melt conditions glass transition Tg crystallisation Tm POLYMER



- Slides: 14

melt conditions glass transition Tg crystallisation Tm POLYMER Tg(o. C) Polydimethylsiloxane -123 Poly(vinyl acetate) 28 Polystyrene 100 Poly(methyl methacrylate) 105 Polycarbonate 150 Polysulfone 190 Poly(2, 6 -dimethyl-1, 4 -phenylene oxide) 220 Polymers with more flexible backbones, and smaller substituent side groups have lower glass transition temperatures

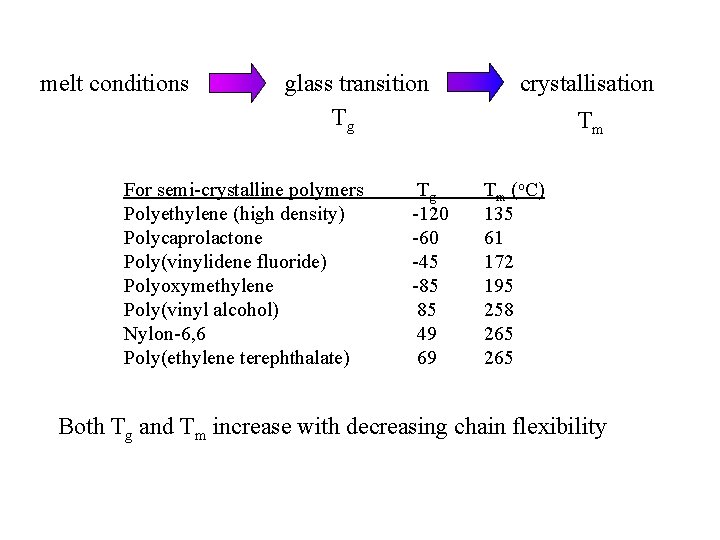
melt conditions glass transition Tg For semi-crystalline polymers Polyethylene (high density) Polycaprolactone Poly(vinylidene fluoride) Polyoxymethylene Poly(vinyl alcohol) Nylon-6, 6 Poly(ethylene terephthalate) Tg -120 -60 -45 -85 85 49 69 crystallisation Tm Tm (o. C) 135 61 172 195 258 265 Both Tg and Tm increase with decreasing chain flexibility

From Fried, Joel R. , “Polymer Science and Technology”, Prentice Hall PTR, Englewood Cliffs, NJ (1995 )

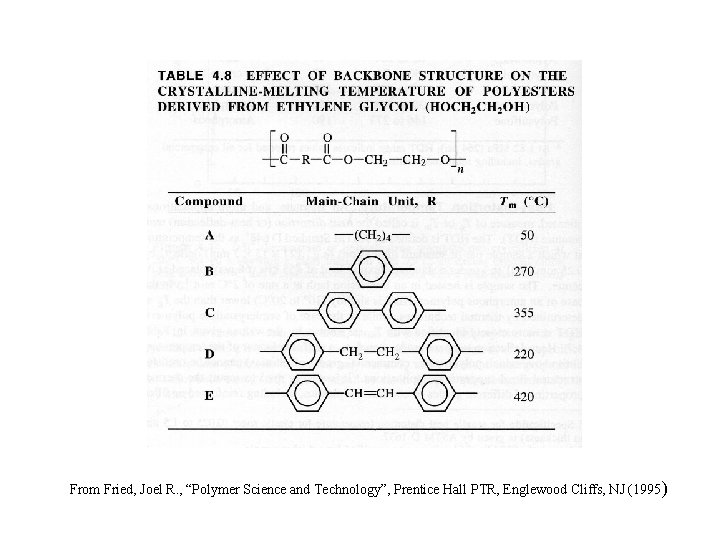
From Fried, Joel R. , “Polymer Science and Technology”, Prentice Hall PTR, Englewood Cliffs, NJ (1995 )

