MEOR Using Facultative Anaerobic Indigenous Consortia and Biosurfactants

MEOR Using Facultative Anaerobic Indigenous Consortia and Bio-surfactants Xiaofang Wei 1 Xingli Li 2 Keyu Liu 3 Yuehui She 4 Jing Wang 1 1 China University of Petroleum 2 GRI, Jiangsu Oilfield 3 CSIRO Petroleum 4 Yangtz University

Acknowledgements n n CSIRO Wf. O Flagship Biochemical Engineering Laboratory, CUP, Beijing n n Yijng Luo, Mang Lu, Peiwen Zheng, Zhongzhi Zhang The Langfang Research Institute of Petroleum Exploration and Development, Petro. China n Core-flooding technical support 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Presentation Outline n n Background of the MEOR study Nutrient stimulation n n Bacteria selection and metabolite analysis n n Oil spreading and blood agar lysis methods Emulsification Interfacial tension (IFT) Core flooding experiments n n n MEOR screening and assessment Experimental procedure Results Conclusions and Future work 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia
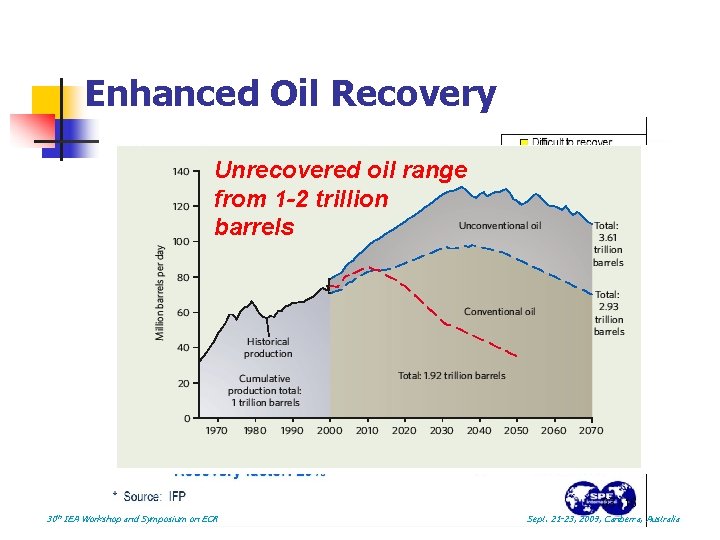
Enhanced Oil Recovery Unrecovered oil range from 1 -2 trillion barrels 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia
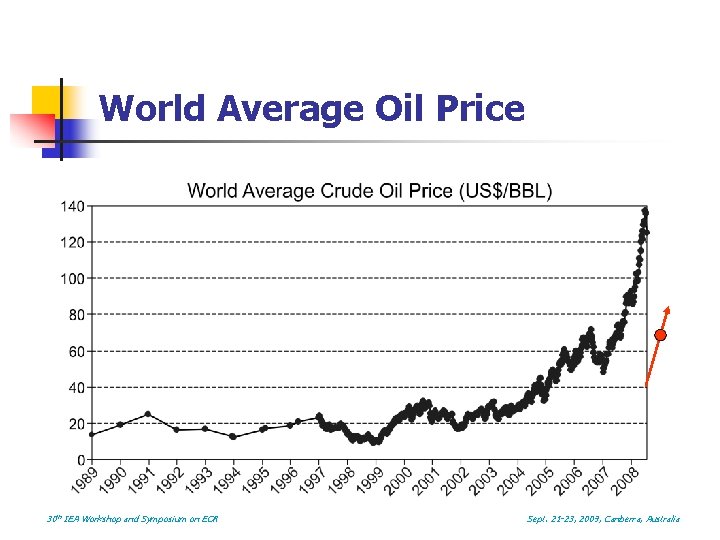
World Average Oil Price 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia
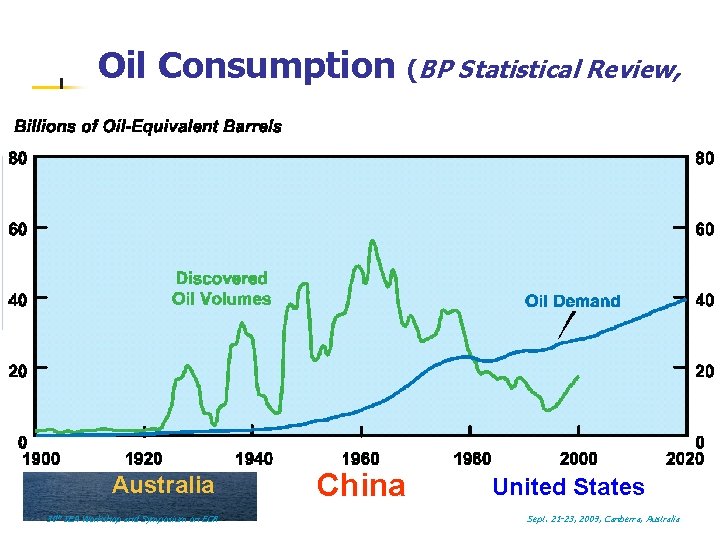
Oil Consumption (BP Statistical Review, 2006) l 1 km l Australia: 0. 9 mbbl/day China: 7. 0 US: 20. 6 World: 82. 5 US 1 cubic km oil / year Australia 30 th IEA Workshop and Symposium on EOR China United States Sept. 21 -23, 2009, Canberra, Australia
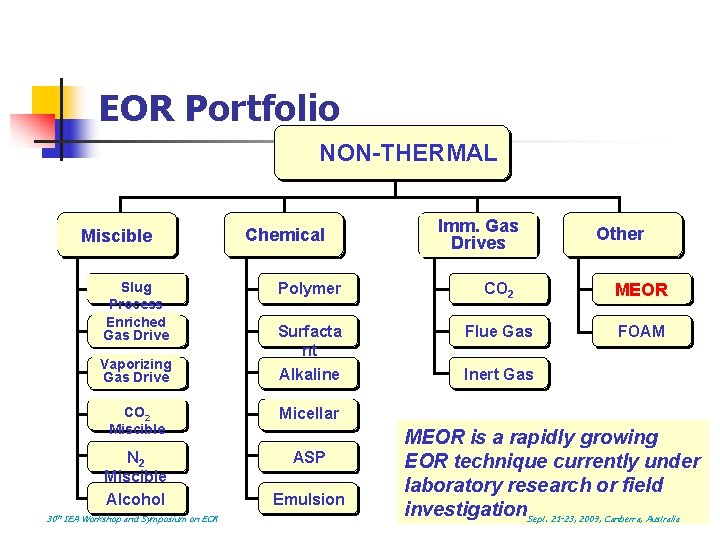
EOR Portfolio NON-THERMAL Miscible Slug Process Enriched Gas Drive Vaporizing Gas Drive 30 th Chemical Other Polymer CO 2 MEOR Surfacta nt Alkaline Flue Gas FOAM CO 2 Miscible Micellar N 2 Miscible Alcohol ASP IEA Workshop and Symposium on EOR Imm. Gas Drives Emulsion Inert Gas MEOR is a rapidly growing EOR technique currently under laboratory research or field investigation. Sept. 21 -23, 2009, Canberra, Australia

