Meningococcal serogroup B defeated Ray Borrow Vaccine Evaluation

Meningococcal serogroup B defeated? Ray Borrow Vaccine Evaluation Unit, Health Protection Agency, Manchester Royal Infirmary, Manchester, U. K. ray. borrow@hpa. org. uk

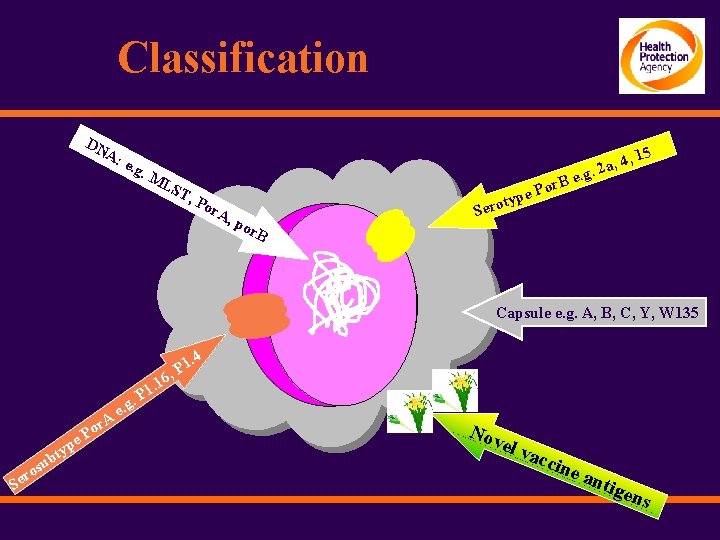
Classification DN A: e. g. M LS T, Po r. A pe , po r. B ty Sero a, 4, 2. g B e. r o P 15 Capsule e. g. A, B, C, Y, W 135 1. 4 P , . r. A u os r Se b Po e typ e. g . 16 1 P Nov el v acc ine a ntig ens
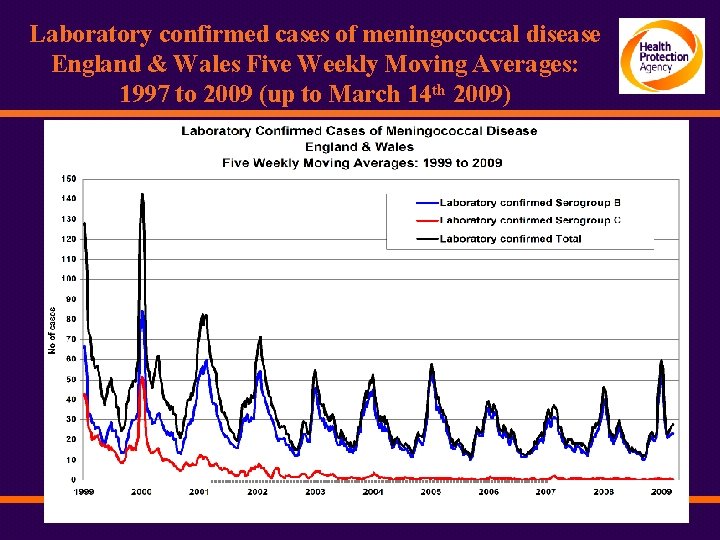
Laboratory confirmed cases of meningococcal disease England & Wales Five Weekly Moving Averages: 1997 to 2009 (up to March 14 th 2009)
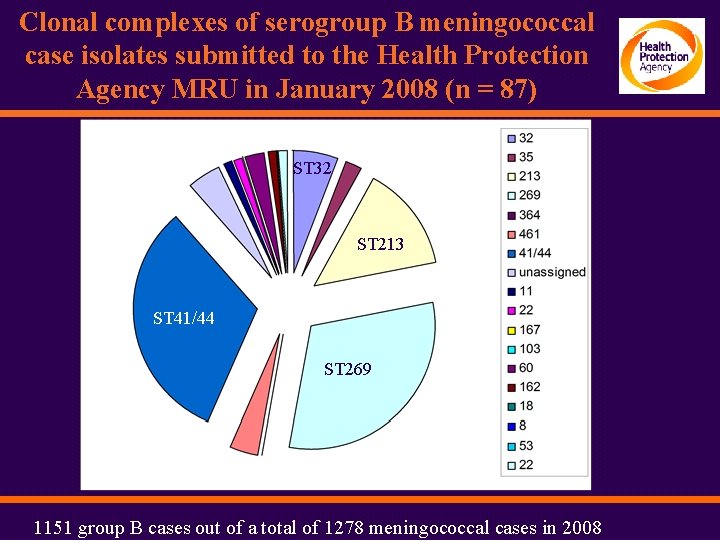
Clonal complexes of serogroup B meningococcal case isolates submitted to the Health Protection Agency MRU in January 2008 (n = 87) N = 189 N = 97 N = 90 ST 32 ST 213 ST 41/44 ST 269 1151 group B cases out of a total of 1278 meningococcal cases in 2008
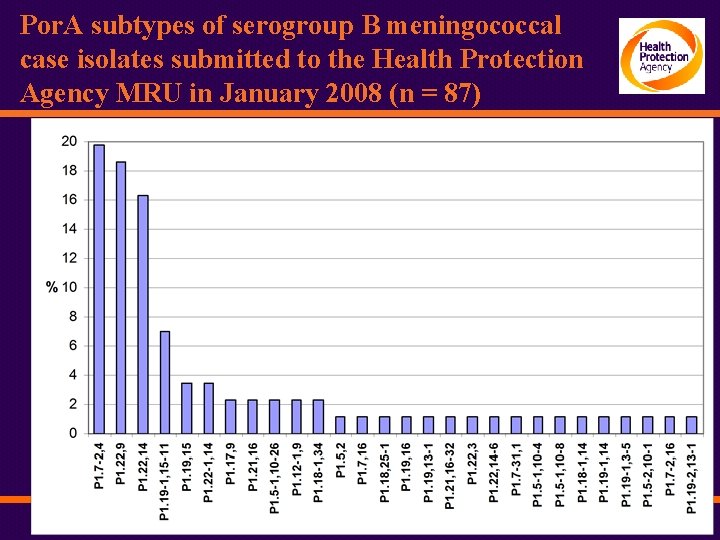
Por. A subtypes of serogroup B meningococcal case isolates submitted to the Health Protection Agency MRU in January 2008 (n = 87)

Correlates of protection • Immunogenicity studies, rather than efficacy studies, are sufficient for licensure of ‘group B’ meningococcal vaccines. • Elevated serum bactericidal antibody (SBA) titres against a broad range of representative strains. • If no SBA activity, efficacy studies will be required. Borrow R et al. Neisseria meningitidis Group B Correlates of Protection and Assay Standardization – International Meeting Report, Atlanta USA, 16 -17 March 2005. Vaccine 2006; 24: 5093 -107.
