Membrane Dynamics contd How many substrates can a

Membrane Dynamics cont’d • • How many substrates can a carrier move? Active Transport Secondary Active Transport Transepithelial transport
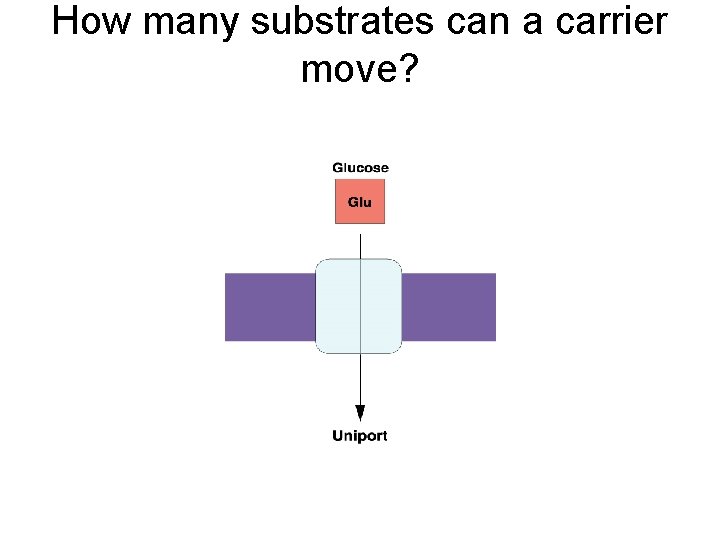
How many substrates can a carrier move?
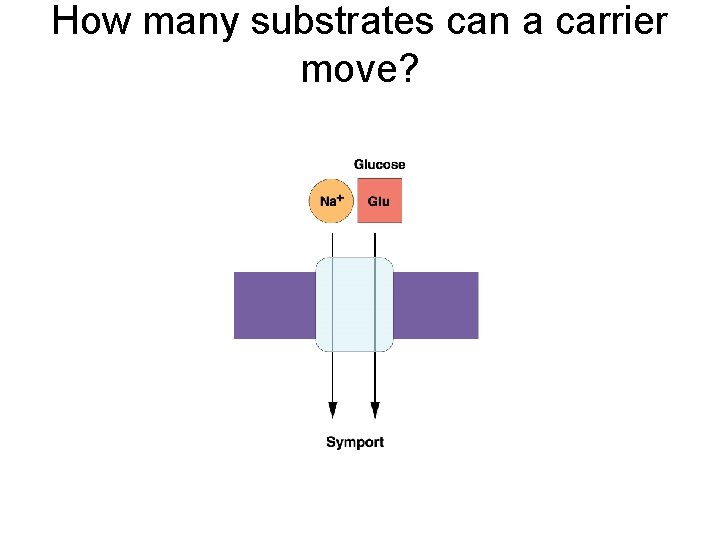
How many substrates can a carrier move?
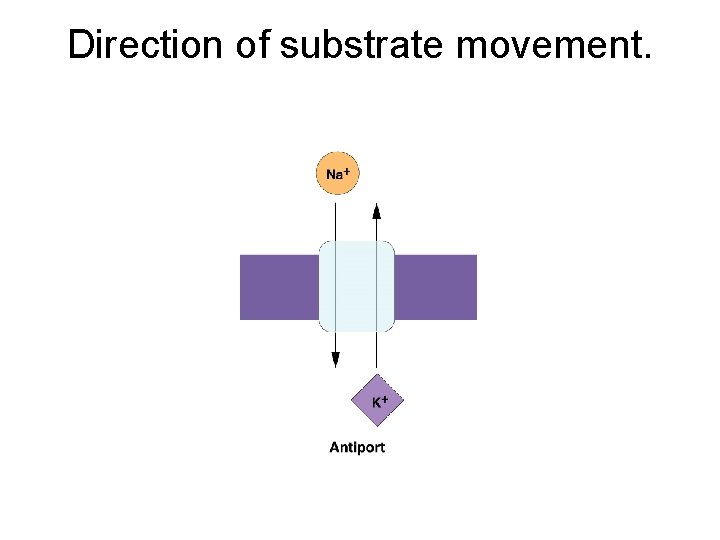
Direction of substrate movement.
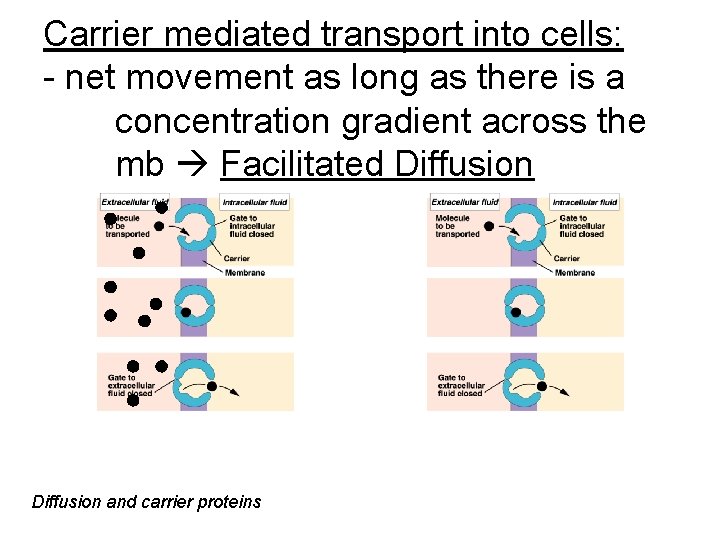
Carrier mediated transport into cells: - net movement as long as there is a concentration gradient across the mb Facilitated Diffusion and carrier proteins
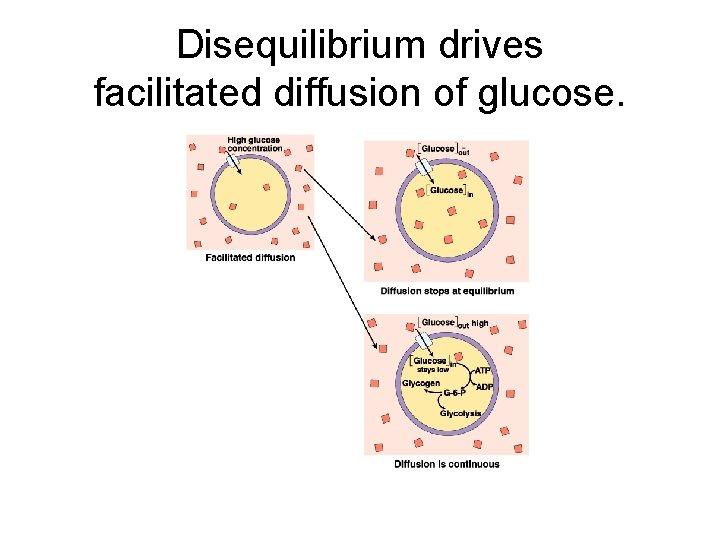
Disequilibrium drives facilitated diffusion of glucose.
