Melanoma Natural History and Principles of Treatment Melanoma








- Slides: 30

Melanoma – Natural History and Principles of Treatment Melanoma Patient Symposium YNHH – Smilow Cancer Hospital Sept 11, 2014

What is Melanoma? • Cancer of cells which are responsible for all types of body pigmentation (melanocytes) • Melanocytes are primarily present in skin but are also present in the eye and mucous membranes (head sinuses, oral cavity, rectum/anus, vulva/vagina) • Some types of melanoma are related to sun exposure and sunburns • Malignant cells gain special properties through genetic (mutations) and other cell changes • • • Uncontrolled growth Ability to travel in blood and lymphatics to other organs Can implant in other organs and divide and grow (metastases) Can remain dormant for years before growth is triggered Dormant state cannot be detected by scans or other tests

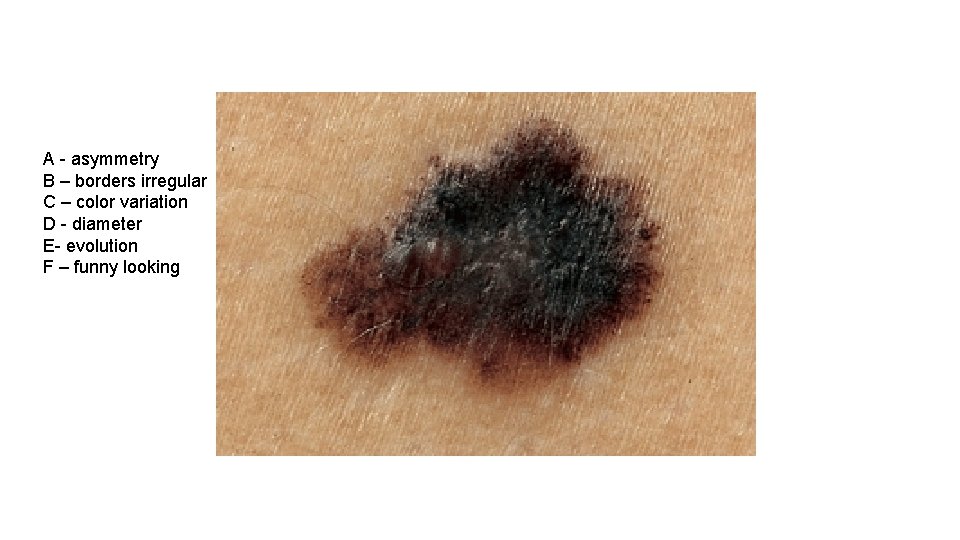
A - asymmetry B – borders irregular C – color variation D - diameter E- evolution F – funny looking

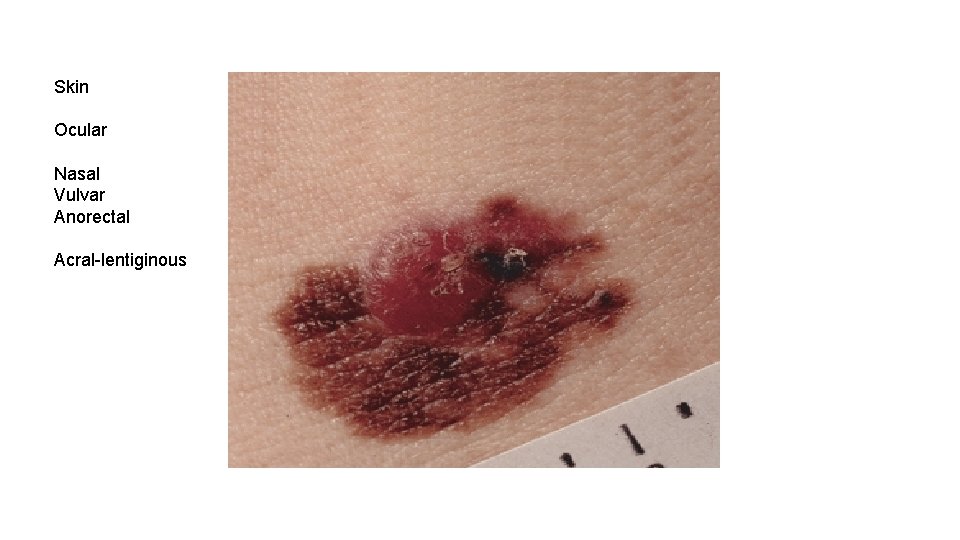
Skin Ocular Nasal Vulvar Anorectal Acral-lentiginous

