Meet the Elements Periodic Table Organizing the Elements

Meet the Elements Periodic Table: Organizing the Elements Science is the tool of the Western mind and with it more doors can be opened than with bare hands. It is part and parcel of our knowledge and obscures our insight only when it holds that the understanding given by it is the only kind there is. -Carl Jung

Learning Target n Define and identify the groups and classes of the Periodic Table n Analyze how the trends of the periodic table relate to subjects we have already learned. Make connections.

Why is the Periodic Table important to me? n The periodic table is the most useful tool to a chemist. n You get to use it on every test. n It organizes lots of information about all the known elements.

Pre-Periodic Table Chemistry … n …was a mess!!! n No organization of elements. n Imagine going to a grocery store with no organization!! n Difficult to find information. n Chemistry didn’t make sense.

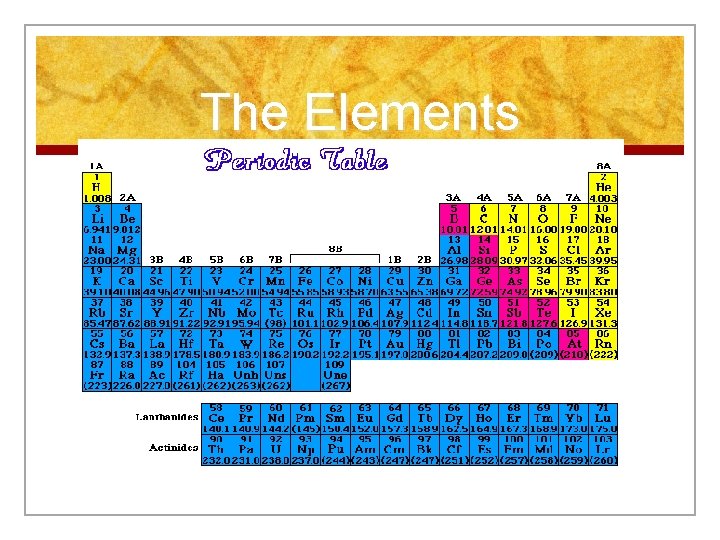
The Genesis n Around 70 elements had been found by mid-1800 s n They were organized in a systematic, logical way. He constructed the first Periodic Table. n Periodic Table: an arrangement of the elements according to similarities in their properties.
The Modern Periodic Table

Organization of Periodic Table n Horizontal rows are called Periods There are 7 periods n The number of elements in a period ranges from 2 to 32. n Within a period the properties of elements change. As you move from period to period the pattern of chemical properties repeats itself. n

Periodic Law n The pattern that the periods provide gives us the Periodic Law which states that when elements are arranged in order of increasing atomic #, there is a periodic repetition of their physical and chemical properties. n Elements that have similar properties end up in the same column as well.

Organization of Periodic Table n The vertical columns of elements are known as Groups; or families. The elements in each group have similar chemical and physical properties. n Each group is identified by a number and a letter (A or B)

White Board Question n. How many periods are there? n. How many groups are there?

Answer n. There are 7 periods n. There are 18 groups

Think Box n. Why do elements in the same group have similar chemical and physical properties?

Answer n Elements in the same group have similar chemical and physical properties because they all have the same amount of valence electrons. Valence electrons control chemical properties (how elements bond and react with other elements) and physical properties (melting/boiling point, volatile, etc…)

Representative Elements (Grp. A) n Group A elements are called the representative elements b/c they exhibit a wide range of both physical and chemical properties. n Can be divided into 3 broad classes. n 1) Metals: electrical conductors, high luster, ductile, and malleable. (1 A, 2 A) n n Group 1 A alkali metals Group 2 A alkaline earth metals
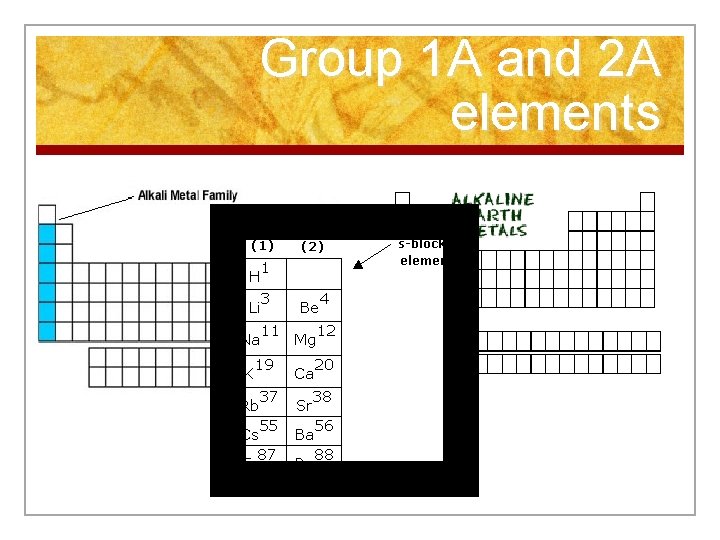
Group 1 A and 2 A elements

