Membrane Transport and the Membrane Potential Cell membrane

Membrane Transport and the Membrane Potential

Cell membrane Extracellular fluid Intracellular fluid Composed primarily phospholipids Separates intracellular fluidoffrom extracellular and proteins fluid
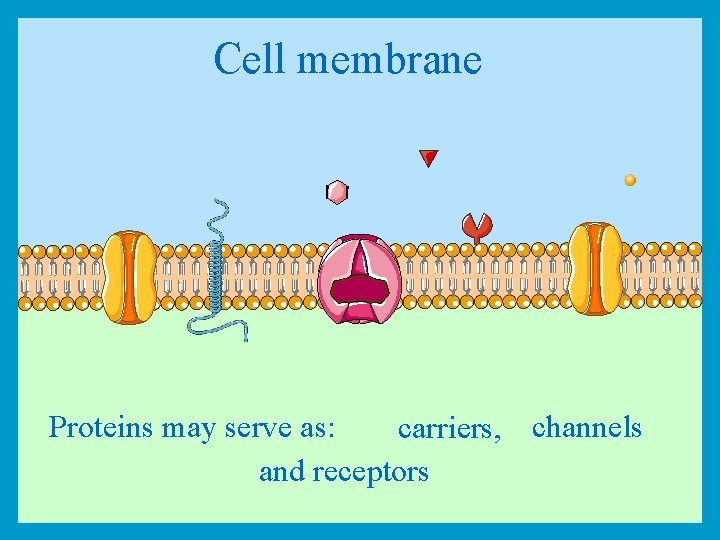
Cell membrane Proteins may serve as: carriers, channels and receptors

Mechanisms of Transport Across Cell Membrane • According to the means of transport there are two categories: 1 - Non-carrier-mediated transport - Simple diffusion Passive 2 - Carrier-mediated transport - Facilitated diffusion - Active transport Passive Active
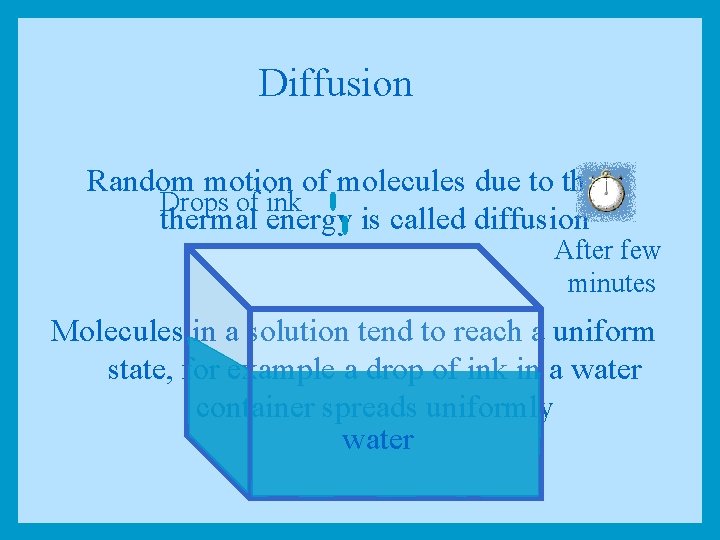
Diffusion Random motion of molecules due to their Drops of ink thermal energy is called diffusion After few minutes Molecules in a solution tend to reach a uniform state, for example a drop of ink in a water container spreads uniformly water

Equal Concentrations Higher Concentration Lower Concentration Netnetdiffusion No diffusion

