Membrane Transport About Cell Membranes 1 All cells
Membrane Transport

About Cell Membranes 1. All cells have a cell membrane 2. Functions: a. Controls what enters and exits the cell to maintain an internal balance called homeostasis b. Provides protection and support for the cell TEM picture of a real cell membrane.

About Cell Membranes (continued) 3. Structure of cell membrane Lipid Bilayer -2 layers of phospholipids a. Phosphate head is polar (water loving) b. Fatty acid tails non-polar (water fearing) c. Proteins embedded in membrane Phospholipid Lipid Bilayer
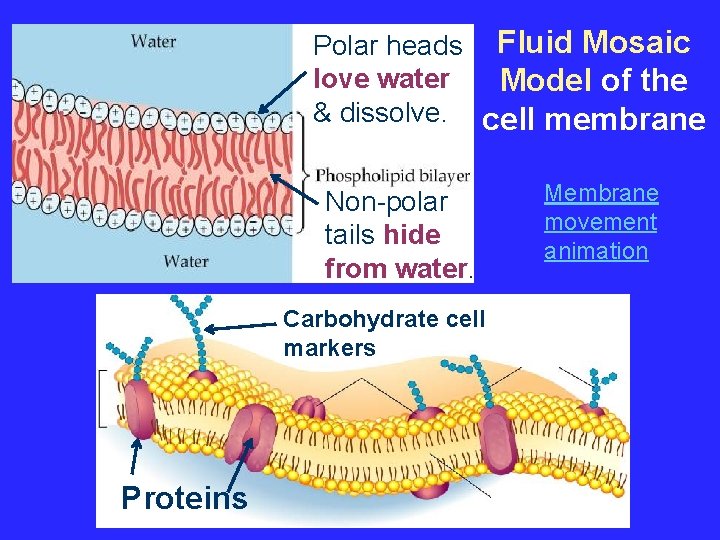
Polar heads Fluid Mosaic love water Model of the & dissolve. cell membrane Non-polar tails hide from water. Carbohydrate cell markers Proteins Membrane movement animation
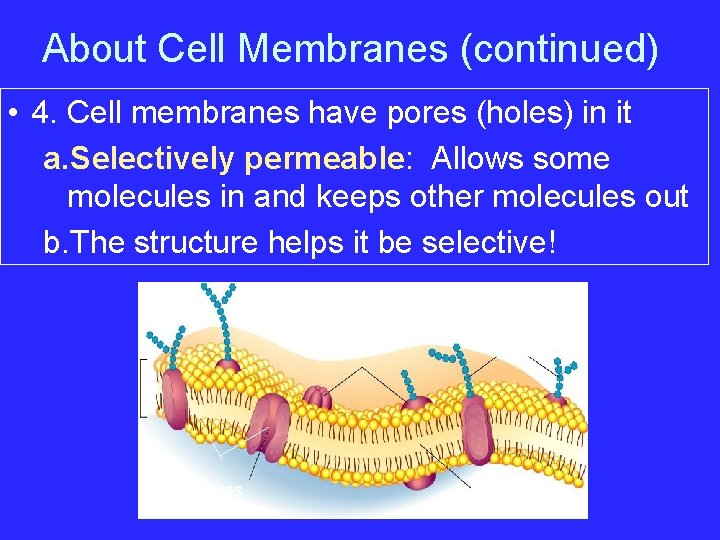
About Cell Membranes (continued) • 4. Cell membranes have pores (holes) in it a. Selectively permeable: Allows some molecules in and keeps other molecules out b. The structure helps it be selective! Pores
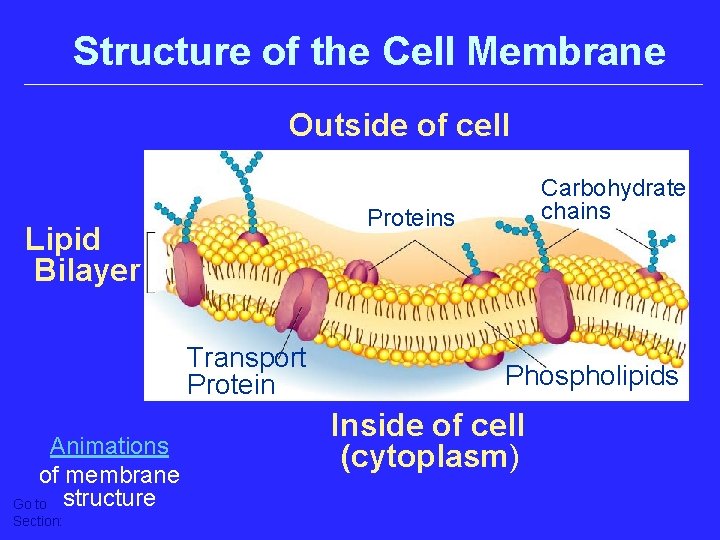
Structure of the Cell Membrane Outside of cell Proteins Lipid Bilayer Transport Protein Animations of membrane Go to structure Section: Carbohydrate chains Phospholipids Inside of cell (cytoplasm)
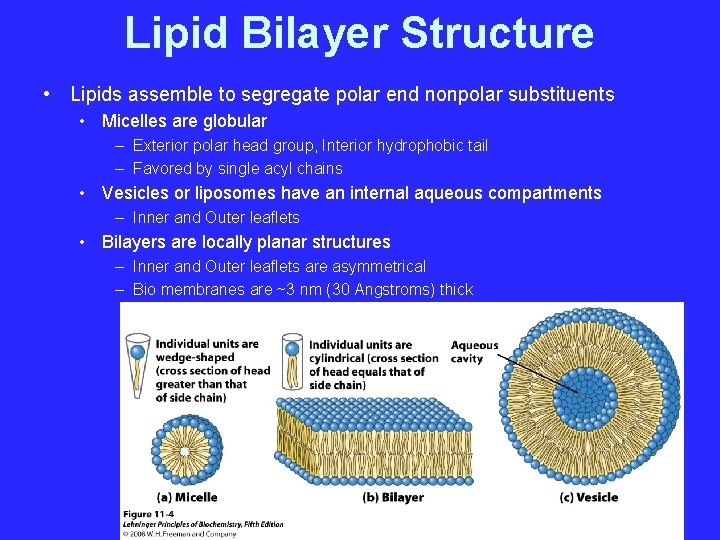
Lipid Bilayer Structure • Lipids assemble to segregate polar end nonpolar substituents • Micelles are globular – Exterior polar head group, Interior hydrophobic tail – Favored by single acyl chains • Vesicles or liposomes have an internal aqueous compartments – Inner and Outer leaflets • Bilayers are locally planar structures – Inner and Outer leaflets are asymmetrical – Bio membranes are ~3 nm (30 Angstroms) thick
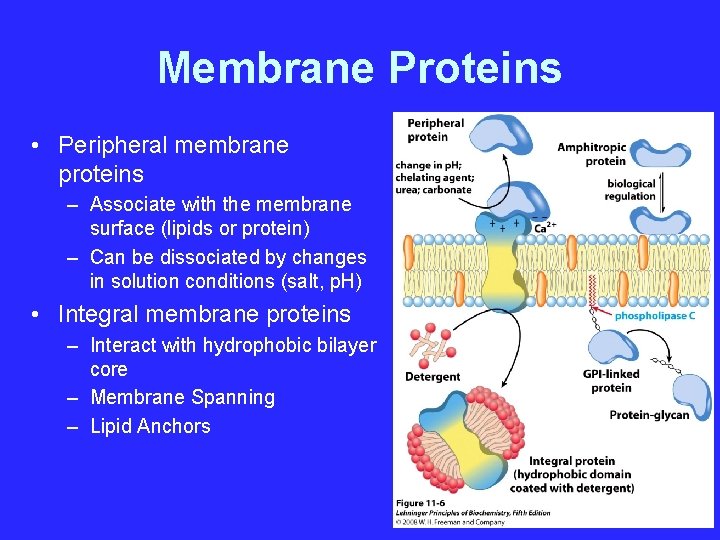
Membrane Proteins • Peripheral membrane proteins – Associate with the membrane surface (lipids or protein) – Can be dissociated by changes in solution conditions (salt, p. H) • Integral membrane proteins – Interact with hydrophobic bilayer core – Membrane Spanning – Lipid Anchors
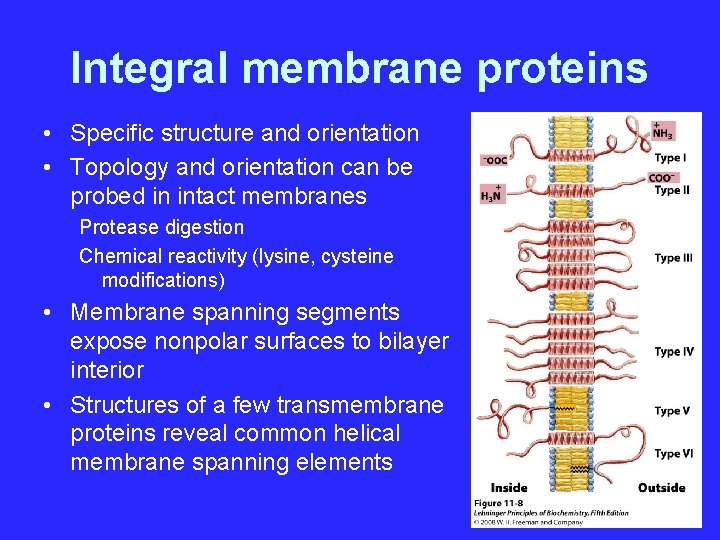
Integral membrane proteins • Specific structure and orientation • Topology and orientation can be probed in intact membranes Protease digestion Chemical reactivity (lysine, cysteine modifications) • Membrane spanning segments expose nonpolar surfaces to bilayer interior • Structures of a few transmembrane proteins reveal common helical membrane spanning elements