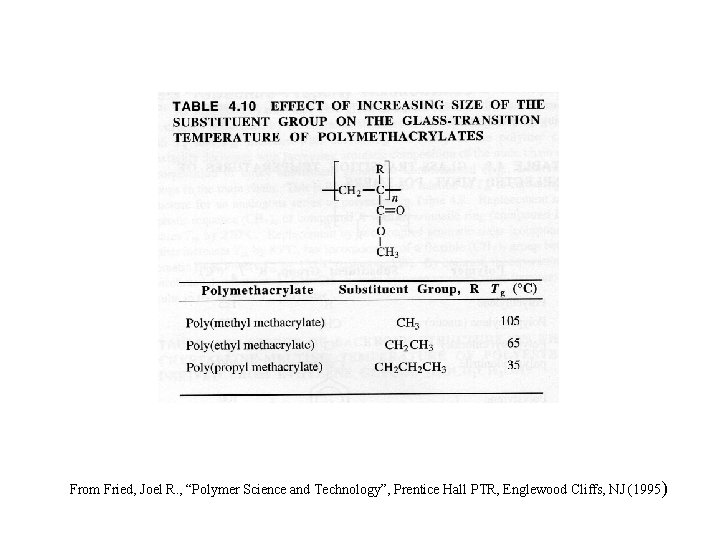
From Fried, Joel R. , “Polymer Science and Technology”, Prentice Hall PTR, Englewood Cliffs, NJ (1995 )

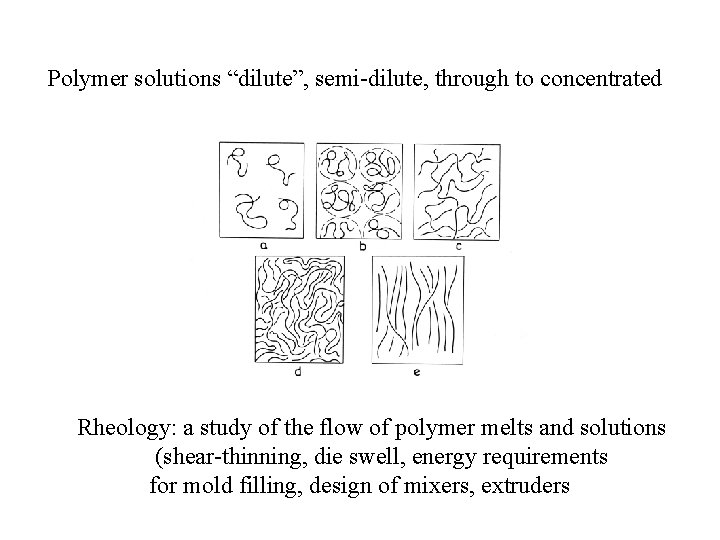
Polymer solutions “dilute”, semi-dilute, through to concentrated Rheology: a study of the flow of polymer melts and solutions (shear-thinning, die swell, energy requirements for mold filling, design of mixers, extruders

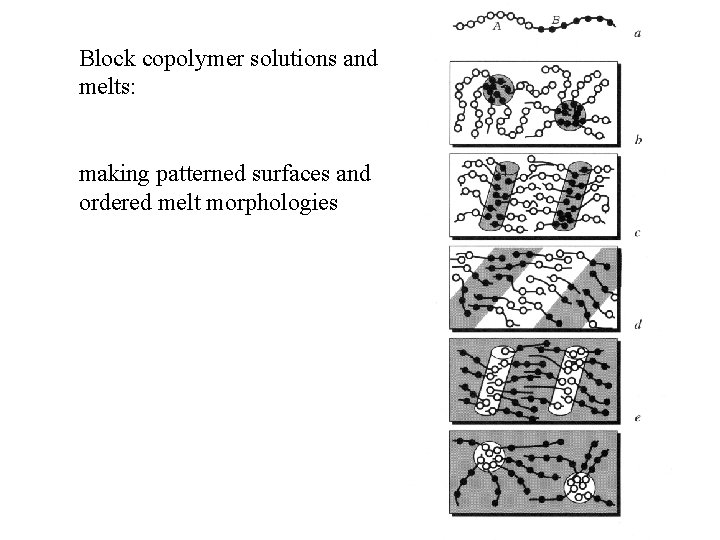
Block copolymer solutions and melts: making patterned surfaces and ordered melt morphologies

