Mercury Awareness and Safety Bureau of Workers Comp
Mercury Awareness and Safety Bureau of Workers’ Comp PA Training for Health & Safety (PATHS) PPT-056 -01 1

Topics • Mercury (Hg) and variations • Properties • Historic uses • In-home uses • Amalgams • Dental use • Health effects • Hg poisoning • Respirator recommendations • Response safety • Bibliography PPT-056 -01 2

Mercury Awareness and Safety • Atomic number 80 • Also known as quicksilver • Only metal that is liquid at standard conditions for temperature and pressure • Only other element that is liquid under these conditions is bromine PPT-056 -01 3

Mercury (Hg) • Mercury occurs as deposits; mostly as cinnabar, known as mercuric sulfide. • Cinnabar: Highly toxic by ingestion or dust inhalation • Elemental Hg can be produced by heating mercury-containing ores and condensing the vapor PPT-056 -01 4

Three (3) Classes of Hg • Mercury (Element) Hg • Organic (Methylmercury & Phenylmercury) • Inorganic Compounds known as Mercurial salts: • Mercury(I) chloride (mercurous chloride) • a. k. a. Calomel • Mercury(II) chloride (mercuric chloride) David Aldridge, his own work with permission PPT-056 -01 5

29 CFR 1910. 1000, Subpart Z From Table Z-1: Limits for Air Contaminants 8 -hr TWA Substance CAS No. mg/m 3 Mercury (aryl and inorganic)(as Hg) 7439 -97 -6 OSHA PEL: C 0. 1 Mercury (organo) alkyl compounds (as Hg) 7439 -97 -6 OSHA PEL: TWA 0. 01 Mercury (vapor)(as Hg) 7439 -97 -6 OSHA PEL: TWA 0. 05 [skin] PPT-056 -01 6

Incompatibilities Reaction Vigorous Material Mixtures of sodium carbide, aluminum, lead, iron Violent/explosive Chlorine dioxide, lithium, rubidium Reactive with Azides, ammonia, ethylene oxide, and other materials PPT-056 -01 7

Hg Uses Hg Compound Used to make • Mercuric sulfate acetaldehye vinyl chloride vinyl acetate • Mercuric chloride disinfectant • Metallic Hg is poisonous • Heavy metals alter enzymatic and protein action and can lead to death. * *Eugene Meyer, Chemistry of Hazardous Materials, Prentice-Hall Inc. , 1977 PPT-056 -01 8
![NIOSH Pocket Guide Information • Mercury compounds [except (organo) alkyls] (as Hg) • Formula: NIOSH Pocket Guide Information • Mercury compounds [except (organo) alkyls] (as Hg) • Formula:](http://slidetodoc.com/presentation_image/48b2d3e490532a2fcd52210277e8bcde/image-9.jpg)
NIOSH Pocket Guide Information • Mercury compounds [except (organo) alkyls] (as Hg) • Formula: Hg (metal) • CAS# 7439 -97 -6 • IDLH: 10 mg/m 3 (as Hg) • DOT: 2809 • ERG Guide page 172 (metal) • OSHA PEL: Ceiling 0. 1 mg/m 3 • Synonyms/Trade names: Mercury metal - Colloidal mercury, Metallic mercury, Quicksilver; other synonyms vary depending upon specific inorganic and aryl Hg compounds PPT-056 -01 9

NIOSH Pocket Guide Information • MW: 200. 6 • Insoluble • Flash point: NA • IP: ? • Specific Gravity: 13. 6 (metal) • UEL: NA • LEL: NA PPT-056 -01 10

Properties of Hg Specific Gravity: • Mercury is 13. 6 times heavier than water • This density enables it to be used in water level gauges measuring inches of Mercury to feet of water; where each inch of Mercury read will support a column of water 1. 13 feet high. Note Mercury’s ability to support heavy objects PPT-056 -01 11

Properties of Hg • Allows us to determine atmospheric pressure PPT-056 -01 12

Historic Uses • Fur removal from pelts to make hats in the mid 1700 s to mid-1800 s • Vapors from process were very toxic • Mercury poisoning was said to make one “mad as a hatter” • Also used in mining to separate gold and silver from ore to create an amalgam for recovery PPT-056 -01 13

