11. 6) – BMI, mean (SD): 34. 6 (7. 3); 35. 0 (7. 3) • • 44/148 clinical and laboratory parameters considered were retained for the model Models were trained to identify fast progressors using 75% of the patient data • • A Light. GBM model showed the best performance, with a ROC-AUC of 0. 77 In 25% of data retained for testing, 77% of fast progressors are correctly predicted: Clinical progression Predicted progression, % (N) Standard Fast TOTAL Standard 66% (109) 34% (57) 100% (166) Fast 23% (42) 77% (77) 100% (180) CONCLUSIONS • • Identification of NAFLD/NASH patients with different progression rates to end-stage liver disease in real-world data was supported A predictive algorithm to identify patients with the highest risk of progression could be developed and may be useful to support physician’s treatment choices Reinhart B, et al. DILC 2020; FRI 008

7. NAFLD: experimental and pathophysiology
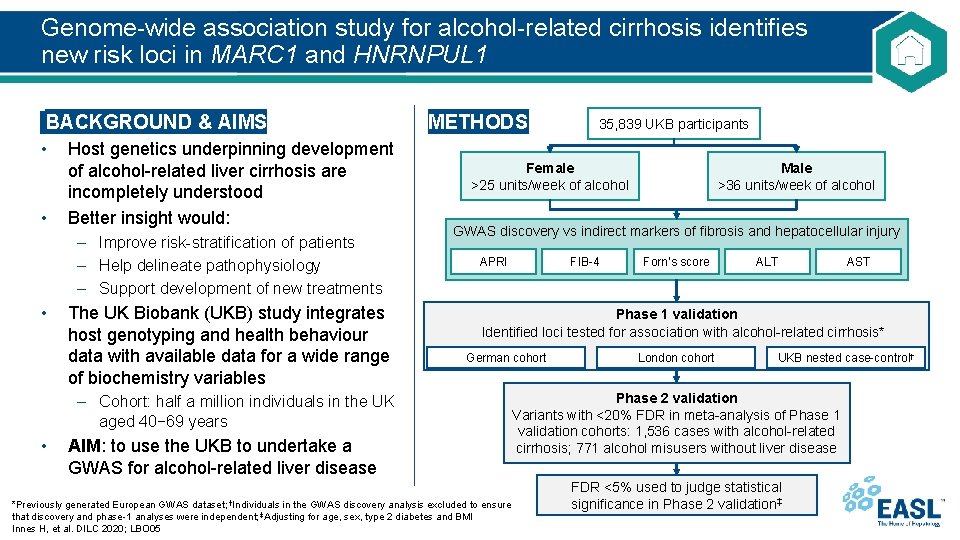
Genome-wide association study for alcohol-related cirrhosis identifies new risk loci in MARC 1 and HNRNPUL 1 BACKGROUND & AIMS • Host genetics underpinning development of alcohol-related liver cirrhosis are incompletely understood • Better insight would: – Improve risk-stratification of patients – Help delineate pathophysiology – Support development of new treatments • The UK Biobank (UKB) study integrates host genotyping and health behaviour data with available data for a wide range of biochemistry variables – Cohort: half a million individuals in the UK aged 40− 69 years • AIM: to use the UKB to undertake a GWAS for alcohol-related liver disease METHODS 35, 839 UKB participants Female >25 units/week of alcohol Male >36 units/week of alcohol GWAS discovery vs indirect markers of fibrosis and hepatocellular injury APRI FIB-4 Forn’s score ALT AST Phase 1 validation Identified loci tested for association with alcohol-related cirrhosis* German cohort London cohort UKB nested case-control† Phase 2 validation Variants with <20% FDR in meta-analysis of Phase 1 validation cohorts: 1, 536 cases with alcohol-related cirrhosis; 771 alcohol misusers without liver disease *Previously generated European GWAS dataset; †Individuals in the GWAS discovery analysis excluded to ensure that discovery and phase-1 analyses were independent; ‡Adjusting for age, sex, type 2 diabetes and BMI Innes H, et al. DILC 2020; LBO 05 FDR <5% used to judge statistical significance in Phase 2 validation‡
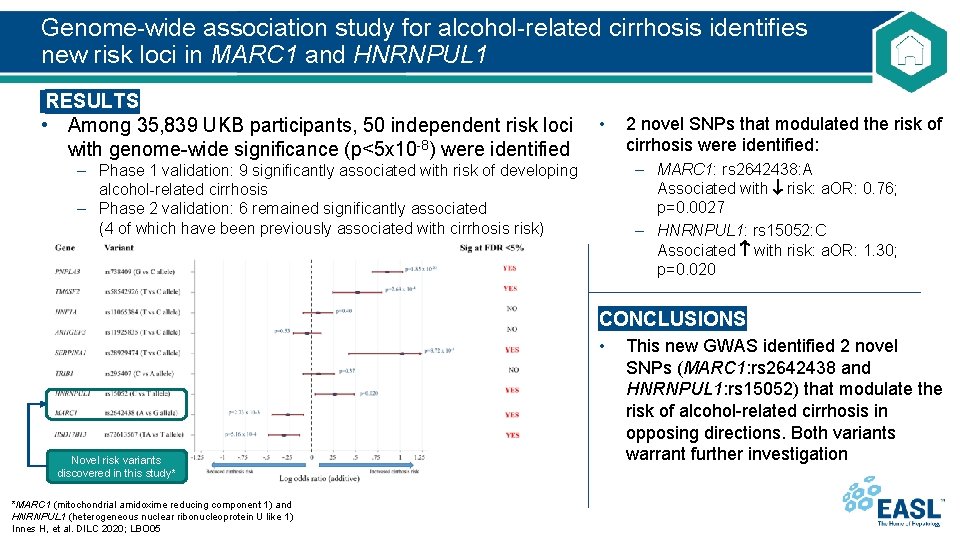
Genome-wide association study for alcohol-related cirrhosis identifies new risk loci in MARC 1 and HNRNPUL 1 RESULTS • Among 35, 839 UKB participants, 50 independent risk loci • with genome-wide significance (p<5 x 10 -8) were identified – Phase 1 validation: 9 significantly associated with risk of developing alcohol-related cirrhosis – Phase 2 validation: 6 remained significantly associated (4 of which have been previously associated with cirrhosis risk) 2 novel SNPs that modulated the risk of cirrhosis were identified: – MARC 1: rs 2642438: A Associated with risk: a. OR: 0. 