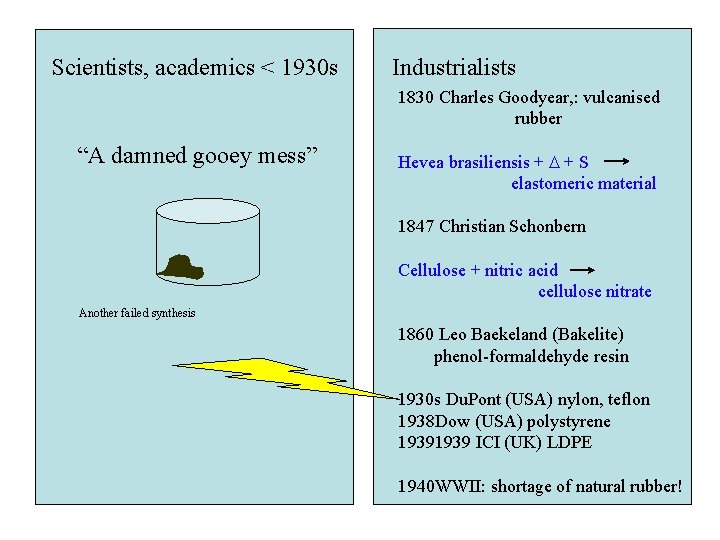
Scientists, academics < 1930 s Industrialists 1830 Charles Goodyear, : vulcanised rubber “A damned gooey mess” Hevea brasiliensis + D + S elastomeric material 1847 Christian Schonbern Cellulose + nitric acid cellulose nitrate Another failed synthesis 1860 Leo Baekeland (Bakelite) phenol-formaldehyde resin 1930 s Du. Pont (USA) nylon, teflon 1938 Dow (USA) polystyrene 1939 ICI (UK) LDPE 1940 WWII: shortage of natural rubber!

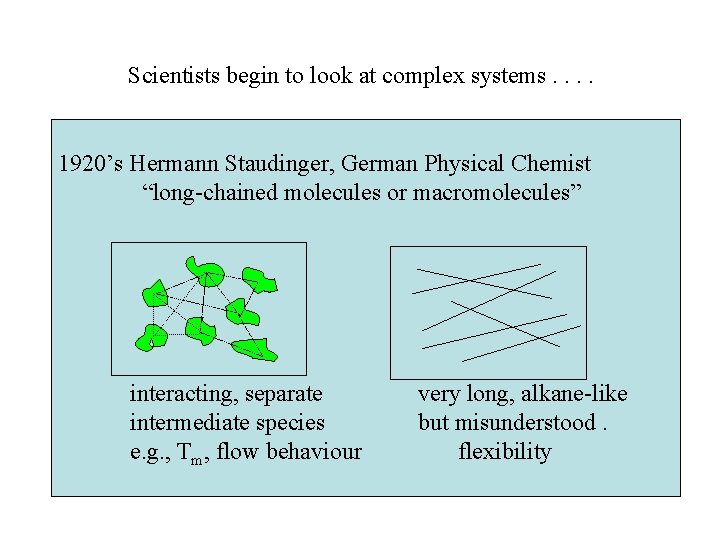
Scientists begin to look at complex systems. . 1920’s Hermann Staudinger, German Physical Chemist “long-chained molecules or macromolecules” interacting, separate intermediate species e. g. , Tm, flow behaviour very long, alkane-like but misunderstood. flexibility

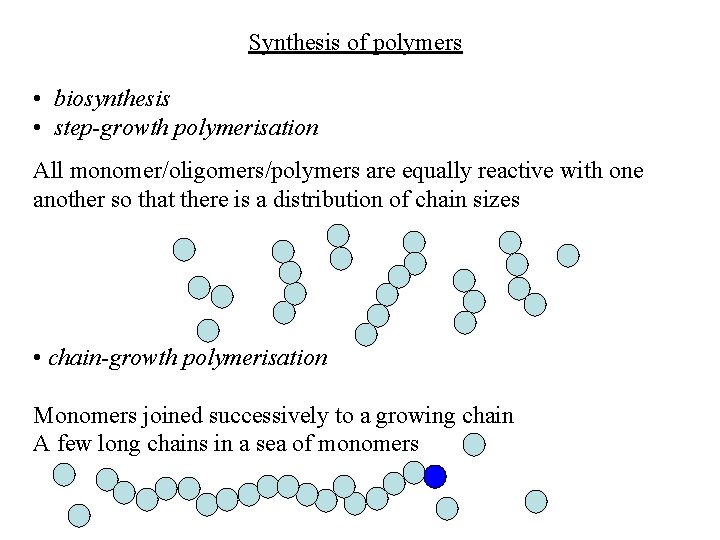
Synthesis of polymers • biosynthesis • step-growth polymerisation All monomer/oligomers/polymers are equally reactive with one another so that there is a distribution of chain sizes • chain-growth polymerisation Monomers joined successively to a growing chain A few long chains in a sea of monomers