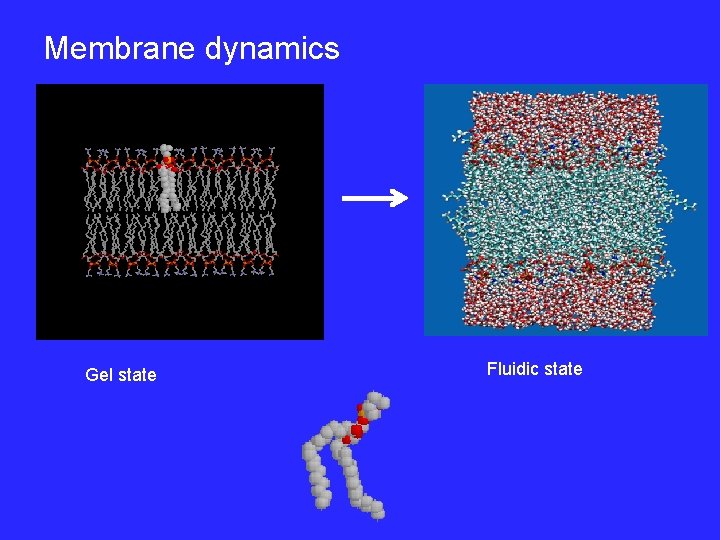
Membrane dynamics Gel state Fluidic state
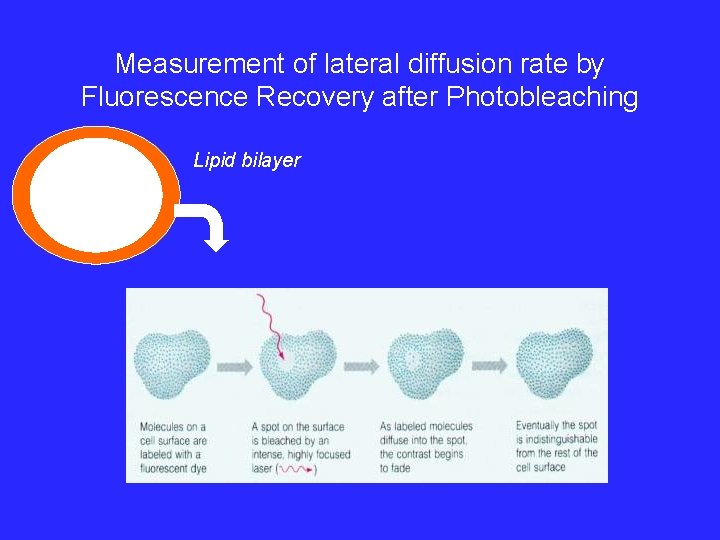
Measurement of lateral diffusion rate by Fluorescence Recovery after Photobleaching Lipid bilayer
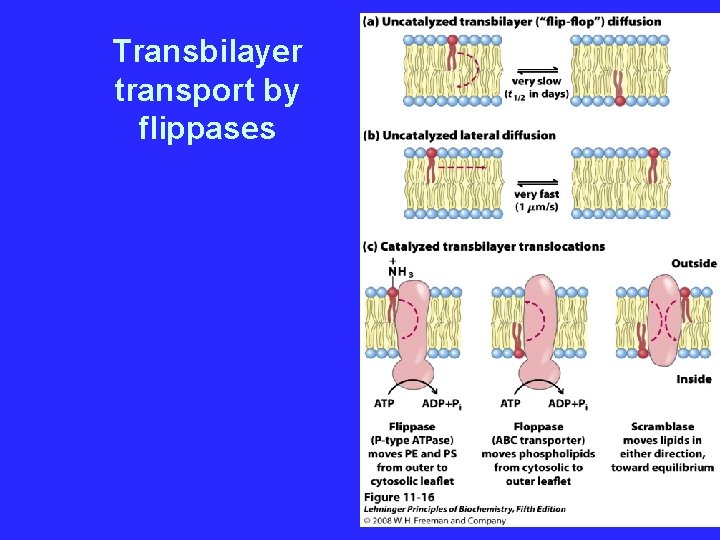
Transbilayer transport by flippases
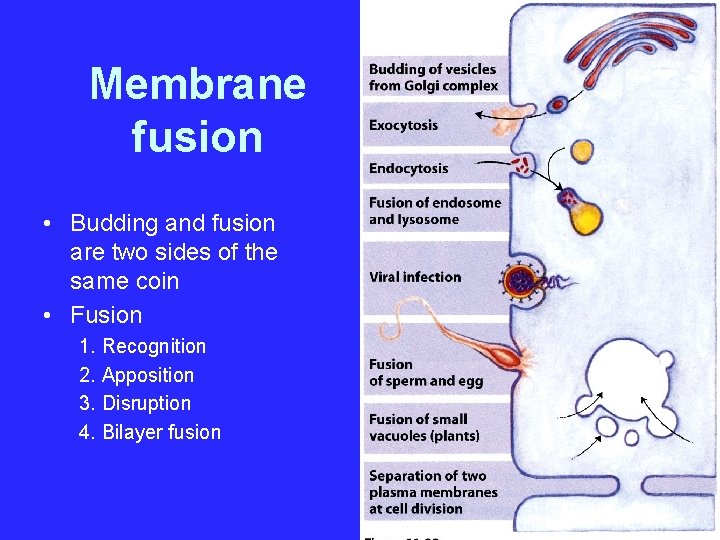
Membrane fusion • Budding and fusion are two sides of the same coin • Fusion 1. Recognition 2. Apposition 3. Disruption 4. Bilayer fusion
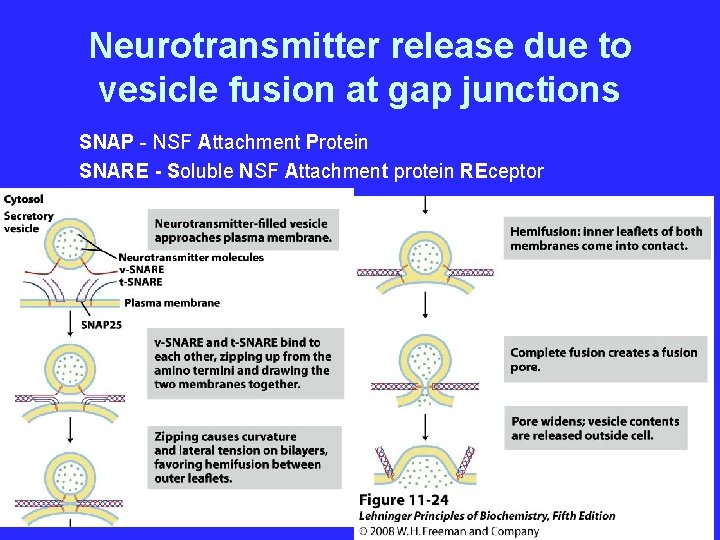
Neurotransmitter release due to vesicle fusion at gap junctions SNAP - NSF Attachment Protein SNARE - Soluble NSF Attachment protein REceptor