76; p=0. 0027 – HNRNPUL 1: rs 15052: C Associated with risk: a. OR: 1. 30; p=0. 020 CONCLUSIONS • Novel risk variants discovered in this study* *MARC 1 (mitochondrial amidoxime reducing component 1) and HNRNPUL 1 (heterogeneous nuclear ribonucleoprotein U like 1) Innes H, et al. DILC 2020; LBO 05 This new GWAS identified 2 novel SNPs (MARC 1: rs 2642438 and HNRNPUL 1: rs 15052) that modulate the risk of alcohol-related cirrhosis in opposing directions. Both variants warrant further investigation
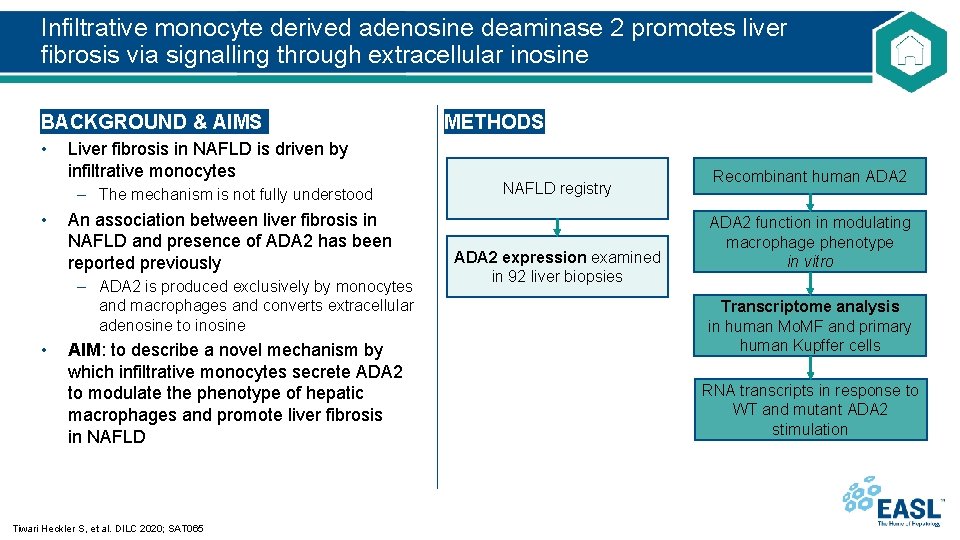
Infiltrative monocyte derived adenosine deaminase 2 promotes liver fibrosis via signalling through extracellular inosine BACKGROUND & AIMS • Liver fibrosis in NAFLD is driven by infiltrative monocytes – The mechanism is not fully understood • An association between liver fibrosis in NAFLD and presence of ADA 2 has been reported previously – ADA 2 is produced exclusively by monocytes and macrophages and converts extracellular adenosine to inosine • AIM: to describe a novel mechanism by which infiltrative monocytes secrete ADA 2 to modulate the phenotype of hepatic macrophages and promote liver fibrosis in NAFLD Tiwari Heckler S, et al. DILC 2020; SAT 065 METHODS NAFLD registry ADA 2 expression examined in 92 liver biopsies Recombinant human ADA 2 function in modulating macrophage phenotype in vitro Transcriptome analysis in human Mo. MF and primary human Kupffer cells RNA transcripts in response to WT and mutant ADA 2 stimulation

Infiltrative monocyte derived adenosine deaminase 2 promotes liver fibrosis via signalling through extracellular inosine RESULTS • ADA 2 expression in human liver – Noted in CD 14+/CD 16+ portal macrophages and Kupffer cells • ADA 2 -positive portal macrophages were significantly associated with degree of liver fibrosis* • Portal macrophages had far more ADA 2 transcripts than Kupffer cells† – Suggests a putative paracrine and autocrine nature of regulation • Transcriptome analysis – ADA 2 pro-inflammatory and -fibrotic genes in human Mo. MF‡ – ADA 2 and its substrate adenosine transcription of PDGF-BB, a key pro-fibrotic cytokine in macrophages – Inosine, the catalytic product of ADA 2 significant PDGF-BB expression in macrophages • Suggests a causal role of deaminase activity – Mutant ADA 2§ enzyme activity and PDGF-BB gene expression in macrophages vs WT ADA 2 • ADA 2 PDGF-BB from primary human Kupffer cells in a human-derived multicellular liver spheroid system CONCLUSION • • Infiltrative monocytes release ADA 2 to promote liver fibrosis by augmenting pro-fibrotic differentiation of hepatic macrophages via signalling through extracellular inosine This pathway may provide alternative targets in modulating liver fibrosis in NAFLD *As shown by a quantitative histological score; †Measured by RNAscope; ‡Validated in U 937 -derived macrophages and human Mo. MF from six independent donors; §Mutation at glutamic acid 359 in the catalytic active site Tiwari Heckler S, et al. DILC 2020; SAT 065
- Slides: 38