Metabolic response to injury We will discuss mediators

Metabolic response to injury

We will discuss: • mediators of the stress response. • physiological and biochemical pathway changes associated with surgical injury. • Changes in body composition that occur following surgical injury.
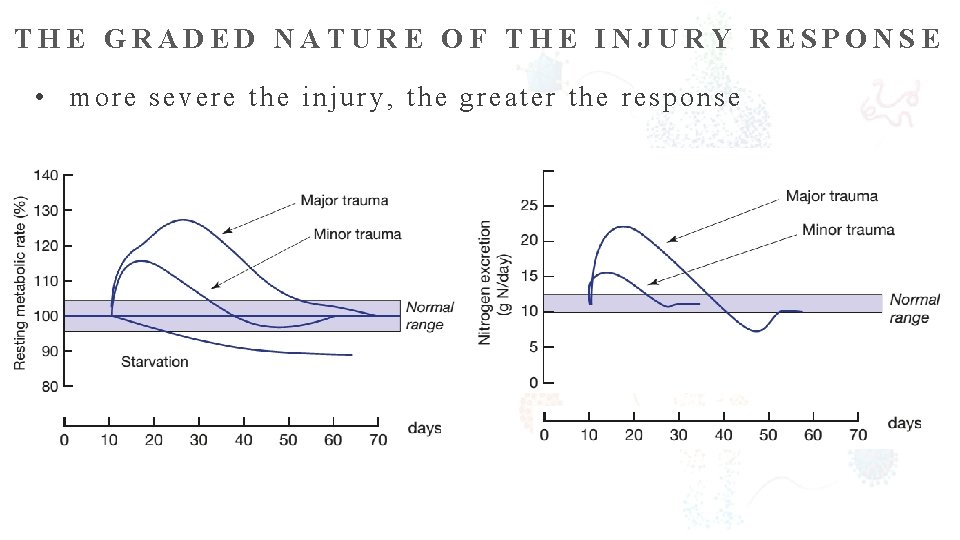
THE GRADED NATURE OF THE INJURY RESPONSE • more severe the injury, the greater the response

Systemic Inflammatory Response Syndrome (SIRS) • clinical response to a nonspecific insult of either infectious or noninfectious origin. • SIRS is nonspecific and can be caused by ischemia, inflammation, trauma, infection, or several insults combined.

Pathophysiology Stage I cytokine production, to promote healing process by induction of the body inflammatory components. rubor, tumor, dolor, calor and functio laesa • Rubor or redness reflects local vasodilation • Tumor or swelling is due to vascular endothelial tight junction disruption and the local extravasation of protein rich fluid into the interstitium. • Dolor is pain and represents the impact inflammatory mediators have on local somatosensory nerves. • Calor is the increased heat primarily due to increased blood flow but also increased local metabolism • Functio laesa is loss of function, a hallmark of inflammation and a common clinical finding of organ dysfunction with the infection Tissue destruction by activated leukocytes, cytokines, and chemokines in form of abscess and pus.

Stage II Small quantities of local cytokines are released into the circulation. This leads to growth factor stimulation and the recruitment of macrophages and platelets. At this stage, some minimal malaise and low grade fever may become manifest. Stage III homeostasis is not restored and if the inflammatory stimuli continue to seed into the systemic circulation. a significant systemic reaction occurs. The cytokine release leads to destruction rather than protection. numerous humoral cascades and the activation of the reticular endothelial system and subsequent loss of circulatory integrity. End Organ dysfunction

