MESOVATS TRIAL SATYA SHANBHAG WAIKATO CARDIOTHORACIC UNIT INTRODUCTION

MESO-VATS TRIAL SATYA SHANBHAG WAIKATO CARDIOTHORACIC UNIT

INTRODUCTION • MALIGNANT PLEURAL MESOTHELIOMA (MPM) HAS CONSISTENT WORSE PROGNOSIS • TREATMENT MODALITIES LIMITED: CHEMO, EPP AND COMBINED THERAPY • VATS DEBULKING WITH PARTIAL PLEURECTOMY EMERGING OPTION • USUAL SYMPTOM CONTROL THROUGH TALC PLEURODESIS

RATIONALE The Meso. VATS trial was designed to establish whether 1) VAT-PP improves survival in patients with pleural effusion secondary to malignant pleural mesothelioma when compared with talc pleurodesis, 2) to provide a full economic analysis of these treatments, 3) and to compare symptom control and quality-of-life outcomes.

METHODS • PATIENTS RANDOMLY ASSIGNED TO UNDERGOVAT-PP OR TALC PLEURODESIS(JUNE 2003 D – EC 2012) • STUDY IN 12 CENTERS FROMUK (MAIN CENTER: PAPWORTH) • F/U PERIOD UP TO 1 YEAR (1, 3, 6 AND 12 MONTHS) • PRIMARY OUTCOME: OVERALL SURVIVAL AFTER 1 YR AFTER RANDOMISATION • SECONDARY OUTCOME: PLEURAL EFFUSION, QUALITY OF LIFE, EXERCISE TOLERANCE AND COST TO HEALTH SERVICE

METHODS • PTS RANDOMISED IN 1: 1 FASHION BY COMPUTER GENERATED BLOCK OF 10 PTS • RANDOMISATION STRATIFIED ACCORDING TO RISK SCORE ( HIGHWCC, NON-EPITHELOID CELL TYPE & MALE SEX) • STAGING DETERMINED BY 2 RADIOLOGISTS USINGIMIG SCORE • ESTIMATED 1 YR SURVIVAL TO BE 37 WITHTP AND 59% WITHVATS-PP RESPECTIVELY (80% POWER- 196 PTS)


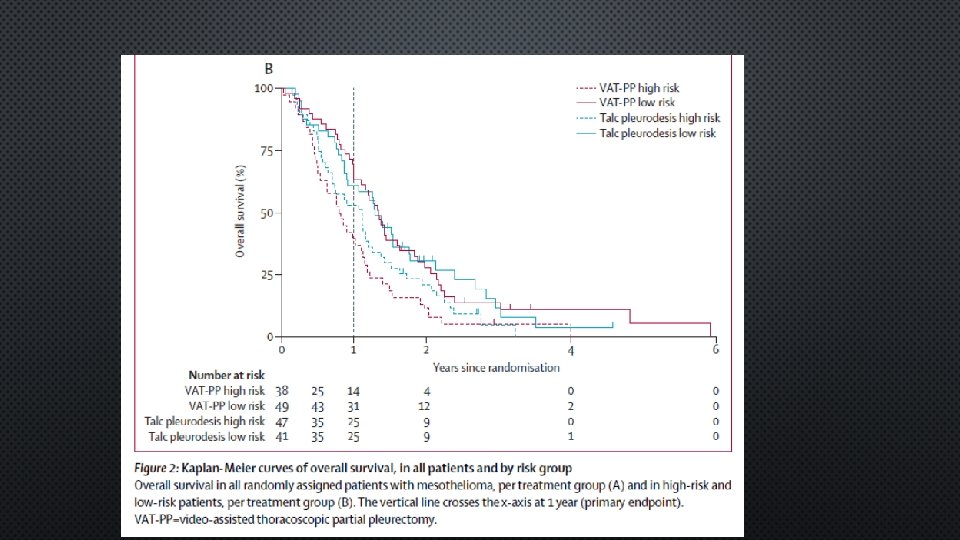
SURVIVAL

QUALITY OF LIFE POST PROCEDURE

COST PER PROCEDURE

AUTHOR CONCLUSIONS • FIRST RT STUDY ABOUTVAT-PP AND TALC PLEURODESIS FOR MPM • AT 1 YR NO DIFFERENCE IN PRIMARY OUTCOME BETWEEN THE TWO GROUP • VATS-PP RESULTED IN MORE COMPLICATIONS, LONGER HOSPITAL STAY AND WAS EXPENSIVE

STUDY STRENGTHS • WELL CONDUCTED MULTI INSTITUTIONAL RANDOMISED STUDY • THE STUDY MET THE REQUIRED NUMBER OF PATIENTS • SOME AMOUNT OF BLINDING PERFORMED FOR SELECTION • PUBLISHED IN A REPUTED JOURNAL

STUDY WEAKNESS • VERY FEW PATIENTS TO COMPARE AT 1 YR, LOGICAL CONCLUSION CANNOT BE DRAWN • COMBINATION THERAPY NOT STANDARDISED, LEFT TO THE TREATING HOSPITAL • BECAUSE OF DIMINISHING NUMBER OF PATIENTS, SECONDARY ENDPOINTS COULD BE DEDUCTED FROM ABOUT 40% OF PATIENTS

STUDY WEAKNESS • OVERALL SURVIVAL CHOSEN AS PRIMARY ENDPOINT FOR AN OPERATION WHERE THE TUMOUR REMAINED • VARYING DEGREE OF DEBULKING INTRODUCED HETEROGENEITY TO THE STUDY • ADDITIONAL CONFOUNDERS: NEOADJUVANT THERAPY IN SOME AND ALSO CHANGE IN METHOD OF TALC INTRODUCTION • TREATMENT NOT STRATIFIED FOR TUMOUR STAGE OR VOLUME

MESOTHELIOMA & RADICAL SURGERY (MARS TRIAL) • UK TRIAL FROM 2003 -2008 WITH 50 PATIENTS RANDOMISED EPP (24 AND 26 NOEPP) • THREE EARLY MORTALITY IN EPP GROUP WITH AHR OF 2. 75 COMPARED TO NON EPP GROUP • WEAKNESS: SMALL STUDY, VERY FAR REACHING CONCLUSIONS Investigator Conclusion: In view of the high morbidity associated with EPP in this trial and in other non-randomised studies a larger study is not feasible. These data, although limited, suggest that radical surgery in the form of EPP within trimodal therapy offers no benefit and possibly harms patients

MARS 2 TRIAL • RANDOMISATION BETWEEN CHEMOTHERAPY ONLY AND EXTENDED PLEURAL DECORTICATION (EPD) • TRIAL STILL ONGOING RECRUITING TOTAL OF 50 PATIENTS. • COMPLETION BY END OF THIS YEAR • EARLY RESULTS FAVOUR SURGERY OVER CHEMOTHERAPY ARM


WHAT ARE THE EVIDENCE/GUIDELINES?
FUTURE PROGRESS

CONCLUSIONS • EPP HAS A ROLE IN MESOTHELIOMA PATIENTS WHO HAVE A FAVOURABLE EARLY STAGE TUMOUR • THESE SHOULD BE OFFERED FOR THE IDEAL CANDIDATE PATIENTS • MULTI-MODAL THERAPY HAS BETTER OUTCOME COMPARED TO MONOTHERAPY/SURGERY • TALC PLEURODESIS IS ADEQUATE FOR ADVANCED STAGE CANCER FOR SYMPTOM RELIEF • NEWER TREATMENT MODALITIES/SURGERY DEVISED FOR PROLONGING LIFE AND QUALITY
- Slides: 24