MELTING BOILING POINTS STRONGER the The forces including

MELTING & BOILING POINTS STRONGER the The forces (including both the intramolecular and intermolecular forces) holding a compound together, the melting &HIGHER boiling points. Ionic bonds are STRONGER making their melting points than covalent bonds, thus. HIGHER Covalent molecules which have intermolecular STRONG forces (such as water, with its strong hydrogen bonding) are more to pull apart from each other, which DIFFICULT than compounds causes their melting points to be without hydrogen bonding or dipole-dipole forces (such as F 2). HIGHER
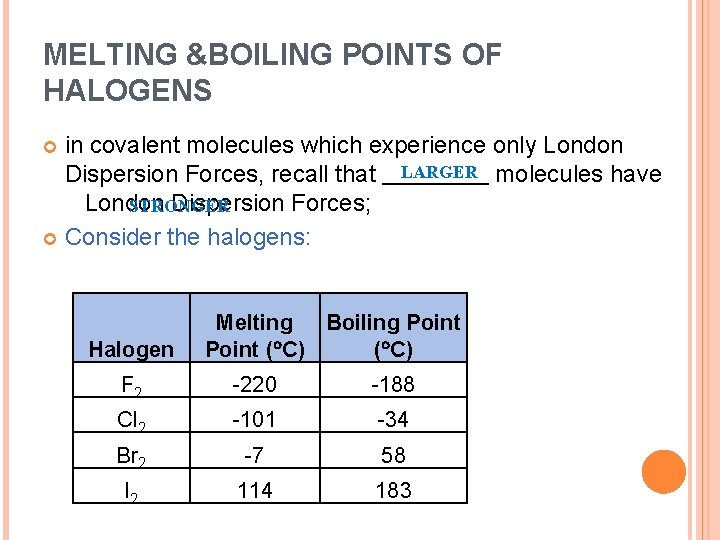
MELTING &BOILING POINTS OF HALOGENS in covalent molecules which experience only London Dispersion Forces, recall that LARGER molecules have London Dispersion Forces; STRONGER Consider the halogens: Halogen Melting Boiling Point ( C) F 2 -220 -188 Cl 2 -101 -34 Br 2 -7 58 I 2 114 183

VOLATILITY Volatility: is the tendency of a liquid at room temperature to evaporate into a gas In order to evaporate, the liquid molecules must possess enough K. E. to overcome the forces holding the molecules together. Among the halogens, volatility decreases as you go down the group, for exactly the same reasons that melting & boiling points increase: greater mass greater stronger (and thus # of e-) van der Waal forces.

CONDUCTIVITY Recall that metallic solids have delocalized electrons, which enables them to readily electricity. By contrast, most covalent and ionic solids conductors, because their valence electrons delocalized. valence conduct are nonare not In covalent molecules, the electrons are shared between just a few atoms. In ionic compounds, the valence electrons are completely lost or gained to other atoms, to satisfy each atom’s octet rule. Ionic substances become conductive in the liquid state, however, because the ions can move (in an ionic liquid, the ions carry the electric current). Likewise, many ionic solutions will conduct electricity, if ionic solid can be dissolved in water, producing an aqueous solution

SOLUBILITY Recall that “ like dissolves like ”. In other words, solvents tend to dissolve solutes with similar properties. Polar solvents tend to dissolve polar solutes. ex #1: NH 3 dissolves in water to make household cleaners Ammonia & water are both polar, having unshared e–s on the central atom of their molecules. Non-polar solvents tend to dissolve non-polar solutes. ex: I 2(s) dissolves in CCl 4(l)
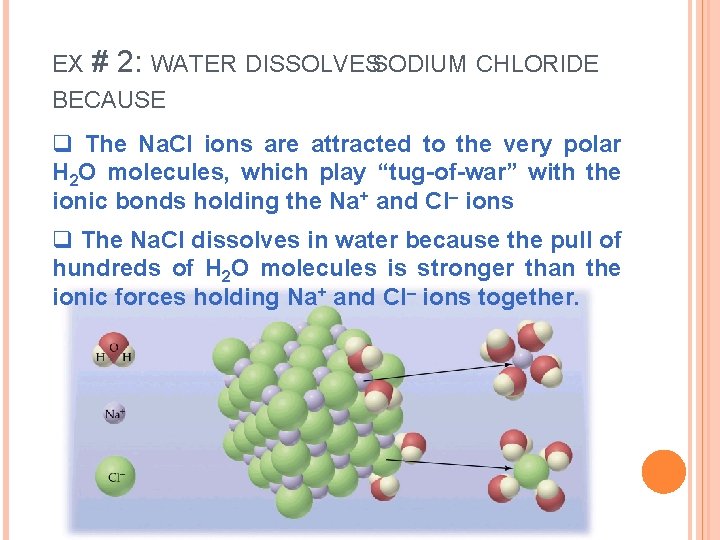
EX # 2: WATER DISSOLVESSODIUM CHLORIDE BECAUSE q The Na. Cl ions are attracted to the very polar H 2 O molecules, which play “tug-of-war” with the ionic bonds holding the Na+ and Cl– ions q The Na. Cl dissolves in water because the pull of hundreds of H 2 O molecules is stronger than the ionic forces holding Na+ and Cl– ions together.

Consider also the solubilities of the following alcohols in water: Alcohol Solubility in Water (mol / 100 g H 2 O) name alcohol solubility methanol CH 3 OH infinite ethanol C 2 H 5 OH Infinite propanol C 3 H 7 OH Infinite butanol C 4 H 9 OH 0. 11 pentanol C 5 H 11 OH 0. 03 hexanol C 6 H 13 OH 0. 0058 heptanol C 7 H 15 OH 0. 0008 Alcohols dissolve in water as the – OH group is able to hydrogen bond with H 2 O molecules, and be pulled in solution Thus, as the length of the alcohol molecule increases, water molecules have less of a chance of hydrogen bonding with the water (more of the interactions with water occur at the non-polar C – C and C – H groups, rather than the polar –OH group). This is why larger alcohol molecules do not dissolve as readily in water.
- Slides: 7