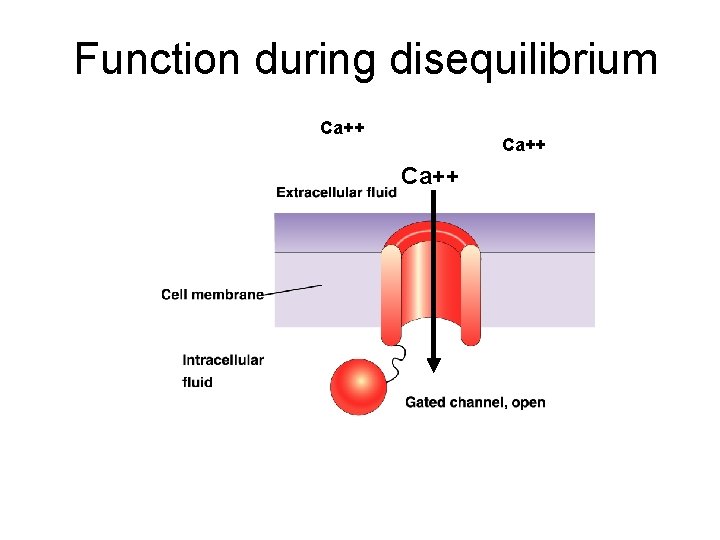
Function during disequilibrium Ca++ Calcium entry
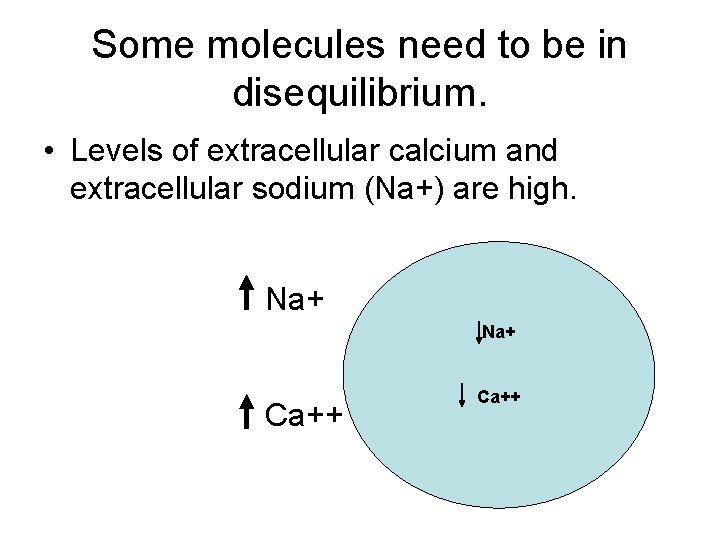
Some molecules need to be in disequilibrium. • Levels of extracellular calcium and extracellular sodium (Na+) are high. Na+ Ca++

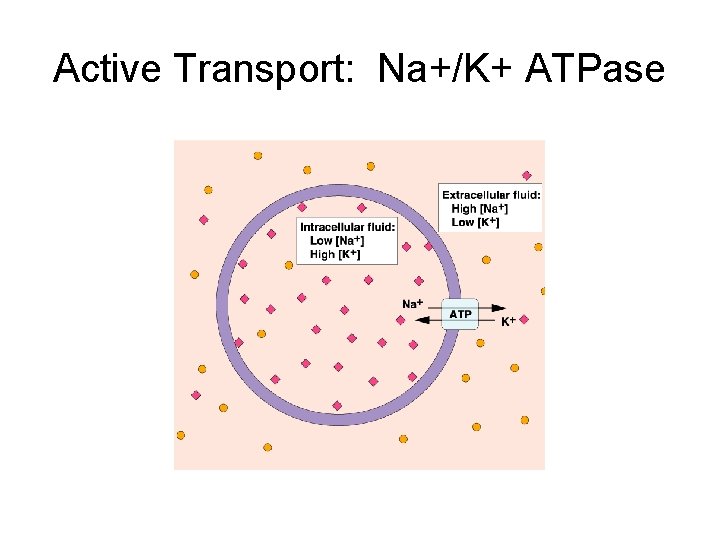
Active Transport • Na+ is removed from cells against its concentration gradient • Need ATP energy for this work
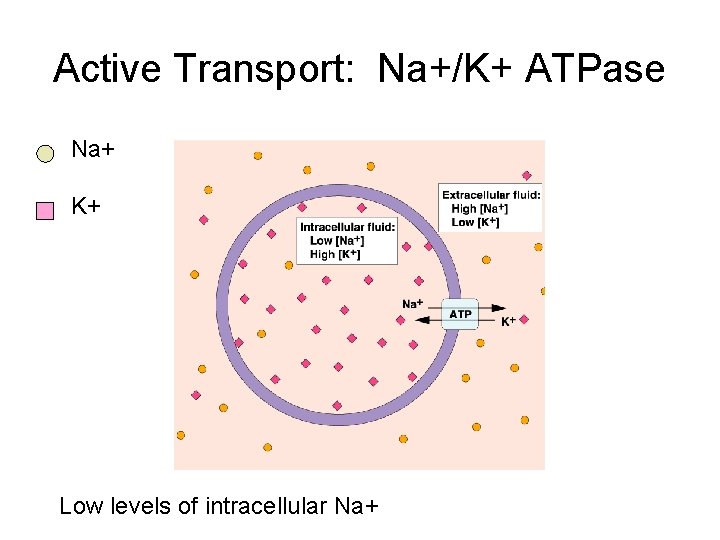
Active Transport: Na+/K+ ATPase Na+ K+ Low levels of intracellular Na+