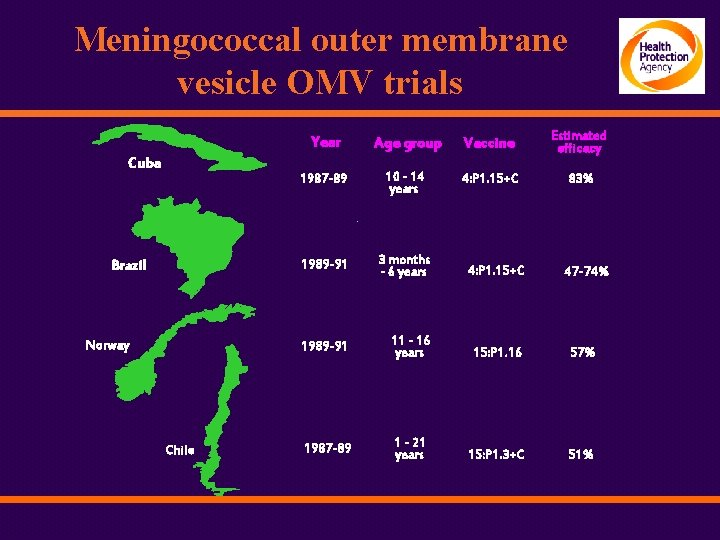
Meningococcal outer membrane vesicle OMV trials Cuba Brazil Norway Chile Year Age group Vaccine Estimated efficacy 1987 -89 10 - 14 years 4: P 1. 15+C 83% 1989 -91 3 months - 6 years 4: P 1. 15+C 1989 -91 11 - 16 years 15: P 1. 16 57% 1987 -89 1 - 21 years 15: P 1. 3+C 51% 47 -74%
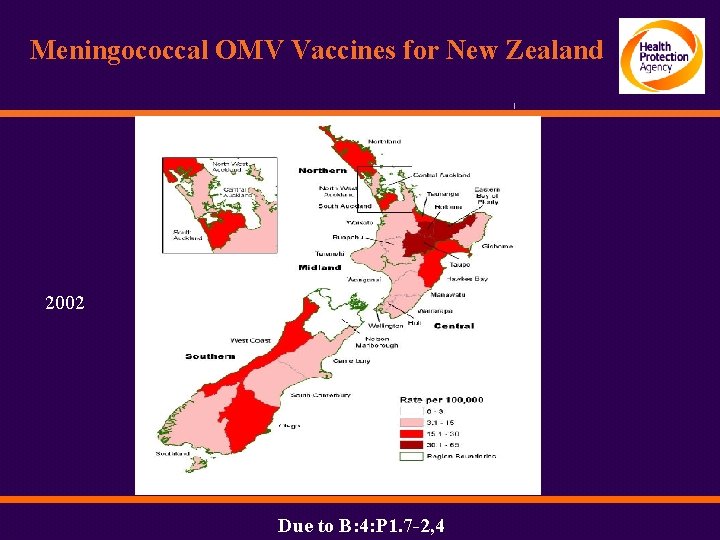
Meningococcal OMV Vaccines for New Zealand 2002 Due to B: 4: P 1. 7 -2, 4
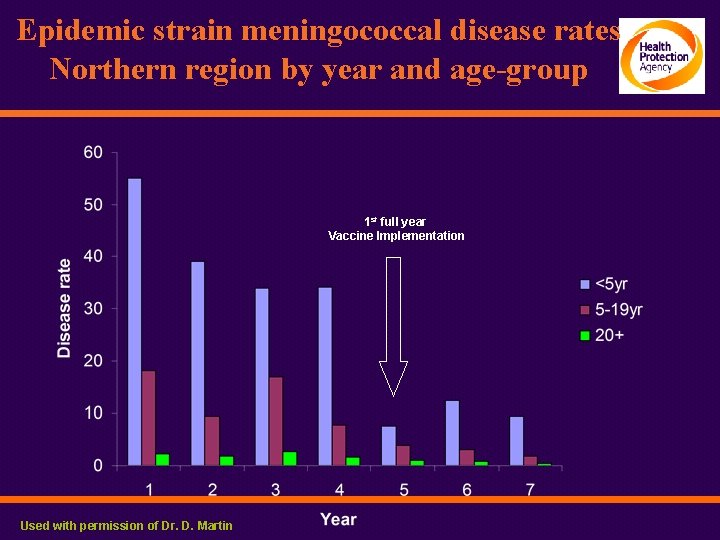
Epidemic strain meningococcal disease rates Northern region by year and age-group 1 st full year Vaccine Implementation Used with permission of Dr. D. Martin
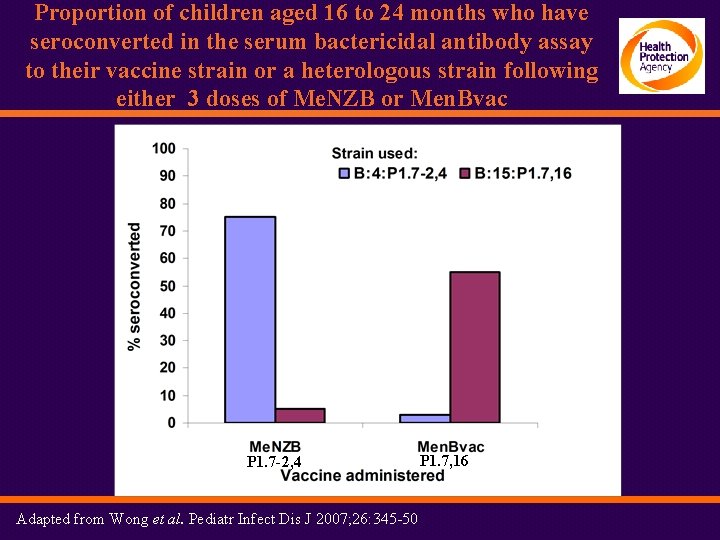
