Medicinal chemistry Third stage Lecture 3 AcidBase Dr

Medicinal chemistry Third stage Lecture 3 Acid-Base Dr. Narmin Hussen B. Sc. In Pharmacy M. Sc. In Ph. chemistry
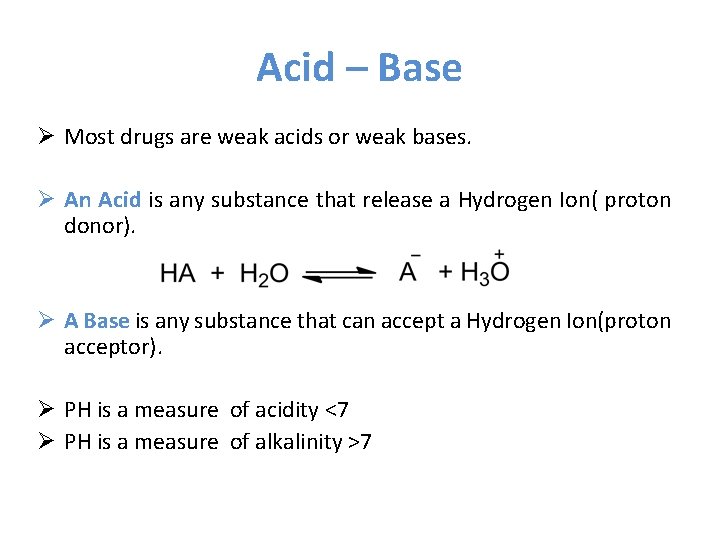
Acid – Base Ø Most drugs are weak acids or weak bases. Ø An Acid is any substance that release a Hydrogen Ion( proton donor). Ø A Base is any substance that can accept a Hydrogen Ion(proton acceptor). Ø PH is a measure of acidity <7 Ø PH is a measure of alkalinity >7
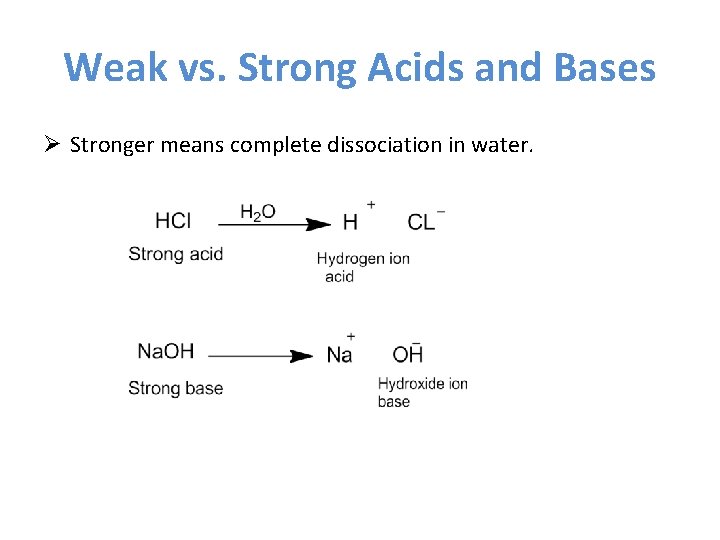
Weak vs. Strong Acids and Bases Ø Stronger means complete dissociation in water.
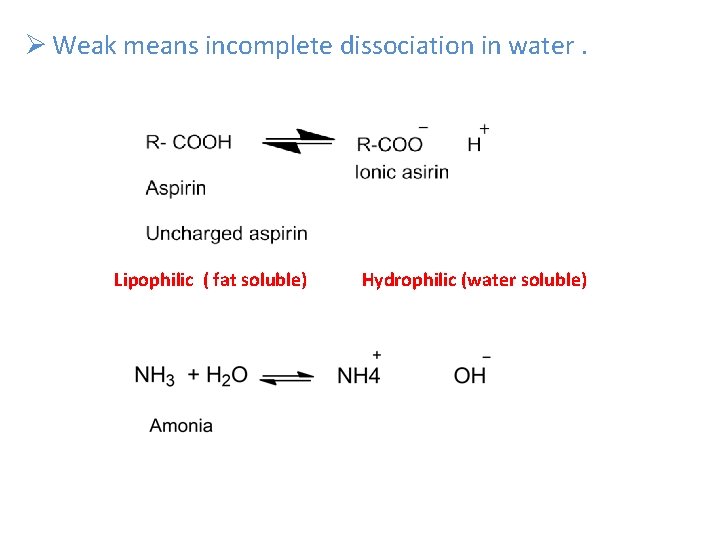
Ø Weak means incomplete dissociation in water. Lipophilic ( fat soluble) Hydrophilic (water soluble)

Acid/Base conjugate Ø Conjugate acid compound formed when an base gains a hydrogen ion. Ø Conjugate base compound formed when an acid loses a hydrogen ion.