MEOR Mechanisms and Strategies n Acids, gas, solvents production n Selective plugging n n Commonly made by carbohydrate fermentation Inject carbohydrates (molasses, etc. ) Create biofilms, cell mass and/or mineral precipitates Any nutrient could work: use nitrate as electron acceptor If reservoir has high divalent cation concentration, CO 2 production may stimulate carbonate formation Hydrocarbon degradation n Inject electron acceptor and/or limiting nutrients (N, S, P, metals) n Biosurfactant production n Emulsifiers n Periodic cycles of nutrient-excess and nutrient limitation 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia
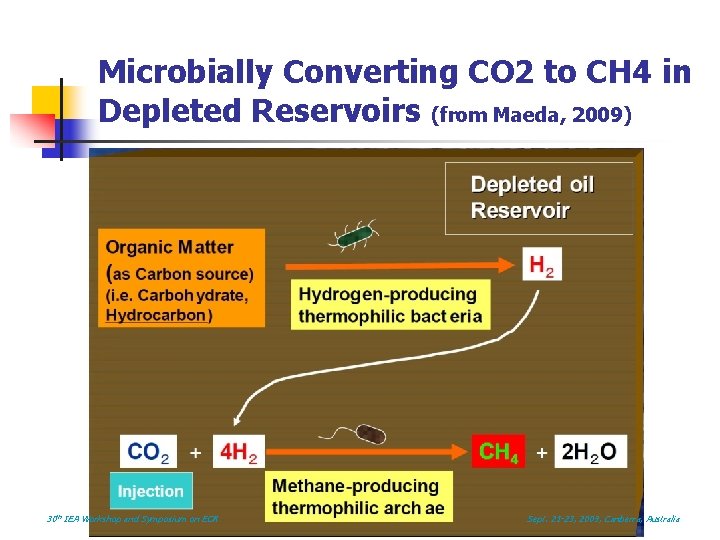
Microbially Converting CO 2 to CH 4 in Depleted Reservoirs (from Maeda, 2009) 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Implementing MEOR (from Mc. Inerney, 2009) n n n Not all reservoirs are candidates for MEOR Environmental conditions limit microbial growth Recommendations: n n n Temperature: <80 o. C p. H: 5 -9 Salinity: <10% Depth: <2500 m API gravity: > 15 o Residual oil saturation: >25% 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Background of the Reservoir Investigated n Characteristics of the Huatugou Oil Reservoir, Qinghai Oilfield, China n n n Production history: 1958 -present EOR: Water-flooding Formation Water Salinity <2 x 104 ppm Reservoir P/T: 45℃ /22 MPa Fm water p. H≤ 7 Low sulfate concentration 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Microbial Screening n Hydrocarbon Degrading Bacteria (HBD) n n Microbes population>105 cells/ml (Bottle test) Sulphate Reducing Bacteria (SRB) n Not Detected (PCR) 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