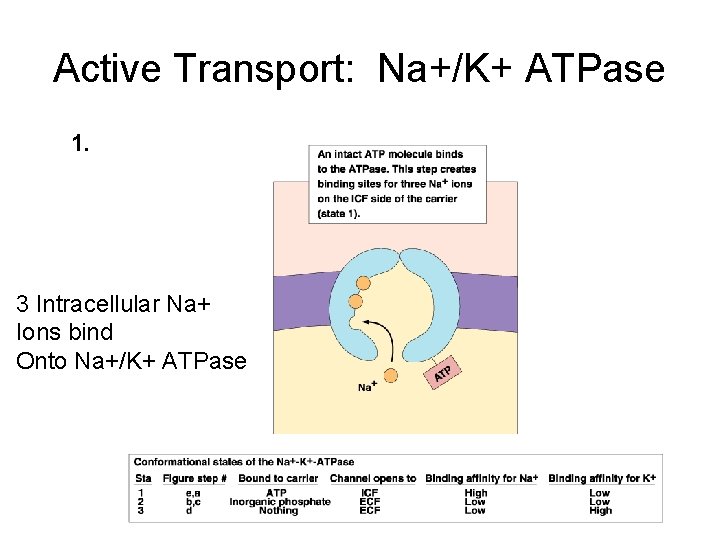
Active Transport: Na+/K+ ATPase 1. 3 Intracellular Na+ Ions bind Onto Na+/K+ ATPase
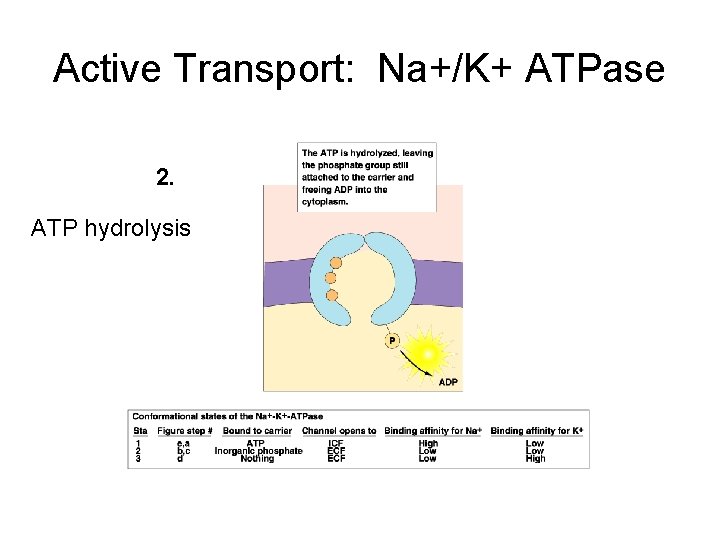
Active Transport: Na+/K+ ATPase 2. ATP hydrolysis
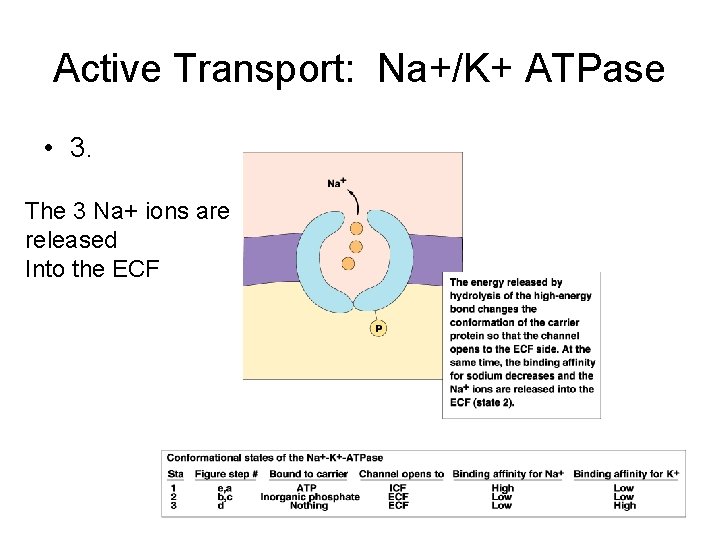
Active Transport: Na+/K+ ATPase • 3. The 3 Na+ ions are released Into the ECF
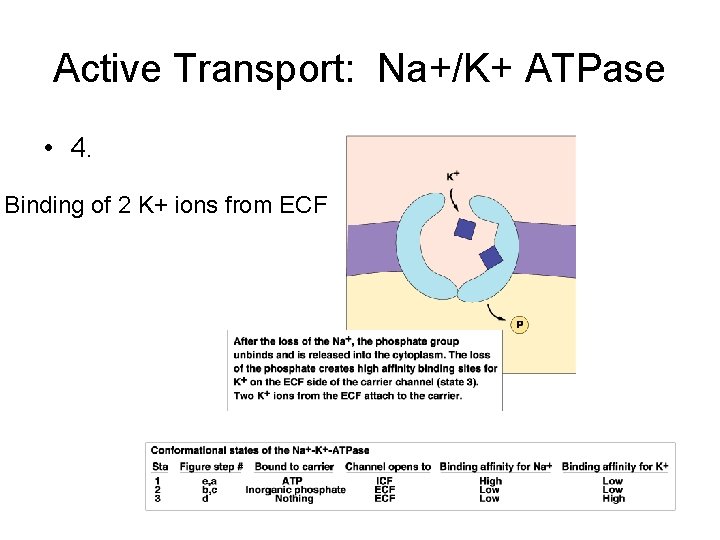
Active Transport: Na+/K+ ATPase • 4. Binding of 2 K+ ions from ECF
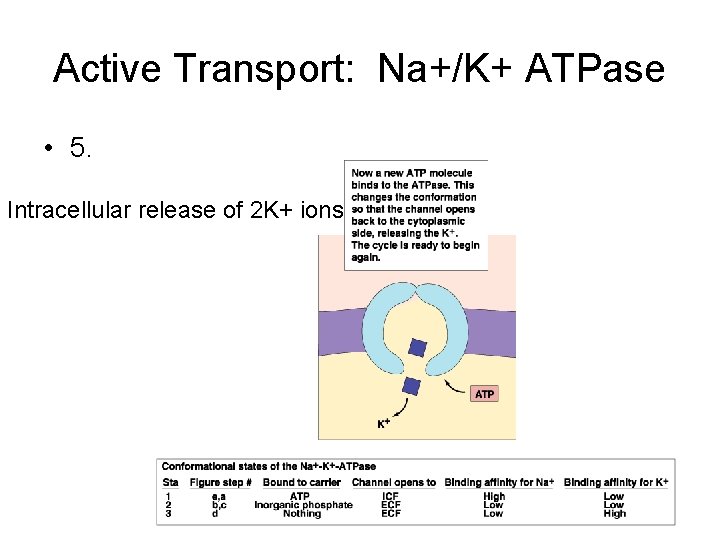
Active Transport: Na+/K+ ATPase • 5. Intracellular release of 2 K+ ions