Diffusion Through the Cell Membrane • Two major groups of molecules can pass the cell membrane by simple diffusion: 1. Molecules that can dissolve in the lipid bilayer membrane, non-polar molecules such as: • O 2, Hormones 2. Small polar molecules which are uncharged such as: • CO 2, alcohol, and urea
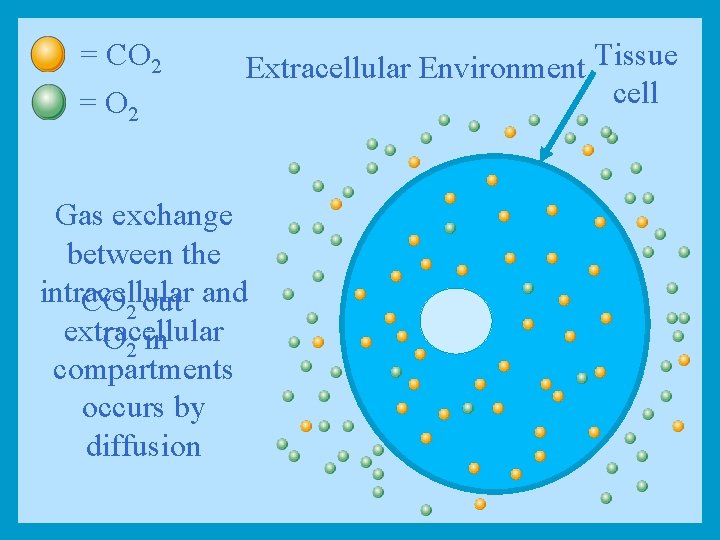
= CO 2 = O 2 Extracellular Environment Tissue cell Gas exchange between the intracellular CO 2 out and extracellular O 2 in compartments occurs by diffusion
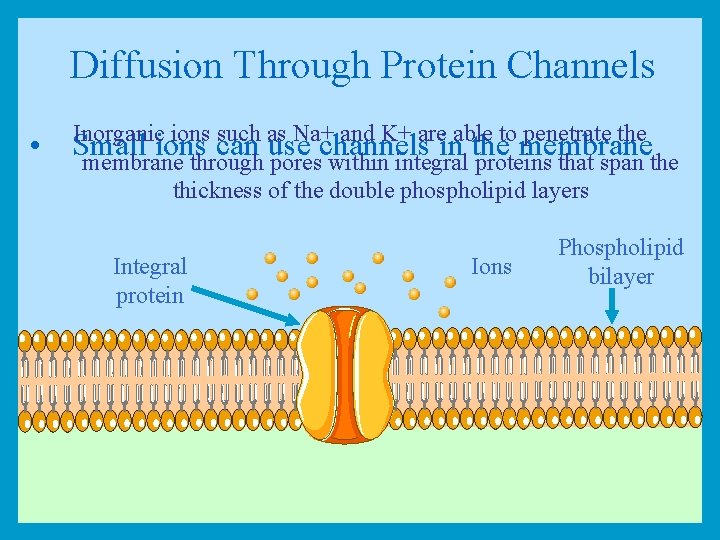
Diffusion Through Protein Channels Inorganic ions such as Na+ and K+ are able to penetrate the • Small ions can use channels in the membrane through pores within integral proteins that span the thickness of the double phospholipid layers Integral protein Ions Phospholipid bilayer

Rate of Diffusion • The number of diffusing molecules passing through the membrane per unit time

Rate of Diffusion • Rate of diffusion depends on: 1. 2. 3. 4. 5. 6. Concentration difference across the membrane Permeability of the membrane to the diffusing molecule Surface area of the membrane Molecular weight of the diffusion molecule Distance Temperature Rate of diffusion a Concentration gradient x Surface area x Temperature MW x distance

Osmosis • The net diffusion of water across the membrane is called osmosis • Osmosis can occur only if the membrane is semipermeable • Semipermeable means that the membrane must be more permeable to water than the solute dissolved in water

= Solute Osmosis More diluted = Water More concentrated


Osmotic Pressure • What is osmotic pressure? The force needed to prevent osmotic movement of water from one area to another across a semipermeable membrane.
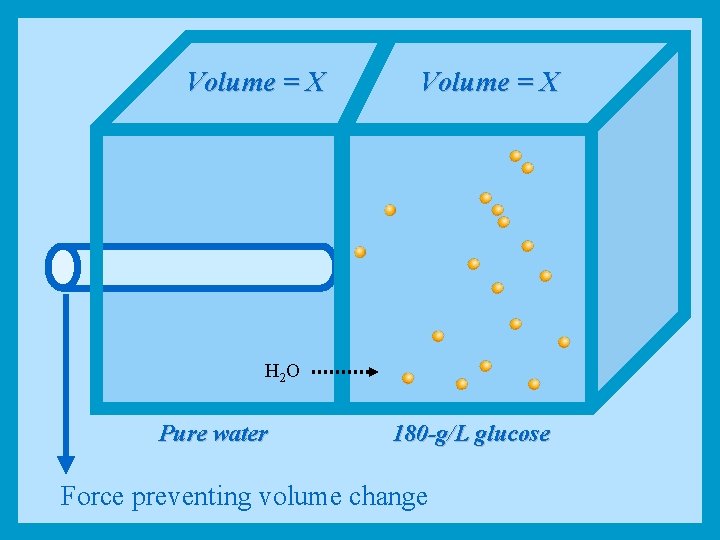
Volume = X H 2 O Pure water 180 -g/L glucose Force preventing volume change