Ka &PKa Ø The equilibrium between unionized and ionized forms by the acidity constant Ka(the acid dissociation constant). Ø The PKa is the negative logarithm of Ka. PKa= - log 10 Ka Ø For an acid
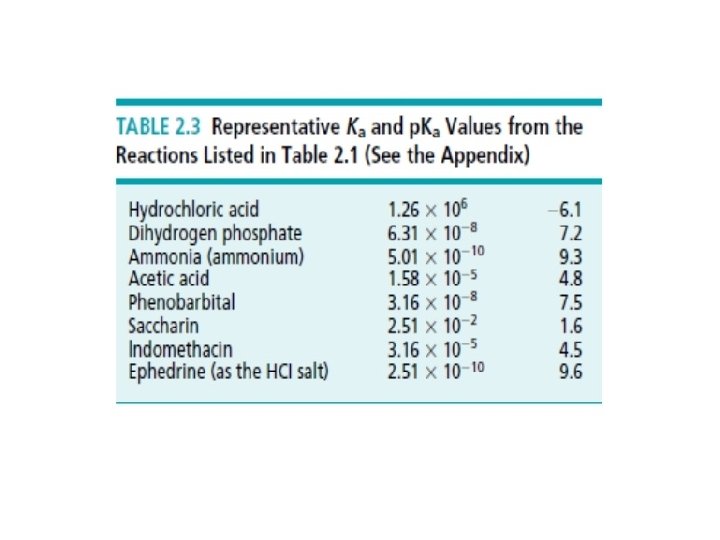

A General rule for determining a chemical is strong or weak acid or base Ø Ø PKa <2: strong acid , PKa 4 to 6 : weak acid , weak conjugate base PKa 8 to 10: very weak acid; conjugate base getting stronger PKa > 12: essentially no acidic properties in water; strong conjugate base Ø The Higher Ka the stronger acid and the lower Ka weaker acid.

Henderson–Hasselbalch equation Ø In chemistry, the Henderson–Hasselbalch equation describes the derivation of p. H as a measure of acidity (using p. Ka, the negative log of the acid dissociation constant).

Examples of calculations 1. What is the ratio of ephedrine to ephedrine HCl (PKa 9. 6) in the intestinal tract at PH 8. 0?

2 - What is the PH of buffer containing 0. 1 M acetic acid (PKa 4. 8) and 0. 8 M sodium acetate ?

Percent Ionization Ø Using the drugs PKa, the formulation or compounding pharmacist can adjust the PH to ensure maximum water solubility(ionic form of the drug) or maximum solubility in non polar media(un-ionic form). Ø Acids can be divided in two types. 1 - 2 -

HA acids include : Ø Ø Inorganic acids (HCl , H 2 SO 4) Enols (barbiturate , hydantoins) Carboxylic acids( low M. wt organic acids, salicylic acids) Amides and imides BH+ acids include : Ø They are all protonated amines.