MEOR Screening The Huatugou reservoir is a good candidate for MEOR n n n Relatively high microbe population: >105 (Cell counting) Low formation Water Salinity: <2 x 104 ppm Low reservoir T: 45℃ Suitable formation water chemistry: p. H≤ 7 Low corrosion risk: no SRB detected 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Nutrient Stimulation n n Proper nutrient (Carbon Source: Reservoir oil) Target facultative anaerobic indigenous consortia Cell Population is up to 108 cells/ml Minor IFT and Viscosity changes Gas production observed 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Bio-surfactant: species selection and identification n Two strains were selected and named as BIOS 682 -1 and BIOS 682 -2 Initial screening by the blood agar lysis method: positive Screening using the oil spreading method n n n Pure culture clear zone diameters : 4. 5 cm and 4. 8 cm Mixed culture clear zone diameter: 5. 0 cm Species identification using 16 s RNA n n BIOS 682 -1: Brevibacillus agri (99% similarity) BIOS 682 -2: Brevibacillus levicki (95%, a possible new species) 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Bio-surfactants: species culture and assessment n n Mixed culture of two selected strains n 5 ml oil/50 ml-medium cultured for 3 weeks at 45℃ n Under facultative anaerobic condition (headspace filled with N 2) Oil-water separation n Oil was centrifuged at 5000 rpm for 10 min at room temperature n Oil was extracted by using CCl 4 (oil in water) The surface tension n Equipment: Contact Angle analyzer (DCA 322) Emulsification (Culture and reservoir oil 1: 1 v/v) n Emulsification maintained for 100 hrs after 15 min u/s bathing n The ratio between emulsified oil and water measured 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia
Bio-surfactant producing species: culture and assessment n Surface tension reduction: 24% n Emulsification improvement: 14% 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Bio-surfactant analysis n Bio-surfactant of lipopeptide extraction protocol n n n n centrifuged twice at 4°C @10, 000 rpm for 20 min the supernatant was mixed with 6 mol HCL @ p. H=2 (24 hrs 4°C) The floccules were collected using centrifuge (4°C @10, 000 rpm for 20 