Cellular Membrane Transport
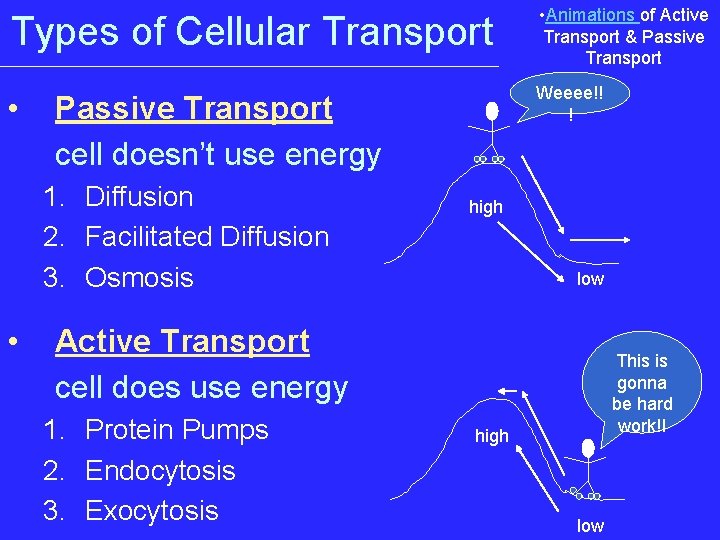
Types of Cellular Transport • Animations of Active Transport & Passive Transport • Weeee!! ! Passive Transport cell doesn’t use energy 1. Diffusion 2. Facilitated Diffusion 3. Osmosis • high low Active Transport cell does use energy 1. Protein Pumps 2. Endocytosis 3. Exocytosis This is gonna be hard work!! high low

