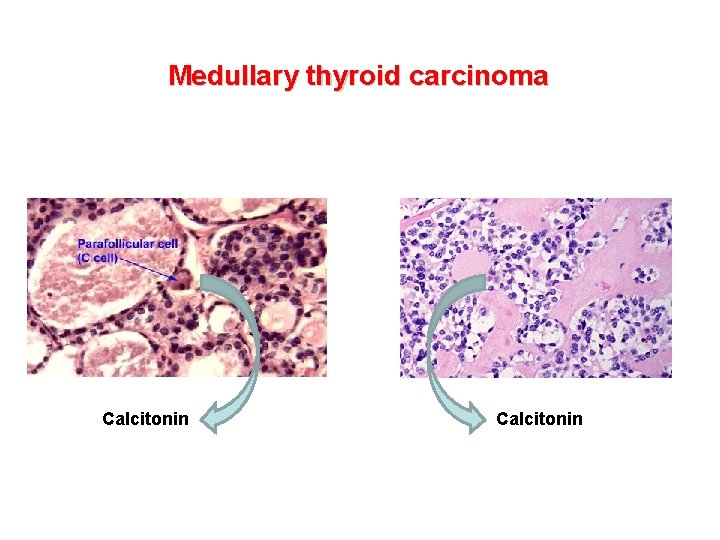
Medullary thyroid carcinoma Calcitonin Medullary thyroid carcinoma MTC


















- Slides: 50

Medullary thyroid carcinoma Calcitonin

Medullary thyroid carcinoma • MTC is a malignant tumor arising from thyroid C cells • It represents about 5% of all thyroid cancers • It is hereditary in 25% of cases and sporadic in the remaining cases • Familial forms develop in the contest of MEN 2 syndrome (Multiple Endocrine Neoplasia type 2) • Activating germline mutations of the proto-oncogene RET confer predisposition to all forms of familial MTC • Prevalence of hereditary MTC: 2. 5 cases per 100, 000

RET gene • At chromosome 10 q 11. 2 • Spans 60 kb • Contains 21 exons in the ORF • Encodes a 1100 aa protein (Marx SJ, Nature Reviews Cancer 2005)

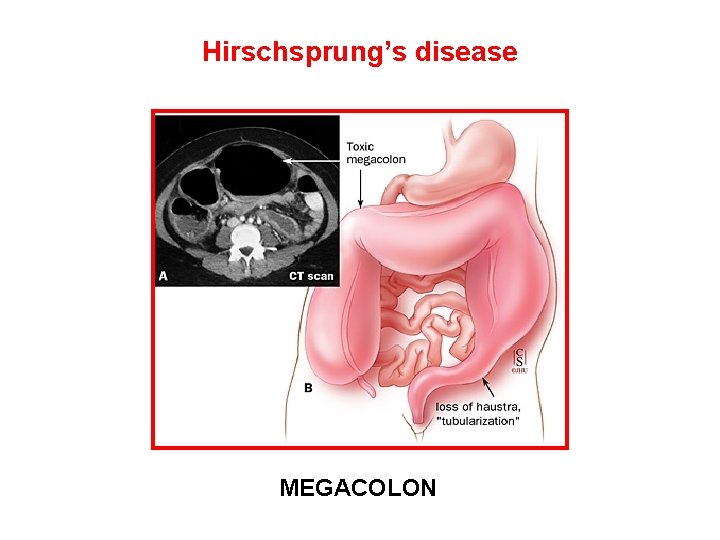
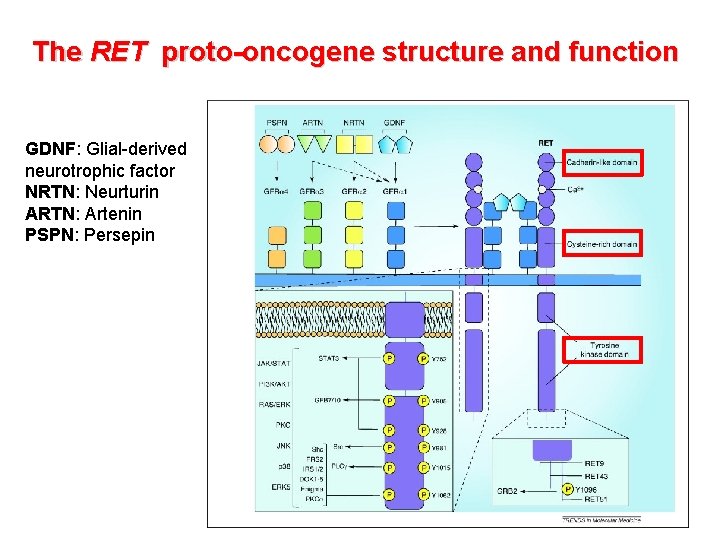
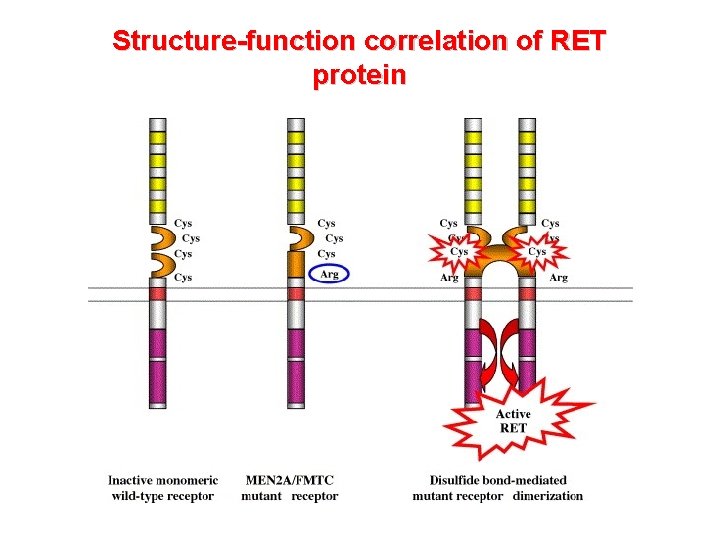
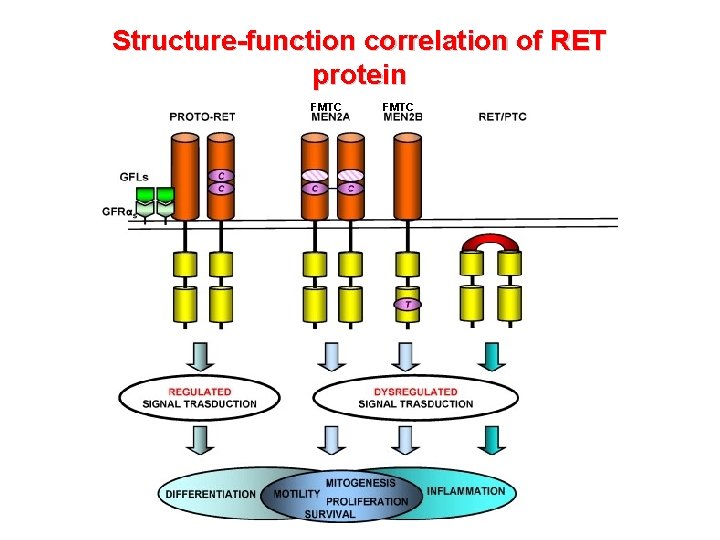
The RET proto-oncogene structure and function • Codes a member of the membrane RTK family • RET protein is a subunit in a plasma-membrane signalling complex that includes 4 ligands and 4 coreceptors • Normaly expressed in developing neural ectoderm and derived tissues of the adult • GDNF/RET is essential for the development of the kidney and the enteric nervous system • RET mutations associated with loss of function are responsible for Hirschsprung’s aganglionosis) disease (intestinal