min) The settlings were washed using acidic solution (p. H=2), then dissolved in weak alkali solution (p. H=8) Extracted with DCM and methanol solution (v/v 3: 1) Dried under vacuum at low temperature The primary products were dissolved in alkali solution (p. H=8) The extraction was then dried under vacuum 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Bio-surfactant analysis n Thin Layer Chromatography (TLC) n n Initial screening Liquid Chromatography-Mass Spectrometry (LC-MS). n n The purified product was hydrolyzed for 14 hrs at 110 ℃ with 6 N HCl in N 2 chamber Equipment: Agilent 1100 system a-Amino acid 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Bio-surfactant analysis n Thin Layer Chromatography (TLC) Metabolit e Standard Sample lipopeptide bio-surfactant (Pink): Qualitative analysis Solution used is a mixture of n-butanol/alcohol/water(4: 1: 1) 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia
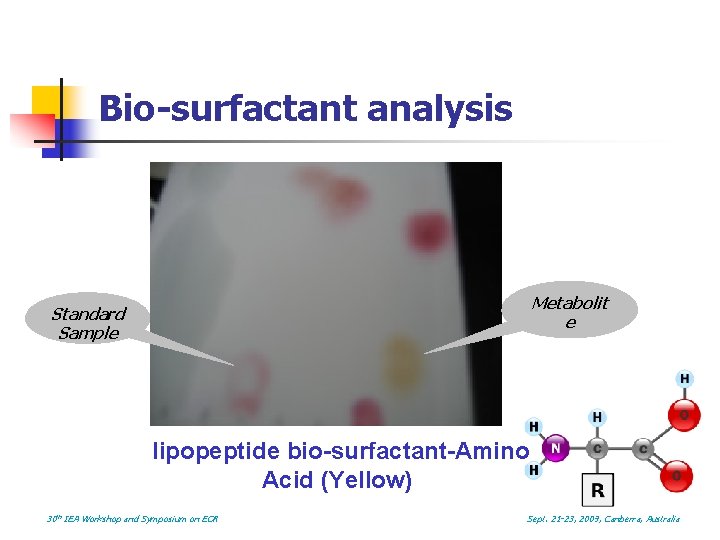
Bio-surfactant analysis Metabolit e Standard Sample lipopeptide bio-surfactant-Amino Acid (Yellow) 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia
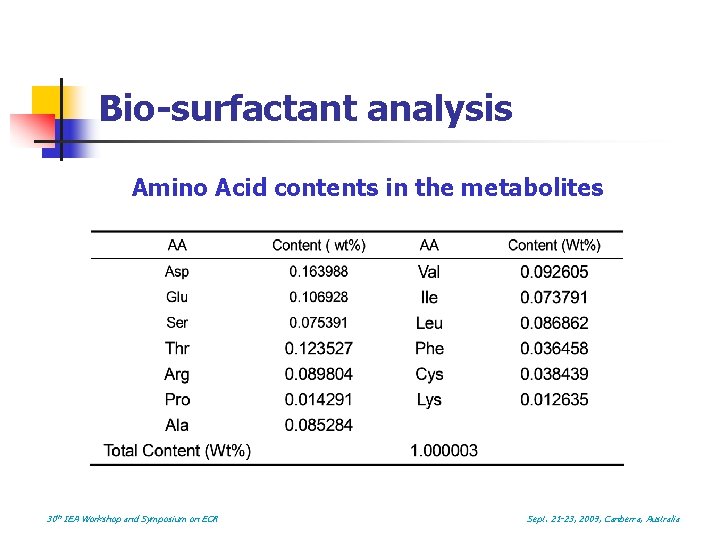
Bio-surfactant analysis Amino Acid contents in the metabolites 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia
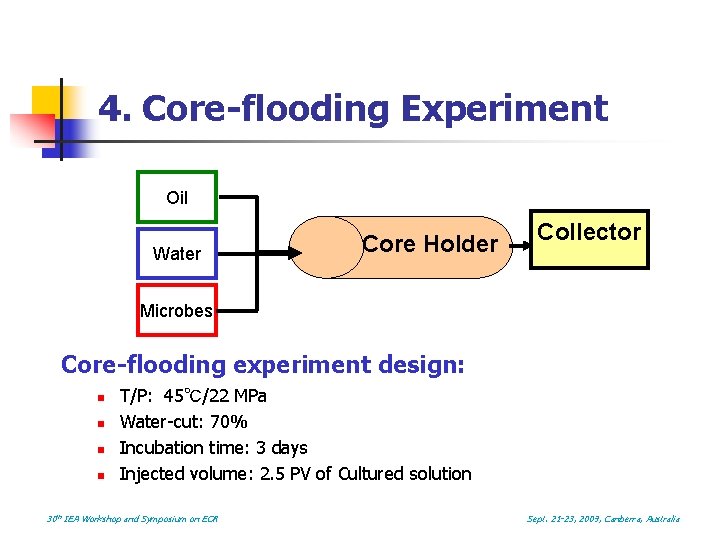
4. Core-flooding Experiment Oil Water Core Holder Collector Microbes Core-flooding experiment design: n n T/P: 45℃/22 MPa Water-cut: 70% Incubation time: 3 days Injected volume: 2. 5 PV of Cultured solution 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Synthetic Core Plugs for Core Flooding Experiments n n Material: oil zone sandstone recovered from the producing well, n Disintegrated Cleaned n Sieved to 30, 80 and 120 meshes, respectively A two-layer heterogeneous sand packs with different permeabilities was constructed n Layer 1: 2: 2: 1 mix of 30 -80, 80 -120 and >120 meshes n Layer 2: 1: 1 mix of 30 -80 and 80 -120 meshes 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Core-flooding Experiment η=(B-A)-C n n B: final oil recovery % A: oil recovery % from 0. 