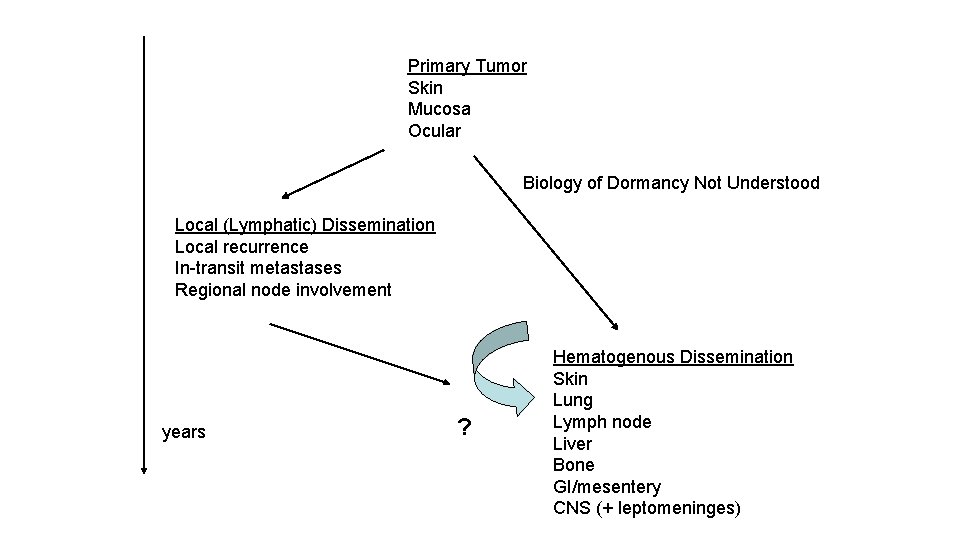
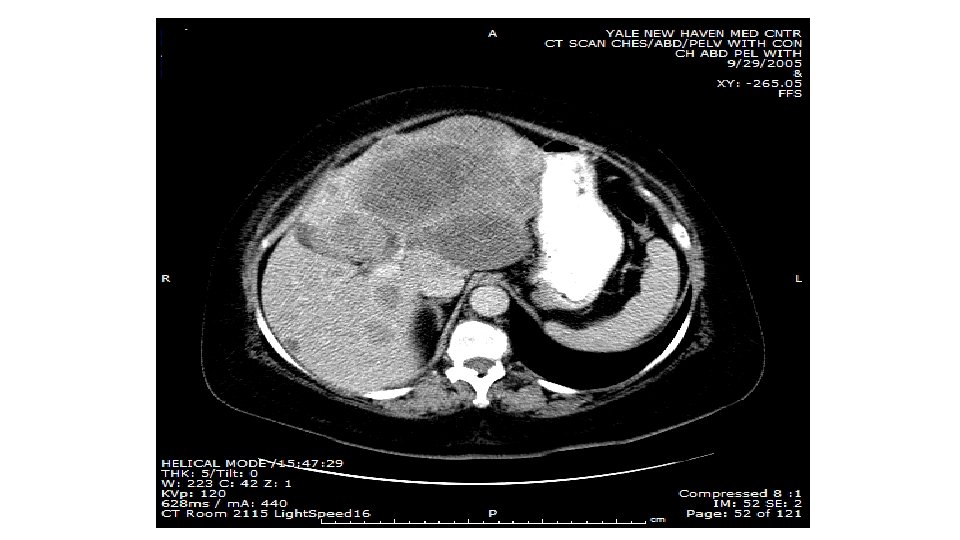
Primary Tumor Skin Mucosa Ocular Biology of Dormancy Not Understood Local (Lymphatic) Dissemination Local recurrence In-transit metastases Regional node involvement years ? Hematogenous Dissemination Skin Lung Lymph node Liver Bone GI/mesentery CNS (+ leptomeninges)




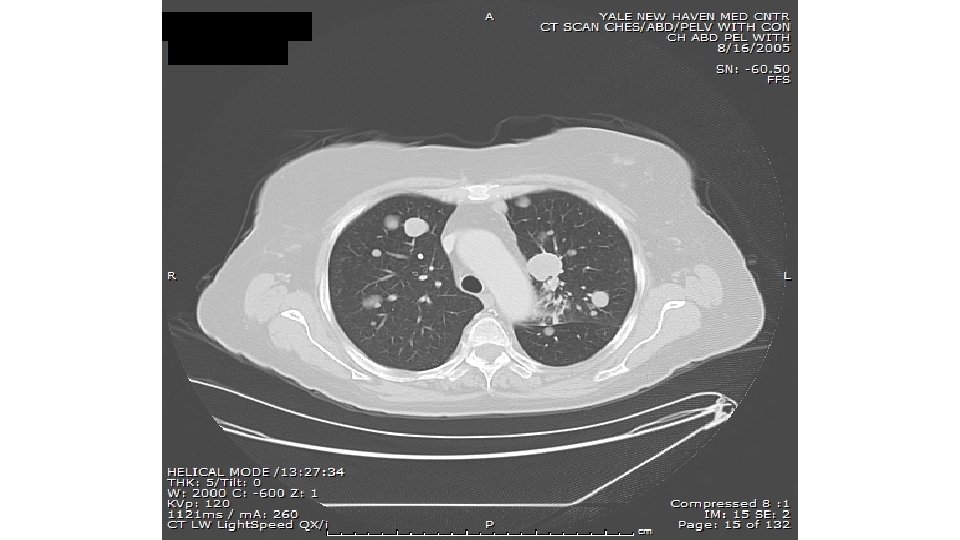
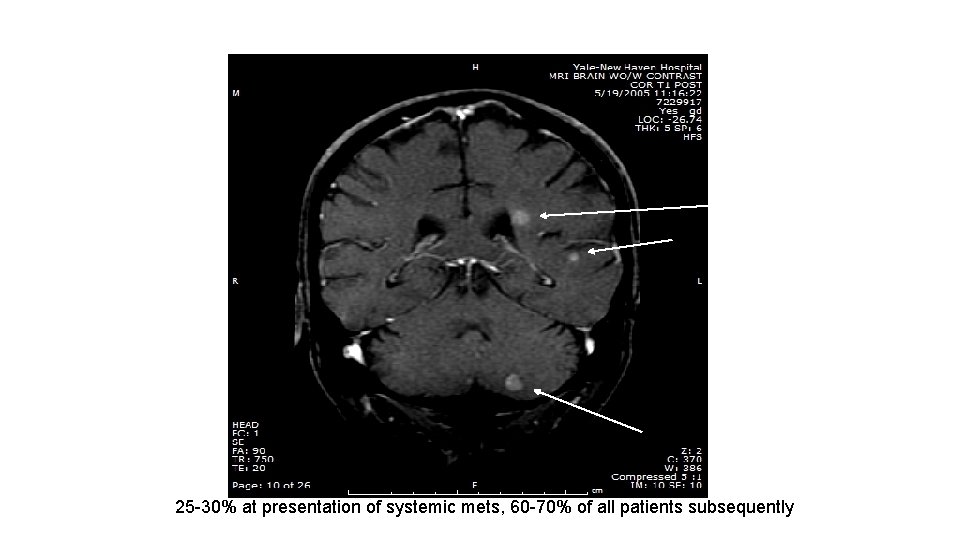
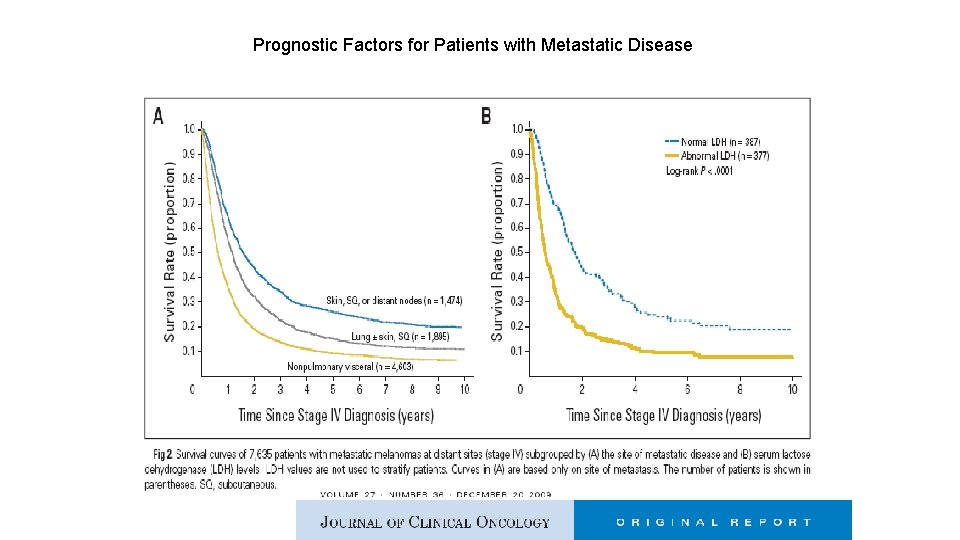
25 -30% at presentation of systemic mets, 60 -70% of all patients subsequently