Osmotic Pressure • • If a selectively permeable membrane separates pure water from a 180 -g/L glucose solution, water will tend to move by osmosis into the glucose solution, thus creating a hydrostatic pressure that will push the membrane to the left and expand the volume of the glucose solution The amount of pressure that must be applied to just counteract this volume change is equal to the osmosis pressure of the glucose solution

Molarity Equivalent of one molecular weight (g) of a substance dissolved in water to make a total one liter solution is called a Molar solution (1 M)
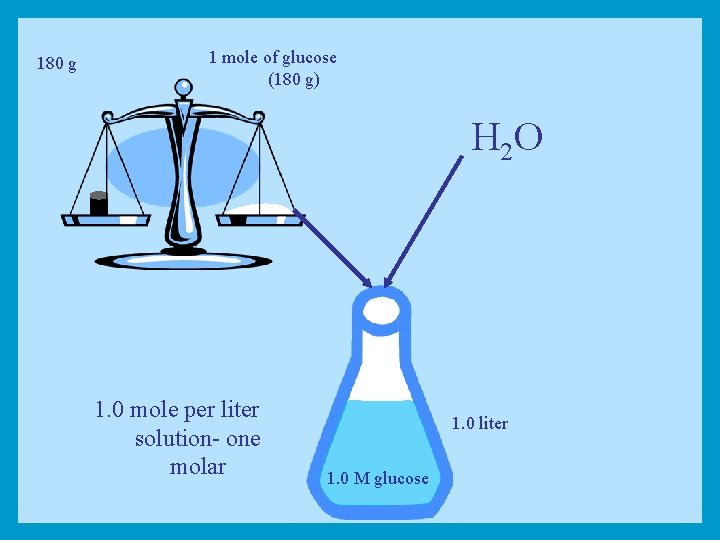
180 g 1 mole of glucose (180 g) H 2 O 1. 0 mole per liter solution- one molar 1. 0 liter 1. 0 M glucose

Molality When equivalent of one molecular weight (g) of a substance is added to one liter (Kg) of water, this solution is called Molal solution (1 m)
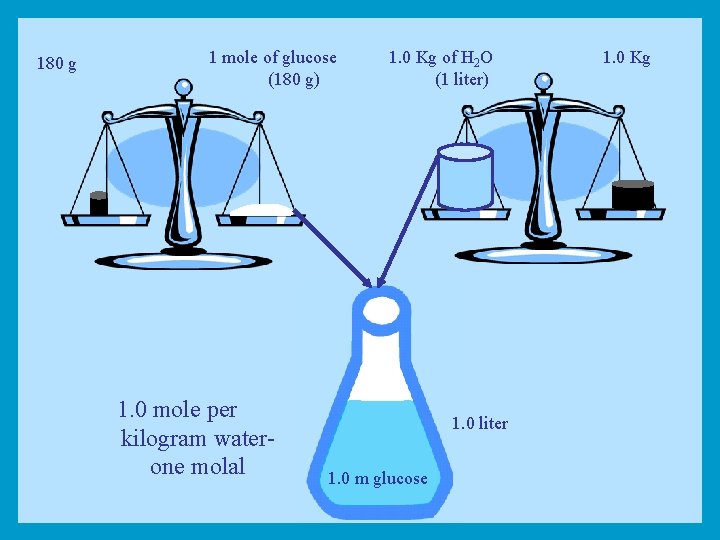
180 g 1 mole of glucose (180 g) 1. 0 mole per kilogram waterone molal 1. 0 Kg of H 2 O (1 liter) 1. 0 liter 1. 0 m glucose 1. 0 Kg

Molarity and Molality • Molal solution is a better indication of solute to solvent ratio, therefore it is a better indicator of osmosis • However, in the body since the difference between Molal and Molar concentration of solutes is very small, Molarity is often used

Osmolality • Total molality of substances in a solution is called osmolality (Osm). • e. g. A solution containing 1 m glucose and 1 m fructose has osmolality of 2 osmol/L (2 Osm). • Electrolytes such as Na. Cl are ionized when in solution, therefore one molecule of Na. Cl in solution yields tow ions. So 1 m of Na. Cl has osmolality of 2 Osm.