Active Transport: Na+/K+ ATPase
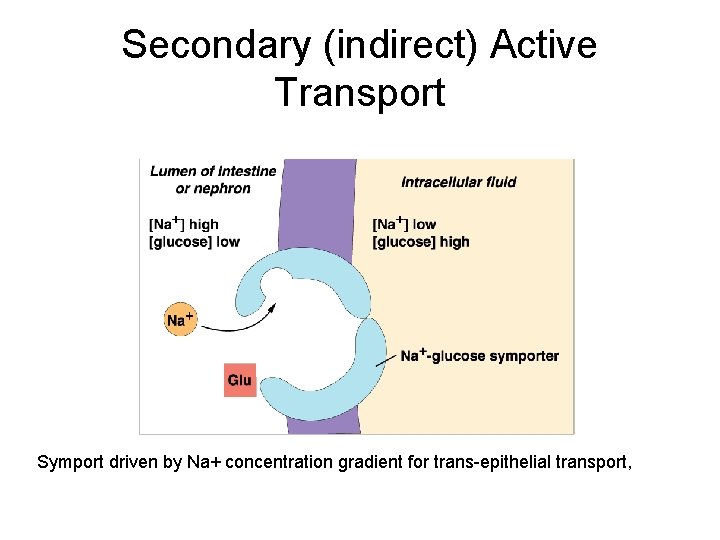
Secondary (indirect) Active Transport Symport driven by Na+ concentration gradient for trans-epithelial transport,
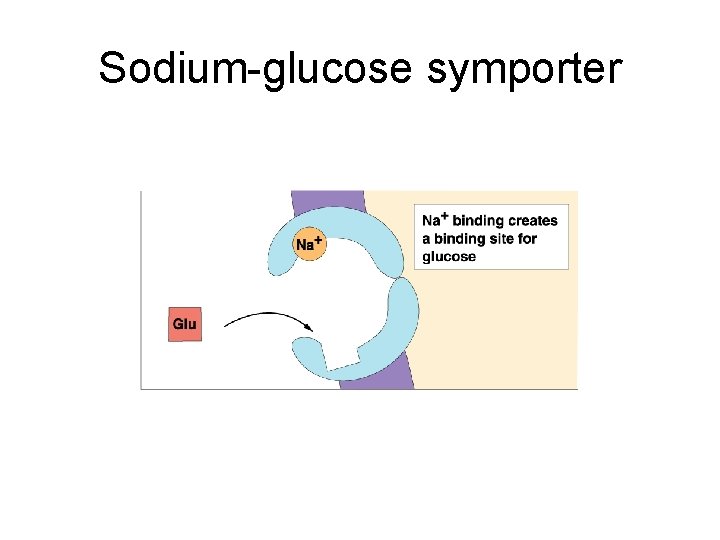
Sodium-glucose symporter
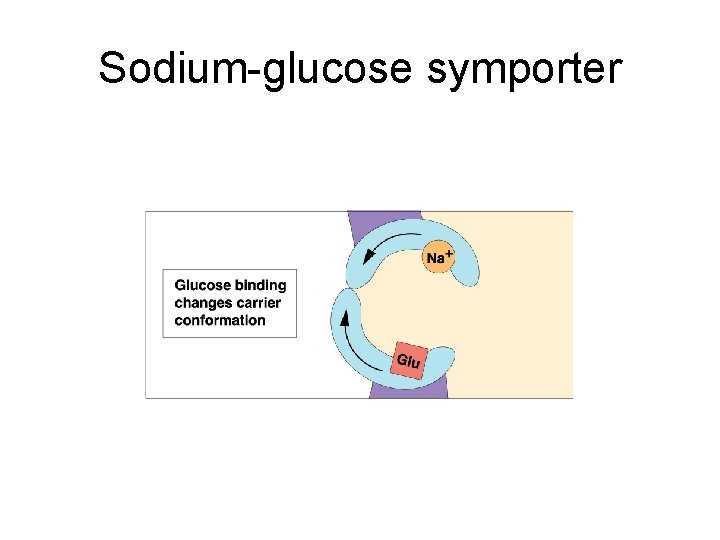
Sodium-glucose symporter
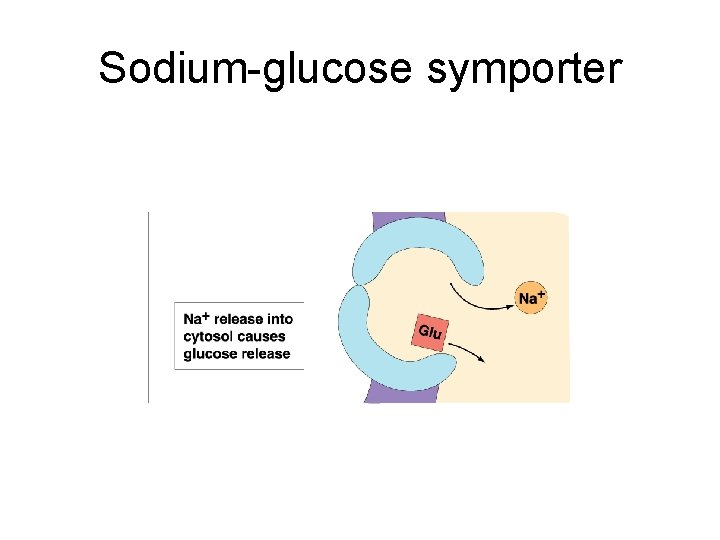
Sodium-glucose symporter
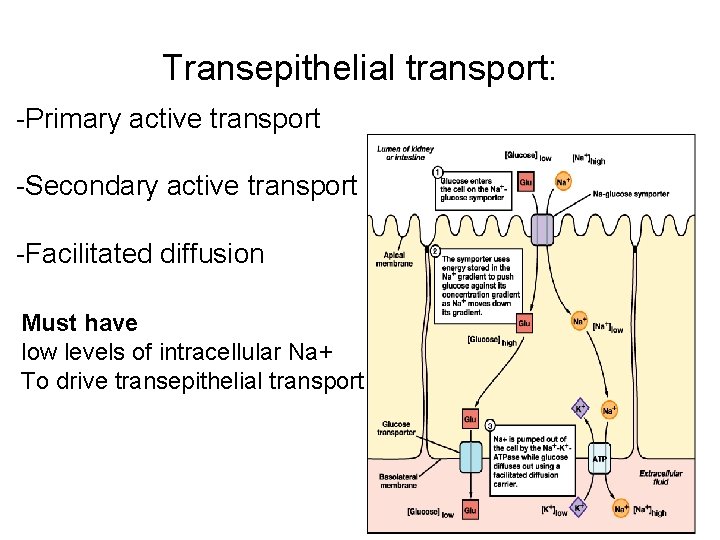