Hirschsprung’s disease MEGACOLON

The RET proto-oncogene structure and function GDNF: Glial-derived neurotrophic factor NRTN: Neurturin ARTN: Artenin PSPN: Persepin

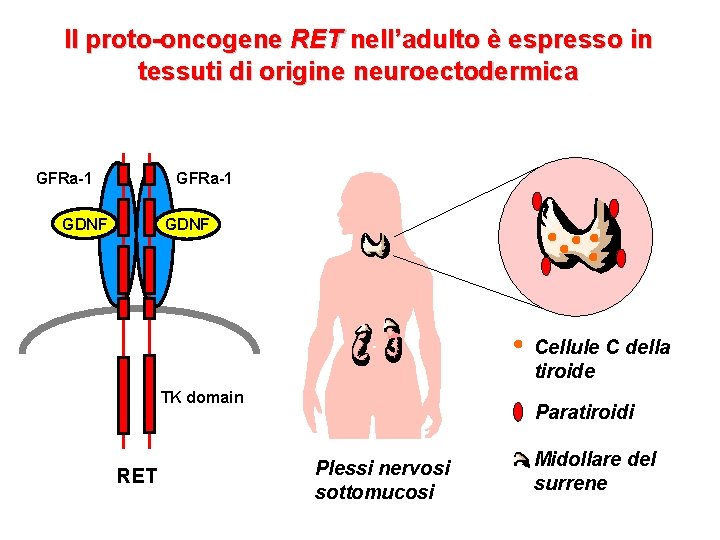
Il proto-oncogene RET nell’adulto è espresso in tessuti di origine neuroectodermica GFRa-1 GDNF Cellule C della tiroide TK domain RET Paratiroidi Plessi nervosi sottomucosi Midollare del surrene

Fenotipi della sindrome MEN 2 FMTC: MTC MEN 2 A: MTC + Feo + Iperplasia o adenoma paratiroidi MEN 2 B: MTC + Feo + Habitus Marfanoide + Neuromatosi MEN 2 A e Lichen Cutaneo Amiloidosico MEN 2 A e Malattia di Hirschsprung

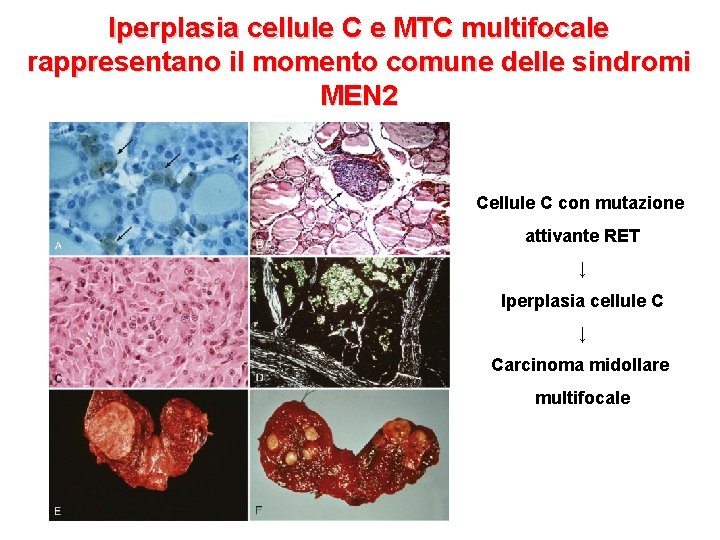
Iperplasia cellule C e MTC multifocale rappresentano il momento comune delle sindromi MEN 2 Cellule C con mutazione attivante RET ↓ Iperplasia cellule C ↓ Carcinoma midollare multifocale

Fenotipi della sindrome MEN 2 FMTC: MTC ≥ 4 membri di una famiglia (penetranza variabile) MEN 2 A: MTC + Feo + Iperplasia o adenoma paratiroidi MEN 2 B: MTC + Feo + Habitus Marfanoide + Neuromatosi MEN 2 A e Lichen Cutaneo Amiloidosico MEN 2 A e Malattia di Hirschsprung

Fenotipi della sindrome MEN 2 FMTC: MTC MEN 2 A: MTC (90 -100%) + Feo (30 -50%) + Iperplasia o adenoma paratiroidi (10 -30%) MEN 2 B: MTC + Feo + Habitus Marfanoide + Neuromatosi MEN 2 A e Lichen Cutaneo Amiloidosico MEN 2 A e Malattia di Hirschsprung