Passive Transport • • • cell uses no energy molecules move randomly Molecules spread out from an area of high concentration to an area of low concentration. • (High Low) • Three types:

3 Types of Passive Transport 1. Diffusion 2. Facilitative Diffusion – diffusion with the help of transport proteins 3. Osmosis – diffusion of water

Passive Transport: 1. Diffusion Simple Diffusion Animation 1. Diffusion: random movement of particles from an area of high concentration to an area of low concentration. (High to Low) • Diffusion continues until all molecules are evenly spaced (equilibrium is reached)-Note: molecules will still move around but stay spread out. http: //bio. winona. edu/berg/Free. htm
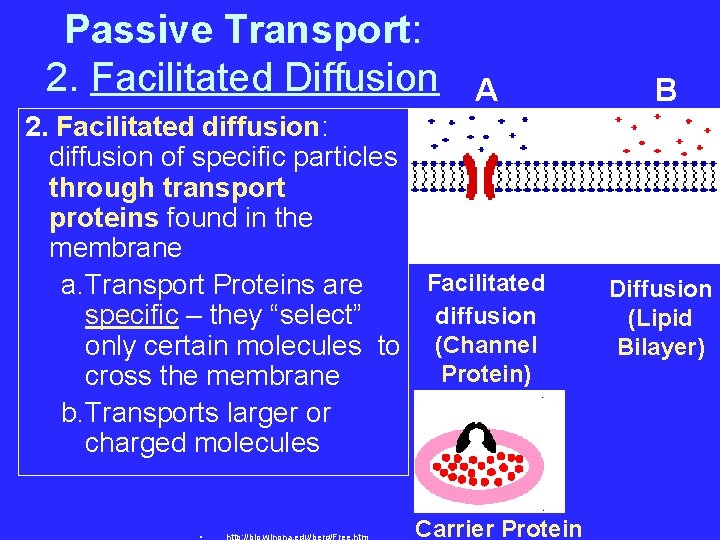
Passive Transport: 2. Facilitated Diffusion A 2. Facilitated diffusion: diffusion of specific particles through transport proteins found in the membrane Facilitated a. Transport Proteins are diffusion specific – they “select” only certain molecules to (Channel Protein) cross the membrane b. Transports larger or charged molecules Carrier Protein B Diffusion (Lipid Bilayer)
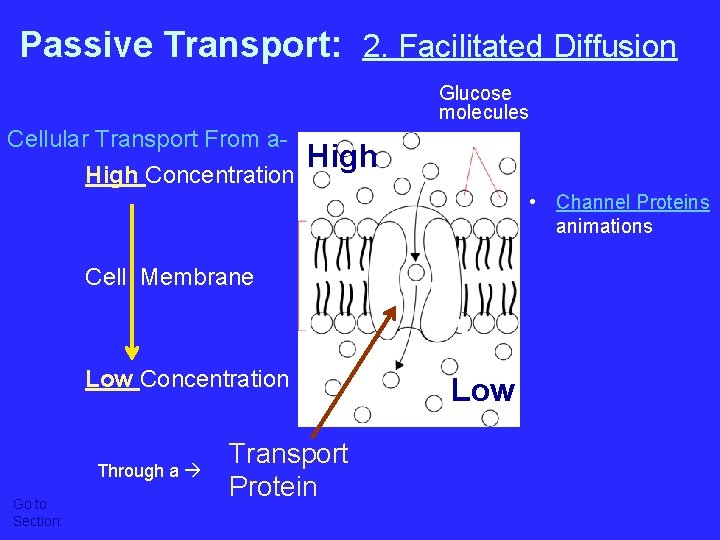
Passive Transport: 2. Facilitated Diffusion Glucose molecules Cellular Transport From a. High Concentration High • Channel Proteins animations Cell Membrane Low Concentration Through a Go to Section: Transport Protein channel Low
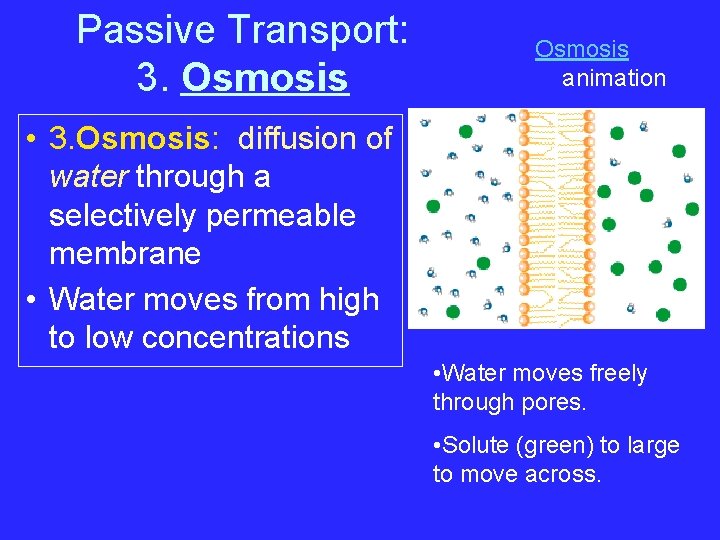
Passive Transport: 3. Osmosis animation • 3. Osmosis: diffusion of water through a selectively permeable membrane • Water moves from high to low concentrations • Water moves freely through pores. • Solute (green) to large to move across.

