Mentis Cura Datadriven brain health April Mentis 2020
Mentis Cura Data-driven brain health April. Mentis 2020 Cura Company presentation May 2020
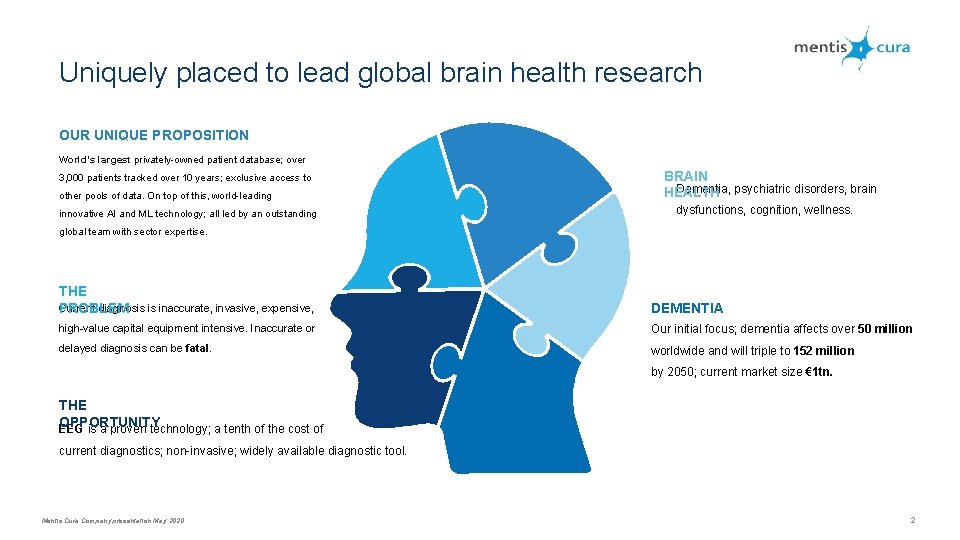
Uniquely placed to lead global brain health research OUR UNIQUE PROPOSITION World’s largest privately-owned patient database; over 3, 000 patients tracked over 10 years; exclusive access to other pools of data. On top of this, world-leading innovative AI and ML technology; all led by an outstanding BRAIN Dementia, psychiatric disorders, brain HEALTH dysfunctions, cognition, wellness. global team with sector expertise. THE Current diagnosis is inaccurate, invasive, expensive, PROBLEM DEMENTIA high-value capital equipment intensive. Inaccurate or Our initial focus; dementia affects over 50 million delayed diagnosis can be fatal. worldwide and will triple to 152 million by 2050; current market size € 1 tn. THE OPPORTUNITY EEG is a proven technology; a tenth of the cost of current diagnostics; non-invasive; widely available diagnostic tool. Mentis Cura Company presentation May 2020 2
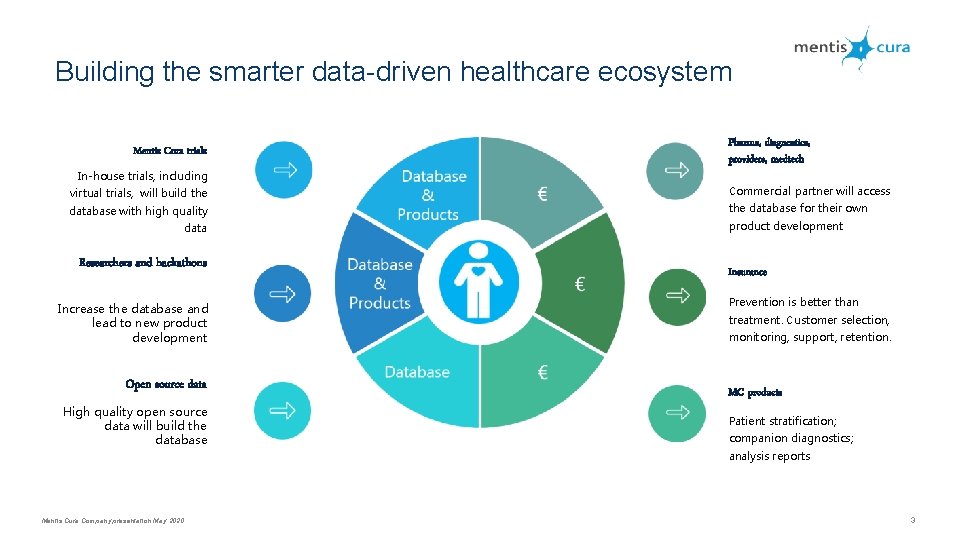
Building the smarter data-driven healthcare ecosystem Mentis Cura trials In-house trials, including Pharma, diagnostics, providers, medtech virtual trials, will build the Commercial partner will access database with high quality the database for their own data Researchers and hackathons Increase the database and lead to new product development Open source data High quality open source data will build the database product development Insurance Prevention is better than treatment. Customer selection, monitoring, support, retention. MC products Patient stratification; companion diagnostics; analysis reports Mentis Cura Company presentation May 2020 3