Historic Uses 1800 s • Treat depression • Toothaches 1900 s • Laxatives • Teething powders • Mercurochrome PPT-056 -01 14

Historic Uses • Through catalytic reactions, Hg is used to make polymers, chlorine and caustic soda, as well as to extract gold from ore PPT-056 -01 15

Historic Uses • Fishing lures • Lighthouse lenses • Fire detectors • Power plants: fossil and nuclear • Separating chlorine and sodium from brine • Thermometers PPT-056 -01 16

In-Home Uses • Precautions are required due to the use of Mercurycontaining materials in each household • Other uses of Hg should instill safety considerations PPT-056 -01 17

In-Home Uses • Mercury switches to control thermostats in our homes PPT-056 -01 18

In-Home Uses • Wood preservative • Making mirrors • Herbicides • Antiseptics • Antidepressants • Mercury battery for hearing aids • Cosmetics PPT-056 -01 19

Fluorescent Lamp • Compact Fluorescent Lamp (CFL) • More in use: less power to generate more light; last longer than conventional bulbs • Injection of Mercury via broken glass may occur PPT-056 -01 20

Mercury Lamps • Breakage from Hg lamps may appear insignificant and easy to remedy, but you should exercise due care and maximize safety when cleaning this up PPT-056 -01 21

Hg Forms • Most toxic forms for Mercury • Organic compounds: • Dimethyl mercury • Methyl mercury • Inorganic compounds (such as cinnabar) highly toxic by ingestion and inhalation PPT-056 -01 22

Amalgams • Hg dissolves to form amalgams with gold, zinc and other metals • Hg reacts with aluminum to form a Mercury/aluminum amalgam • This reacts with air resulting in aluminum oxide; corrodes aluminum, not allowed on aircraft because it may react with aircraft metal, thereby weakening it PPT-056 -01 23
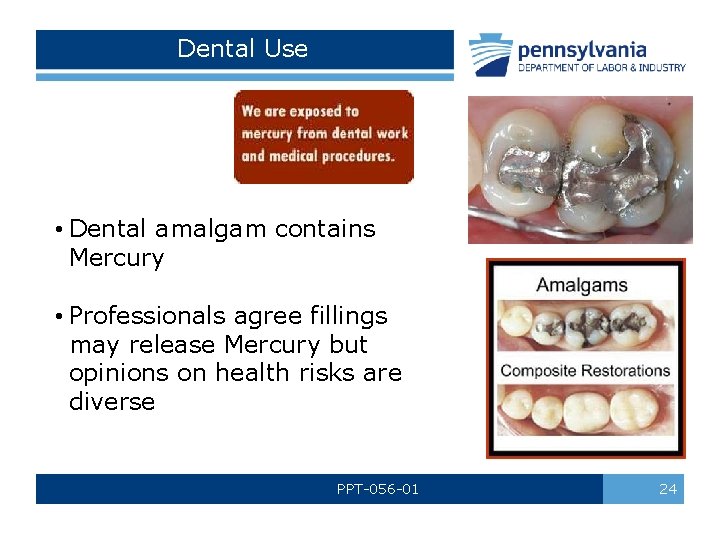
Dental Use • Dental amalgam contains Mercury • Professionals agree fillings may release Mercury but opinions on health risks are diverse PPT-056 -01 24
Routes of Exposure Inhalation • Primary route for elemental Mercury • 75 percent to 80 percent is absorbed by lungs • Vapors are heavier than air (vapor density: 6. 9) Skin or Eye Contact • Absorbed slowly through skin • Causes irritation to skin and eyes and possibly contact dermatitis Ingestion • Methyl mercury completely absorbed and not readily eliminated PPT-056 -01 25

Health Effects • Inhalation of vapor; major exposure route • Symptoms may include corrosive bronchitis progressing to pulmonary edema or fibrosis • Mercury can cross the blood, brain and placental barriers, posing an increased risk to children Agency for Toxic Substances and Disease Registry, Medical Management Guidelines for Mercury, found at http: //www. atsdr. cdc. gov/MMG. asp? id=106&tid=24 PPT-056 -01 26