Active Transport • cell uses energy • actively moves molecules to where they are needed • Movement from an area of low concentration to an area of high concentration • (Low High) • Three Types:

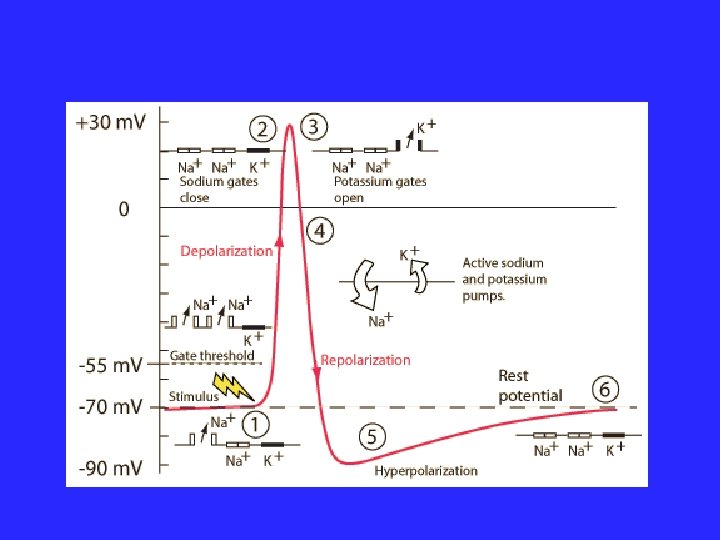
Types of Active Transport 1. Protein Pumps transport proteins that require energy to do work • Example: Sodium / Potassium Pumps are important in nerve responses. Sodium Potassium Pumps (Active Transport using proteins) Protein changes shape to move molecules: this requires energy!
Types of Active Transport • 2. Endocytosis: taking bulky material into a cell • Uses energy • Cell membrane in-folds around food particle • “cell eating” • forms food vacuole & digests food • This is how white blood cells eat bacteria!
Endocytosis

Types of Active Transport 3. Exocytosis: Forces material out of cell in bulk • membrane surrounding the material fuses with cell membrane • Cell changes shape – requires energy • EX: Hormones or wastes released from cell Endocytosis & Exocytosis animations

Exocytosis

Osmosis • Osmosis- diffusion of water through a selectively permeable membrane • Water is so small and there is so much of it the cell can’t control it’s movement through the cell membrane.
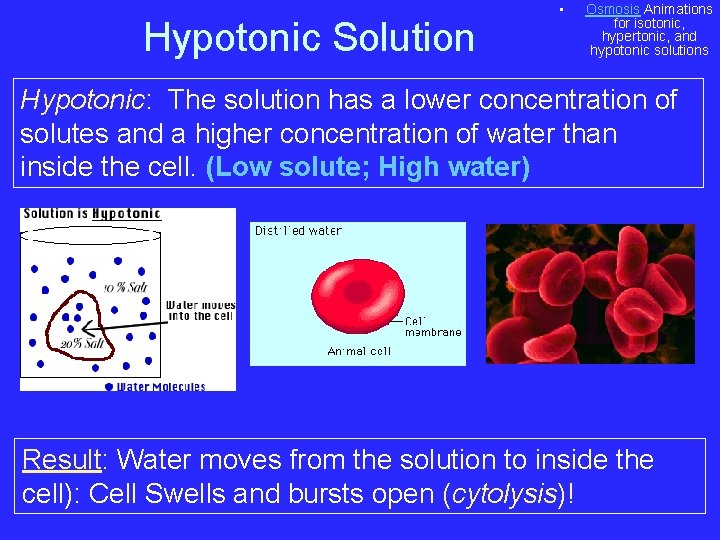
Hypotonic Solution • Osmosis Animations for isotonic, hypertonic, and hypotonic solutions Hypotonic: The solution has a lower concentration of solutes and a higher concentration of water than inside the cell. (Low solute; High water) Result: Water moves from the solution to inside the cell): Cell Swells and bursts open (cytolysis)!
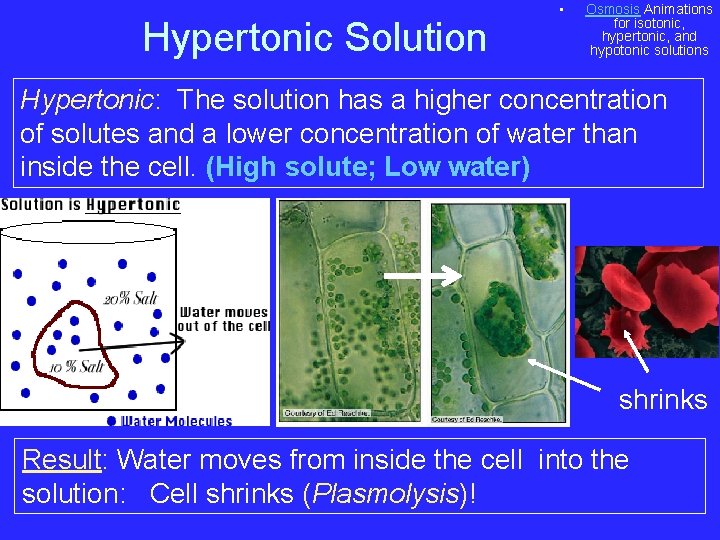
Hypertonic Solution • Osmosis Animations for isotonic, hypertonic, and hypotonic solutions Hypertonic: The solution has a higher concentration of solutes and a lower concentration of water than inside the cell. (High solute; Low water) shrinks Result: Water moves from inside the cell into the solution: Cell shrinks (Plasmolysis)!