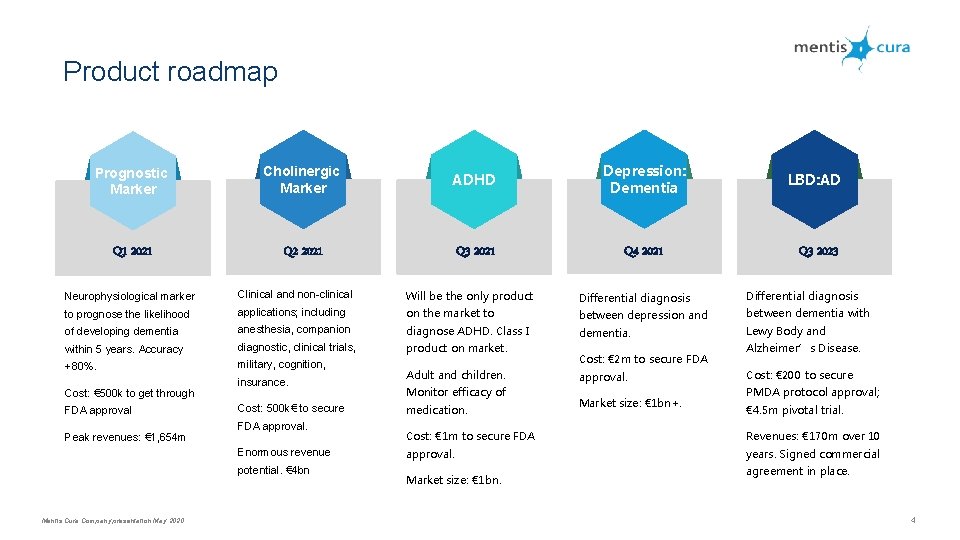
Product roadmap Prognostic Marker Cholinergic Marker ADHD Depression: Dementia LBD: AD Q 1 2021 Q 2 2021 Q 3 2021 Q 4 2021 Q 3 2023 Neurophysiological marker Clinical and non-clinical Will be the only product Differential diagnosis to prognose the likelihood applications; including on the market to between depression and between dementia with of developing dementia anesthesia, companion diagnose ADHD. Class I dementia. Lewy Body and within 5 years. Accuracy diagnostic, clinical trials, product on market. +80%. military, cognition, Cost: € 500 k to get through FDA approval Peak revenues: € 1, 654 m insurance. Cost: 500 k€ to secure FDA approval. Enormous revenue potential. € 4 bn Mentis Cura Company presentation May 2020 Adult and children. Monitor efficacy of medication. Cost: € 2 m to secure FDA approval. Market size: € 1 bn+. Alzheimer’s Disease. Cost: € 200 to secure PMDA protocol approval; € 4. 5 m pivotal trial. Cost: € 1 m to secure FDA Revenues: € 170 m over 10 approval. years. Signed commercial Market size: € 1 bn. agreement in place. 4

Data platform Enables new novel drugs and medical More patients diagnosed and treated with devices to be developed faster and less improved quality of life expensively Real World Database for Discovery $10 bn global healthcare analytic market and precision medicine Clinical Trials precision medicine demands development of companion diagnostics for patient stratification Data Analysis software that can detect genes linked to preventable medical conditions e. g. cancer, stroke Pipeline of potential partners Mentis Cura Company presentation May 2020 5
- Slides: 5