Proportion of children aged 16 to 24 months who have seroconverted in the serum bactericidal antibody assay to their vaccine strain or a heterologous strain following either 3 doses of Me. NZB or Men. Bvac P 1. 7 -2, 4 Adapted from Wong et al. Pediatr Infect Dis J 2007; 26: 345 -50 P 1. 7, 16
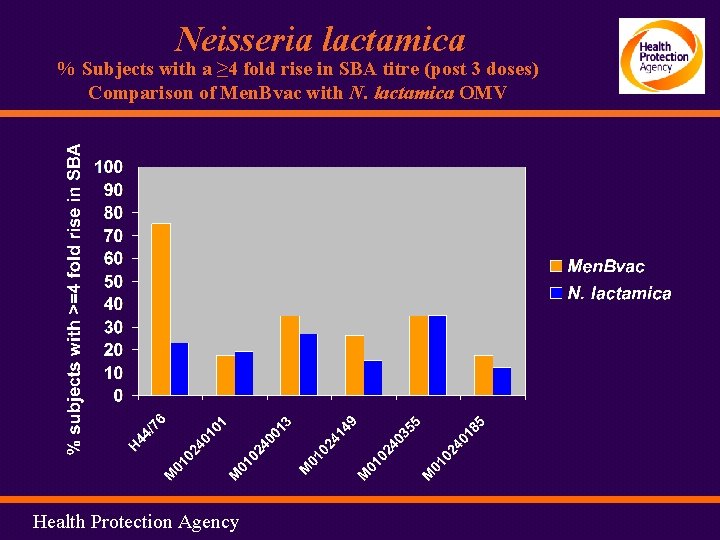
Neisseria lactamica % Subjects with a ≥ 4 fold rise in SBA titre (post 3 doses) Comparison of Men. Bvac with N. lactamica OMV Health Protection Agency

Por. A OMP • Meningococci are classified into serosubtypes based upon variation in VR 1 & VR 2. • Mabs against VR 1 & VR 2 are bactericidal and protective in animal protection studies. • Have long been perceived as potential vaccine components despite variation. • Efficacy studies of OMV vaccines have shown a link between Por. A antibodies & SBA activity.