• Isotonic Solution Osmosis Animations for isotonic, hypertonic, and hypotonic solutions Isotonic: The concentration of solutes in the solution is equal to the concentration of solutes inside the cell. Result: Water moves equally in both directions and the cell remains same size! (Dynamic Equilibrium)
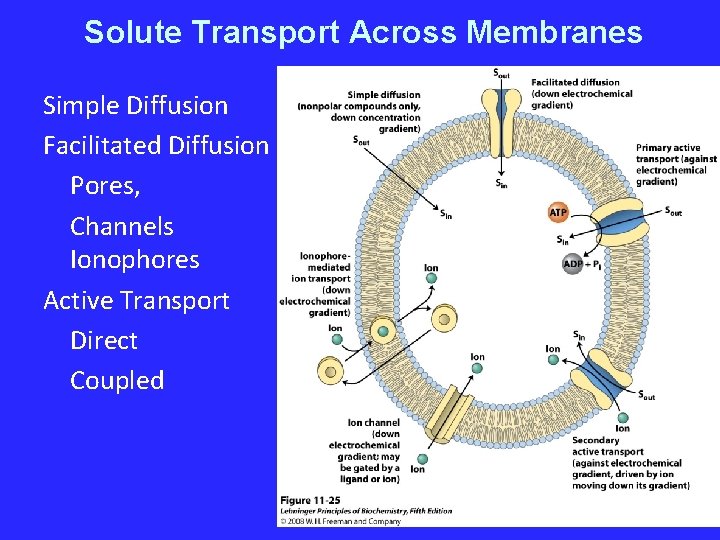
Solute Transport Across Membranes Simple Diffusion Facilitated Diffusion Pores, Channels Ionophores Active Transport Direct Coupled
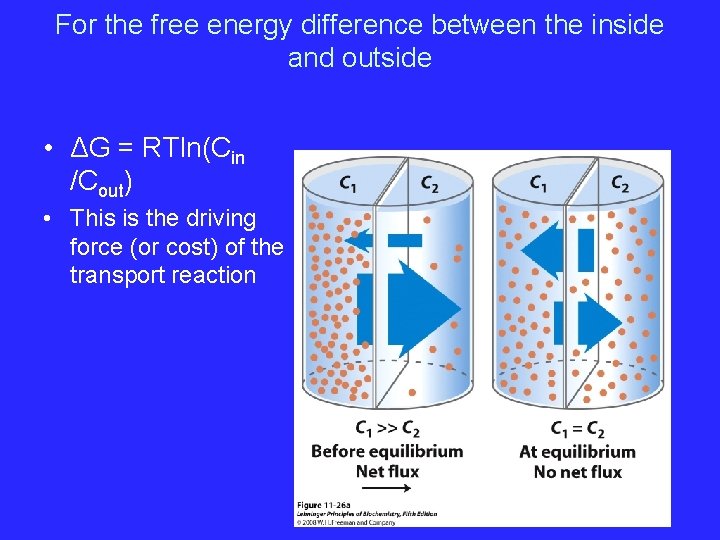
For the free energy difference between the inside and outside • ΔG = RTln(Cin /Cout) • This is the driving force (or cost) of the transport reaction

Energetics of Transport Electrical Potential • The energetic driving force (or cost) of transporting a charged object across an electric field ΔGel = Z F ΔΦ ΔΦ is the electrical potential in volts Z is the charge on the ion F is the Faraday constant 96, 480 J/(V mole) For Electro-chemical gradient, the Free energy change is: ΔG = RTln(Cinn/Cout) + ZF ΔΦ

Nernst Equation: It describes the flux of the ion under combined influence of concentration gradient and the electric gradient field Jconc. grad Jtotal = Jconc. grad + Jelec. grad Jtotal = d. C - Dion ─ d. X Z F Dion - C dΔФ d. X RT At equilibrium Jtotal = 0 Jconc. grad = ΔФ(volt) = Фin - Фout = Jelec. grad - RT ZF ln d. C = - Dion ─ d. X Cin Cout Z F Dion Jelec. . grad = - RT C dΔФ d. X D - diffusion coefficent of ion C - conc. of ion Ф - electrical gradient(membrane potential) X- thickness of membrane Z-valency of ion F-Faraday constant R-gas constant T-temp.

Nernst Equation : Equilibrium ionic potential of individual ion( Eion), when ion concentration gradient is equivalent to electrical gradient • • • Eion = 2. 303 RT/z. F log [ion]o/[ion]in Eion = ionic equilibrium potential Z= charge of ion F= Faraday’s constant T= absolute temperature (0 Kelvin/-273°C) R= gas constant • ENa = 61. 54 m. V log [Na]o/[Na]I = 62 m. V • EK = 61. 54 m. V log [K]o/[K]I = -80 m. V • ECa = 30. 77 m. V log [Ca]o/[Ca]I = 123 m. V • CCl = -61. 54 m. V log [Cl]o/[Cl]I = - 65 m. V Ion Inside (m. M) Outside (m. M) K+ 125 5 Na+ 12 120 Cl- 5 125

Goldman equation ►The membrane potential experimentally measured is often different than the Nernst potential for any given cell, due to the existence of other ions. ►The Goldman equation (also called the Goldman-Hodgkin-Katz equation)which quantitatively describes the relationship between membrane potential and permeable ions. ►According to the Goldman equation, the membrane potential is a compromise between various equilibrium potentials, each dependent on the membrane permeability and absolute ion concentration. For multiple ions, the ion flux through the membrane at the resting state is zero, consider the three major ions involved in the nerve cell stimulation, Na+, K+, and Cl− ions. where PNa, PK, and PCl are the relative membrane permeabilities (D/L) of Na+, K+, and Cl- ions, respectively. [D-diffusion coefficient of ion, L-thickness of ion channel] PNa, PK, and PCl = 1: 0. 1: 1 Goldman prediction of membrane potential = – 84 m. V (almost equivalent to equilibrium ionic potential of K+ )

Problems: 1. Assume: a concentration of glucose inside the cell of 0. 5 millimolar and a concentration of glucose outside the cell of 5 millimolar (m. M) at body temperature of 37°C, so an absolute temperature of 37 + 273 = 310°K, and the plasma membrane is permeable to glucose. Calculate the amount of free energy released. ΔG = (2)(273+37) x ln (0. 5/5) = (2)(310) x ln (0. 1) = (620)(− 2. 3) = − 1426 cal/mole = − 1. 4 kcal/mole 2. What is the resting ΔΦ for Na+ ions, if the outside concentration is 140 m. M and the inside concentration is 50 m. M?

3. How many sodium ions are needed to provide the free energy to transport a molecule of glutamic acid from a concentration of 0. 1 m. M outside the cell to 20 m. M inside the cell? Again, assume a temperature of 37°C (310°K) at p. H 7. 0. The resting membrane potential is -70 m. V. Note: One Na+ ion releases 3. 3 kcal/mole free energy for transport across the membrane. Solution At p. H of ~7, glutamic acid molecules carry a net charge of minus 1. So, once again, we have a problem of determining the movement of a molecule against an electrochemical gradient; that is, against both a concentration gradient (20/0. 1 = 200) and a electrostatic gradient (moving a negative charge against a voltage of − 70 m. V). ΔG = (R)(T) x ln(20/0. 1) + (z)(F)(Vm) = [(2)(310) x ln(200)] + [(− 1)(23, 062)(− 0. 070) = (620) x (5. 3) + 1614 = 3286 + 1614 = 4900 or 4. 9 kcal/mole Because sodium ions release only 3. 3 kcal/mole, at least 2 Na+ are needed to cotransport one molecule of glutamic acid.