Food Chain Consequences • Understanding the food chain allows us to understand how we might internalize Mercury • State fishing laws are specific regarding the amount of fish which may be eaten on a daily basis after waterway studies have been made PPT-056 -01 27

Acute Health Effects System: Symptoms: Respiratory Cough, sore throat, shortness of breath Gastrointestinal Metallic taste, nausea, vomiting, diarrhea, abdominal pain Central Nervous Headache, weakness, visual System disturbances PPT-056 -01 28
Chronic Exposure Mercury may accumulate in the body, resulting in permanent damage and changes to: • Kidneys • Nervous system • Muscles PPT-056 -01 29

Atmospheric Hg in U. S. 1998 • Due to air pollution and other releases of Hg into the atmosphere PPT-056 -01 30

Mercury Poisoning may result from: • Exposure to water-soluble forms of Mercury, i. e. Mercuric chloride or Methylmercury • Inhalation of Mercury vapor or eating contaminated seafood Symptoms of Hg poisoning include: • Mild gastritis to severe pain with vomiting • Convulsions and numbness in mouth and limbs • Visual field constriction • Speaking difficulty PPT-056 -01 31

Mercury Poisoning • In 1953, poisoning occurred in Japan when industrial waste was discharged into Minamata Bay; the waste contained Methyl mercury salts • Aquatic microorganisms can convert mercury salts into methylated compounds (a methyl group attaches to Mercury atoms); these are easily absorbed into animal tissues • Minamata residents ate the fish containing Mercury PPT-056 -01 32

Mercury Poisoning • In the 1960’s and 1970’s in Iraq, wheat treated with Methyl mercury as a preservative due to being seed grain was fed to animals and consumed by people • Resulted in neurological symptoms: • • • Speech difficulties Narrowing of visual field Hearing impairment Blindness Death PPT-056 -01 33

Respirator Recommendations (NIOSH, Table 3 & 4) • Mercury Vapor: Mercury compounds [except (organo) alkyls] (as Hg) Levels for concern: Up to 0. 5 mg/m 3 Up to 1. 25 mg/m 3 Up to 2. 5 mg/m 3 Up to 10 mg/m 3 Emergency or planned entry into unknown concentrations or IDLH conditions • Escape • • • PPT-056 -01 34

Respirators • Up to 0. 5 mg/m 3 • Any air-purifying half-mask respirator equipped with cartridge(s) providing protection against the compound of concern (APF 10) or • Any supplied - air respirator (APF 10) PPT-056 -01 35

Respirators • Up to 1. 25 mg/m 3 • Supplied-air respirator operated in a continuous -flow mode (APF 25) or • Any powered air-purifying respirator with cartridge(s) providing protection against the compound of concern (APF 25) Canister with end of service life indicator PPT-056 -01 36

Respirators • Up to 2. 5 mg/m 3 • Any air-purifying full-face piece respirator equipped with cartridge(s) providing protection against the compound of concern (APF 50) or • Any air-purifying, full-face piece respirator (gas mask) with a chin-style, front- or back-mounted canister providing protection against the compound of concern (APF 50) ESLI required for canister, or PPT-056 -01 37

Respirators • Up to 2. 5 mg/m 3 (continued) • Any supplied-air respirator that has a tight-fitting face piece and is operated in a continuous-flow mode (APF 50) or • Any powered air-purifying respirator with a tightfitting face piece and cartridge(s) providing protection against the compound of concern (APF 50) or PPT-056 -01 38

Respirators • Up to 2. 5 mg/m 3 (continued) • Any self-contained breathing apparatus with a full face piece (APF 50) or • Any supplied-air respirator with a full face piece (APF 50) PPT-056 -01 39

Respirators • Up to 10 mg/m 3 • Any supplied-air respirator operated in a pressure-demand or other positive-pressure mode (APF 1, 000) PPT-056 -01 40

Respirators • Emergency or planned entry into unknown concentrations or IDLH conditions • Any self-contained breathing apparatus that has a full face piece and is operated in a pressuredemand or other positive-pressure mode (APF 10, 000) or PPT-056 -01 41

Respirators • Any supplied-air respirator that has a full-face piece and is operated in a pressure-demand or other positive-pressure mode in combination with an auxiliary self-contained positivepressure breathing apparatus (APF 10, 000) PPT-056 -01 42