Transepithelial transport: -Primary active transport -Secondary active transport -Facilitated diffusion Must have low levels of intracellular Na+ To drive transepithelial transport
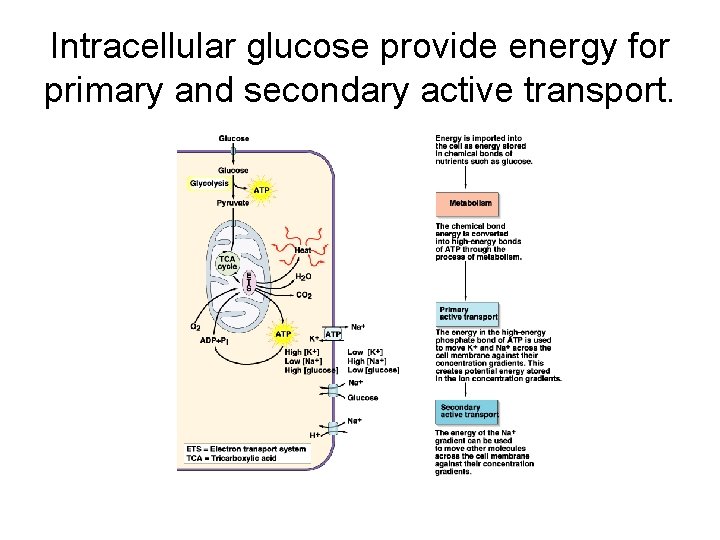
Intracellular glucose provide energy for primary and secondary active transport.

Where does transepithelial transport occur? • Glucose absorption in the intestine • Glucose absorption in the nephron • Glucose is moved from the mucosal surface of the epithelium to the serosal surface. • Glucose is moved from the apical surface of the cells to the basal surface of the cells.

