Melissa PMTCT case study Melissas background Melissa is

Melissa PMTCT case study

Melissa’s background Melissa is a 30 year old women known to be living with HIV, and currently on TEE for 18 months. She attends for her routine ART follow-up visit at her PHC clinic • Her VL at 12 months on ART was < 50 c/ml • Her CD 4 at 12 months was 490. • She is no longer on CPT. • She completed a 12 months course of TPT 2 years ago

On history taking today: • TB and STI screen: She is well and has no TB or genital symptoms. • Adherence: She has a good routine and rarely misses a dose • Side effects: She has had no side effects form her ART. • Drug Interactions: She is not taking any new drugs or over the counter medications • Mental Health screen: She has no symptoms of depression or substance abuse or dementia. • NCDs: She has no CVD risk factors.

What else needs to be included in the history taking?

What else needs to be included in the history taking? Family Planning and fertility intentions: • She uses condoms with her partner some of the time, and is not using any other form of hormonal contraception or IUCD. • Her last period was 4 months ago • Her urine pregnancy test is positive Index case testing • Has her partner been tested for HIV? • Have all her biological children been tested for HIV? Melissa has disclosed to her partner. He has been tested and is also on ART. Her other biological child (age 2 years) tested HIV negative

On examination • Melissa is clinically well, and has no danger signs. • Her vitals and clinical examination are normal. • She wants to keep the baby • She is provided with 1 months supply of her current regimen (TEE), and accompanied to the antenatal clinic to meet the sister and to register for antenatal care

Melissa has her booking visit at the antenatal clinic • She is provided with all routine antenatal care according to the BANC-plus guideline including BP, HB, urine dipstix, syphilis test, nutritional assessment, and counselling on infant feeding. • Her uterus is just palpable, and she is assessed to be at 16 weeks gestation. • She is connected to Mom-connect, and assigned a CHW who will visit her at home and provide her with additional support. Discuss in your groups (5 minutes): • Pertaining to her HIV and ART, what additional management would you like to provide for Melissa, and what counseling would you give her?

Additional Management for Melissa related to her HIV and ART • A VL should be done at booking visit in ANC for all women already on ART • Pregnant women already on ART should continue their current ART regimen pending the result of their 1 st VL • Only if her VL is <50 c/ml, and she is no longer in the 1 st trimester, offer her the option of switching to DTG (see next slide). • If her VL is ≥ 50 c/ml, manage her as per the VL Non-suppression algorithm • Explain that she has the option to collect her ART at ANC as a “one-stop service”. If she will now collect her ART at ANC, ensure that she is documented as a transfer-out from her former clinic, and not classified as lost-to-follow-up. • Ensure that the mother understands the importance of VL suppression for her own health and for that of her baby, and check that she has the skills to be adherent to her ART
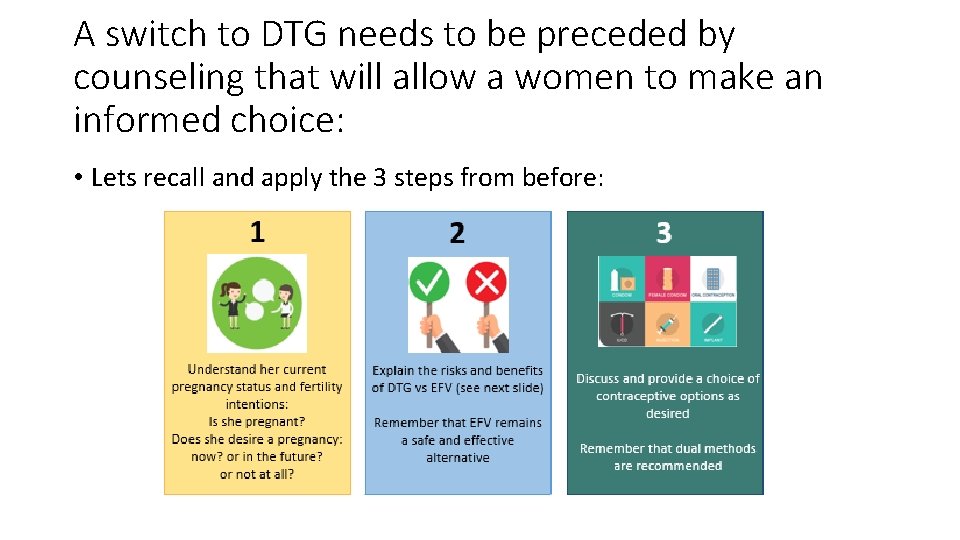
A switch to DTG needs to be preceded by counseling that will allow a women to make an informed choice: • Lets recall and apply the 3 steps from before:

Enabling a Client to Make an Informed Choice • Step 1: • We know that she is currently pregnant and in the 2 nd trimester • Ask if her family would be complete after this baby, or if she would desire to have more children in the future. • Step 2: • Explain the risks and benefits of DTG vs EFV, including the risk of NTDs: • For this pregnancy (the high risk time has passed; there is no longer a risk for NTDs for this pregnancy) • For subsequent pregnancies (due to risk of NTDs around the time of conception, she should be changed from TLD back to TEE before she stops her contraception) • Inform her of the new side-effects that may be experienced when switching to a new drug • Explain about potential drug interactions with calcium iron, and antacids • Step 3: Discuss options and recommendations for postpartum contraception. Remember that dual methods are recommended
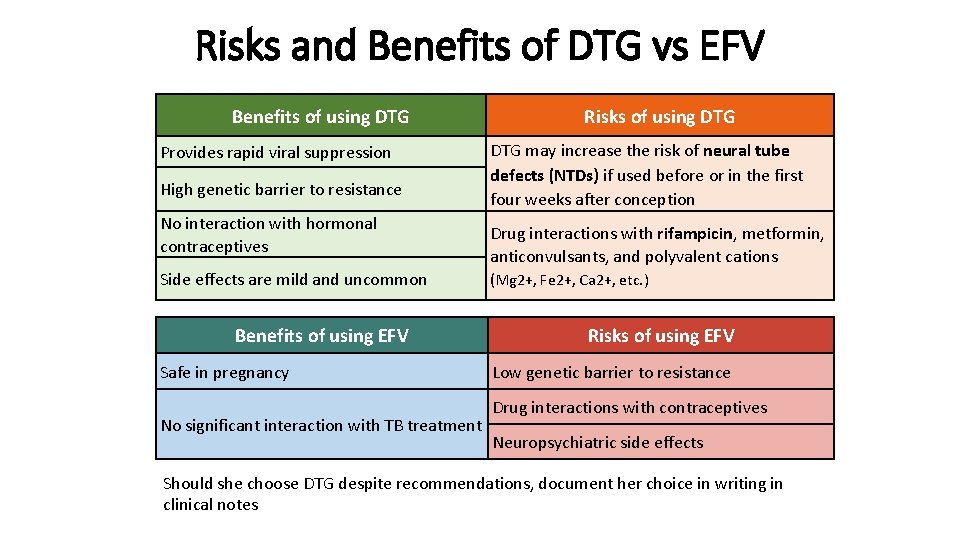
Risks and Benefits of DTG vs EFV Benefits of using DTG Provides rapid viral suppression High genetic barrier to resistance No interaction with hormonal contraceptives Side effects are mild and uncommon Benefits of using EFV Safe in pregnancy No significant interaction with TB treatment Risks of using DTG may increase the risk of neural tube defects (NTDs) if used before or in the first four weeks after conception Drug interactions with rifampicin, metformin, anticonvulsants, and polyvalent cations (Mg 2+, Fe 2+, Ca 2+, etc. ) Risks of using EFV Low genetic barrier to resistance Drug interactions with contraceptives Neuropsychiatric side effects Should she choose DTG despite recommendations, document her choice in writing in clinical notes
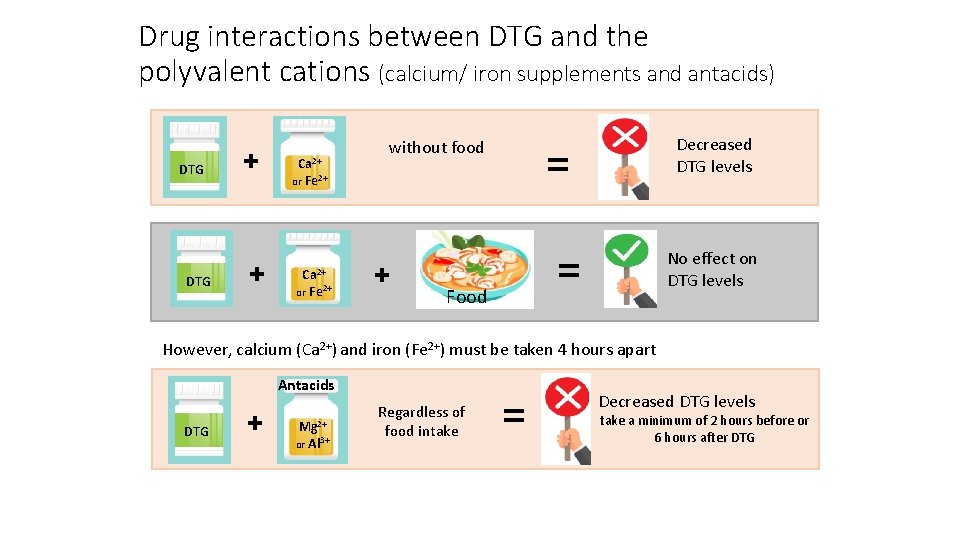
Drug interactions between DTG and the polyvalent cations (calcium/ iron supplements and antacids) DTG + + Ca 2+ or Fe 2+ = without food + Decreased DTG levels = Food No effect on DTG levels However, calcium (Ca 2+) and iron (Fe 2+) must be taken 4 hours apart Antacids DTG + Mg 2+ or Al 3+ Regardless of food intake = Decreased DTG levels take a minimum of 2 hours before or 6 hours after DTG