• Elective surgeries considered interpreted by the body as trauma/ injury • transient and modest rise in • Temperature (Normal 37°C) • heart rate (Normal 60 100 b/min) • respiratory rate (Normal 12 20 b/min) • energy expenditure (Require Calculations) • peripheral white cell count (4 11 × 10 × 9).
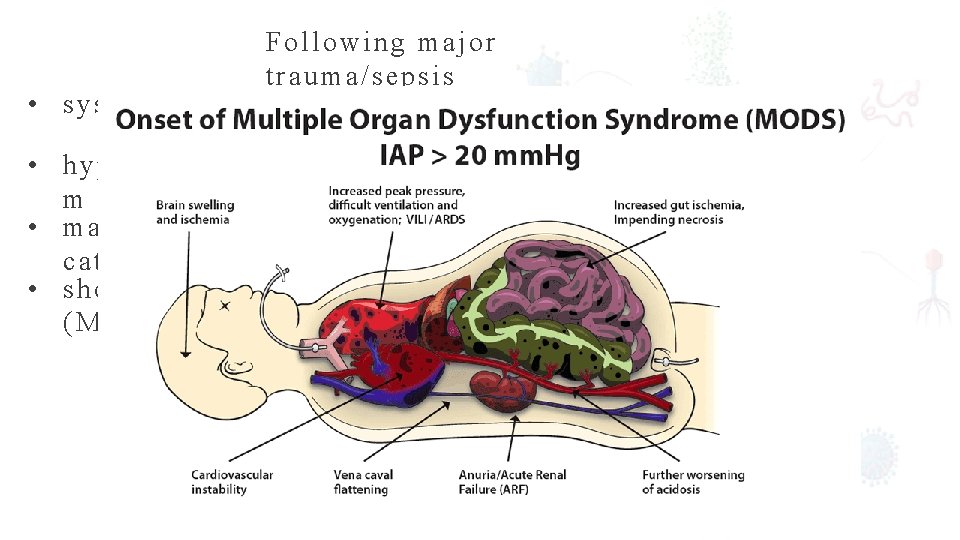
Following major trauma/sepsis • systemic inflammatory response syndrome (SIRS) • hypermetabolis m • marked catabolism • shock and even multiple organ dysfunction (MODS)

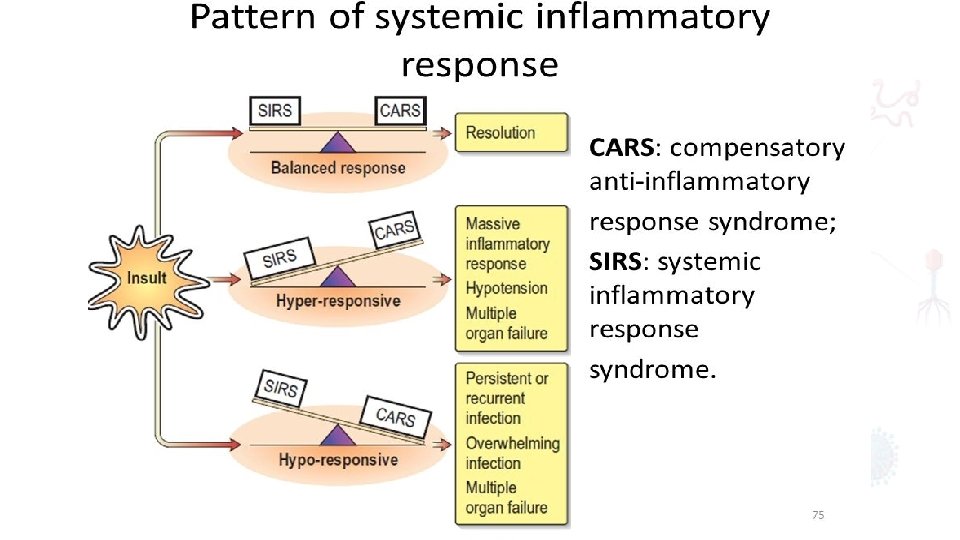
• Response evolves with time • evolve from a proinflammatory state driven primarily by the innate immune system • The compensatory anti inflammatory response syndrome (CARS) characterized by suppressed immunity • diminished resistance to infection