Treatment of Primary Melanoma • Excisional biopsy by dermatologist – Greater than 90% present without distant metastases • Referral to surgeon • Wide local excision – margins ≥ 2 cm for lesions > 1 mm thick • Sentinel Node Biopsy in regional basin – For lesions > 0. 75 thick – For prognostic information – does not affect outcome • In very high risk patients, CT scans or PET scans to rule out distant metastases • Completion lymph node dissection (for positive SLNB)

The Three Important Questions After Complete Resection of Primary Melanoma (and Regional Nodes) • What is my risk that distant metastases will be found in the future? • What can be done to lower the risk that my cancer might recur? • How will I be monitored to detect the cancer if it recurs?

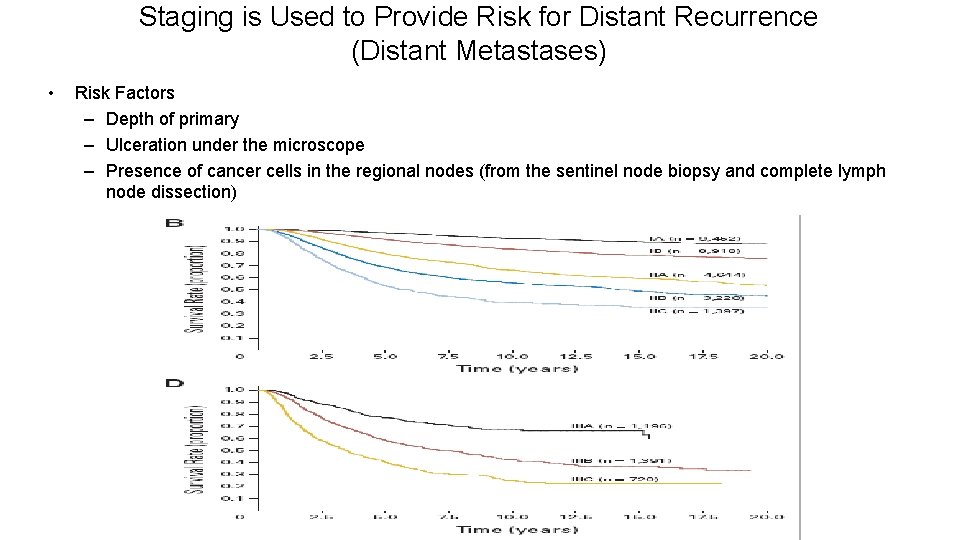
Staging is Used to Provide Risk for Distant Recurrence (Distant Metastases) • Risk Factors – Depth of primary – Ulceration under the microscope – Presence of cancer cells in the regional nodes (from the sentinel node biopsy and complete lymph node dissection)

Prognostic Factors for Patients with Metastatic Disease

Principles of Monitoring for Recurrence • No (good) data to understand the impact of frequency of type of monitoring on outcome – Both determined by risk • Views on monitoring may change as more effective therapies are introduced for advanced disease • Evaluation by oncologist every 3 months to 1 year • History, exam, blood work (CBC, liver function, LDH) • CT scans and/or PET-CT scans in high risk individuals every 6 to 12 months • Usually stop monitoring at 5 -7 years • Dermatology evaluation 2 -4 x yearly for detection of second primaries (10% risk)