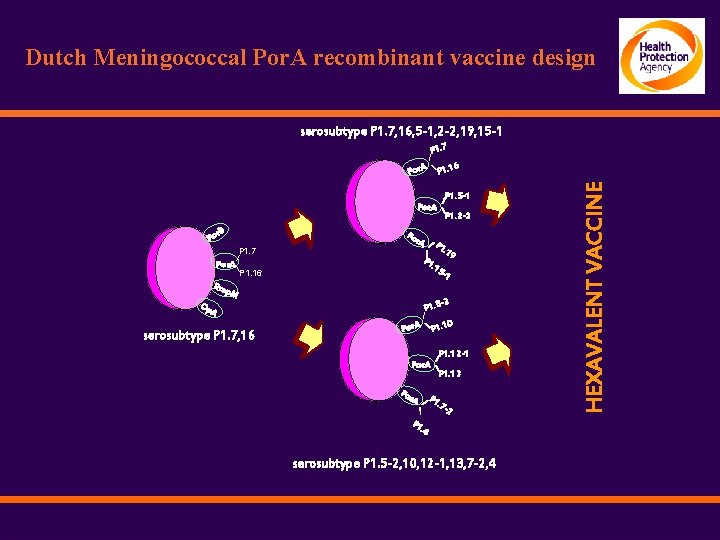
Dutch Meningococcal Por. A recombinant vaccine design serosubtype P 1. 7, 16, 5 -1, 2 -2, 19, 15 -1 /P 1. 5 -1 Por. A P 1. 2 -2 r. B Po P 1. 7 / Por. A P 1. 16 Rm p. M Op A serosubtype P 1. 7, 16 Po r. A / P 1. 1 9 P 1. 15 -1 -2 P 1. 5 / 0 Por. A P 1. 1 /P 1. 12 -1 Por. A P 1. 13 Po r. A / P 1. 72 P 1. 4 serosubtype P 1. 5 -2, 10, 12 -1, 13, 7 -2, 4 HEXAVALENT VACCINE P 1. 7 / 6 Por. A P 1. 1
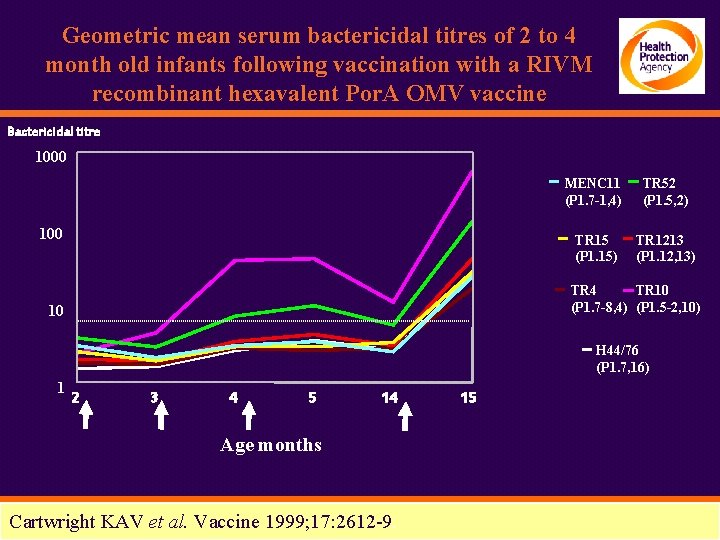
Geometric mean serum bactericidal titres of 2 to 4 month old infants following vaccination with a RIVM recombinant hexavalent Por. A OMV vaccine Bactericidal titre 1000 100 MENC 11 (P 1. 7 -1, 4) TR 52 (P 1. 5, 2) TR 15 (P 1. 15) TR 1213 (P 1. 12, 13) TR 4 TR 10 (P 1. 7 -8, 4) (P 1. 5 -2, 10) 10 H 44/76 (P 1. 7, 16) 1 2 3 4 5 14 Age months Cartwright KAV et al. Vaccine 1999; 17: 2612 -9. 15
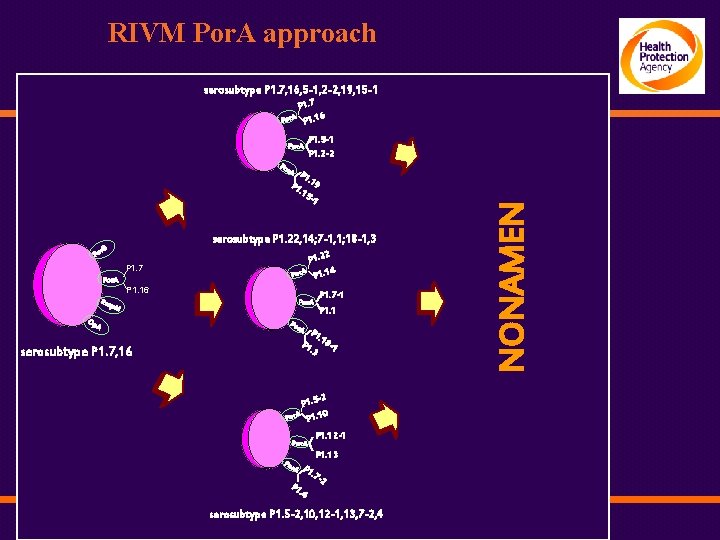
RIVM Por. A approach serosubtype P 1. 7, 16, 5 -1, 2 -2, 19, 15 -1 P 1. 7 / P 1. 16 Por. A P 1. 5 -1 P 1. 2 -2 Por. A / Po 1. P1 19. 15 -1 serosubtype P 1. 22, 14; 7 -1, 1; 18 -1, 3 r. B Po Por. A 2 P 1. 2 / Por. A P 1. 14 P 1. 7 / P 1. 16 Rm Por. A p. M Op A Po /P 1. 7 -1 P 1. 1 r. A /P serosubtype P 1. 7, 16 1. P 1 18 -1. 3 -2 P 1. 5 / Por. A P 1. 10 Por. A / Po P 1. 12 -1 P 1. 13 r. A P 1 / . 7 -2 P 1. 4 serosubtype P 1. 5 -2, 10, 12 -1, 13, 7 -2, 4 NONAMEN r. A P /