Transport superfamilies • Carriers – – Bind specific ligands Catalyze transport across the membrane In a sense they are enzymes Transport is saturable • Channels – Less specific (often size specific) – Can be fluid filled – Transport may not be easily saturable
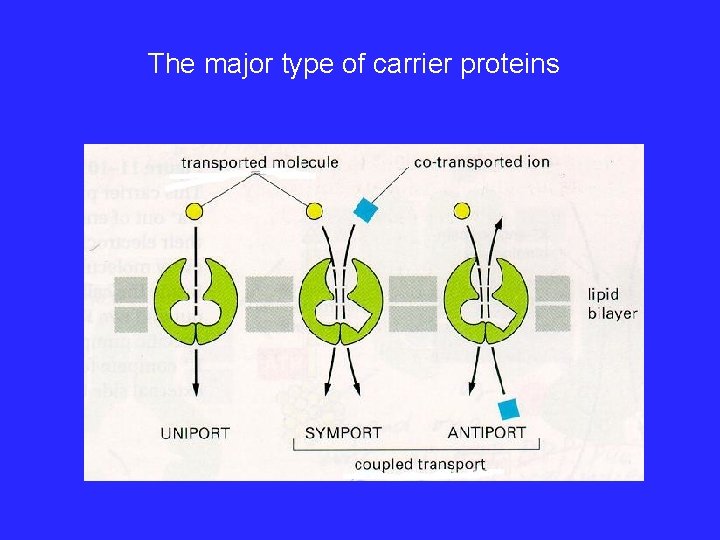
The major type of carrier proteins
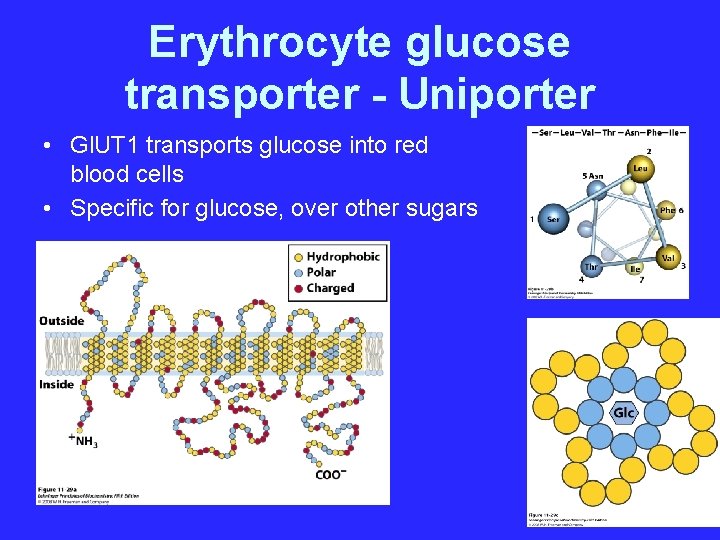
Erythrocyte glucose transporter - Uniporter • Gl. UT 1 transports glucose into red blood cells • Specific for glucose, over other sugars
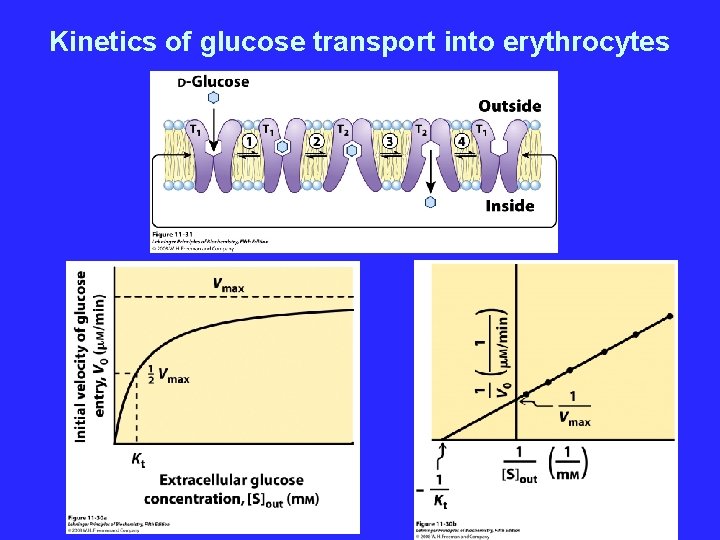
Kinetics of glucose transport into erythrocytes
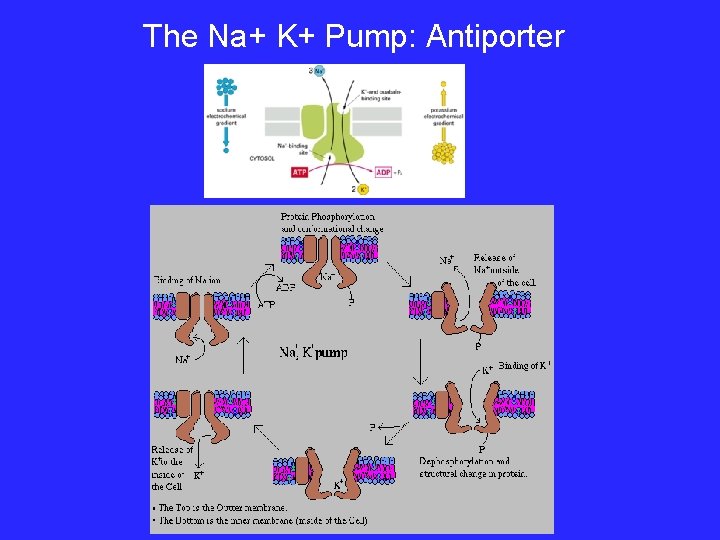
The Na+ K+ Pump: Antiporter
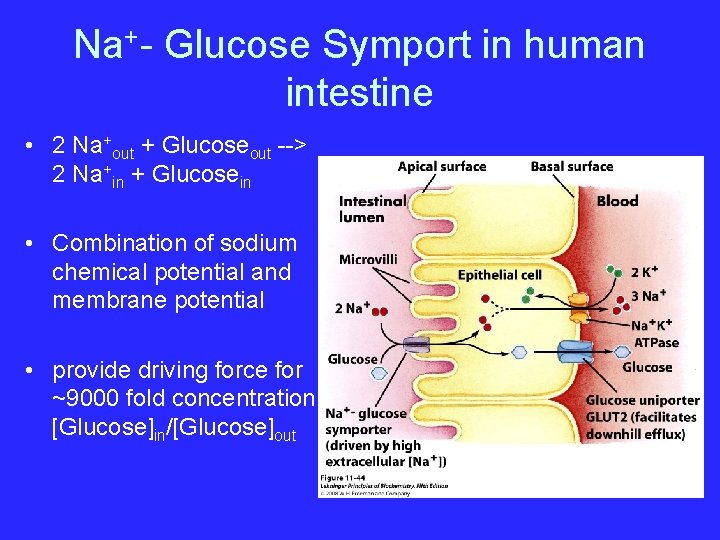
Na+- Glucose Symport in human intestine • 2 Na+out + Glucoseout --> 2 Na+in + Glucosein • Combination of sodium chemical potential and membrane potential • provide driving force for ~9000 fold concentration [Glucose]in/[Glucose]out