Options to Reduce Recurrence Risk • Observation • Interferon-alfa – Different dose and schedules – Administration for up to one year – Increases time to recurrence – Reduces overall risk of recurrence by about 10% – Can induce moderate to severe toxicity in some (fever, chills, fatigue, loss of appetite, depression, difficulty in concentration) • Possible new options – Ipilimumab (Yervoy) – not yet approved, data so far similar to interferon, potential for severe toxicity • Clinical Trials – compare potential better agents to standard of care

Management of Advanced Disease • Treat both the lesions seen on scans and areas of disease that have not yet appeared on scans – Surgery, local injection, or radiation not sufficient to eliminate the disease – Requires systemic (intravenous or oral) medications • Use systemic therapies first that can induce long term remissions • Control pain and manage lesions early that may cause early morbidity (pain, bleeding, limitation of function, unacceptable cosmetic appearance) • Screen the brain at baseline and every 8 -12 weeks • CT scans of chest/abd/pelvis or CT chest + MRI abdomen/pelvis to ‘stage’ disease • Repeat scans every 6 -12 weeks (depends of treatment)

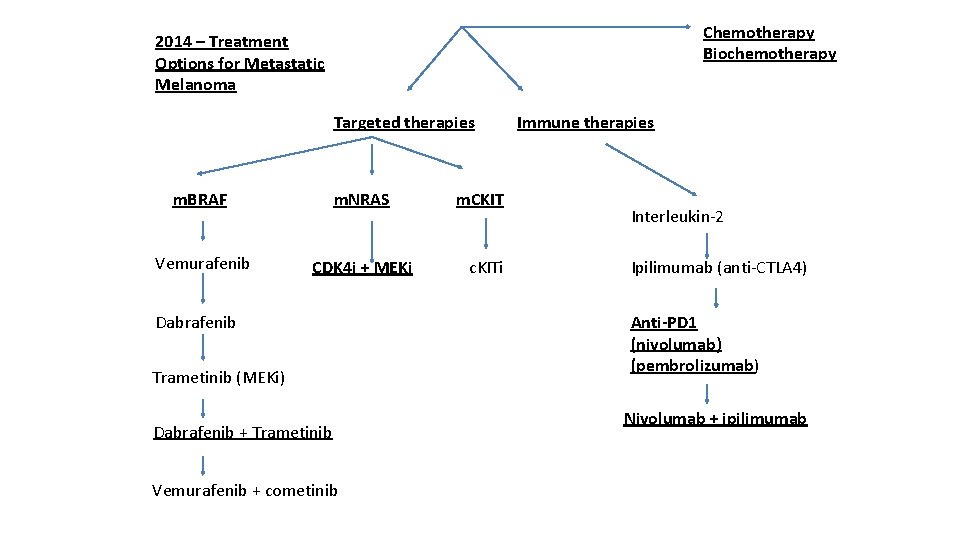
Options for systemic therapies • Clinical Trials • Immune therapies (can give long term remissions) – High dose interleukin-2 (Proleukin) – Ipilimumab (anti-CTLA-4) (Yervoy) – Pembrolizumab (anti-PD-1) (Keytruda) – Nivolumab (anti-PD-1) (Optiva) – approval pending • Targeted therapies (rapid response in most but few have long term control) – BRAF mutation– dabrafenib (dafinlar)/trametinib (mekinist), vemurafenib (zelboraf) – NRAS mutation – investigation MEK + CDK 4 inhibitors – C-kit (mucosal and acral-lentiginous melanomas) – imatinib, dasatinib, sorafenib, others • Cytotoxic Chemotherapy (can work rapidly but only in a few and rarely achieve long term control) – Temozolomide (temodar) or dacarbazine – Carboplatin and paclitaxel – Biochemotherapy

Chemotherapy Biochemotherapy 2014 – Treatment Options for Metastatic Melanoma Targeted therapies m. BRAF m. NRAS Vemurafenib CDK 4 i + MEKi Dabrafenib Trametinib (MEKi) Dabrafenib + Trametinib Vemurafenib + cometinib m. CKIT c. KITi Immune therapies Interleukin-2 Ipilimumab (anti-CTLA 4) Anti-PD 1 (nivolumab) (pembrolizumab) Nivolumab + ipilimumab

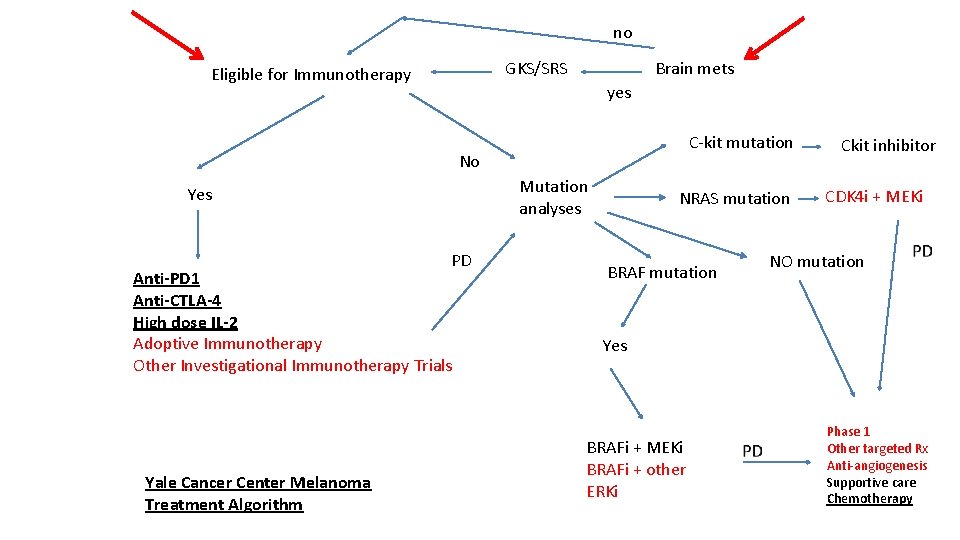
no GKS/SRS Eligible for Immunotherapy Brain mets yes C-kit mutation No Mutation analyses Yes PD Anti-PD 1 Anti-CTLA-4 High dose IL-2 Adoptive Immunotherapy Other Investigational Immunotherapy Trials Yale Cancer Center Melanoma Treatment Algorithm NRAS mutation BRAF mutation Ckit inhibitor CDK 4 i + MEKi NO mutation Yes BRAFi + MEKi BRAFi + other ERKi Phase 1 Other targeted Rx Anti-angiogenesis Supportive care Chemotherapy