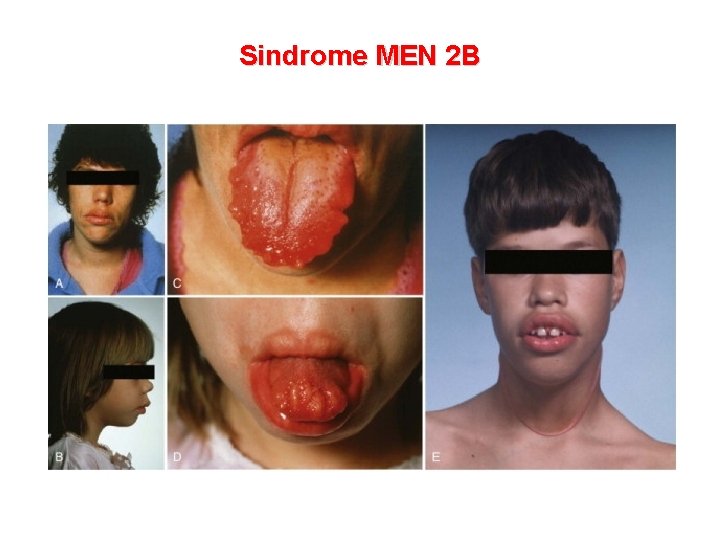
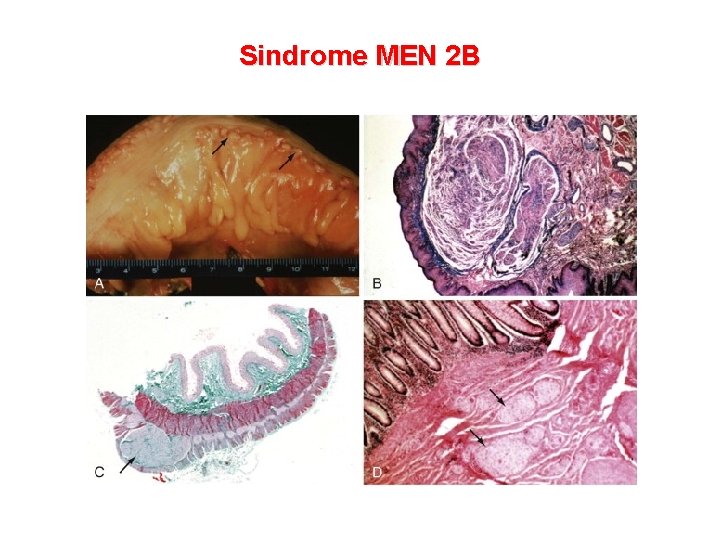
Sindrome MEN 2 B Caratteristiche cliniche Frequenza Neuromi mucosi labbra, palpebre, mucosa orale, lingua, palato e mucosa intestinale 100% Ganglioneuromatosi enterica 100% MTC (molto precoce nell’infanzia) 90 -100% Anomalie scheletriche della colonna (lordosi, cifosi, scoliosi) e pes cavus Comune Nervi corneali inspessiti Comune Facies con labbra ingrossate Comune Habitus marfanoide 65 -75% Feocromocitoma 45 -50%

Sindrome MEN 2 B

Sindrome MEN 2 B

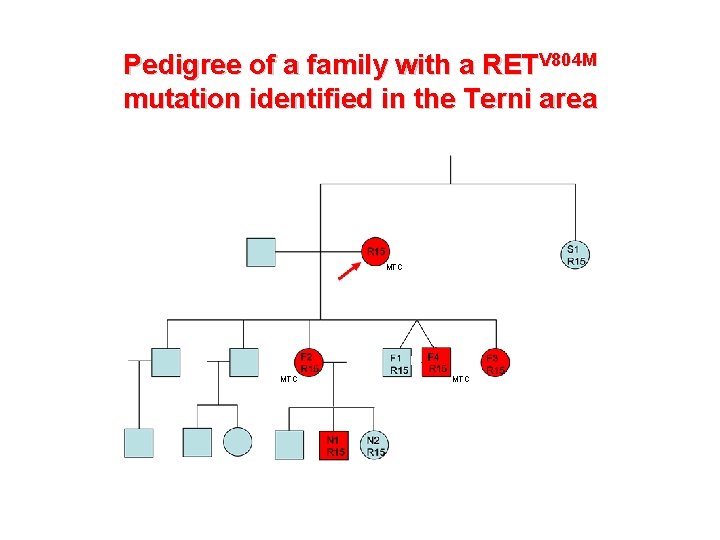
Pedigree of a family with a RETV 804 M mutation identified in the Terni area MTC MTC

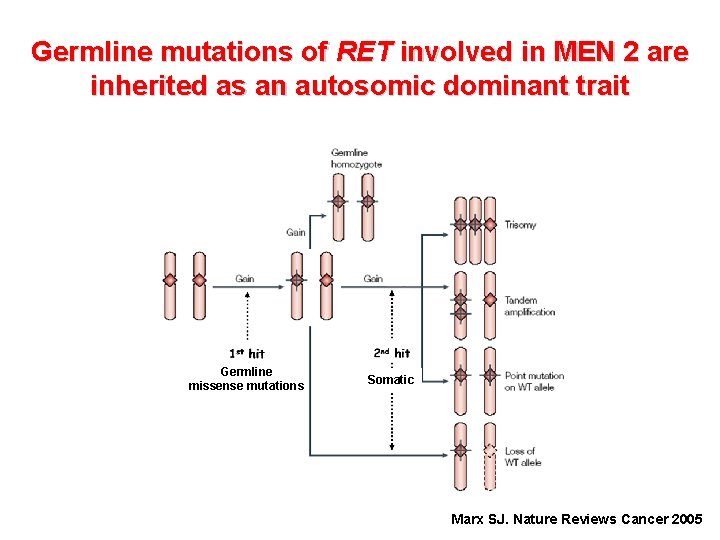
Germline mutations of RET involved in MEN 2 are inherited as an autosomic dominant trait Germline missense mutations 95 -97% of MEN 2 patients harbor a RET mutation Marx SJ. Nature Reviews Cancer 2005

Germline mutations of RET involved in MEN 2 are inherited as an autosomic dominant trait Germline missense mutations Somatic Marx SJ. Nature Reviews Cancer 2005

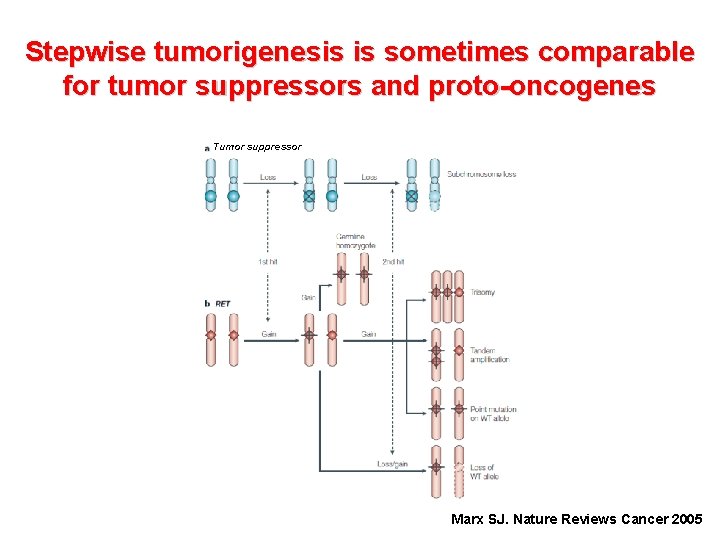
Stepwise tumorigenesis is sometimes comparable for tumor suppressors and proto-oncogenes Tumor suppressor Marx SJ. Nature Reviews Cancer 2005