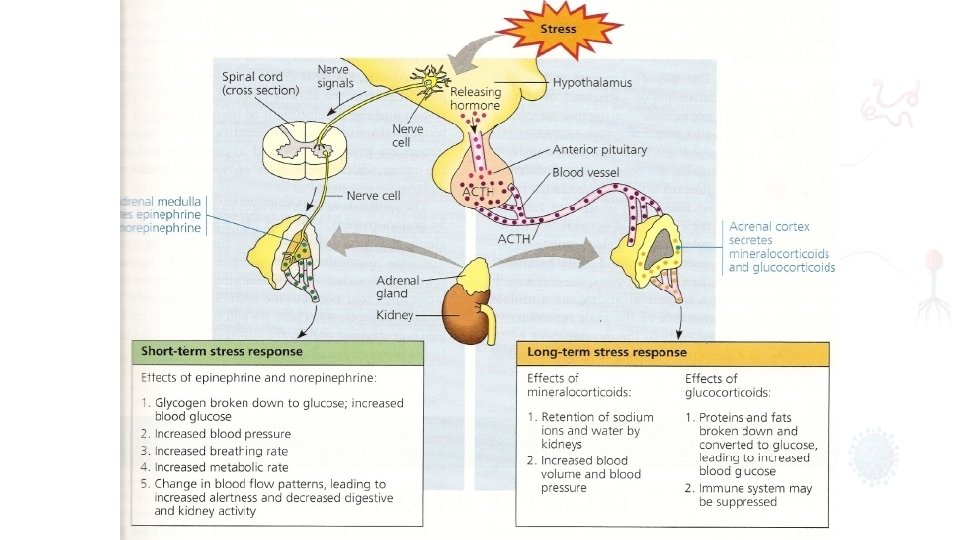
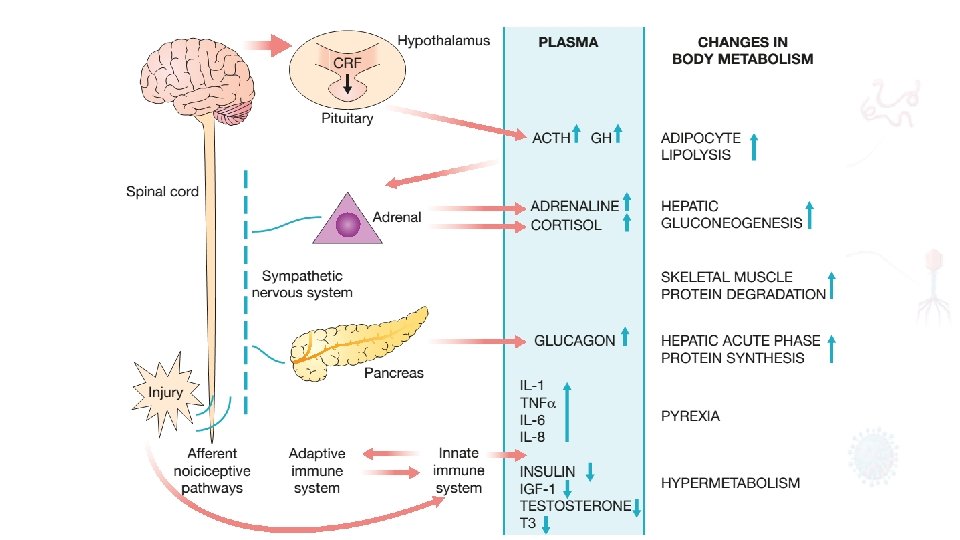
MEDIATORS OF THE METABOLIC RESPONSE TO INJURY neuroendocrine pathways of the stress response consist of afferent nociceptive neurons, the spinal cord, thalamus, hypothalamus and pituitary

• Corticotrophin releasing factor (CRF) released from the hypothalamus increases adrenocorticotrophic hormone (ACTH) release from the anterior pituitary. • ACTH then acts on the adrenals to increase the secretion of cortisol. • stimulates release of glucagon • including alterations in insulin release and sensitivity, hypersecretion of prolactin and growth hormone (GH) in the presence of low circulatory insulin like growth factor 1 (IGF 1) and inactivation of peripheral thyroid hormones and gonadal function. • GH has direct lipolytic, insulin antagonising and proinflammatory properties.
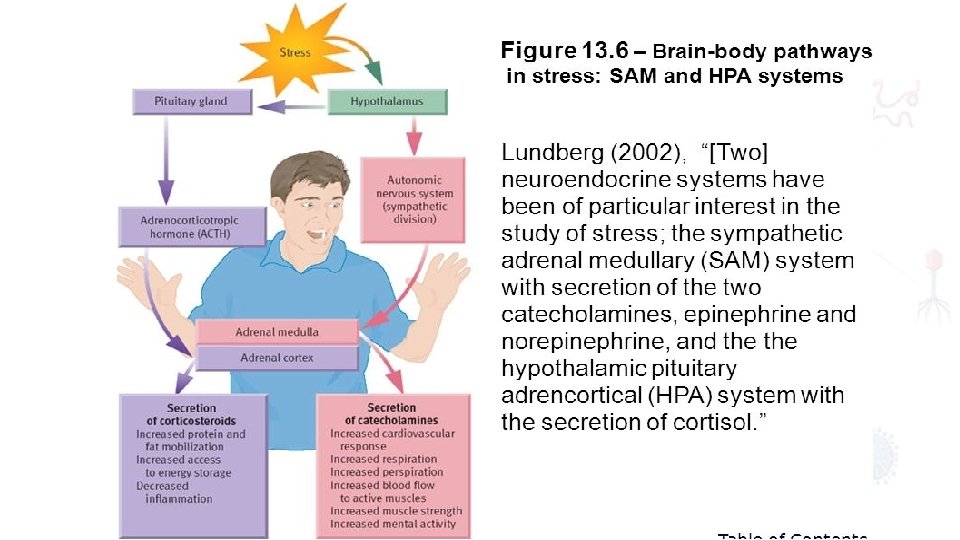