Suggested Dosing Schedule DTG (TLD) can be taken in the morning or evening with calcium and with food If insomnia is experienced, dose in the morning Option 1 • DTG + Calcium 500 mg + food in the morning • Ferrous sulphate 200 mg at lunch with food (better tolerated) • Calcium 500 mg in the evening Option 2 • Calcium 500 mg + food in the morning • Ferrous sulphate 200 mg at lunch with food (better tolerated) • DTG + Calcium 500 mg with food in the evening

Melissa’s VL at booking into ANC was < 50 c/ml. After counselling, she made an informed choice to switch to TLD. She did not experience any new side-effects, and is happy on her new regimen When should Melissa’s VL be repeated? • At delivery

Melissa delivers a healthy male infant at 38 weeks gestation. She is motivated to exclusively breastfeed the baby. Discuss the following 3 questions in your groups (5 minutes) • Would you classify this infant as high-risk or low-risk? • What infant prophylaxis would you provide? • Under what circumstances might you amend your initial assessment of risk and/or the duration of infant prophylaxis?
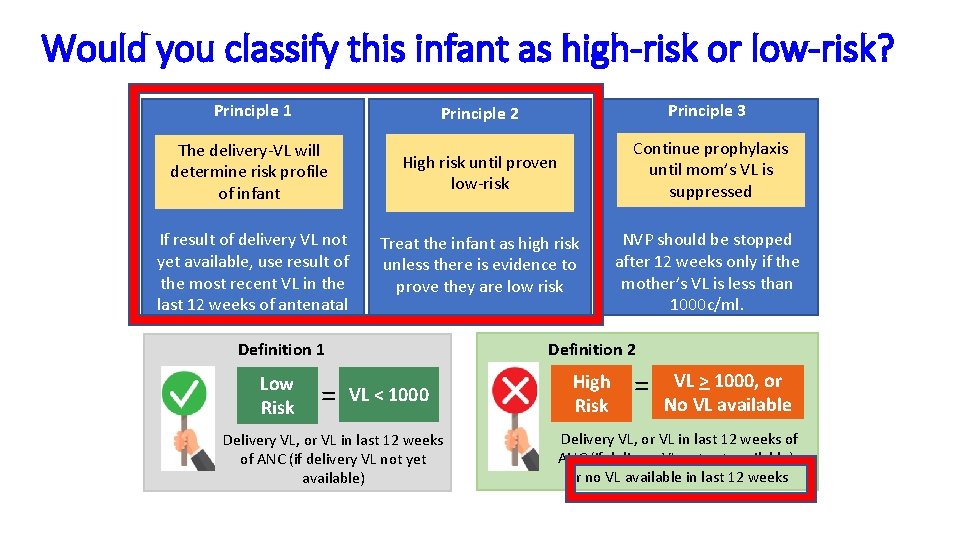
Would you classify this infant as high-risk or low-risk? Principle 1 Principle 2 Principle 3 The delivery-VL will determine risk profile of infant High risk until proven low-risk Continue prophylaxis until mom’s VL is suppressed Treat the infant as high risk unless there is evidence to prove they are low risk NVP should be stopped after 12 weeks only if the mother’s VL is less than 1000 c/ml. If result of delivery VL not yet available, use result of the most recent VL in the last 12 weeks of antenatal care Definition 1 Low Risk = Definition 2 VL < 1000 Delivery VL, or VL in last 12 weeks of ANC (if delivery VL not yet available) High Risk = VL > 1000, or No VL available Delivery VL, or VL in last 12 weeks of ANC (if delivery VL not yet available), or no VL available in last 12 weeks
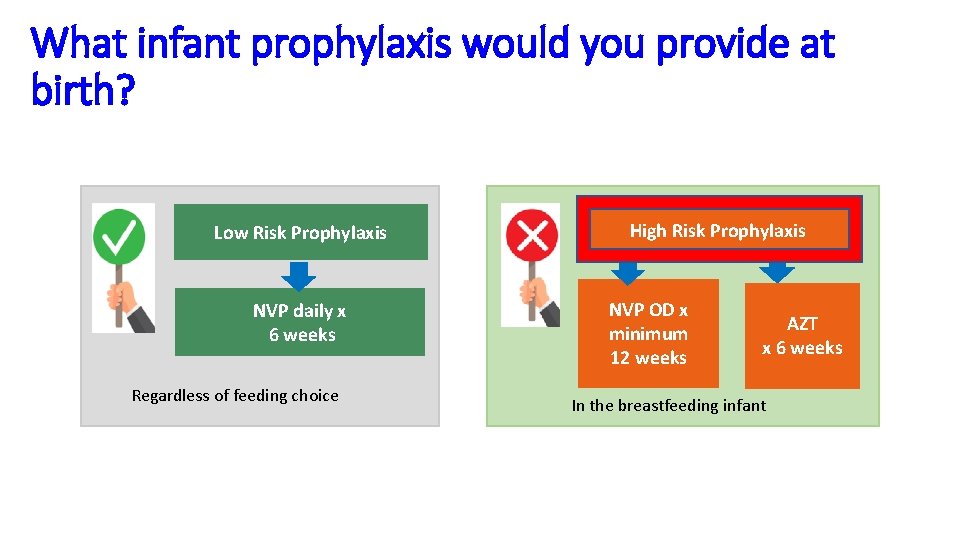
What infant prophylaxis would you provide at birth? Low Risk Prophylaxis NVP daily x 6 weeks Regardless of feeding choice + High Risk Prophylaxis NVP OD x minimum 12 weeks AZT x 6 weeks In the breastfeeding infant