How does water move in the body? • The cell membrane is semi-permeable • Water can move freely • Water is in equilibrium between cells and extracellular fluids (osmotic equilibrium) • Ions and solutes are disequilibrium • Osmosis water moves along its concentration gradient across a semipermeable membrane
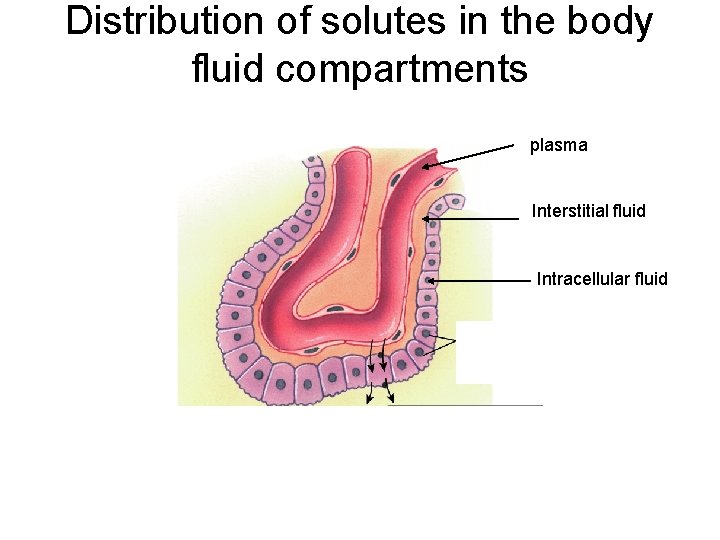
Distribution of solutes in the body fluid compartments plasma Interstitial fluid Intracellular fluid
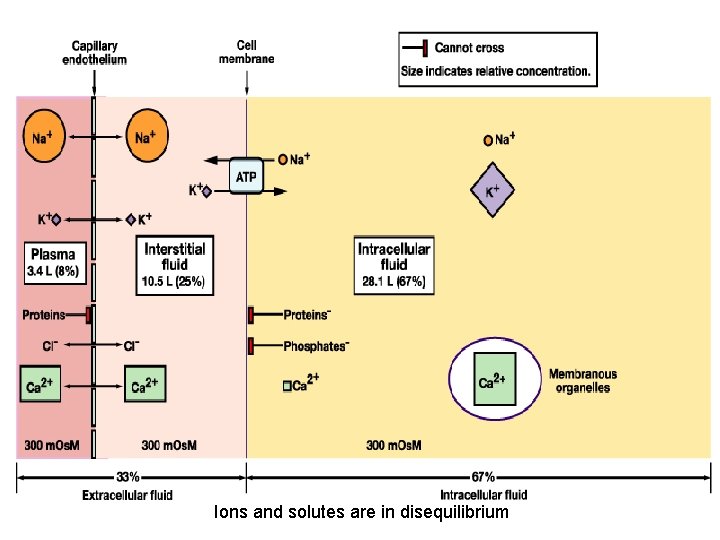
Ions and solutes are in disequilibrium
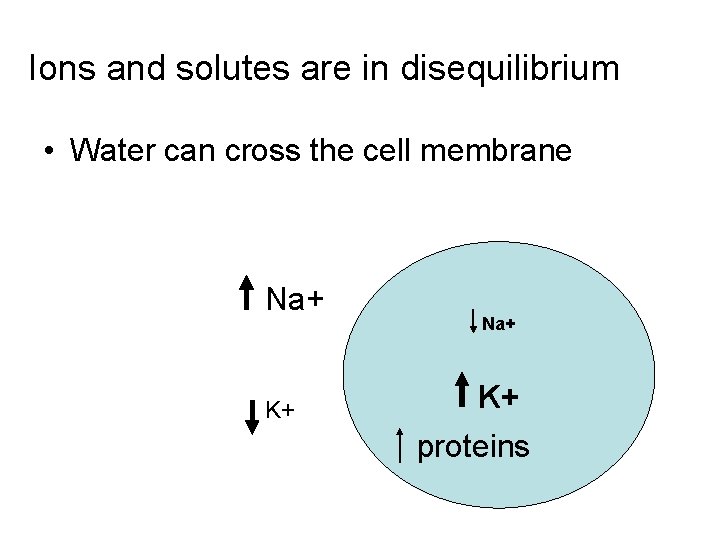
Ions and solutes are in disequilibrium • Water can cross the cell membrane Na+ K+ proteins

Osmosis • water moves along its concentration gradient across a semi-permeable membrane • Water moves to dilute a solute
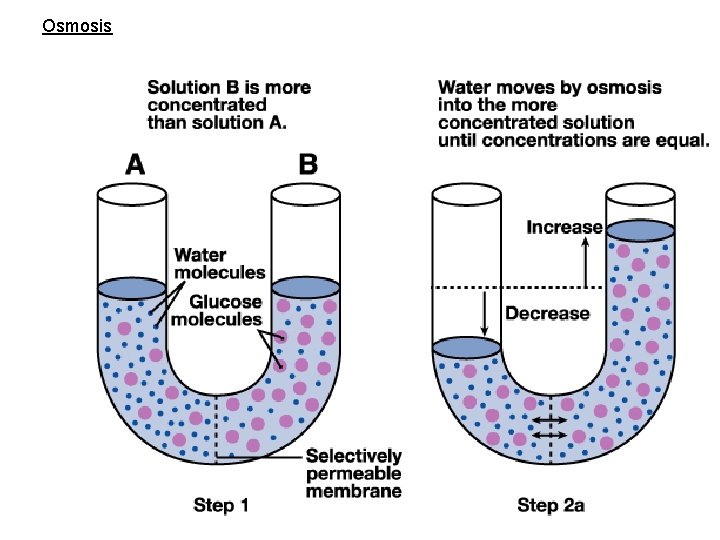
Osmosis
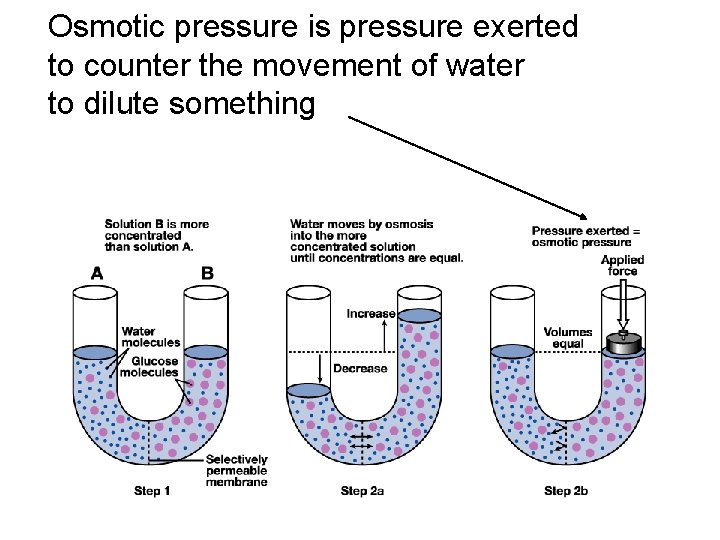
Osmotic pressure is pressure exerted to counter the movement of water to dilute something

Osmolarity • Describes the number of particles in solution • Know this and the direction of water movement can be predicted • • # of particles in 1 liter of solution Is expressed as osmoles/L, or Os. M If very dilute: milliosmoles/L, or m. Os. M Human body, approx 300 m. Os. M

Osmolarity: number of particles in 1 L • 1 M glucose = 1 Os. M glucose • 1 M Na. Cl = 2 Os. M Na. Cl, because Na. Cl disassociates to 2 ions in solution. Na+ Cl-

Compare the osmolarity of 2 solutions: • Solution A • Solution B • 1 Os. M glucose • 2 Os. M glucose • A is hyposmotic to B • B is hyperosmotic to A • (A has fewer particles than B) • (B has more particles than A)

Compare the osmolarity of 2 solutions: • Solution B • Solution C • 2 Os. M glucose • 1 Os. M Na. Cl • B is hyperosmotic to C • C is hypotonic to B • (B has more particles/L than A) • (C has fewer particles/L than B)
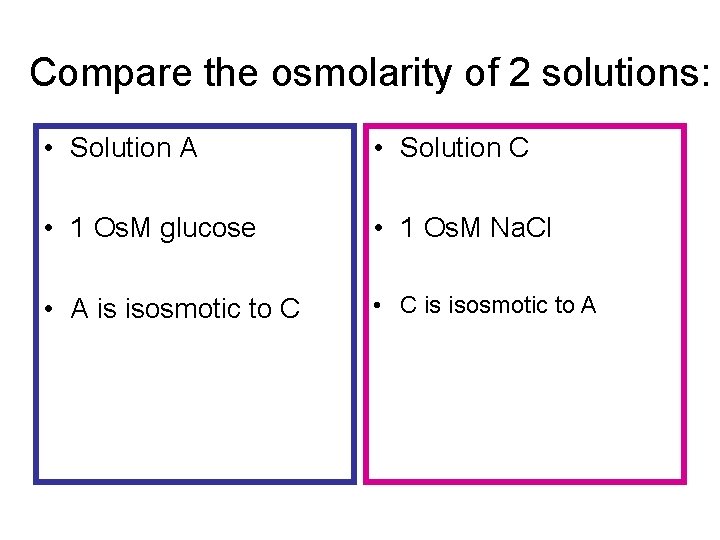
Compare the osmolarity of 2 solutions: • Solution A • Solution C • 1 Os. M glucose • 1 Os. M Na. Cl • A is isosmotic to C • C is isosmotic to A
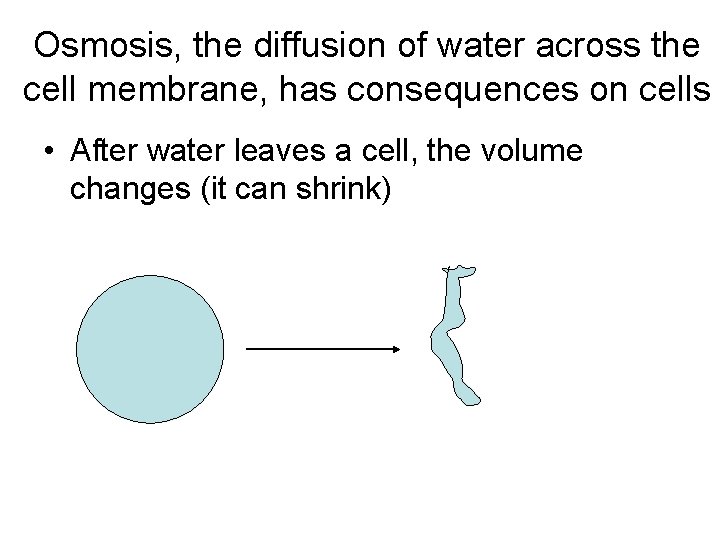
Osmosis, the diffusion of water across the cell membrane, has consequences on cells • After water leaves a cell, the volume changes (it can shrink)