Alkali Metals n 1 st column on the periodic table (Group 1) not including hydrogen. n Very reactive metals, always combined with something else in nature (like in salt). n Soft enough to cut with a butter knife

Alkaline Earth Metals n Second column on the periodic table. (Group 2) n Reactive metals that are always combined with nonmetals in nature. n Several of these elements are important mineral nutrients (such as Mg and Ca

Justify-Think-Pair-Share n During WWII the atomic bomb was dropped on Hiroshima and Nagasaki. After the devastation of the explosion the nuclear radiation fallout consisted of mostly Strontium -90. Why was Sr-90 so harmful to humans?

Answer n Sr-90 was so harmful because when it entered the body our vital systems identified it as calcium. It was identified as calcium because it is in the same group as calcium and we know that within a group the elements have similar chemical and physical properties.

Representative Elements 2) Non metals: non lustrous, poor conductors -Two groups of non-metals are given special names -Group 7 A = Halogens -Group 0 or 18 A = Noble Gases (undergo few chemical reactions)
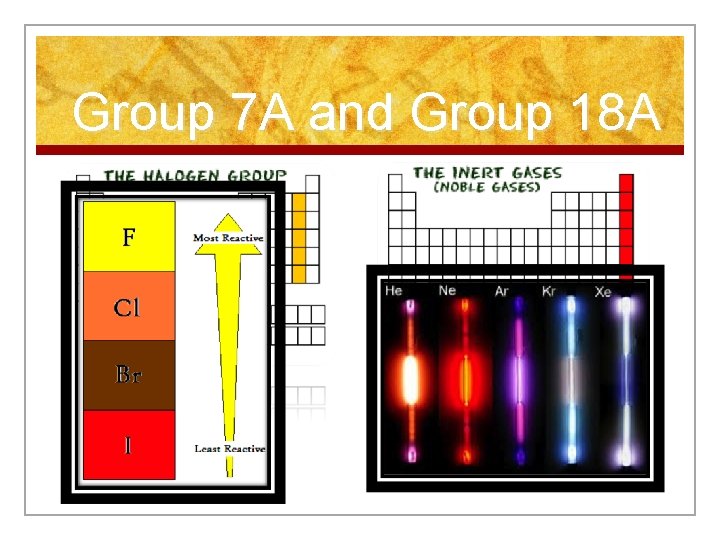
Group 7 A and Group 18 A

Halogens n Elements in group 17 n Very reactive, volatile, diatomic, nonmetals n Always found combined with other element in nature. n Used as disinfectants and to strengthen teeth.

The Noble Gases n Elements in group 18 n VERY unreactive, monatomic gases n Used in lighted “neon” signs n Have shell. a full valence

Representative Elements n 3) Metalloids- elements with properties that are intermediate between those of metals and nonmetals n They are on the staircase!

Metalloids (Semi-metals)

White Board Question n. Is the element phosphorus a metal or a non-metal?

Answer n. Phosphorus metal is a non-

Elements that are not in Group A n Transition n Inner Metals Transition metals (rare earth metals) n These two classes of metals make up the Group B Elements.
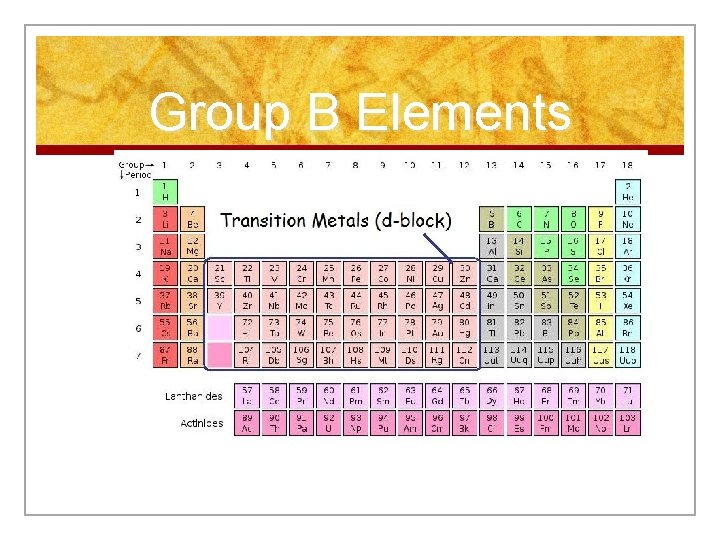
Group B Elements
Transition Metals n Elements in groups 312 n Less reactive harder metals n Includes metals used in jewelry and construction. n Metals used “as metal. ”
The Elements
- Slides: 31