Tonicity • Solutions that have the same total concentration of osmotically active solutes and the same osmotic pressure as plasma* are said to be isotonic * In the body plasma has osmolarity of 0. 28 Osm (280 m. Osm)
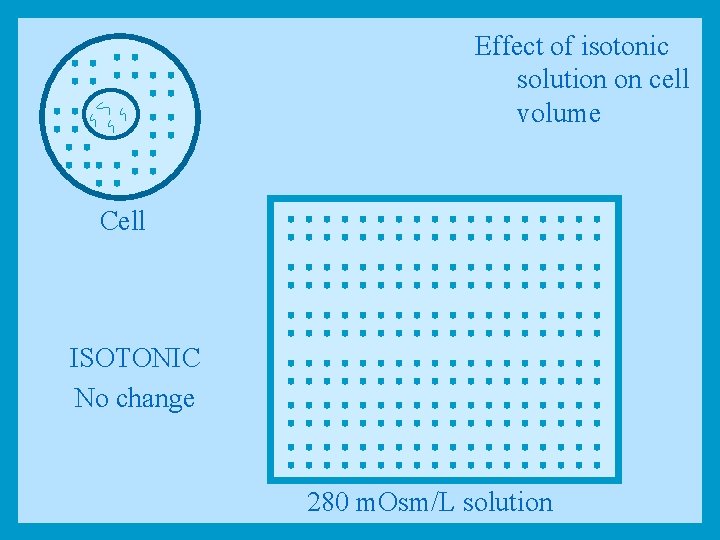
Effect of isotonic solution on cell volume Cell ISOTONIC No change 280 m. Osm/L solution

Tonicity • Solutions that have a lower total concentration of osmotically active solutes and a lower osmotic pressure than plasma are said to be hypotonic

Effect of hypotonic solution on cell volume Cell HYPOTONIC Cell swells 200 m. Osm/L solution

Tonicity • Solutions that have a higher total concentration of osmotically active solutes and a higher osmotic pressure than plasma are said to be hypertonic

Effect of hypertonic solution on cell volume Cell HYPERTONIC Cell shrinks 360 m. Osm/L solution
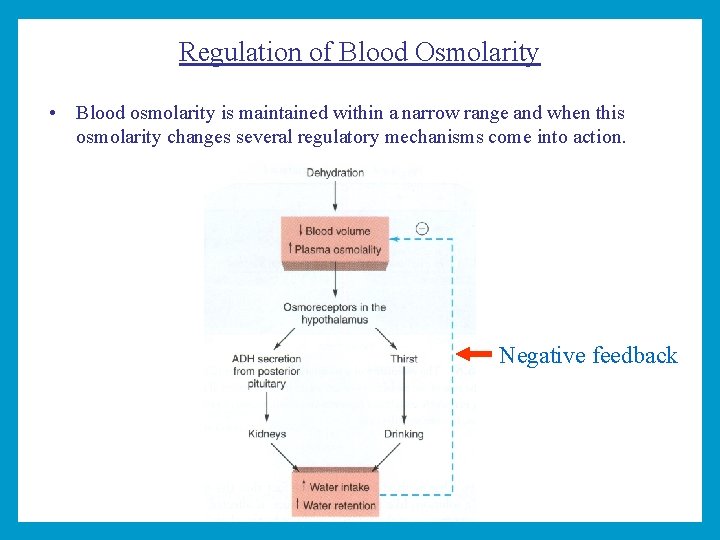
Regulation of Blood Osmolarity • Blood osmolarity is maintained within a narrow range and when this osmolarity changes several regulatory mechanisms come into action. Negative feedback
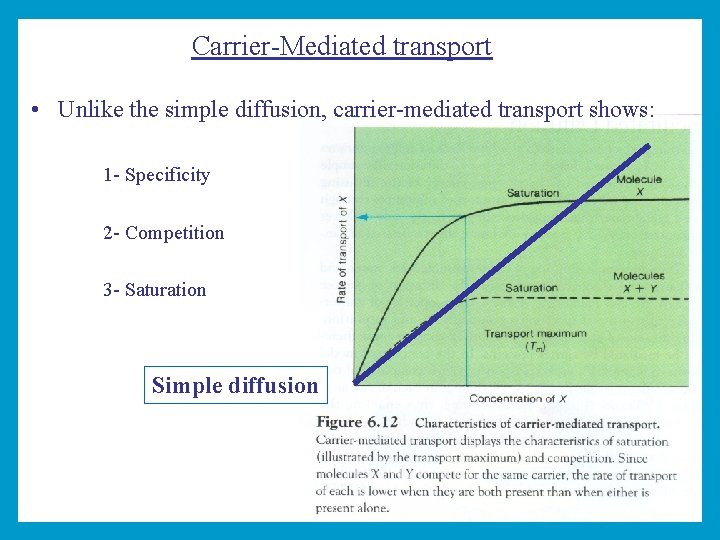
Carrier-Mediated transport • Unlike the simple diffusion, carrier-mediated transport shows: 1 - Specificity 2 - Competition 3 - Saturation Simple diffusion