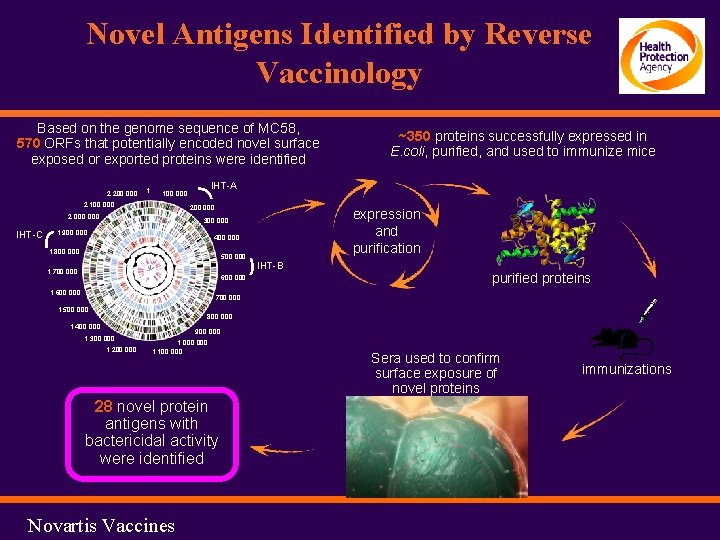
Novel Antigens Identified by Reverse Vaccinology Based on the genome sequence of MC 58, 570 ORFs that potentially encoded novel surface exposed or exported proteins were identified 2, 200, 000 1 2, 100, 000 IHT-C IHT-A 100, 000 2, 000 ~350 proteins successfully expressed in E. coli, purified, and used to immunize mice expression and purification 300, 000 1, 900, 000 400, 000 1, 800, 000 500, 000 1, 700, 000 600, 000 1, 600, 000 IHT-B purified proteins 700, 000 1, 500, 000 800, 000 1, 400, 000 1, 300, 000 1, 200, 000 900, 000 1, 100, 000 28 novel protein antigens with bactericidal activity were identified Novartis Vaccines Sera used to confirm surface exposure of novel proteins immunizations
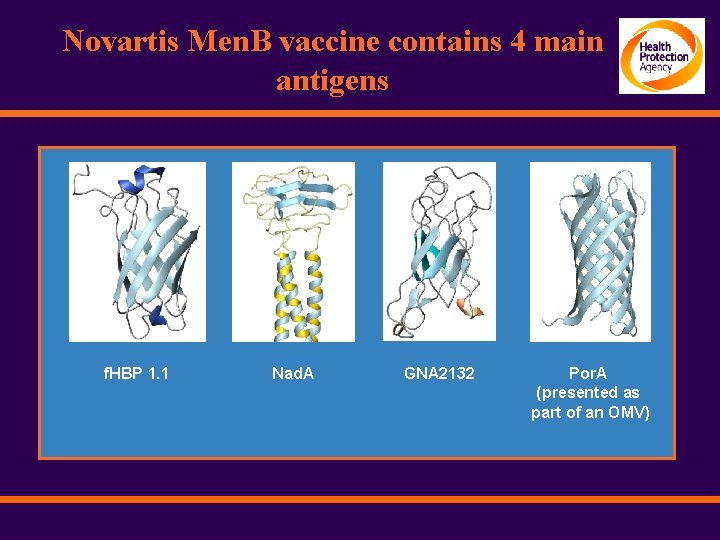
Novartis Men. B vaccine contains 4 main antigens f. HBP 1. 1 Nad. A GNA 2132 Por. A (presented as part of an OMV)
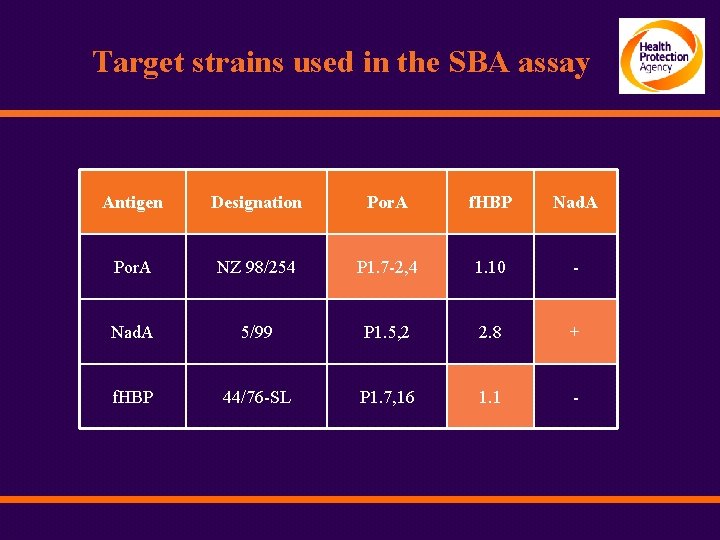
Target strains used in the SBA assay Antigen Designation Por. A f. HBP Nad. A Por. A NZ 98/254 P 1. 7 -2, 4 1. 10 - Nad. A 5/99 P 1. 5, 2 2. 8 + f. HBP 44/76 -SL P 1. 7, 16 1. 1 -