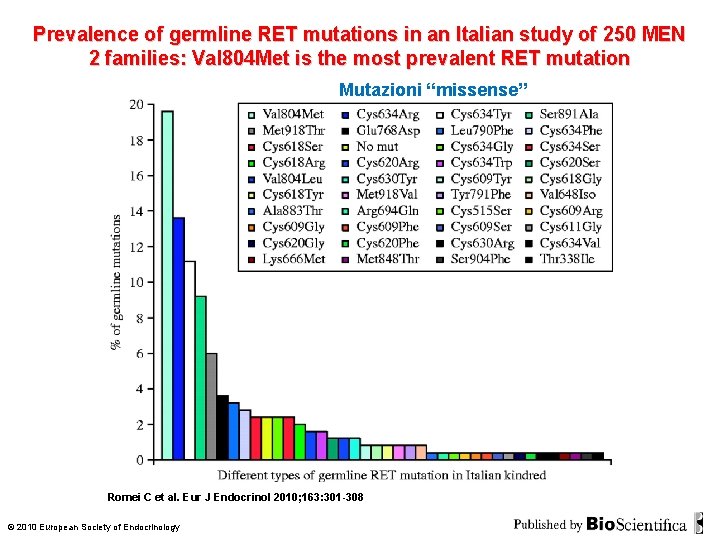
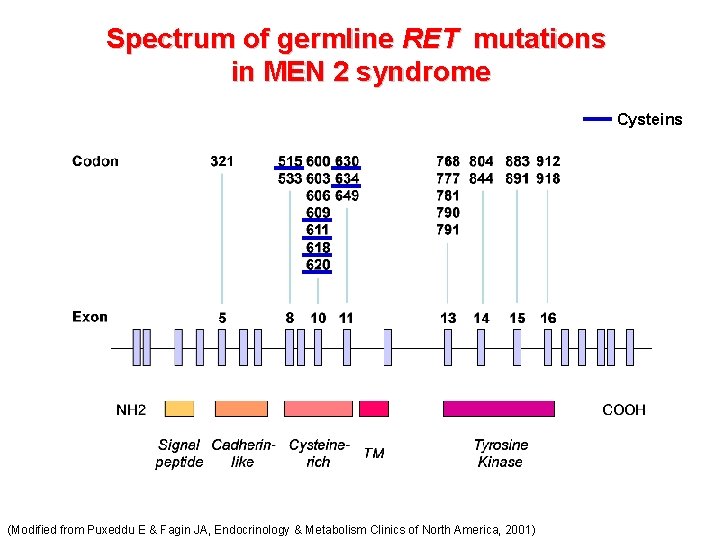
Prevalence of germline RET mutations in an Italian study of 250 MEN 2 families: Val 804 Met is the most prevalent RET mutation Mutazioni “missense” Romei C et al. Eur J Endocrinol 2010; 163: 301 -308 © 2010 European Society of Endocrinology

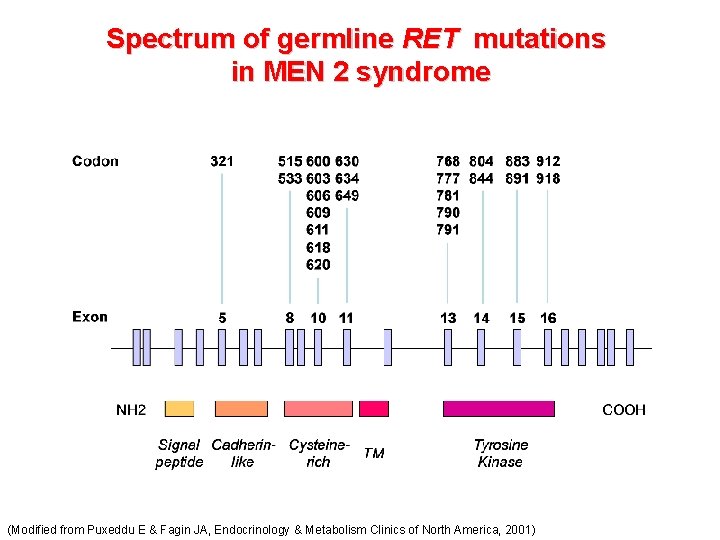
Spectrum of germline RET mutations in MEN 2 syndrome (Modified from Puxeddu E & Fagin JA, Endocrinology & Metabolism Clinics of North America, 2001)

Spectrum of germline RET mutations in MEN 2 syndrome Cysteins (Modified from Puxeddu E & Fagin JA, Endocrinology & Metabolism Clinics of North America, 2001)

Structure-function correlation of RET protein

Structure-function correlation of RET protein FMTC

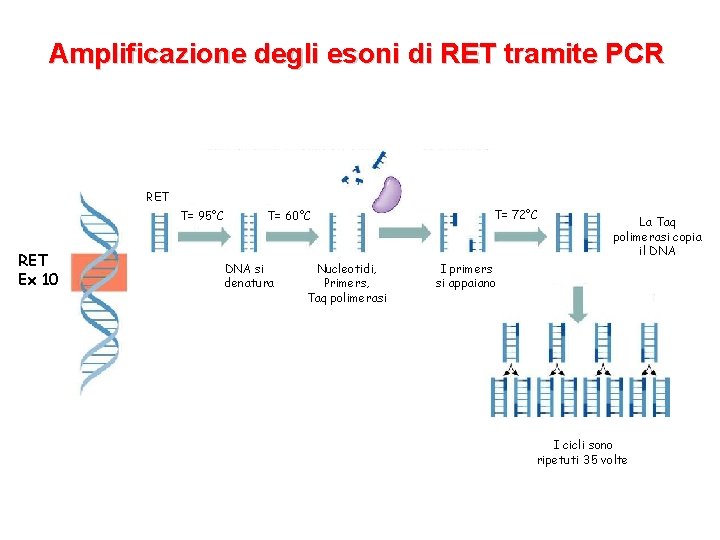
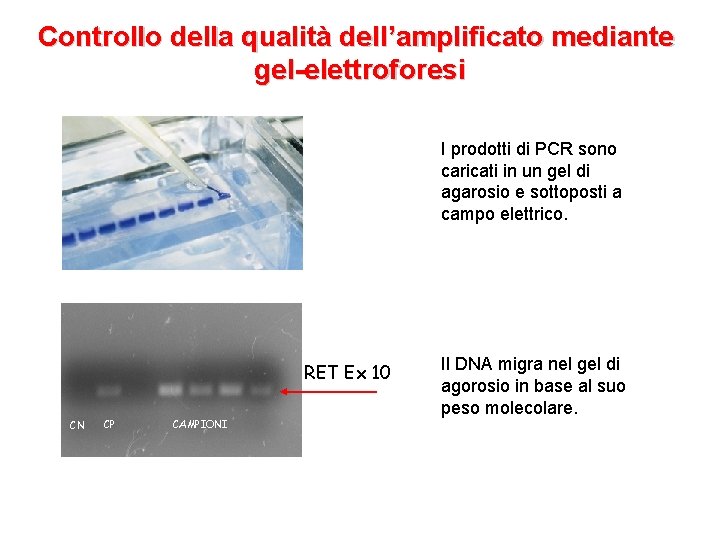
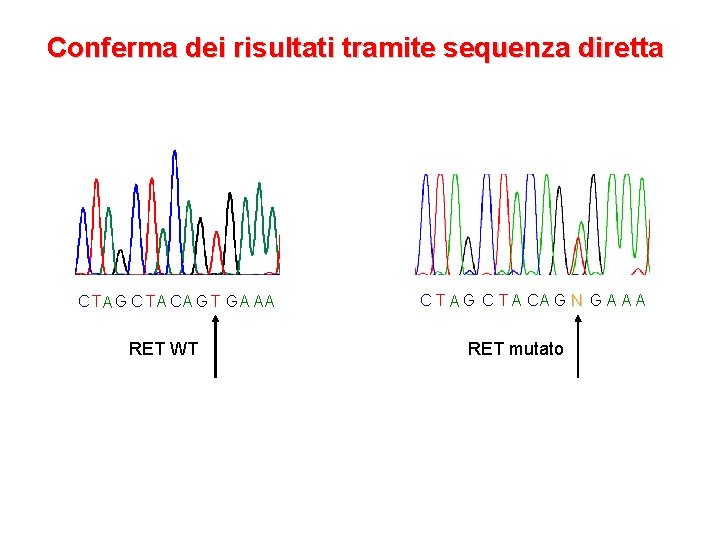
Il test genetico per MEN 2 • Estrazione del DNA da sangue periferico • Amplificazione degli esoni (5, 8), 10, 11, 13, 14, 15 e 16 di RET tramite PCR • Controllo della qualità dell’amplificato mediante elettroforesi su gel di agarosio • Sequenziamento diretto degli esoni del gene RET