• Proinflammatory cytokines including interleukin 1 (IL 1), tumour necrosis factor alpha (TNFα), IL 6 and IL 8 are produced within the first 24 hours and act directly on the hypothalamus to cause pyrexia. • Proinflammatory cytokines • Pyrexia • Muscular Destruction • Insulin resistance • Nitric Oxide receptors on the wall of the vessels will cause vasoconstriction.
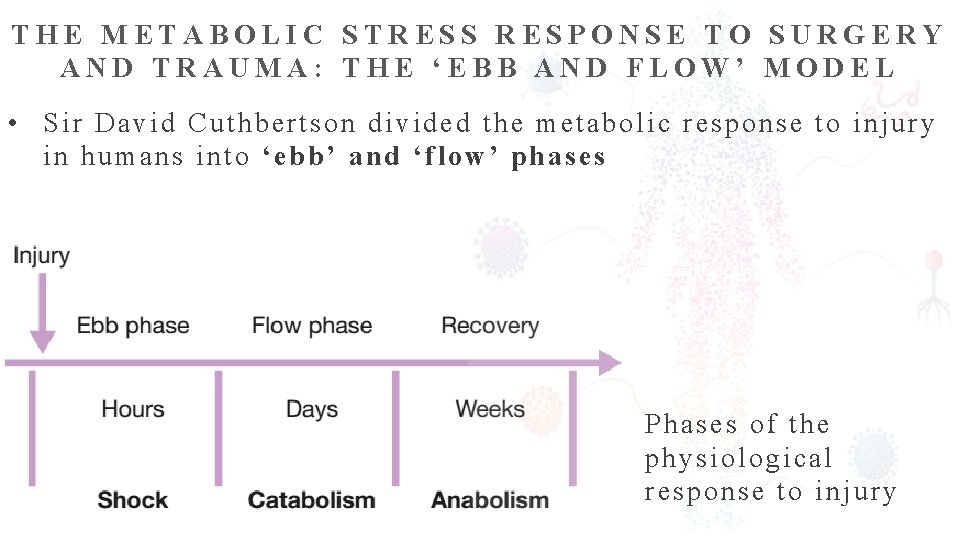
THE METABOLIC STRESS RESPONSE TO SURGERY AND TRAUMA: THE ‘EBB AND FLOW’ MODEL • Sir David Cuthbertson divided the metabolic response to injury in humans into ‘ebb’ and ‘flow’ phases Phases of the physiological response to injury

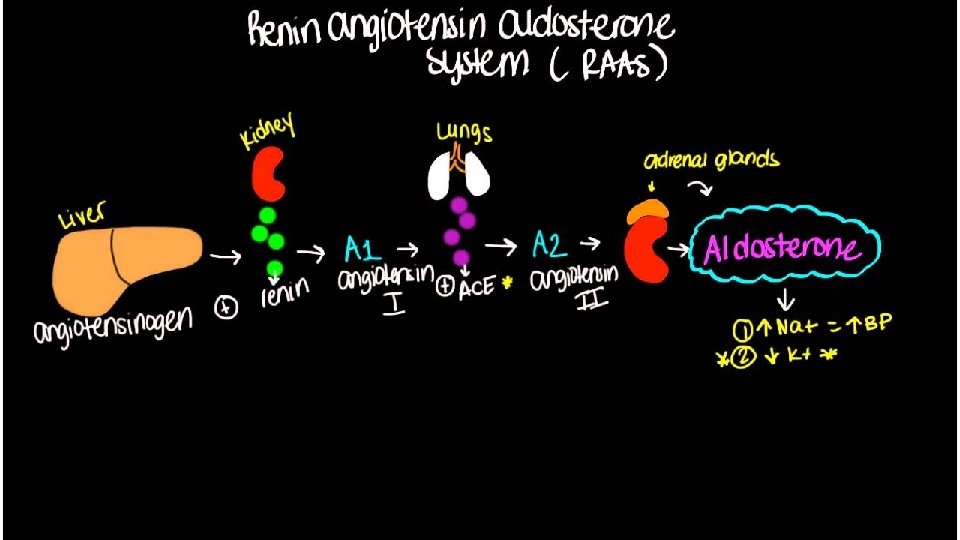
• ebb phase begins at the time of injury and lasts for approximately 24– 48 hours. • It can be reduced by proper resuscitation / management but never abolished. • Hypovolemia • Decrease Basic Metabolic Rate. • Reduced Cardiac Output. • Hypothermia • lactic acidosis • predominant hormones regulating the ebb phase • Catecholamines • Cortisol • Aldosterone & Activation of Renin Angiotensin Pathway