Tonicity • Describes how the cell volume will change in a solution
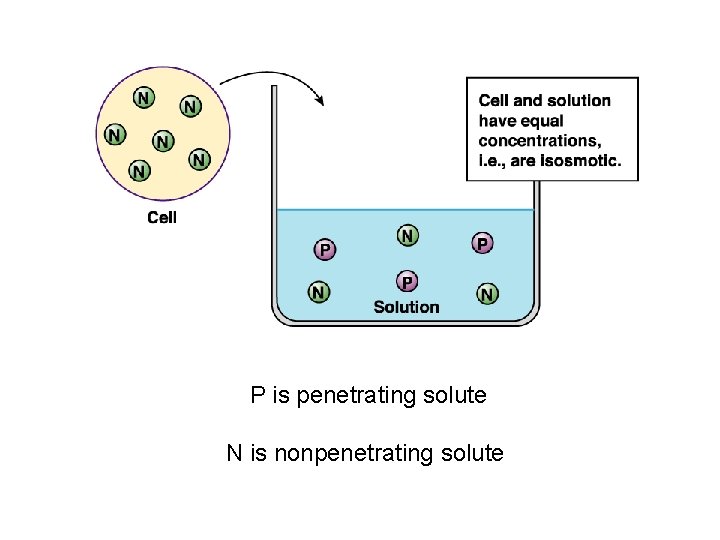
P is penetrating solute N is nonpenetrating solute
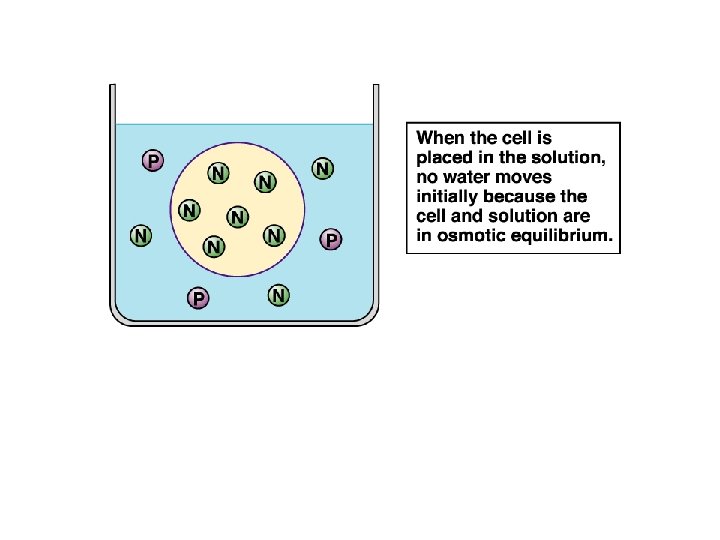
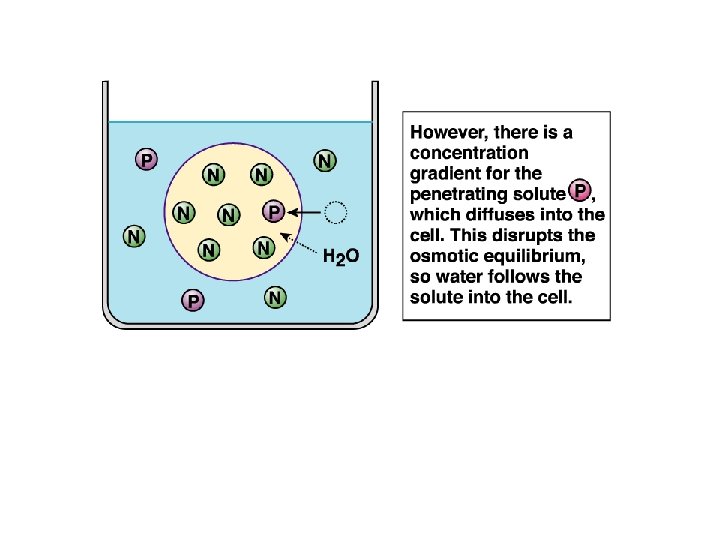
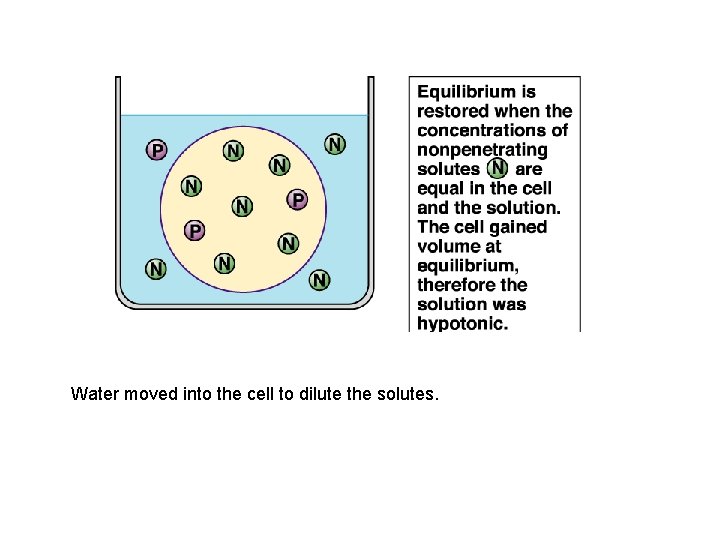
Water moved into the cell to dilute the solutes.

• Cell gains volume in a hypotonic solution • Cell looses volume in a hypertonic solution • Cell keeps the same volume in an isotonic solution.

Tonicity indicates how the cell volume will change in a solution • In a hypotonic solution, the cell has a higher concentration of a nonpenetrating solute than the solution, water moves in. • In a hypertonic solution, the cell has a lower concentration of nonpenetrating solute than the solution, water leaves the cell

During intavenous injection: • • 0. 9% (normal) saline isotonic D 5 --. 9% saline (5% dextrose) isotonic D 5 W hypotonic 0. 45% saline hypotonic • Vs dehydration hypotonic • Vs blood loss isotonic
- Slides: 44