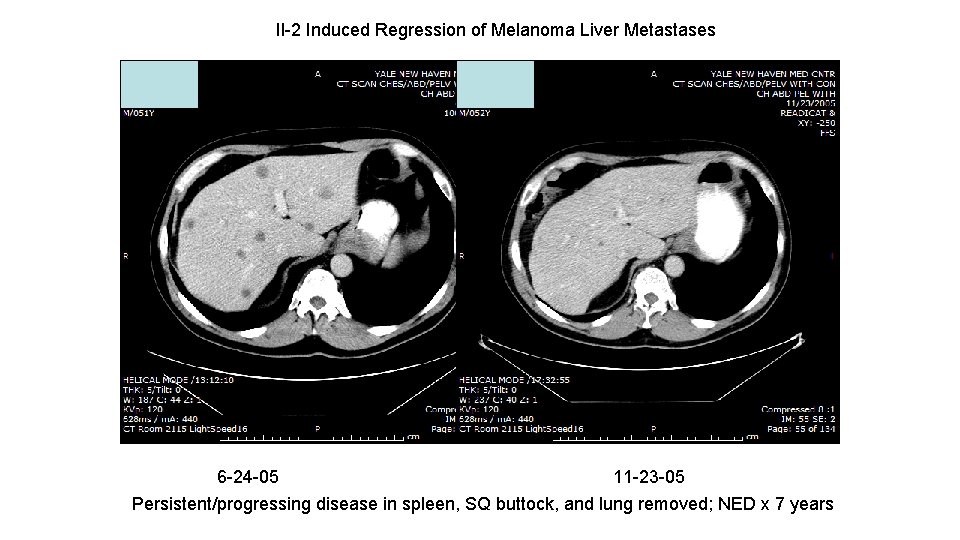
Il-2 Induced Regression of Melanoma Liver Metastases 6 -24 -05 11 -23 -05 Persistent/progressing disease in spleen, SQ buttock, and lung removed; NED x 7 years

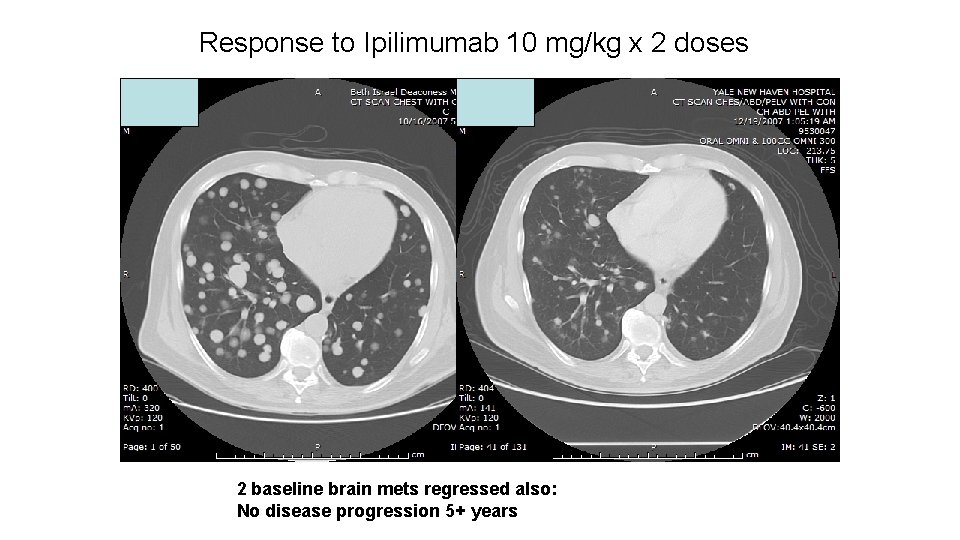
Response to Ipilimumab 10 mg/kg x 2 doses 2 baseline brain mets regressed also: No disease progression 5+ years