UK infant trial r. Men. B vaccine with or without OMV at 2, 4, 6 and 12 months of age. Control group received a single dose of r. Men. B at 12 months of age. Miller E, Pollard AJ, Borrow R, Findlow J, Dawson T, Morant A, John T, Snape M, Southern J, Morris R, Cartwright K, Oster P. 26 th Meeting of the European Society for paediatric Infectious diseases – ESPID, Graz, Austria, 14 th May 2008
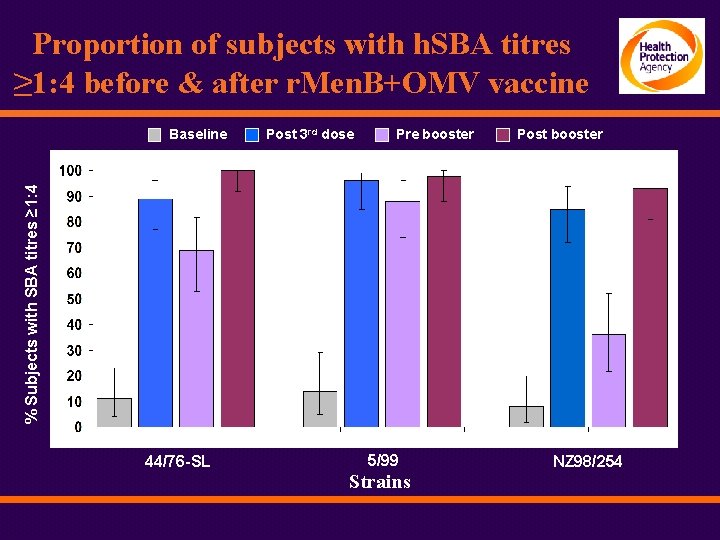
Proportion of subjects with h. SBA titres ≥ 1: 4 before & after r. Men. B+OMV vaccine Post 3 rd dose Pre booster Post booster % Subjects with SBA titres ≥ 1: 4 Baseline 44/76 -SL 5/99 Strains NZ 98/254
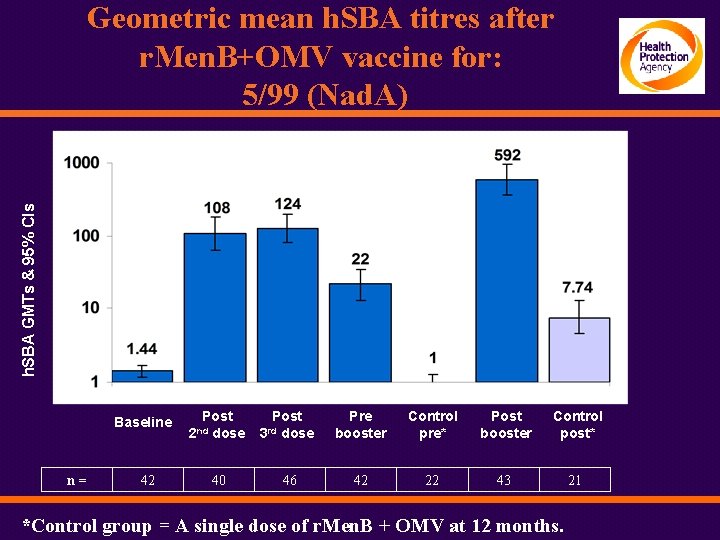
h. SBA GMTs & 95% CIs Geometric mean h. SBA titres after r. Men. B+OMV vaccine for: 5/99 (Nad. A) Baseline n= 42 Post dose 2 nd 40 Post dose Pre booster Control pre* Post booster Control post* 46 42 22 43 21 3 rd *Control group = A single dose of r. Men. B + OMV at 12 months.
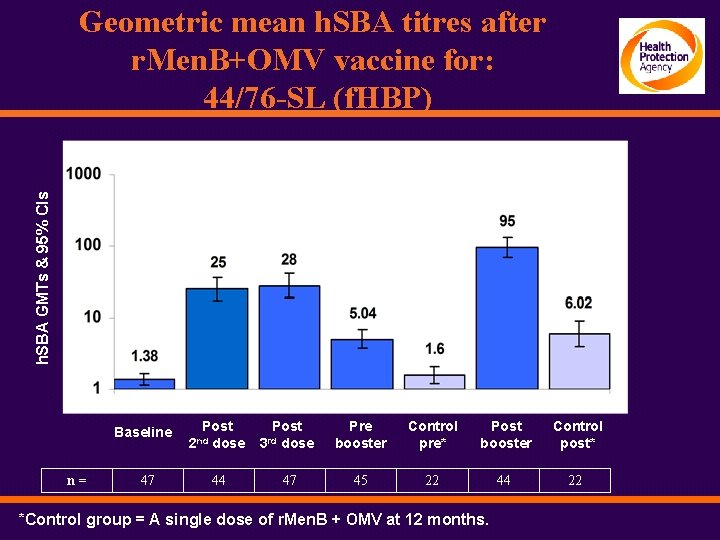
h. SBA GMTs & 95% CIs Geometric mean h. SBA titres after r. Men. B+OMV vaccine for: 44/76 -SL (f. HBP) Baseline n= 47 Post dose 2 nd 44 Post dose Pre booster Control pre* Post booster Control post* 47 45 22 44 22 3 rd *Control group = A single dose of r. Men. B + OMV at 12 months.