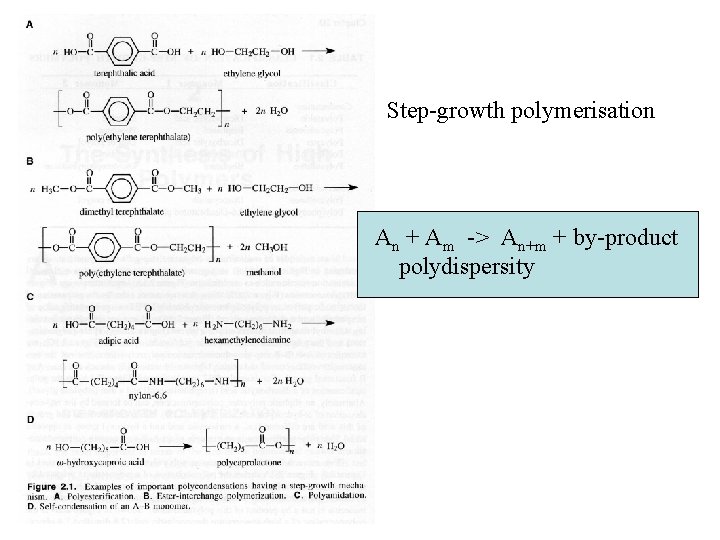
Step-growth polymerisation An + Am -> An+m + by-product polydispersity

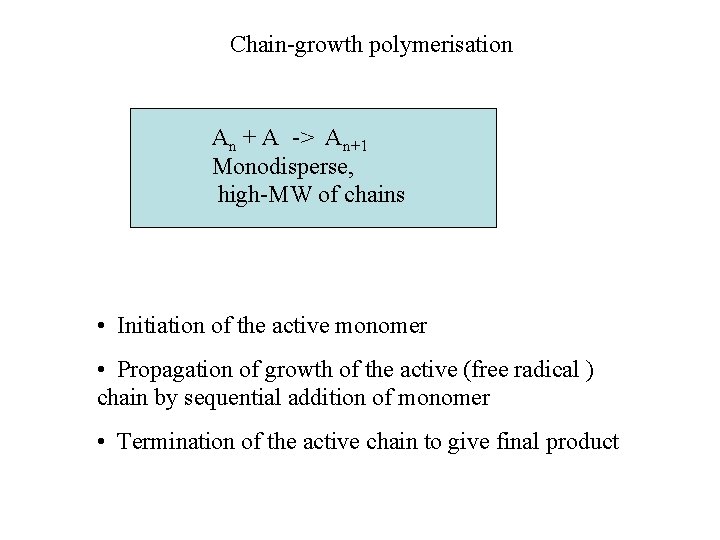
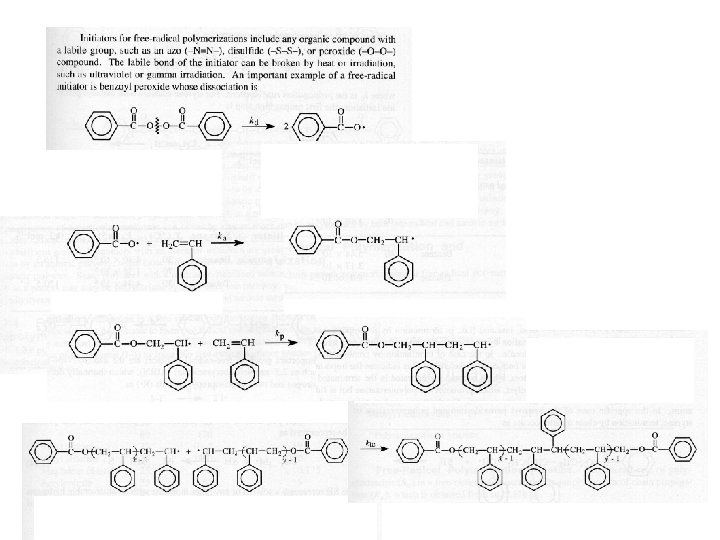
Chain-growth polymerisation An + A -> An+1 Monodisperse, high-MW of chains • Initiation of the active monomer • Propagation of growth of the active (free radical ) chain by sequential addition of monomer • Termination of the active chain to give final product


Q 8: Contrast step-growth and chain-growth mechanisms in the synthesis of linear polymers and include statements comparing the final products of these two classes of synthetic mechanisms.