• EBB phase conserve both circulating volume and energy stores for recovery and repair. • The magnitude of this neuroendocrine response depends on the degree of blood loss • The brief duration of this phase limits clinical relevance.

• the ebb phase evolves into a hypermetabolic flow phase, which corresponds to SIRS. • mobilization of body energy stores for recovery and repair. • replacement of lost or damaged tissue. • tissue oedema (from vasodilatation and increased capillary leakage) • increased basal metabolic rate (hypermetabolism) • increased cardiac • or auitspeudt b o d y t e m p e r a t u r e • increased oxygen consumption and increased gluconeogenesis
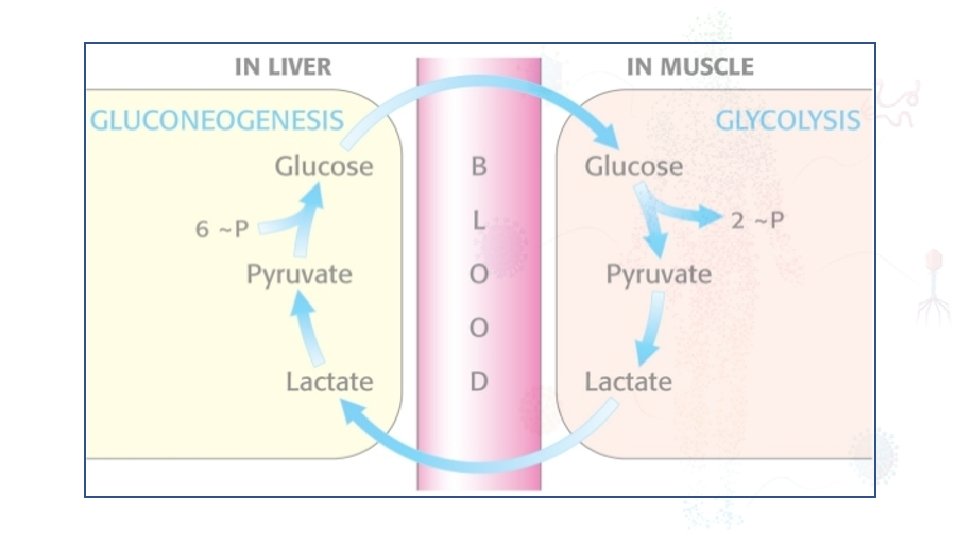

• Flow Phase will have 2 stages • Catabolic stage (3 10 days) • Anabolic Stage (Lasts for weeks). • Catabolic Phase will include. • Initiation of hormones and cytokines (IL 1 & 6, TNF). • Weight Loss. • Increase Urinary Nitrogen (Due to protein destruction). • Increase Insulin production; however, increase in resistance to insulin which will results in poor glycemic control).

I. Hypermetabolism • • 15– 25% above predicted healthy resting values. The predominant cause appears to be a complex interaction between the central control of metabolic rate and peripheral energy utilization. (central thermodysregulation mainly due cytokines cascades), increased sympathetic activity, abnormalities circulation which can cause changes in cardiac output. II. Alterations in skeletal muscle protein metabolism • • Normal rate of muscles brake down with a turnover rate in humans of 1– 2% per day. The major site of protein loss is peripheral skeletal muscle. Nitrogen losses also occur in the respiratory muscles (increase risk of chest infection). Under extreme conditions of catabolism, urinary nitrogen losses can reach 14– 20 g/day; this is equivalent to the loss of 500 g of skeletal muscle per day.

• muscle catabolism cannot be inhibited fully by providing artificial nutritional support as long as the stress response continues. Clinically, a patient with skeletal muscle wasting will experience asthenia, increased fatigue, reduced functional ability, decreased quality of life and an increased risk of morbidity and mortality. • III. Alterations in hepatic protein metabolism • The liver and skeletal muscle together account for >50% of daily body protein turnover. • Skeletal muscle has a large mass but a low turnover rate (1– 2% per day), whereas the liver has a relatively small mass (1. 5 kg) but a much higher protein turnover rate (10– 20% per day). • mononuclear cells secrete a range of proinflammatory cytokines, including IL 1, IL 6 and TNFα. IL 6, promote the hepatic synthesis of positive acute phase proteins (ex. C reactive protein). [Acute phase proteins are a class of proteins whose plasma concentrations increase or decrease in response to inflammation].

CHANGES IN BODY COMPOSITION FOLLOWING INJURY The average 70 kg male. • lean body mass: 57 kg. • 13 kg Fat. • The protein mass can be c o n s i d e r e lean d abody s t mass: w o 57 b kg asic compartments, skeletal muscle (4 kg) and non skeletal muscle (8 kg), which includes the visceral protein mass. • The water mass (42 liters) • Most of the mineral mass is contained in the bony skeleton.

main labile energy reserve in the body is fat, and the main labile protein reserve is skeletal muscle. Within lean issue, each 1 g of nitrogen is contained within 6. 25 g of protein, which is contained in approximately 36 g of wet weight tissue. normal human ingests about 70– 100 g protein per day, which is metabolized and excreted in urine as ammonia and urea
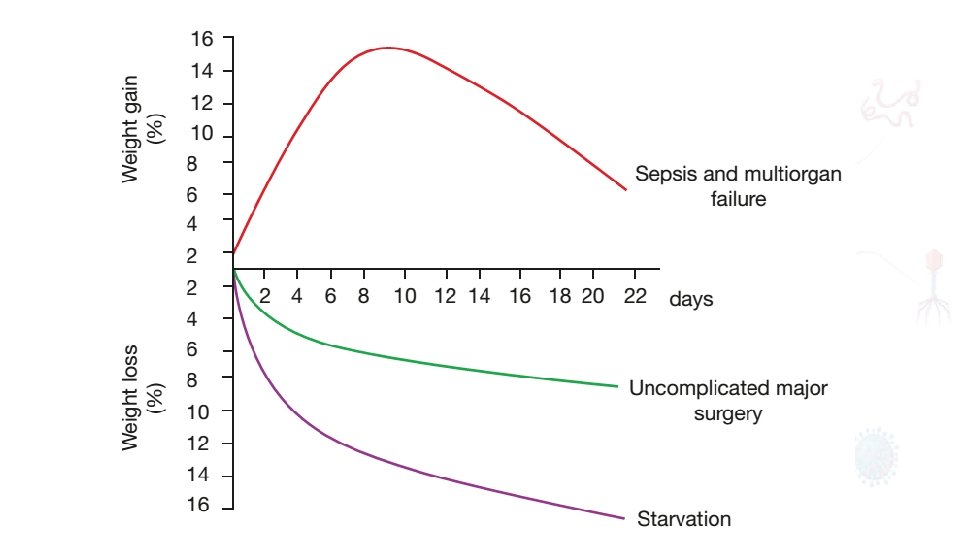

Critically ill patients admitted to the ICU with severe sepsis or major blunt trauma undergo massive changes in body composition Body weight increases immediately on resuscitation with an expansion of extracellular water by 6– 10 liters within 24 hours it is now possible to maintain body weight and nitrogen equilibrium following major elective surgery. Achieved by blocking the neuroendocrine stress response with epidural analgesia/other related techniques and providing early oral/ enteral feeding.

AVOIDABLE FACTORS THAT COMPOUND THE RESPONSE TO INJURY metabolic stress response may be further exacerbated by anaesthesia, dehydration, starvation (including preoperative fasting), sepsis, acute medical illness or even severe psychological stress
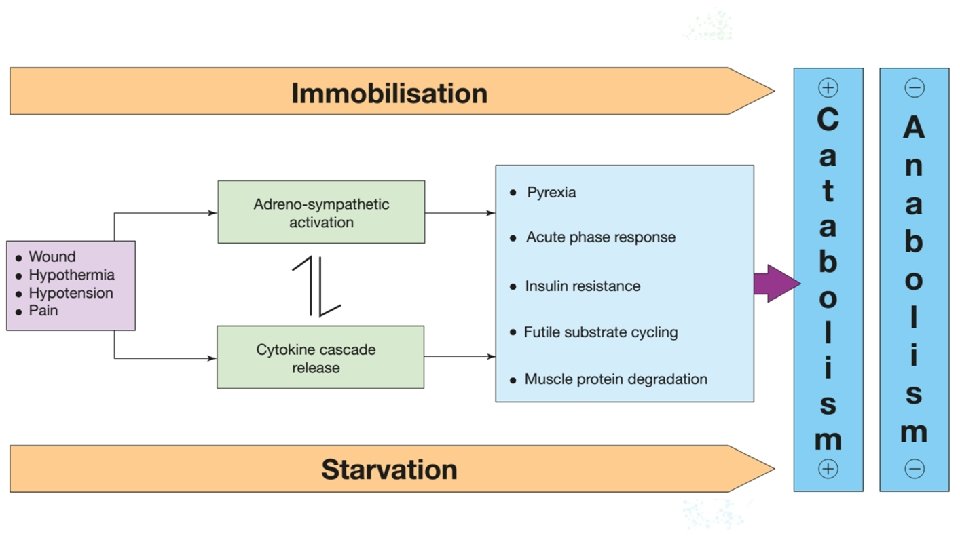

I. Volume loss simple haemorrhage, pressor receptors in the carotid artery and aortic arch, and volume receptors in the wall of the left atrium, initiate afferent nerve input to the central nervous system (CNS), resulting in the release of both aldosterone and antidiuretic hormone (ADH). ADH acts directly on the kidney to cause fluid retention. Decreased pulse pressure stimulates the juxtaglomerular apparatus in the kidney and directly activates the renin–angiotensin system, which in turn increases aldosterone release. Careful limitation of intraoperative administration of balanced crystalloids so that there is no net weight gain
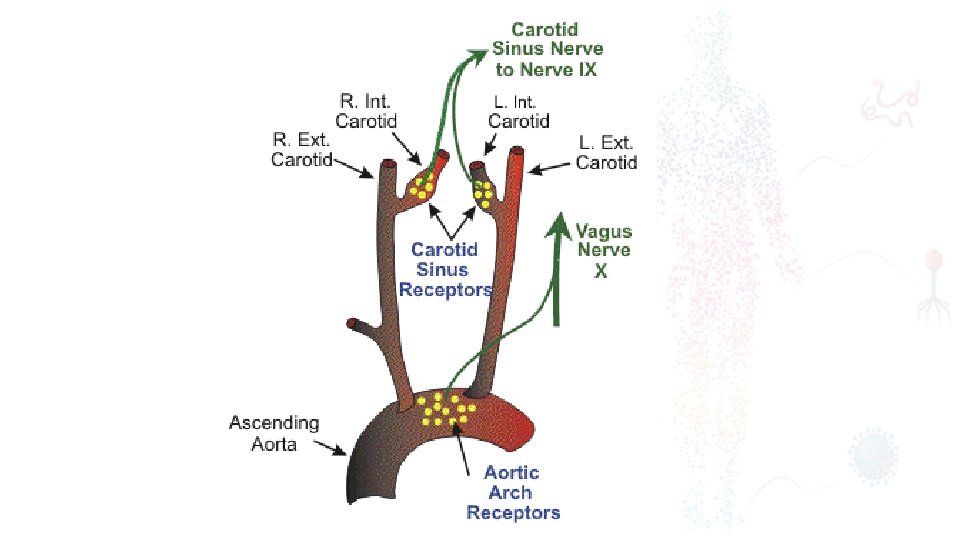
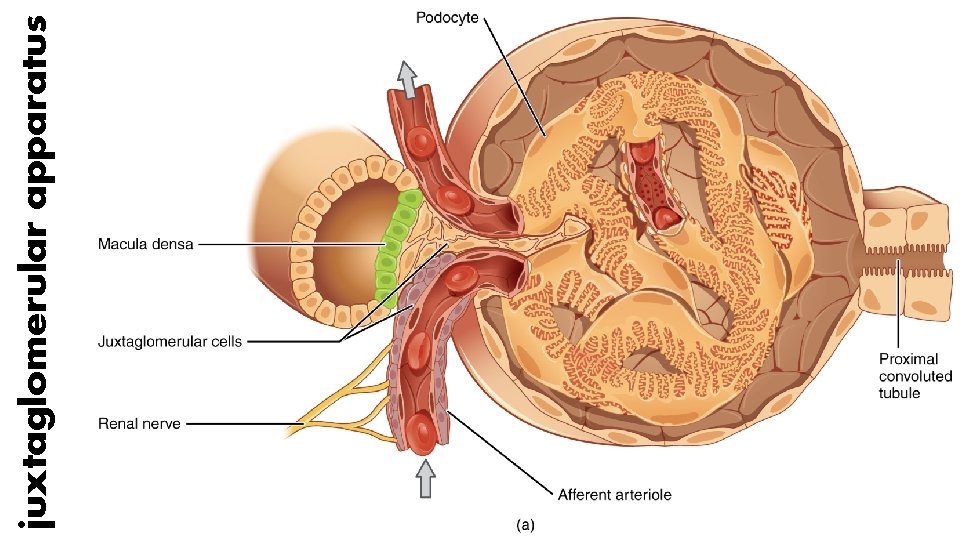
juxtaglomerular apparatus

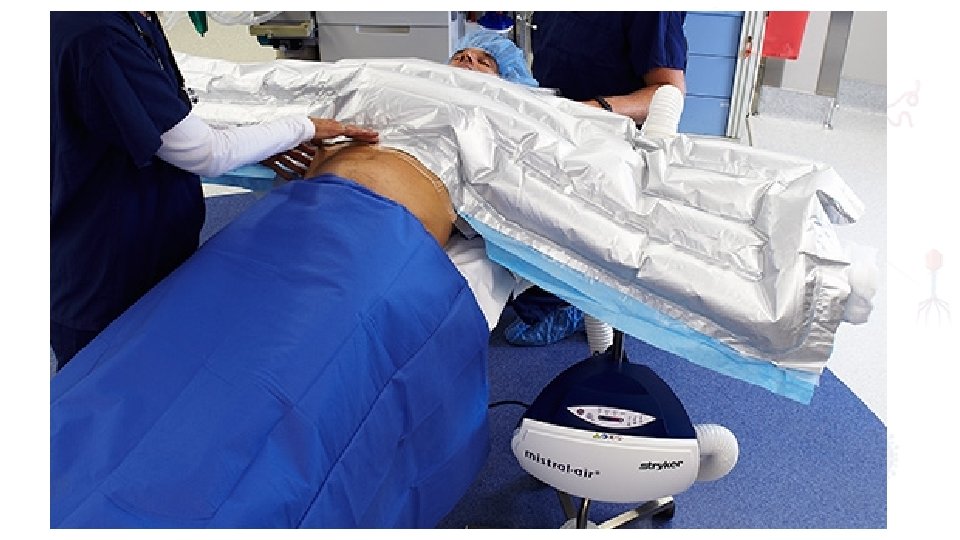
II. Hypothe rmia Hypothermia results in increased elaboration of adrenal steroids and catecholamines. cardiac arrhythmias and increased catabolism maintaining normothermia by an upper body forced air heating cover reduces wound infections, cardiac complications and bleeding and transfusion requirements.

III. Starvation During starvation, the body is faced with an obligate need to generate glucose to sustain cerebral energy metabolism (100 g of glucose per day). This is achieved in the first 24 hours by mobilising glycogen stores and thereafter by hepatic gluconeogenesis from amino acids, glycerol and lactate accelerated loss of lean tissue (the main source of amino acids for hepatic gluconeogenesis) is reduced as a result of the liver converting free fatty acids into ketone bodies, which can serve as a substitute for glucose for cerebral energy metabolism. 2 litres of intravenous 4% dextrose/0. 18% sodium chloride as maintenance intravenous fluids for surgical patients who are fasted provides 80 g of glucose per day and has a significant protein sparing effect.

IV. Immobi Il imt ym o b i l i t y h a s l o n g b e e n r e c o g n i s e d a s a p o t e n t s t i m u l u s f o r i n d u c i n g muscle wasting. Inactivity impairs the normal meal derived amino acid stimulation of protein synthesis in skeletal muscle. Avoidance of unnecessary bed rest and active early mobilisation are essential measures to avoid muscle wasting as a consequence of immobility.
- Slides: 38