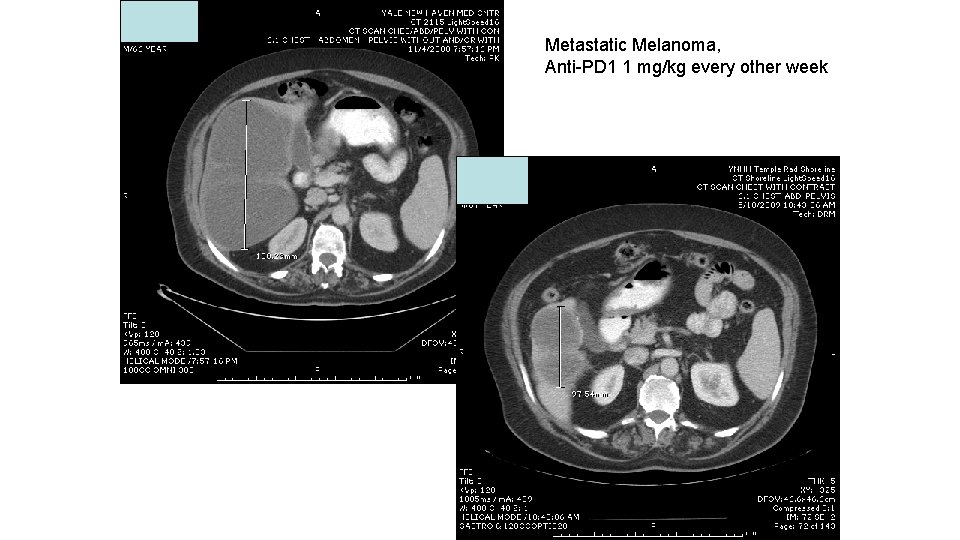
Metastatic Melanoma, Anti-PD 1 1 mg/kg every other week




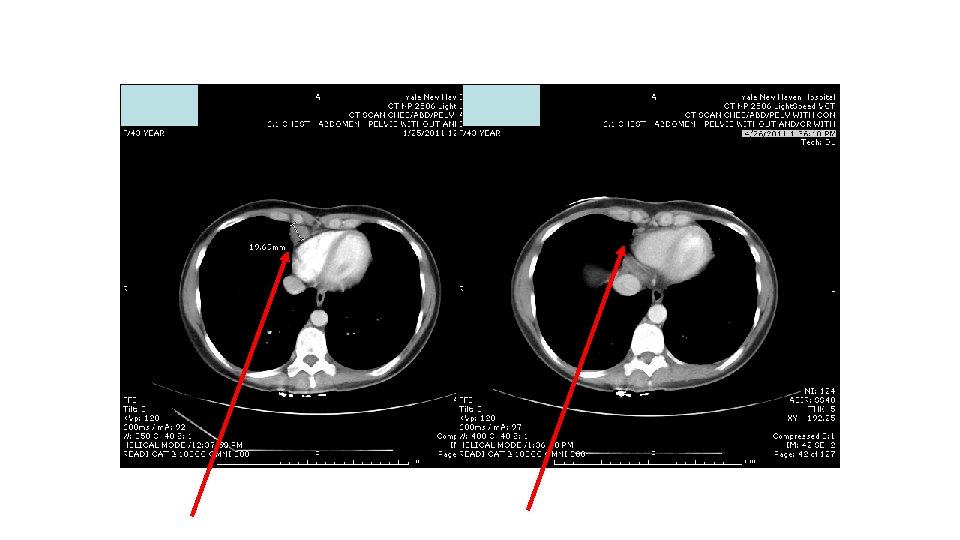
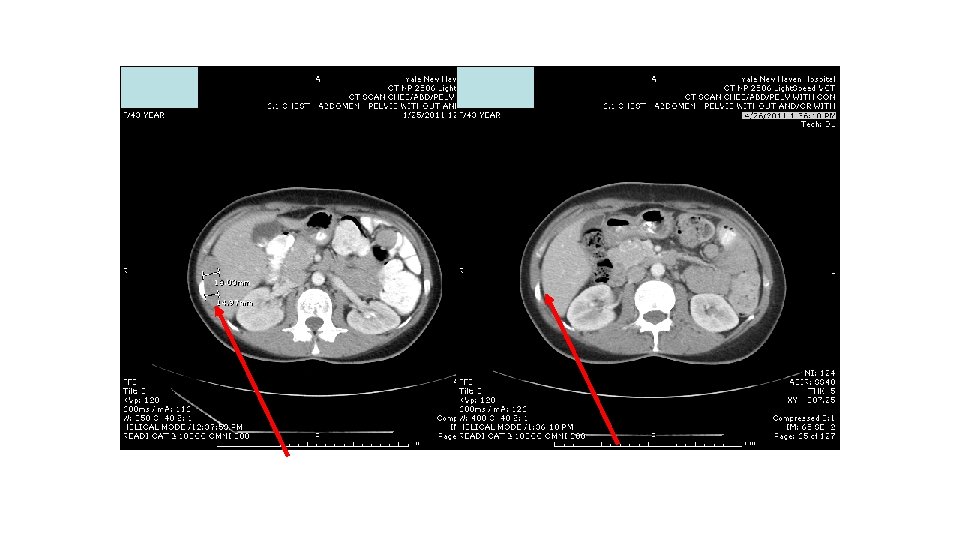
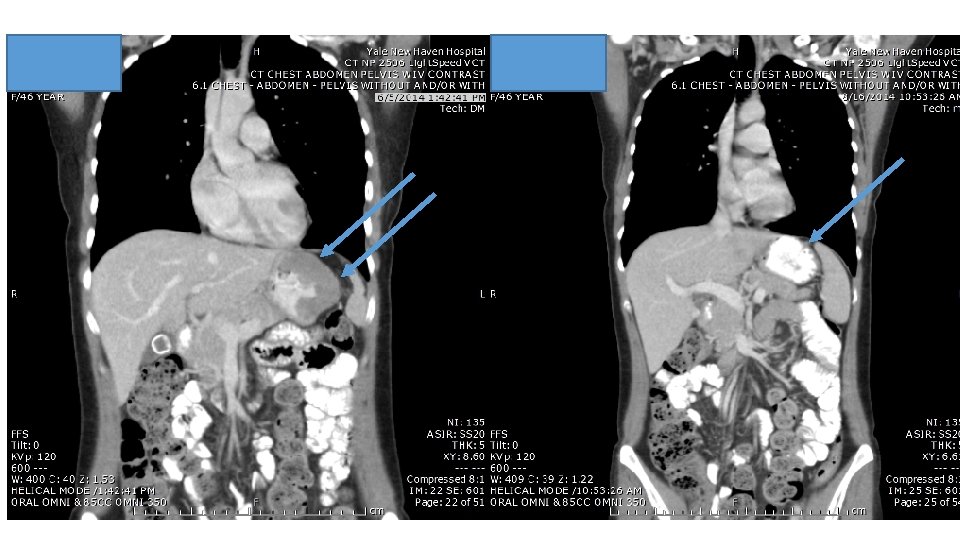
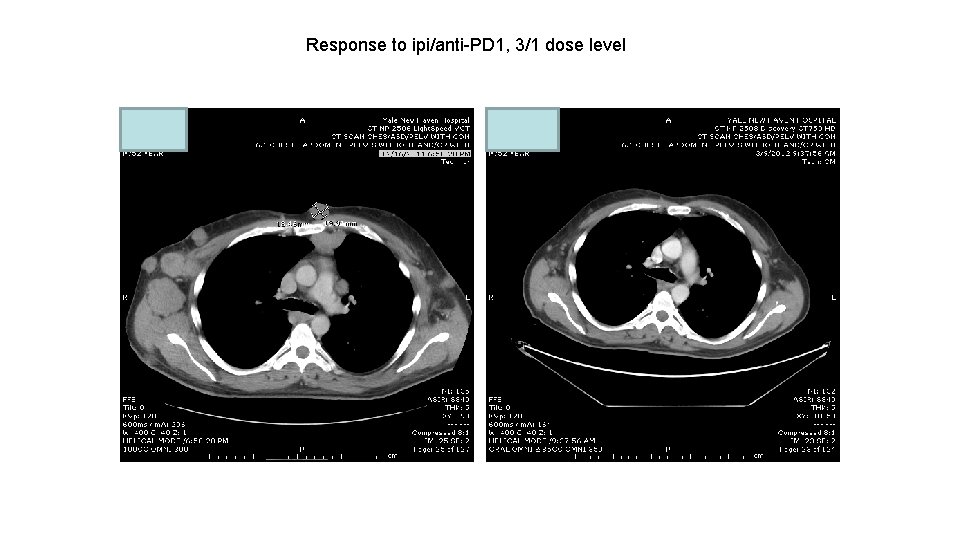
Response to ipi/anti-PD 1, 3/1 dose level