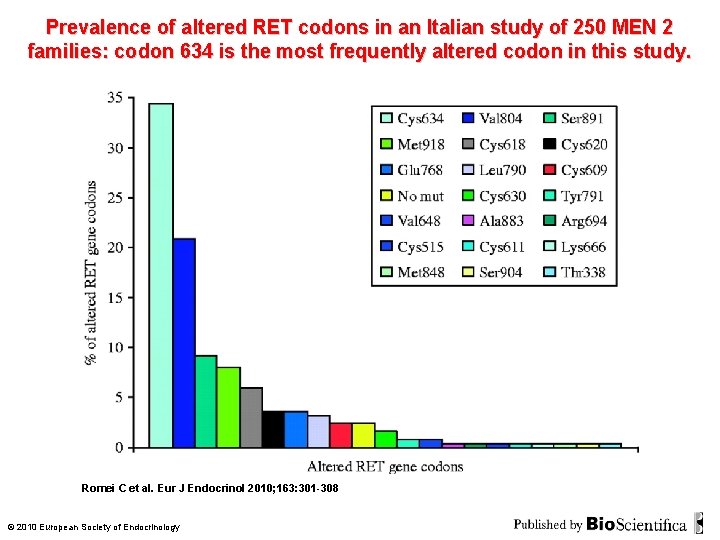
Prevalence of altered RET codons in an Italian study of 250 MEN 2 families: codon 634 is the most frequently altered codon in this study. Romei C et al. Eur J Endocrinol 2010; 163: 301 -308 © 2010 European Society of Endocrinology

Estrazione del DNA

Amplificazione degli esoni di RET tramite PCR RET T= 95°C RET Ex 10 T= 60°C DNA si denatura Nucleotidi, Primers, Taq polimerasi T= 72°C La Taq polimerasi copia il DNA I primers si appaiano I cicli sono ripetuti 35 volte

Controllo della qualità dell’amplificato mediante gel-elettroforesi I prodotti di PCR sono caricati in un gel di agarosio e sottoposti a campo elettrico. RET Ex 10 CN CP CAMPIONI Il DNA migra nel gel di agorosio in base al suo peso molecolare.

Conferma dei risultati tramite sequenza diretta C TA G C TA CA G T GA AA RET WT C T A G C T A CA G N G A A A RET mutato

Valore del test genetico per MEN 2 • Va eseguito in tutti pazienti affetti da MTC o nei familiari di I grado di un soggetto affetto da MEN 2 • Permette di distinguere i pazienti affetti da MEN 2 nell’ambito dei soggetti con MTC • All’interno di una famiglia MEN 2 permette di distinguere i soggetti portatori della mutazione dai non affetti • Permette la diagnosi precoce (già nell’infanzia) dello stato di portatore della malattia, prima dello sviluppo delle neoplasie

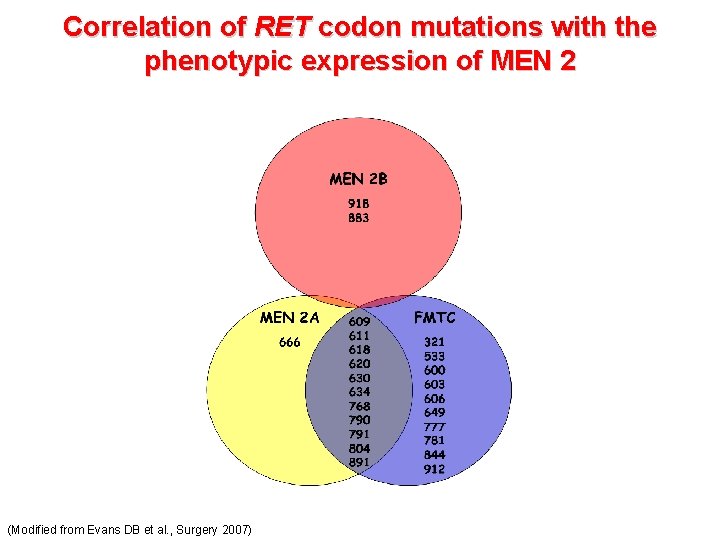
Genotype-phenotype correlation in MEN 2 syndrome • 85% of MEN 2 A patients harbor a germline codon 634 mutation in exon 11 • Hyperparathyroidism in MEN 2 A is associated with mutation at codon 634 (especially Cys 634 Arg substitutions) • Mutation at codon 918 in exon 16 is exclusively associated with MEN 2 B (95% of cases) and represents usually a de novo mutation (Brandi ML et al. , J Clin Endocrinology & Metabolism 2001)

Correlation of RET codon mutations with the phenotypic expression of MEN 2 (Modified from Evans DB et al. , Surgery 2007)