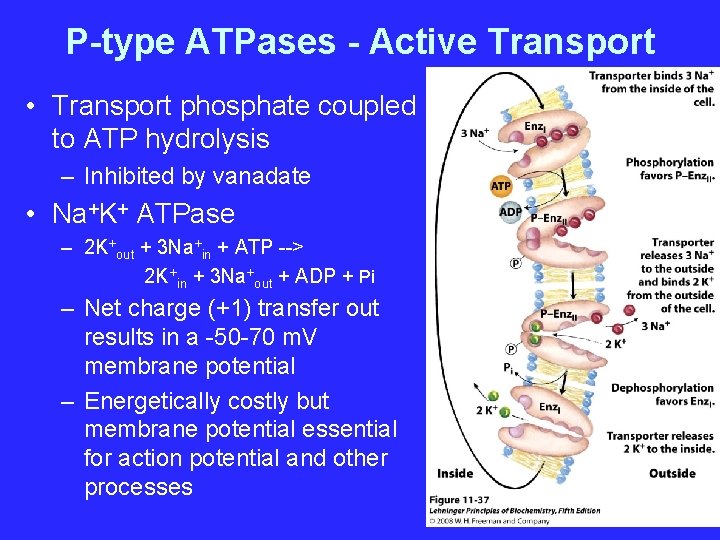
P-type ATPases - Active Transport • Transport phosphate coupled to ATP hydrolysis – Inhibited by vanadate • Na+K+ ATPase – 2 K+out + 3 Na+in + ATP --> 2 K+in + 3 Na+out + ADP + Pi – Net charge (+1) transfer out results in a -50 -70 m. V membrane potential – Energetically costly but membrane potential essential for action potential and other processes
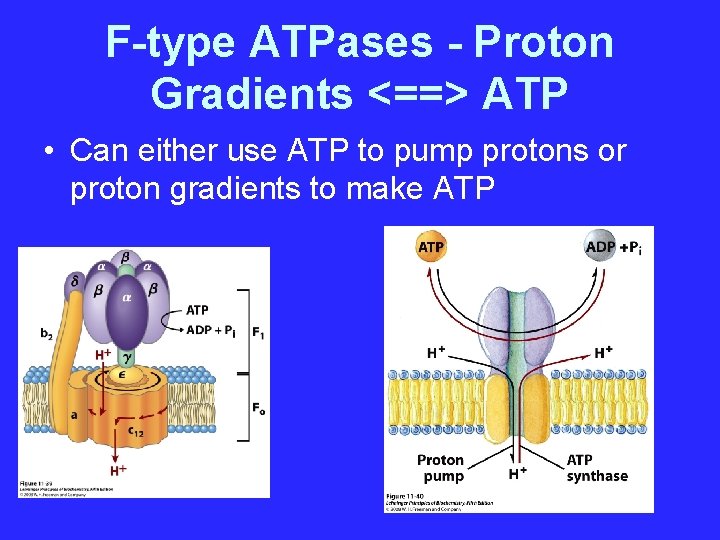
F-type ATPases - Proton Gradients <==> ATP • Can either use ATP to pump protons or proton gradients to make ATP
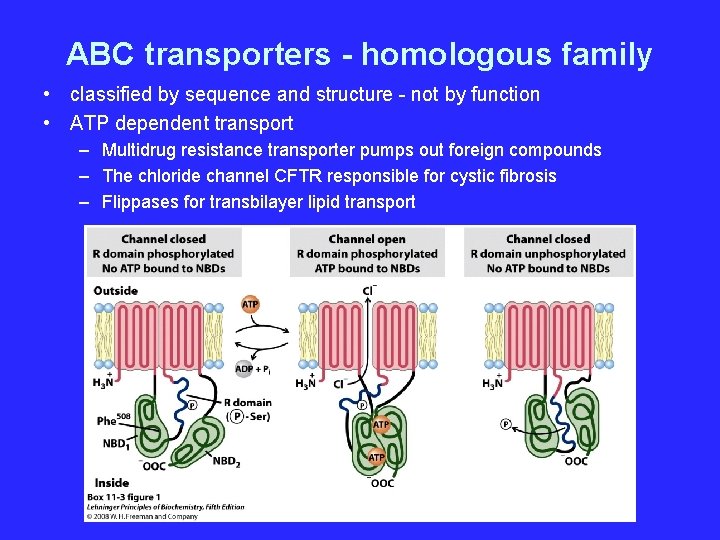
ABC transporters - homologous family • classified by sequence and structure - not by function • ATP dependent transport – Multidrug resistance transporter pumps out foreign compounds – The chloride channel CFTR responsible for cystic fibrosis – Flippases for transbilayer lipid transport
- Slides: 52