5 pv nutrient solution injection C: oil recovery % (Blank) from 0. 5 pv synthetic formation water injection η: incremental oil recovery (%) Core plug properties 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia
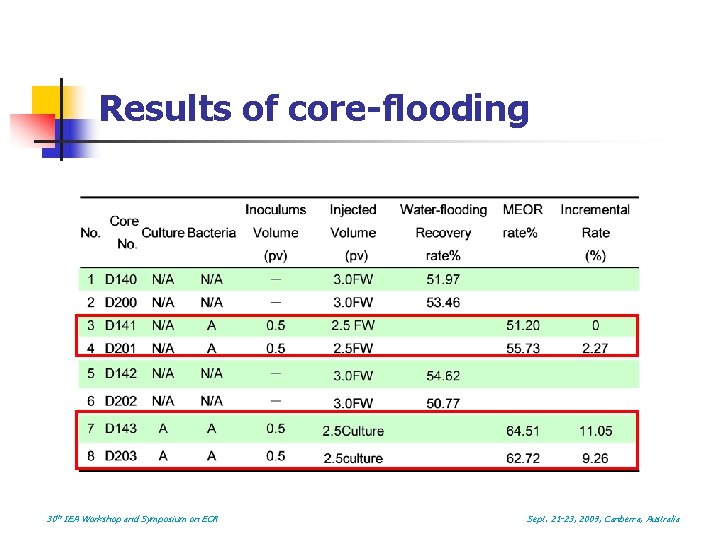
Results of core-flooding 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Conclusions n n The Huatugou reservoir in the Qinghai Oilfield is a good candidate for MEOR The indigenous facultative anaerobic flora from the reservoir was stimulated by adding proper nutrients to promote their activities The two indigenous aerobic species selected can cometabolize well and produced more bio-surfactant than from a single strain in the reservoir The two species identified by 16 s RNA are Brevibacillus agri (BIOS 682 -1); and a possible new species in genus of Bacillus brevis (BIOS 682 -2). 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Conclusions n n The metabolites of BIOS 682 contain low concentration of amino acids An 11% incremental recovery was achieved using a combined microbial and biosurfactant solution injection. The facultative anaerobic flora play an important role in peeling the oil form rock or breaking down the large oil drops. The use of cultured solution with bio-surfactant and other metabolites is much more effective than the use of the indigenous microbes alone in recovering residual oil. 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Future work n In-situ experiments (Field Trials) n Further analysis of the metabolites 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia

Dr Xiaofang Wei Dr Keyu Liu Research Institute of Petroleum Exploration and Development, Petro. China weixfanf@163. com Research Team Leader CSIRO Petroleum Keyu. Liu@csiro. au Thank You 30 th IEA Workshop and Symposium on EOR Sept. 21 -23, 2009, Canberra, Australia
- Slides: 30