Risk stratification of RET mutations in MEN 2 syndrome • Level D: includes mutations at codons 918 and 883 associated with MEN 2 B. Highest risk for aggressive MTC, youngest age of onset, highest risk of metastases and disease specific mortality • Level C: include mutations at codon 634. Lower, yet still high risk of aggressive MTC. • Level B: include mutations at codons 609, 611, 618, 620 and 630. Lower risk for aggressive MTC. • Level A: include mutations at codon 768, 790, 791, 804, and 891. Carry the ‘‘least high’’ risk. Lower serum Ct levels, lower tumor stage, and a higher rate of biochemical cure than age-matched level B mutation carriers. (Kloos RT et al. , Thyroid 2009 – Medullary thyroid cancer: Management Guidelines of the American Thyroid Association)

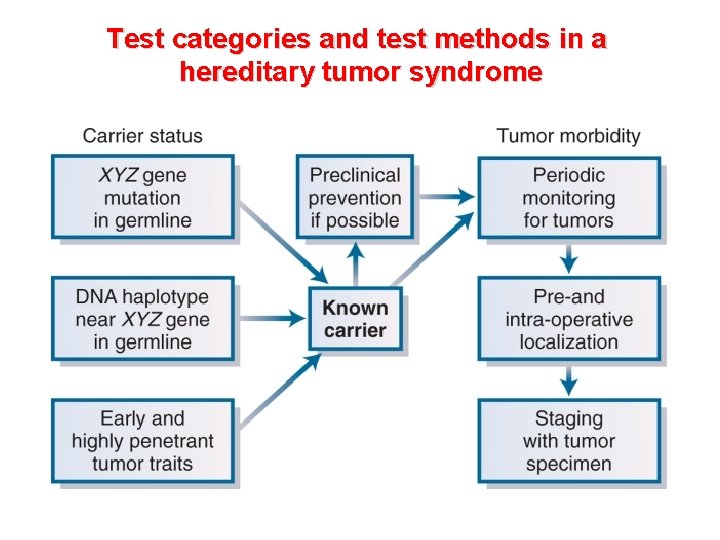
Test categories and test methods in a hereditary tumor syndrome

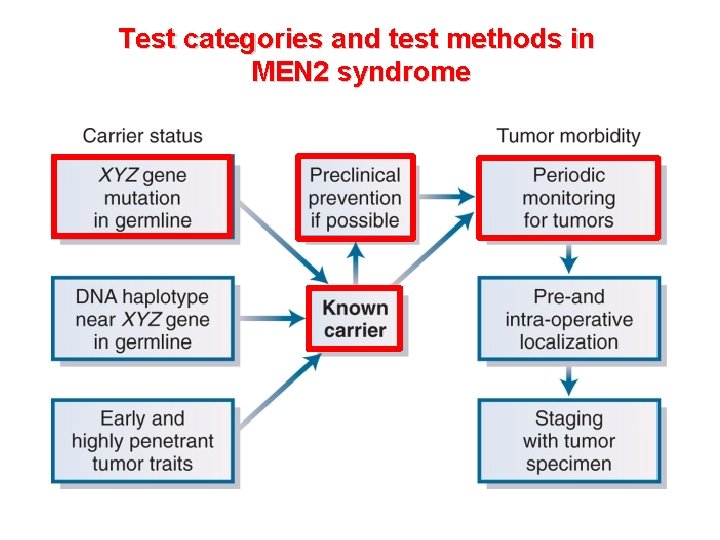
Test categories and test methods in MEN 2 syndrome

Reccomendations for prophylactict thyroidectomy in RET mutations carriers • Carriers of level D mutations: prophylactict total thyroidectomy (PTT) + central compartment lymphectomy (CCL) ASAP or within year 1 of age • Carriers of level C mutation: PTT before age 5 years. CCL after year 10 of age • Carriers of level B mutations: PTT before age 5 years. CCL after year 20 of age • Carriers of level A mutations: PTT beyond age 5 years in the setting of normal annual basal ± stimulated serum calcitonin, normal annual neck US, less aggressive MTC family history, and family preference (Kloos RT et al. , Thyroid 2009 – Medullary thyroid cancer: Management Guidelines of the American Thyroid Association)

Representative protocol of tests and schedules to survey for tumor expression in a (highly likely) carrier of multiple endocrine neoplasia type 2 Tumor MTC PHEO Hyper. PTH Age to Begin Testing (yr) Biochemical Imaging Tests Annually Basal calcitonin MEN 2 A 1 yr Calcium Thyroid US FMTC 3 -5 yrs stimulated calcitonin Urine or plasma metanephrines 5 -7 yrs Urine or plasma None catecholamines Urine VMA Ca++ 10 yrs None PTH

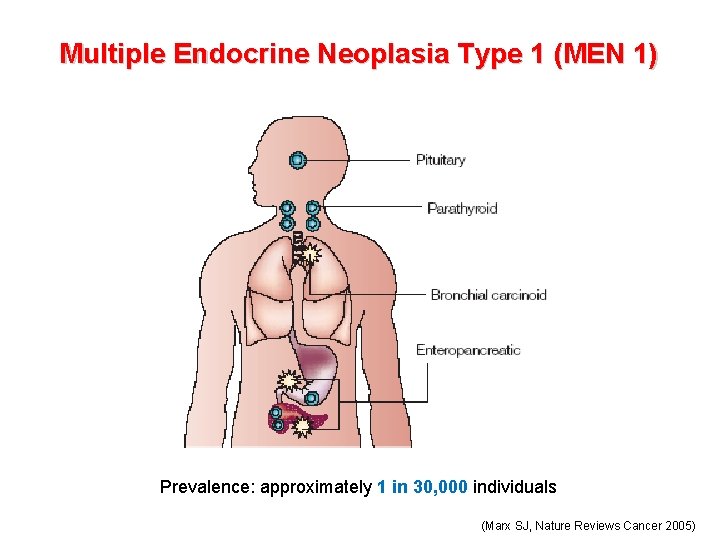
Multiple Endocrine Neoplasia Type 1 (MEN 1) Prevalence: approximately 1 in 30, 000 individuals (Marx SJ, Nature Reviews Cancer 2005)

MEN 1 gene is involved in the development of MEN 1 syndrome • At chromosome 11 q 13 • Spans 9. 8 kb • Contains 10 exons in the ORF • Encodes a 610 aa protein termed Menin (Marx SJ, Nature Reviews Cancer 2005)