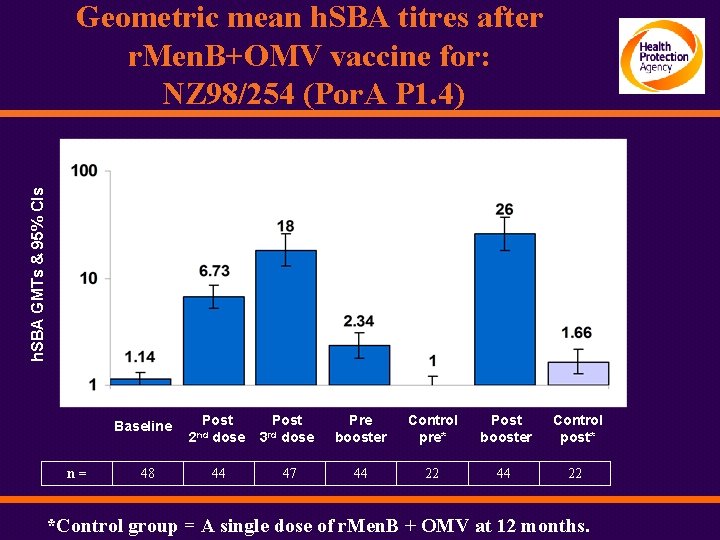
h. SBA GMTs & 95% CIs Geometric mean h. SBA titres after r. Men. B+OMV vaccine for: NZ 98/254 (Por. A P 1. 4) Baseline n= 48 Post dose 2 nd 44 Post dose Pre booster Control pre* Post booster Control post* 47 44 22 3 rd *Control group = A single dose of r. Men. B + OMV at 12 months.

Wyeth r. LP 2086 vaccine LP 2086 recombinantly expressed in E. coli and purified to homogeneity. Vaccine formulation developed for clinical studies contains two r. LP 2086 proteins- one from Subfamily A and one from Subfamily B. Induce bactericidal antibodies cross-reactive against all f. Hbp variants, depending on expression level. Phase I and Phase II (18 to 25 year olds, 8 to 14 year olds and 18 to 36 month olds).

Wyeth Men. B • Encouraging phase 1 trial results • Vaccine relatively well tolerated • Dose-dependent reactogenicity & SBA responses • % of responders approaching 100% for some strains, varied by definition of responder, and by strain tested • GMTs varied by strain tested, expression critical • Moving ahead with Phase 2 studies 16 th International Pathogenic Neisseria Conference, Sept 2008, Rotterdam, the Netherlands

Multivalent group B meningococcal vaccine based on outer membrane vesicles Walter Reed Army Institute of Research, Maryland, USA - Native OMVs - Each strain modified to express a different Por. A, a different core LOS and an increased expression of one conserved antigen (f. Hbp 1, f. Hbp 2, Nad. A, Opc). - Phase I completed, safe and induced SBA GSK - Recombinant OMV technology - Upregulate key protective antigens - Down regulate variable immunodominant antigens - Modify LOS