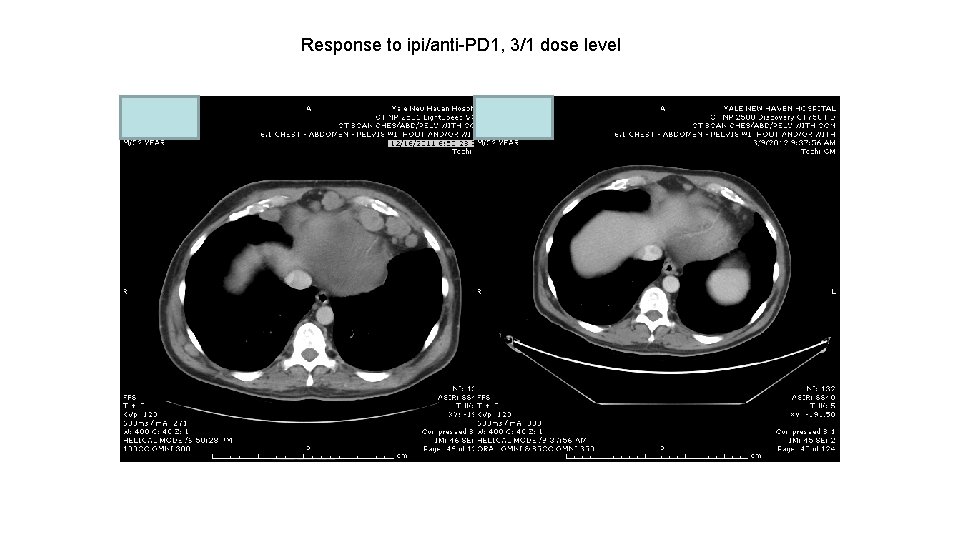
Response to ipi/anti-PD 1, 3/1 dose level

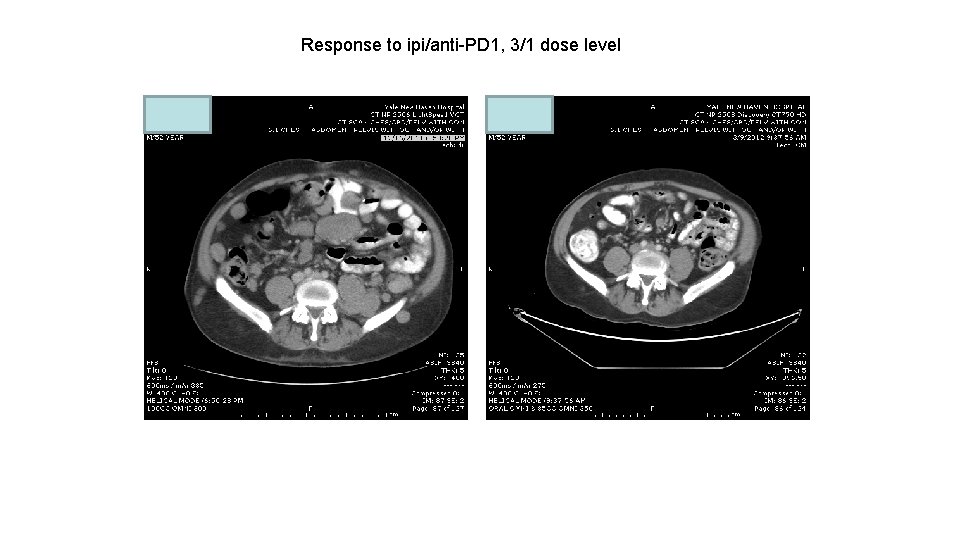
Response to ipi/anti-PD 1, 3/1 dose level