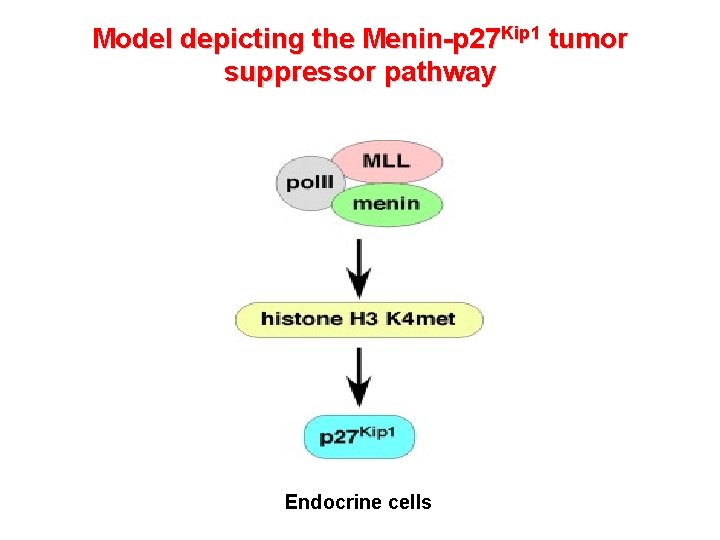
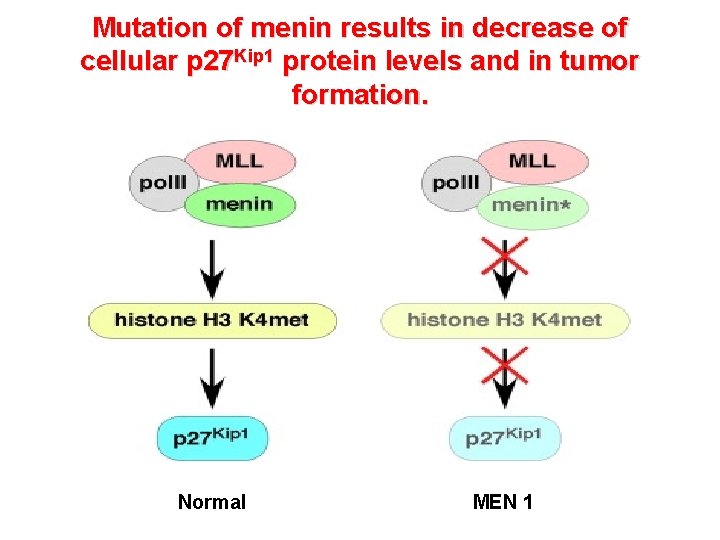
Model depicting the Menin-p 27 Kip 1 tumor suppressor pathway Endocrine cells

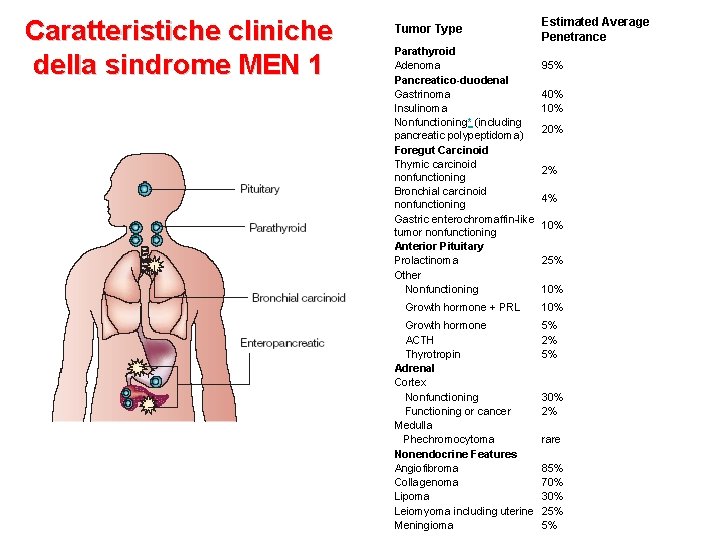
Caratteristiche cliniche della sindrome MEN 1 Tumor Type Estimated Average Penetrance Parathyroid Adenoma Pancreatico-duodenal Gastrinoma Insulinoma Nonfunctioning* (including pancreatic polypeptidoma) Foregut Carcinoid Thymic carcinoid nonfunctioning Bronchial carcinoid nonfunctioning Gastric enterochromaffin-like tumor nonfunctioning Anterior Pituitary Prolactinoma Other Nonfunctioning 95% 40% 10% Growth hormone + PRL 10% Growth hormone ACTH Thyrotropin Adrenal Cortex Nonfunctioning Functioning or cancer Medulla Phechromocytoma Nonendocrine Features Angiofibroma Collagenoma Lipoma Leiomyoma including uterine Meningioma 5% 2% 5% 30% 2% 20% 2% 4% 10% 25% 10% rare 85% 70% 30% 25% 5%

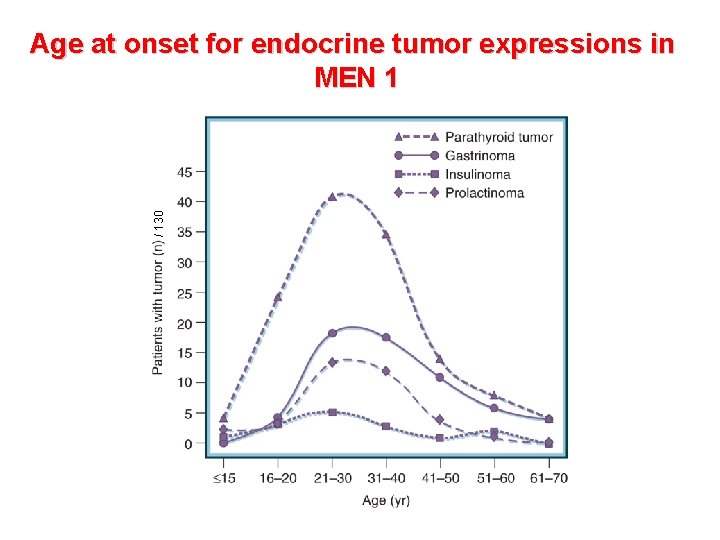
/ 130 Age at onset for endocrine tumor expressions in MEN 1