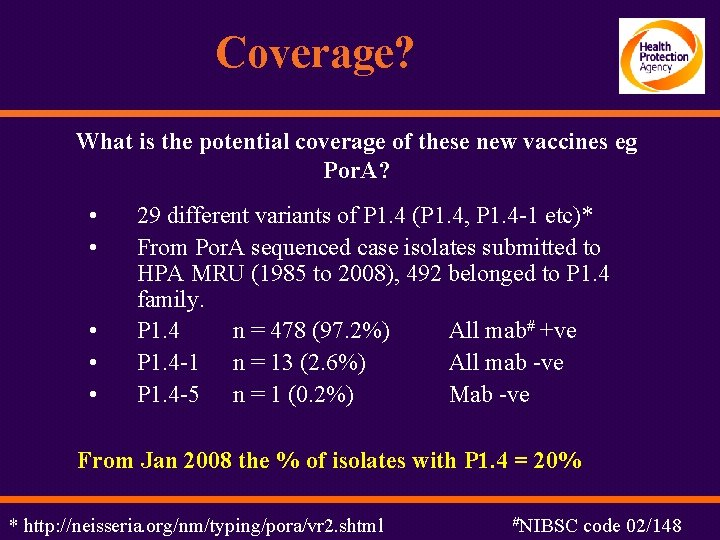
Coverage? What is the potential coverage of these new vaccines eg Por. A? • • • 29 different variants of P 1. 4 (P 1. 4, P 1. 4 -1 etc)* From Por. A sequenced case isolates submitted to HPA MRU (1985 to 2008), 492 belonged to P 1. 4 family. P 1. 4 n = 478 (97. 2%) All mab# +ve P 1. 4 -1 n = 13 (2. 6%) All mab -ve P 1. 4 -5 n = 1 (0. 2%) Mab -ve From Jan 2008 the % of isolates with P 1. 4 = 20% * http: //neisseria. org/nm/typing/pora/vr 2. shtml #NIBSC code 02/148
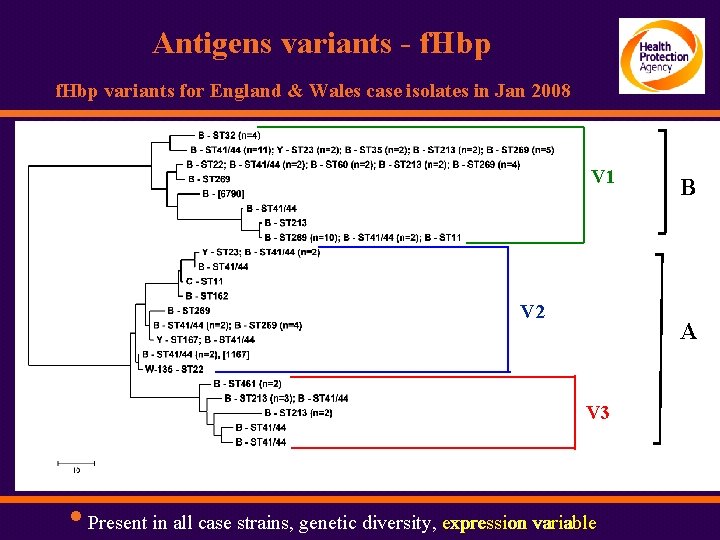
Antigens variants - f. Hbp variants for England & Wales case isolates in Jan 2008 V 1 V 2 }B B }A A V 3 • Present in all case strains, genetic diversity, expression variable
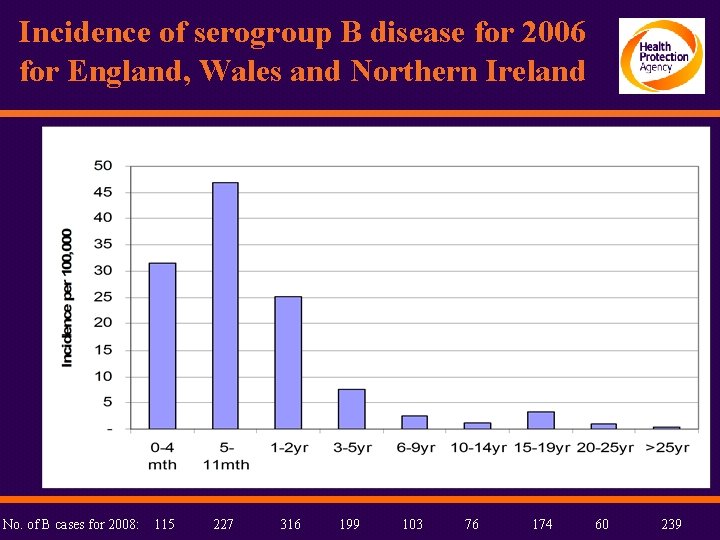
Incidence of serogroup B disease for 2006 for England, Wales and Northern Ireland No. of B cases for 2008: 115 227 316 199 103 76 174 60 239
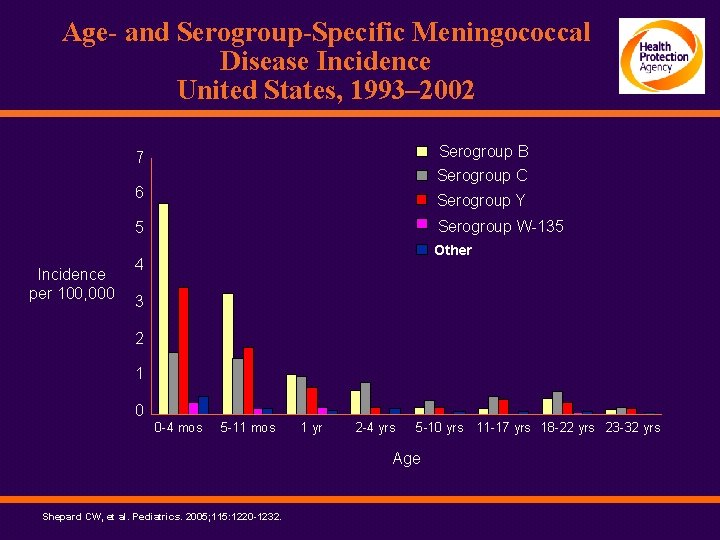
Age- and Serogroup-Specific Meningococcal Disease Incidence United States, 1993– 2002 Serogroup B Serogroup C 7 Incidence per 100, 000 6 Serogroup Y 5 Serogroup W-135 Other 4 3 2 1 0 0 -4 mos 5 -11 mos 1 yr 2 -4 yrs 5 -10 yrs Age Shepard CW, et al. Pediatrics. 2005; 115: 1220 -1232. 11 -17 yrs 18 -22 yrs 23 -32 yrs
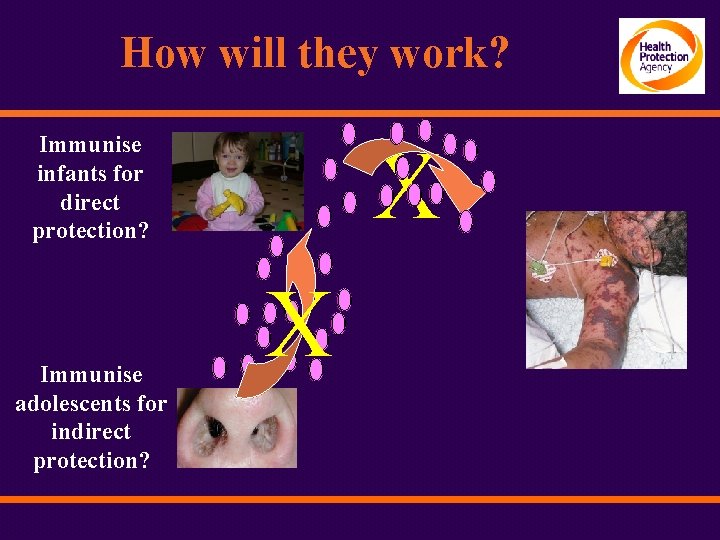
How will they work? Immunise infants for direct protection? Immunise adolescents for indirect protection? X X

Conclusions Ø Group B vaccine trials are underway after a hiatus of a decade. Ø Need to determine immunogenicity to predict effectiveness. Ø Need to determine potential coverage. Ø Which age groups should be vaccinated? Ø Are carriage studies required?
- Slides: 33