Carrier-Mediated transport • There are two major types of carrier-mediated transport: a) Facilitated diffusion: like simple diffusion facilitated diffusion is powered by Passive thermal energy of the diffusing molecules. But the transport of molecules across the membrane is helped by a carrier protein. For example glucose is transported to the cells of the body by facilitated diffusion. the net transport is along the concentration gradient. Active b) Active transport: Movement of molecules against their concentration gradient which requires energy (ATP). For example movement of calcium from inside to outside of the cell.

Facilitated Diffusion
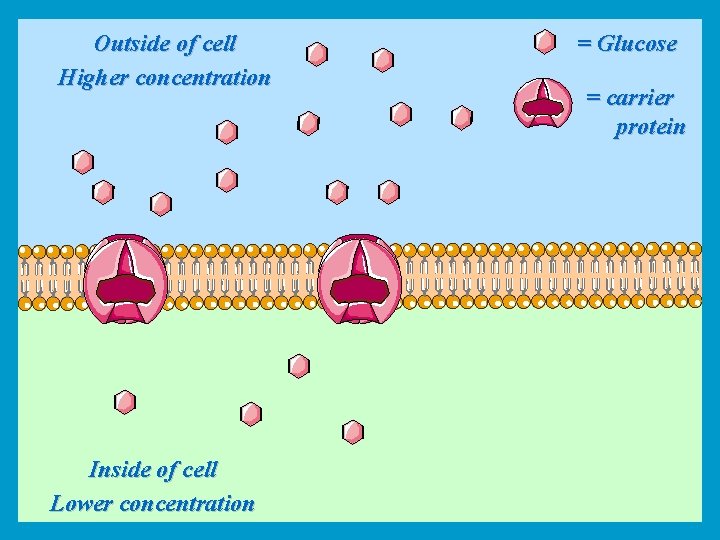
Outside of cell Higher concentration Inside of cell Lower concentration = Glucose = carrier protein

Active Transport a) Primary active transport: ATP is directly needed for the carrier protein in the following sequences: 1 - Binding of molecule to the carrier protein 2 - ATP is hydrolysed to provide energy for transport. 3 - Carrier changes its shape and moves the molecule across the membrane.

a) Primary active transport: b) ++ c)e. g Transport of Ca from inside to outside of the cell
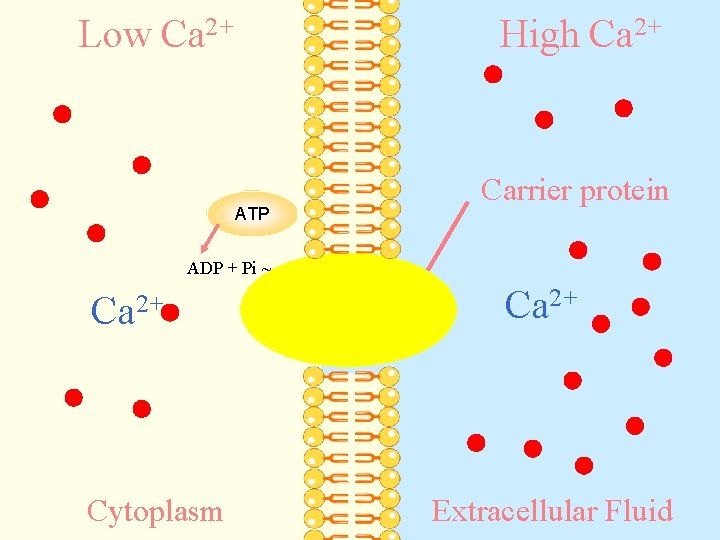
Low Ca 2+ High Ca 2+ ATP Carrier protein ADP + Pi ~ Ca 2+ Cytoplasm Ca 2+ Extracellular Fluid

a) Primary active transport: b) e. g Na/K pump.
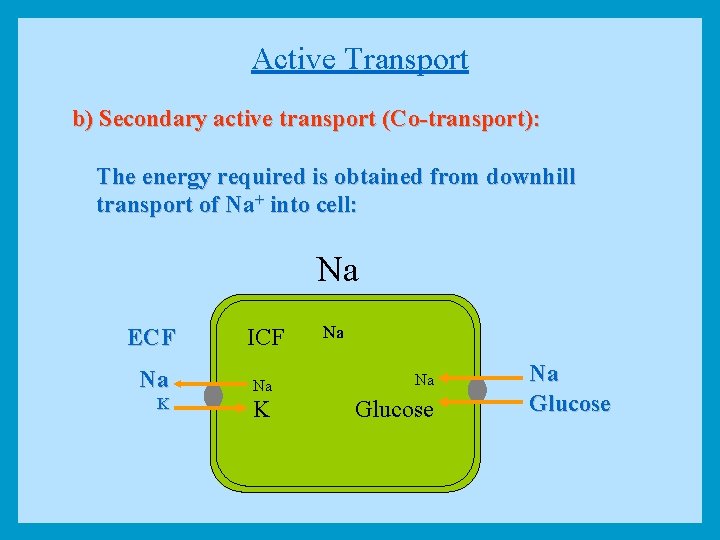
Active Transport b) Secondary active transport (Co-transport): The energy required is obtained from downhill transport of Na+ into cell: Na Na ECF ICF Na Na Na K K Glucose Na Glucose
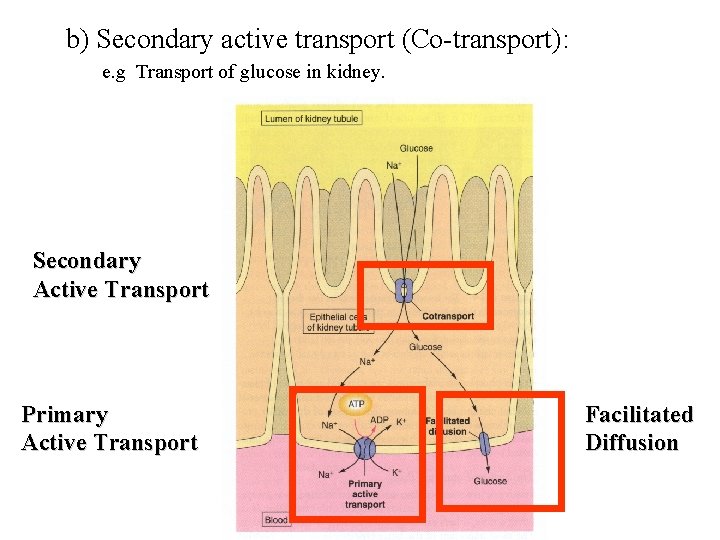
b) Secondary active transport (Co-transport): e. g Transport of glucose in kidney. Secondary Active Transport Primary Active Transport Facilitated Diffusion

b) Secondary active transport (Co-transport): e. g Co-transport of Na+ and glucose.

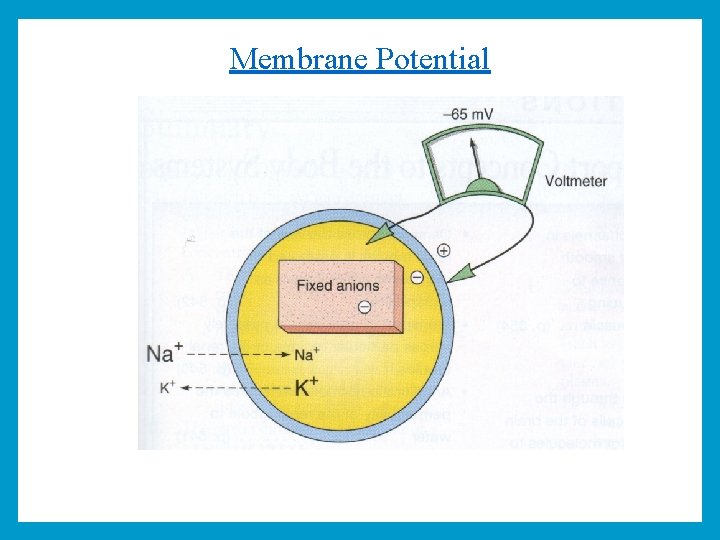
Membrane Potential The difference in ionic distribution between inside and outside of the cell result in electrical potential difference across the cell membrane which is called membrane potential. Membrane potential is produced by: 1 - The action of Na/K pump at the cell membrane is essential for the production of membrane potential. 2 - Proteins, ATP and other organic molecules in the cell are negatively charged, and can not cross the cell membrane therefore this makes inside of the cell negative.
Membrane Potential
- Slides: 43