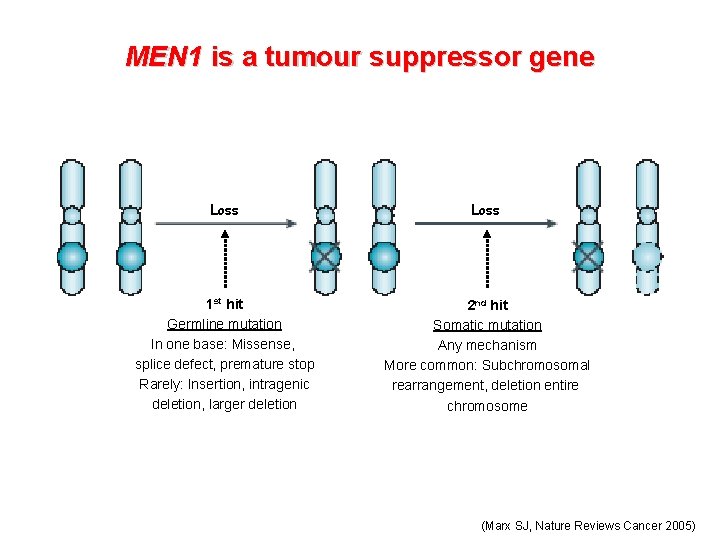
MEN 1 is a tumour suppressor gene Loss 1 st hit Germline mutation In one base: Missense, splice defect, premature stop Rarely: Insertion, intragenic deletion, larger deletion 2 nd hit Somatic mutation Any mechanism More common: Subchromosomal rearrangement, deletion entire chromosome (Marx SJ, Nature Reviews Cancer 2005)

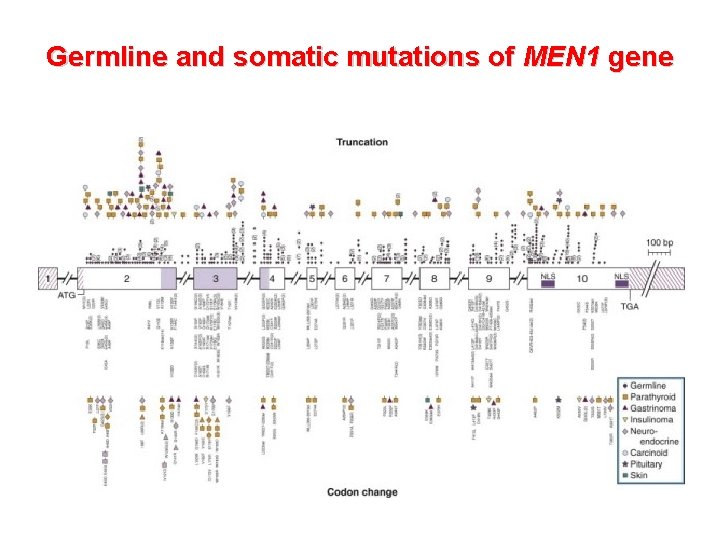
Germline and somatic mutations of MEN 1 gene

Mutation of menin results in decrease of cellular p 27 Kip 1 protein levels and in tumor formation. Normal MEN 1

Genetica sindrome MEN 1 • Ereditarietà autosomica dominante • Identificate oltre 300 mutazioni germinali con penetranza variabile • Grande eterogeneità interfamiliare ma omogenetità in una stessa famiglia • Fenomeno dell’anticipazione

Il test genetico per MEN 1 • Estrazione del DNA da sangue periferico • Amplificazione degli esoni 2 -10 di Menina tramite PCR • Controllo della qualità dell’amplificato mediante elettroforesi su gel di agarosio • Analisi della sequenza degli esoni del gene Menina

Valore del test genetico per MEN 1 • Va eseguito quando c’è il sospetto di MEN 1 (2 tumori tipici) o nei familiari di I grado di un soggetto MEN 1 • Nell’ambito dei soggetti affetti da neoplasie tipiche della sindrome permette di distinguere quelli affetti da MEN 1 • Nell’ambito di una famiglia MEN 1 permette di distinguere i soggetti portatori della mutazione dai non affetti • Permette la diagnosi precoce (già nell’infanzia) dello stato di portatore della malattia, prima dello sviluppo delle neoplasie

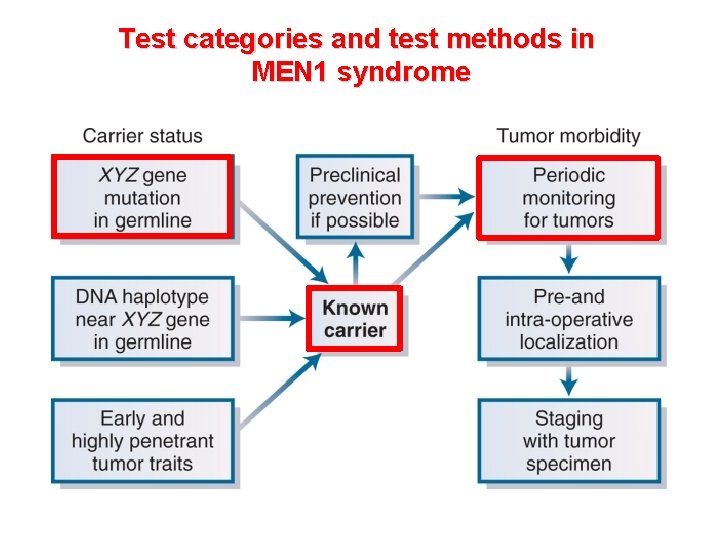
Test categories and test methods in MEN 1 syndrome

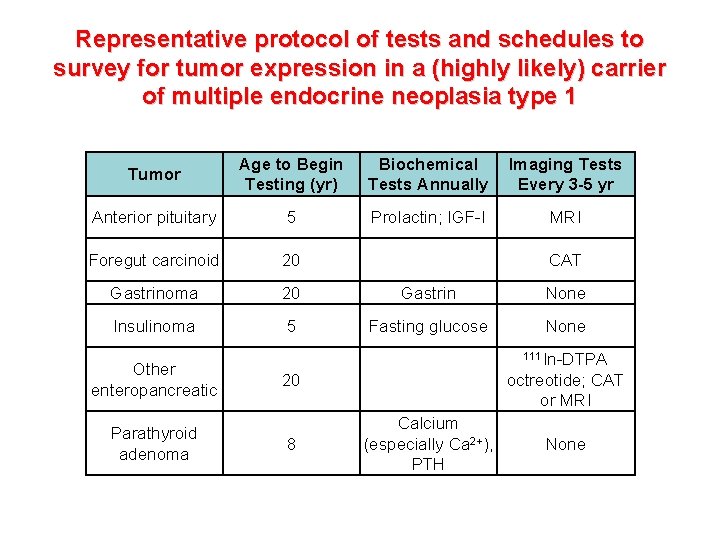
Representative protocol of tests and schedules to survey for tumor expression in a (highly likely) carrier of multiple endocrine neoplasia type 1 Tumor Age to Begin Testing (yr) Biochemical Tests Annually Imaging Tests Every 3 -5 yr Anterior pituitary 5 Prolactin; IGF-I MRI Foregut carcinoid 20 CAT Gastrinoma 20 Gastrin None Insulinoma 5 Fasting glucose None Other enteropancreatic Parathyroid adenoma 111 In-DTPA 20 8 Calcium (especially Ca 2+), PTH octreotide; CAT or MRI None