Under what circumstances might you amend your initial assessment of risk and /or the duration of infant prophylaxis? • We made an initial assessment that the infant is high risk, as no VL was available in the last 12 weeks prior to delivery • Accordingly, we initiated the infant on high risk prophylaxis • The results of the delivery VL should be checked at 3 -6 -day postnatal visit. When the delivery-VL result is known, the infant can be re-classified as high/ low-risk and prophylaxis adjusted accordingly.

Scenario 1: At the 3 -6 day post-natal visit, the delivery VL is found to be undetectable • If the delivery VL is < 1000 c/ml, re-classify the infant as lowrisk stop AZT, and complete NVP for a total of 6 weeks

Scenario 2: At the 3 -6 day post-natal visit, the delivery VL is found to be 2600 c/ml If VL > 1000 c/ml, the classification of high-risk is confirmed • continue high risk prophylaxis in the infant • assess mother according to “ABCDE” • implement interventions accordingly to re-suppress the mother’s VL as a matter of urgency • repeat the maternal VL at the 6 weeks integrated visit for the motherinfant pair • results will be reviewed at the 10 week visit • If mother’s VL is confirmed to be less than 1000, infant NVP can be stopped at 12 weeks • If her VL is still above 1000, continue NVP prophylaxis until the mothers VL is suppressed again on 1 st-line, or she has switched to second line, or until one week after all breastfeeding has stopped

Melissa’s 6 month follow-up • Melissa’s delivery VL was undetectable. • Her infant was re-classified as low risk, and completed NVP prophylaxis for 6 weeks • She attended all follow-up visits. Her infant received all his immunisations, growth monitoring, and other routine care. The baby’s HIV PCR tests were negative at birth, at 10 weeks, and at 6 months • She managed to exclusively breastfeed for 6 months, and plans to continue breastfeeding for at least 1 year and hopefully for longer

Melissa’s 6 month follow-up • The result of her VL done at 6 months is 3000 c/ml • Melissa reports that it has been a difficult as the baby wakes a lot at night. Her sleep patterns have been very disrupted, and has been feeling sad a lot lately. As a result, she does sometimes forget to take her tablets Discuss in your groups (5 -10 minutes): • How will you manage Melissa? • Should Melissa stop breastfeeding? • How will you manage her infant?

How will you manage Melissa? Do a thorough assessment of possible causes of her elevated VL • A, B, C, D, E • Melissa’s may be developing post-partum depression. That, together with her chaotic routine may be affecting her adherence. • Implement interventions: • assess and manage her post-partum depression • provide enhanced adherence support, and ensure that she has mechanisms to help her remember to take her treatment (e. g. using an alarm on her cell phone, using her treatment supporter, what to do about missed doses, keeping spare doses in her handbag for emergency situations) • Repeat VL in 4 -6 weeks to see if interventions have worked

Should Melissa stop breastfeeding? • No, she should not stop breastfeeding • Benefits of breastfeeding for child survival balance the risks of possible MTCT • Need to reduce MTCT via BF as far as possible by Strategy 1 (1 st prize) Maternal VL suppression If the mom’s VL is > 1000 we need to get it re-suppressed again as a matter of urgency Strategy 2 (2 nd prize) Infant post-exposure prophylaxis High-risk prophylaxis for baby if mother’s VL not suppressed during breastfeeding Buys time to get mother’s VL suppressed again

How will you manage her infant? • Provide high-risk prophylaxis until mother’s VL is re-suppressed, or until 1 week after all breastfeeding has stopped High-risk prophylaxis during breastfeeding NVP daily for a minimum of 12 w AZT twice daily for 6 w
- Slides: 25