Respirators • Escape • Any air-purifying, full-face piece respirator (gas mask) with a chin-style, front- or back-mounted canister providing protection against the compound of concern. (APF 50) or • Any appropriate escape-type, self-contained breathing apparatus. • NIOSH has respirator recommendations for other mercury compounds as well: Mercury (organo) alkyl compounds (as Hg) PPT-056 -01 43

Personal Safety • 29 CFR 1910. 252(c)(10) • “In confined spaces or indoors, welding or cutting operations involving metals coated with mercurybearing materials, including paint, must be done using local exhaust ventilation or airline respirators unless atmospheric tests under the most adverse conditions show that employee exposure is within the acceptable concentrations specified by 29 CFR 1910. 1000. Such operations, when done outdoors, must be done using respirators approved for this purpose by NIOSH under 42 CFR part 84. ” PPT-056 -01 44

Mercury Detectors • Gas analyzer • Direct Mercury analyzer • Mercury vapor PPT-056 -01 45

Response Safety Plan for response to: • Hg liquid • Hg vapor events • Have contingencies for: • Rescuer safety • Victim treatment • Multi-casualty situations PPT-056 -01 46

Response Safety Personal protective equipment (PPE) for staff working in each zone: • Hot Zone • Warm Zone (Decon) • Support Zone PPT-056 -01 47

Response Safety Having a response plan for smaller as well as larger types of potential emergencies will promote: • Personnel safety • Reduced downtime • Reduced costs PPT-056 -01 48

Response Safety • Elemental Hg is toxic in both the liquid and vapor form • Although not as readily absorbed through the skin, the inhalation hazard is more severe • Gloves and foot protection are recommended when dealing with liquid form PPT-056 -01 49

Response Safety • However, self-contained breathing apparatus (SCBA) and chemical protective clothing (CPC) are recommended if dealing with heated vapor • Decontaminate or dispose of clothing contacting liquid mercury PPT-056 -01 50

Decontamination • Review response policies before a potential event • Decon or remove and double-bag contaminated clothing • Also bag all personal belongings (if you were on the Emergency Response Team, you shouldn’t have taken any personal items into the Hot Zone) • Wash hair and exposed skin thoroughly • Eyes should be flushed with water or saline for at least 5 minutes if exposed • Initiate proper medical protocols PPT-056 -01 51

Chemical Substitutes for Hg • Due to problems previously cited with the use of Mercury, chemical substitutes are sought • One such substitute, called New. Merc by its originators, is a “nontoxic, electrically-conductive, liquid alloy alternative” • It is based on gallium and indium alloyed with tin • Various applications exist including float switches for waste water, bilge pumps on ships and sump pumps for homes • Further study should result in additional uses Alan Brown, “Nontoxic Liquid Metal Alloy Could Substitute For Mercury, ” from Chemical Online, April 19, 1999, found at http: //www. chemicalonline. com/doc. mvc/Nontoxic-Liquid-Metal-Alloy-Could-Substitute 0001 PPT-056 -01 52

Texts to Assist Your Planning PPT-056 -01 53

Selected Bibliography § Eugene Meyer, Chemistry of Hazardous Materials, Prentice-Hall Inc. , 1977 § NIOSH Pocket Guide to Chemical Hazards §http: //www. cdc. gov/niosh/npgd 0383. html § Agency for Toxic Substances and Disease Registry, Medical Management Guidelines for Mercury §http: //www. atsdr. cdc. gov/MMG. asp? id=106&tid=24 § Philippe Grandjean, Hiroshi Satoh, Katsuyuki Murata, and Komyo Eto, “Adverse Effects of Methylmercury: Environmental Health Research Implications, ” published in Environmental Health Perspectives, August 2010 (published online June 8, 2010) §http: //www. ncbi. nlm. nih. gov/pmc/articles/PMC 2920086/ § Alan Brown, “Nontoxic Liquid Metal Alloy Could Substitute For Mercury, ” from Chemical Online, April 19, 1999 §http: //www. chemicalonline. com/doc. mvc/Nontoxic-Liquid-Metal-Alloy-Could. Substitute-0001 PPT-056 -01 54

Questions PPT-056 -01 55
- Slides: 55