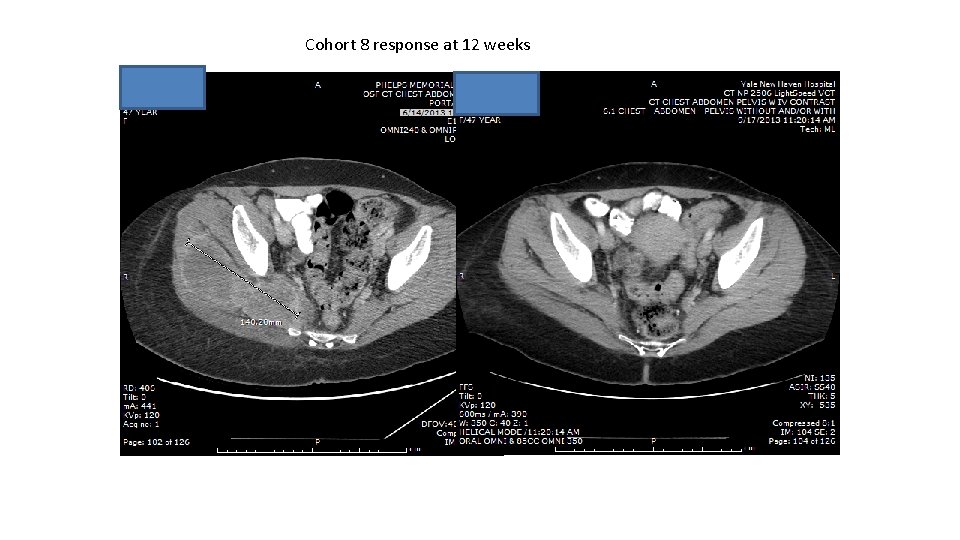
Cohort 8 response at 12 weeks

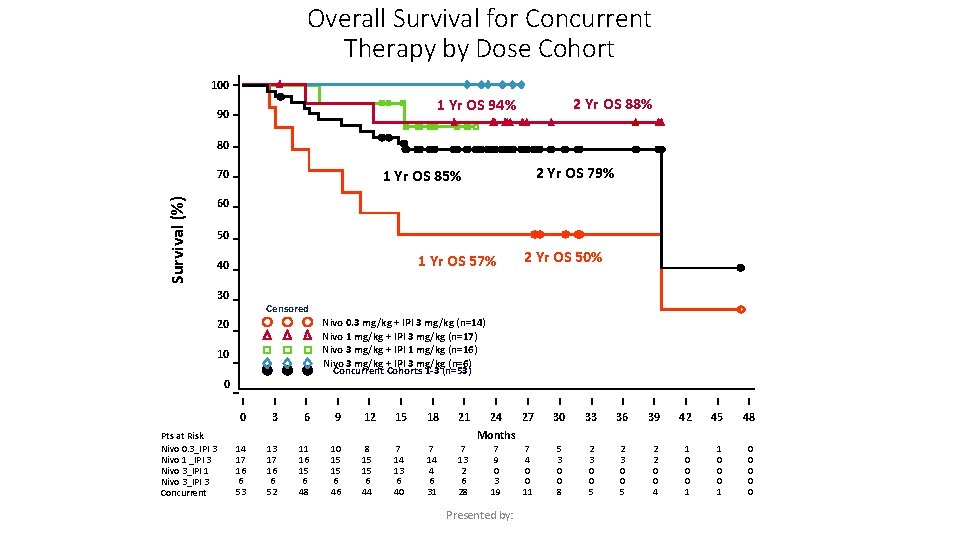
Overall Survival for Concurrent Therapy by Dose Cohort 100 2 Yr OS 88% 1 Yr OS 94% 90 80 Survival (%) 60 50 1 Yr OS 57% 40 30 2 Yr OS 50% Censored Nivo 0. 3 mg/kg + IPI 3 mg/kg (n=14) Nivo 1 mg/kg + IPI 3 mg/kg (n=17) Nivo 3 mg/kg + IPI 1 mg/kg (n=16) Nivo 3 mg/kg + IPI 3 mg/kg (n=6) Concurrent Cohorts 1 -3 (n=53) 20 10 0 0 Pts at Risk Nivo 0. 3_IPI 3 Nivo 1 _IPI 3 Nivo 3_IPI 1 Nivo 3_IPI 3 Concurrent 2 Yr OS 79% 1 Yr OS 85% 70 3 6 9 12 15 18 21 24 27 30 33 36 39 42 45 48 7 4 0 0 11 5 3 0 0 8 2 3 0 0 5 2 2 0 0 4 1 0 0 0 Months 14 17 16 6 53 13 17 16 6 52 11 16 15 6 48 10 15 15 6 46 8 15 15 6 44 7 14 13 6 40 7 14 4 6 31 7 13 2 6 28 7 9 0 3 19 Presented by: