Metabolic Liver Disease in Children Part 2 James

Metabolic Liver Disease in Children Part 2 James Squires, MD James Heubi, MD Cincinnati Children’s Hospital Medical Center 2013 Resident Education Series Reviewed by Sunny Hussain, MD of the Professional Education Committee

Part 2: • Disorders of Amino Acid and Organic Acid Metabolism • Fatty Acid Oxidation Defects • Urea Cycle Defects • Bile Acid and Biliary Transport Defects

Disorders of Amino Acid and Organic Acid Metabolism - Introduction • Although rare, these disorders may be encountered in practice and an approach to the diagnosis, management and outcomes is essential • Most common and important disorders with hepatic manifestations include: – Disorders of tyrosine – Methylmalonic acid – Isovaleric acid – Maple syrup urine disease
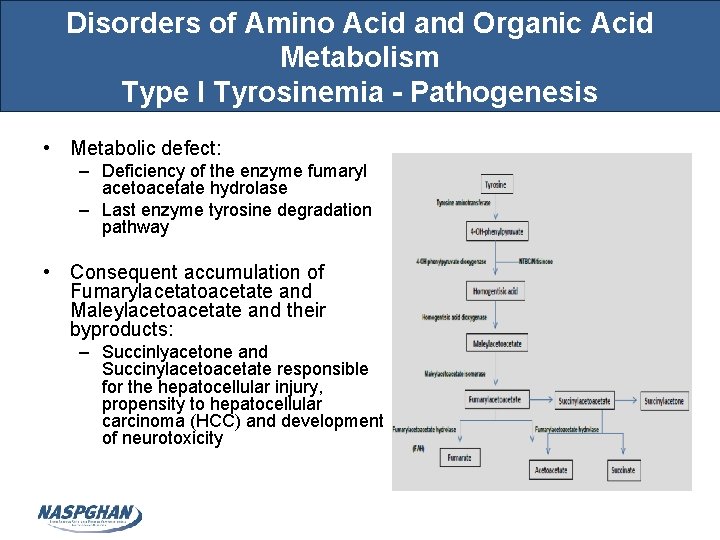
Disorders of Amino Acid and Organic Acid Metabolism Type I Tyrosinemia - Pathogenesis • Metabolic defect: – Deficiency of the enzyme fumaryl acetoacetate hydrolase – Last enzyme tyrosine degradation pathway • Consequent accumulation of Fumarylacetatoacetate and Maleylacetoacetate and their byproducts: – Succinlyacetone and Succinylacetoacetate responsible for the hepatocellular injury, propensity to hepatocellular carcinoma (HCC) and development of neurotoxicity

Disorders of Amino Acid and Organic Acid Metabolism Type I Tyrosinemia – Clinical Presentation & Diagnosis • Characterized by findings of acute liver failure with poor growth, vomiting, ascites, coagulopathy, hypoglycemia, hypoalbminemia, and hyperbilirubinemia • Labs – Serum α-fetoprotein may be markedly elevated – Hemolytic anemia may be present – Renal tubular dysfunction is common with increased excretion of phosphate, glucose, protein and amino acids • Diagnosis: – Elevated urinary succinylacetone

Disorders of Amino Acid and Organic Acid Metabolism Type I Tyrosinemia – Treatment and Outcomes • Neonates presenting with acute liver failure should be treated aggressively to prevent and treat complications – Bleeding should be treated with fresh frozen plasma, platelets and cryoprecipitate – Infusions of intravenous glucose should be given to prevent hypoglycemia • Dietary restriction of tyrosine: – Formulas such as Tyrex® or Tyros® should be used • NTBC ((2 -(2 -Nitro-4 -trifluromethylbenzoyl)-1, 3 -cyclohexanedione, Nitisinone) which inhibits 4 -hydroxyphenyl dioxygenase, the enzyme proximal to the deficient enzyme in Type I tyrosinemia, is effective in the majority of patients with reversal of hepatic and renal dysfunction • Liver transplantation should be considered for patients unresponsive to NTBC

Fatty Acid Oxidation Defects. Introduction • FAO provides most of the energy supplied to the heart and skeletal muscle • Crucial in the maintenance of glucose homeostasis during periods of fasting • End product of FAO is ketones that constitute an important secondary energy source for tissues when glucose supplies are depleted • Mitochondrial fatty acid oxidation (FAO) defects are a group of inherited metabolic disorders that contribute greatly to pediatric morbidity and mortality

Fatty Acid Oxidation Defects: MCAD (Medium-chain acyl-Co. A dehydrogenase) Deficiency - Pathogenesis • MCAD is a nucleus-encoded mitochondrial matrix enzyme that catalyzes the initial dehydrogenation step in the -oxidation of medium-chain fatty acids • MCAD is responsible for the initial metabolism of acyl-Co. As with chain lengths of 4 -12 carbon atoms • MCAD deficiency leads to fasting-induced hypoglycemia and accumulation of toxic acyl-Co. A compounds • MCAD deficiency is now part of newborn screening therefore classic clinical presentation (recurrent emesis, lethargy, coma, and even death provoked by fasting or illness) is rare

Fatty Acid Oxidation Defects: MCAD Deficiency – Clinical Presentation & Diagnosis • Acute decompensation characterized by: – – – Hypoketotosis and hypoglycemia Anion gap acidosis Hyperuricemia Elevated liver enzymes Hyperammonemia • Characteristic urine gas chromatographic findings include elevated levels of sebacic, sebaric, and adipic acids • Fibroblast enzymatic assays and/or genetic testing of the MCAD gene can make a confirmatory diagnosis

Fatty Acid Oxidation Defects: MCAD Deficiency – Treatment and Outcomes • Mainstay of treatment for patients with MCAD deficiency is avoidance of fasting • Unidentified patients with this disorder have a significant risk of sudden death in early childhood • Survivors have a significant risk of developmental disability and chronic somatic illnesses

Fatty Acid Oxidation Defects: LCHAD (Longchain 3 -hydroxyacyl-Co. A dehydrogenase) Deficiency - Pathogenesis • LCHAD is an enzyme that constitutes part of the mitochondrial trifunctional (TFP) complex – TFP is composed of 3 separate enzymes whose main function is to metabolize long-chain fatty acids often found in milk and oils • Pathology in patients with LCHAD deficiency results from inadequate energy supply and toxic accumulation of metabolites

Fatty Acid Oxidation Defects: LCHAD Deficiency – Clinical Presentation & Diagnosis • Classic presentation is newborn with acute onset: – – Feeding difficulties Lethargy Hypotonia Hypoketotic hypoglycemia • Neuropathy and retinopathy are two unique sequelae of LCHAD deficiency • LCHAD deficiency in a fetus is now well recognized to predispose mothers to gestational complications such as HELLP (hemolysis, liver dysfunction, and low platelets) syndrome and AFLP (acute fatty liver of pregnancy) • Newborn screening programs now test for LCHAD deficiency • Confirmation of the diagnosis is possible by measuring LCHAD activity in lymphocytes, fibroblasts, and muscle or liver biopsies and by genetic mutation analysis
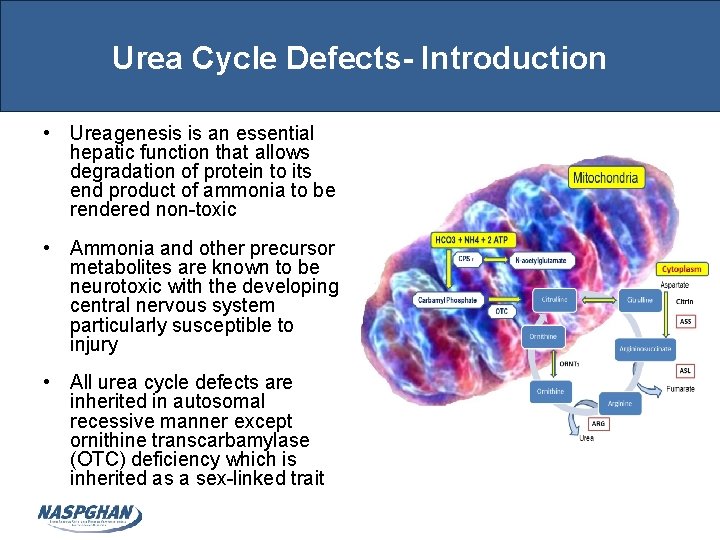
Urea Cycle Defects- Introduction • Ureagenesis is an essential hepatic function that allows degradation of protein to its end product of ammonia to be rendered non-toxic • Ammonia and other precursor metabolites are known to be neurotoxic with the developing central nervous system particularly susceptible to injury • All urea cycle defects are inherited in autosomal recessive manner except ornithine transcarbamylase (OTC) deficiency which is inherited as a sex-linked trait
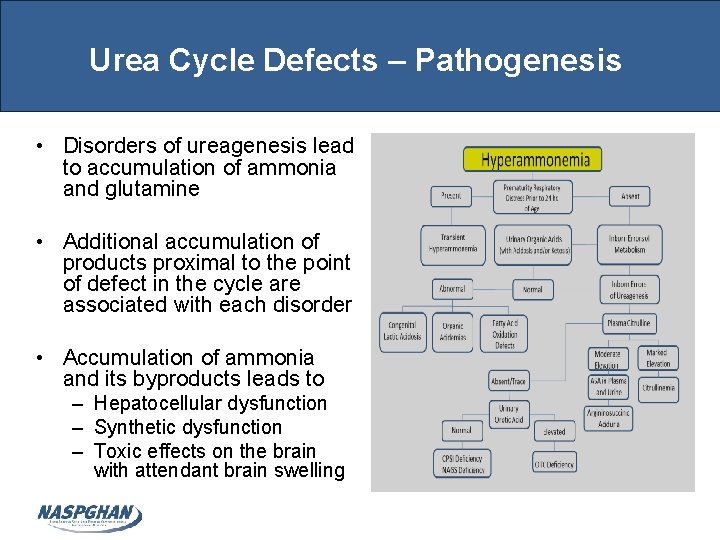
Urea Cycle Defects – Pathogenesis • Disorders of ureagenesis lead to accumulation of ammonia and glutamine • Additional accumulation of products proximal to the point of defect in the cycle are associated with each disorder • Accumulation of ammonia and its byproducts leads to – Hepatocellular dysfunction – Synthetic dysfunction – Toxic effects on the brain with attendant brain swelling

Urea Cycle Defects – Clinical Presentation & Diagnosis • Most common of disorders of ureagenesis include – Ornithine transcarbamylase deficiency (OTC) – Carbamyl phosphate synthetase (CPS) – Arginosuccinate synthetase (AS) or citrullinemia • Infants are normal at birth and rapidly develop hyperammonemia and cerebral edema and progressive coma in the first few days of life • Commonly mistaken to be septic • Diagnosis: – – • High index of suspicion Elevated plasma ammonia levels Reduced blood urea nitrogen and serum amino acid patterns typical of specific defects Presence of orotic acid with OTC deficiency Definitive diagnosis of a urea cycle defect can be made by molecular genetic testing or measurement of enzyme activity in the liver

Urea Cycle Defects – Treatment and Outcomes • Initial therapy should be directed at returning elevated plasma ammonia levels to the normal range • Reversing catabolism is essential and nutritional support with hypertonic glucose should be initiated immediately • If rapid improvement in plasma ammonia is not observed hemodialysis should be considered – If dialysis is initiated, it should be continued until plasma ammonia falls below 150 μmol/l • Once the acute hyperammonemic crisis is resolved, feeding should be commenced with a protein restricted formula • In patients with severe, recurrent episodes of hyperammonemia, liver transplantation is an effective means to prevent future episodes

Biliary Transport Defects- Introduction • Normal bile flow is dependent in part upon specific membrane transporters found in the liver and intestine • Inherited defects in the genes for some of these transporters lead to cholestasis and as a group comprise conditions termed Progressive Familial Intrahepatic Cholestasis (PFIC)

Biliary Transport Defects- Pathogenesis • Three conditions comprise the currently known group of biliary transport defects – PFIC I: Mutations in the FIC 1 gene (ATP 8 B 1) • FIC 1 mediates the flipping of aminophospholipids from outer to inner hemi-leaflet of the canalicular membrane • FIC 1 is located on other tissues including the pancreas and intestine leading to other extrahepatic signs and symptoms – PFIC II have defects in the canalicular bile salt export pump (BSEP) caused by mutation in ABCB 11 • BSEP is responsible for transporting bile acids from inside the hepatocyte into the bile canaliculus – PFIC III caused by mutations in ABCB 4 • Encodes multidrug resistance-associated protein 3 (MDR 3) and mediates the flopping of aminophospholipids from inner to outer hemi-leaflet of the canalicular lipid bi-layer • Liver disease in PFIC results from the effects of hepatocellular accumulation of bile acids

Biliary Transport Defects- Clinical Presentation & Diagnosis • Patients with forms of these diseases present with – Jaundice and/or pruritus – Life-threatening hemorrhage, secondary to cholestasis-related vitamin K deficiency • Patients with PFIC I and II have markedly elevated serum bile acid levels with mildly elevated serum bilirubin, normal serum gamma glutamyl transpeptidase (GGT) • PFIC III have high serum GGT levels • Definitive diagnosis of a specific form of PFIC is dependent upon identification of characteristic genetic defects

Biliary Transport Defects- Treatment and Outcomes • Supplement fat-soluble vitamins • Pruritis – Ursodeoxycholic acid, Cholestyramine, Rifampin, and opioid antagonists may not be successful – Surgical interruption of the enterohepatic circulation of bile acids with either ileal bypass or diversion may be effective • Severe defects in BSEP are associated with a high risk of hepatocellular carcinoma • Severe defects in BSEP and MDR 3 deficiency are typically associated with an unremitting form of cholestasis that are unresponsive to medical and surgical therapies necessitate liver transplantation • FIC 1 is a systemic disease and the post-transplant course can be problematic including intractable diarrhea, liver steatosis leading to cirrhosis, and recurrent pancreatitis • Overall, with optimal surgical intervention the long-term prognosis for children with PFIC is excellent

Inborn Errors of Bile Acid Metabolism Introduction • Bile acids are natural detergents that liver makes that help with bile flow and efficient fat and fat soluble vitamin absorption • The primary bile acids, cholic acid and chenodeoxycholic acid, are synthesized from cholesterol through a series of 17 enzymatic steps that result in the production taurine or glycine conjugates • Any alteration in the pathway can lead to blockage of the bile acid production with failure to produce “normal bile acids” and the accumulation of intermediary metabolites

Inborn Errors of Bile Acid Metabolism Pathogenesis • Accumulation of metabolites in the bile acid synthesis may result in mild to severe liver disease depending upon which of the enzymes is affected • There are two principal causes of liver injury associated with inborn errors of bile acid metabolism 1. Failure to make “normal” bile acids which are a major force propelling bile out of the liver • 2. Leads to impaired bile flow with attendant reduction of biliary excretion of individual components out of the liver Intermediary metabolites produced because of the block in the bile acid production pathway may themselves be toxic to the liver • Combination of poor bile flow coupled with the production of potential toxic bile acid intermediates most likely is responsible for the injury to the liver seen in these conditions.

Inborn Errors of Bile Acid Metabolism Clinical Presentation & Diagnosis • Most common defects including 3 -HSD and 4 -3 -oxosteroid- 5 -β reductase deficiencies commonly present with – – Neonatal jaundice Poor growth Hepatosplenomegaly Bleeding • Pruritus is usually absent • Serum bile acids measured by standard clinical methods are either normal or low • If suspected - urine must be sent for analysis to specialized laboratories for measurement of urinary bile acids by methods such as fast atom bombardment-mass spectrometry (FAB-MS) – Allows identification of the “profile” of the bile acids in urine that would determine the potential presence of an inborn error of bile acid metabolism

Inborn Errors of Bile Acid Metabolism - Treatment and Outcomes • Treatment for inborn errors of bile acid metabolism focuses on restoring normal bile flow with reduction in the injury to the liver by the toxic intermediates of the bile acid production pathway • Liver disease may progress to end stage liver disease with cirrhosis and a liver transplant may be required
- Slides: 24