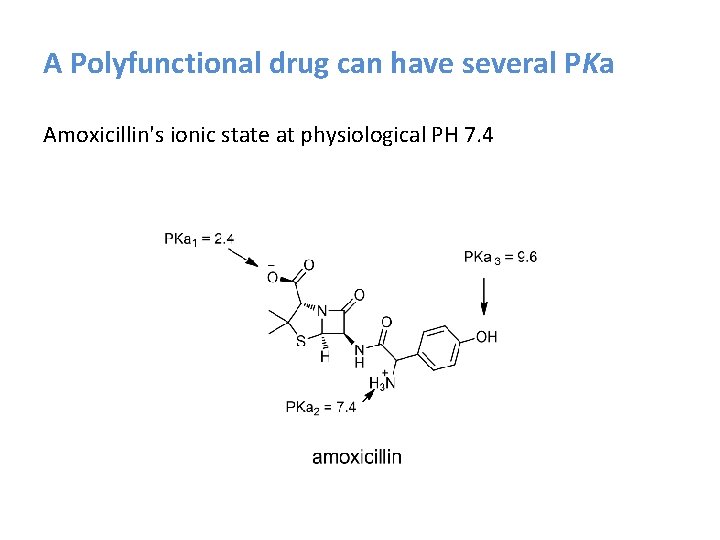
A Polyfunctional drug can have several PKa Amoxicillin's ionic state at physiological PH 7. 4
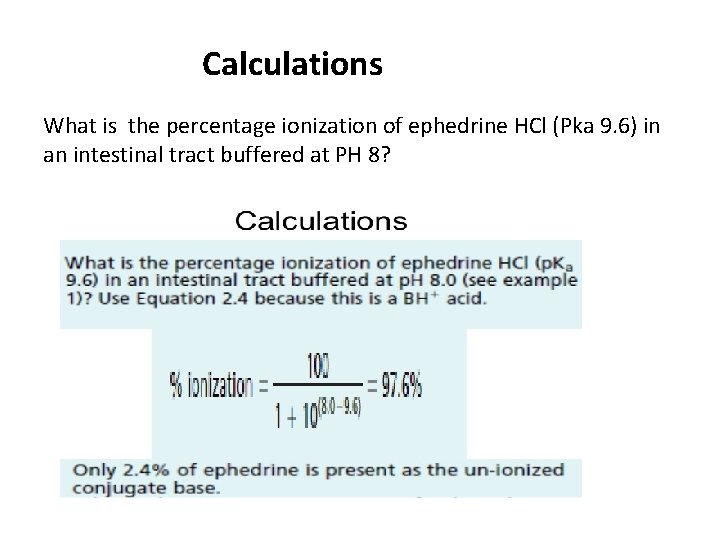
Calculations What is the percentage ionization of ephedrine HCl (Pka 9. 6) in an intestinal tract buffered at PH 8?
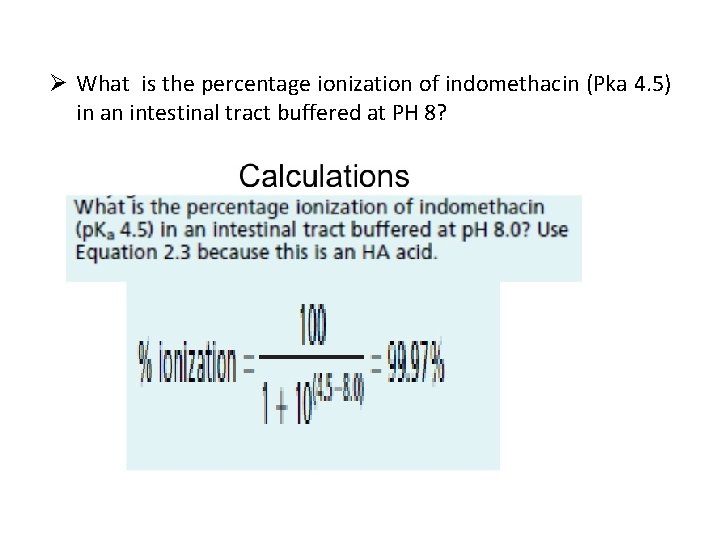
Ø What is the percentage ionization of indomethacin (Pka 4. 5) in an intestinal tract buffered at PH 8?

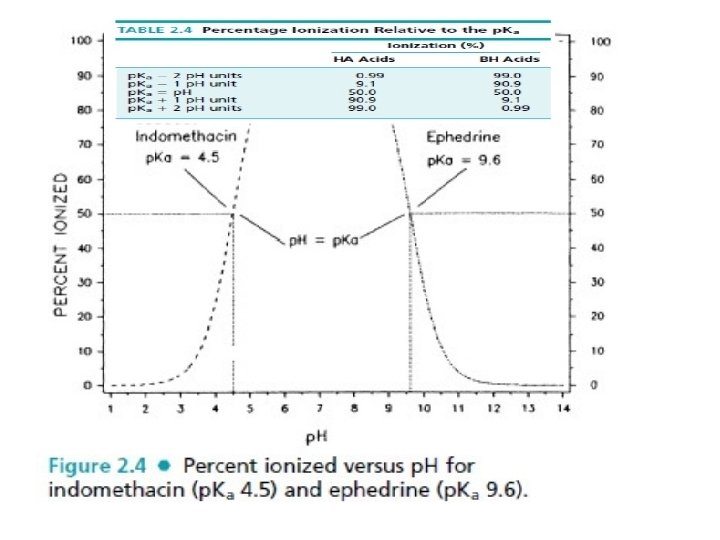
PH vs. PKa? PH= PKa Ø When the PH=PKa , the compound is 50% ionized (or 50% unionized). Ø When the PKa =PH , the molar concentration of the acid equals to molar concentration of its conjugate base. Ø When log �conjugate base�/[acid] =1

Phenytoin

Tropicamide

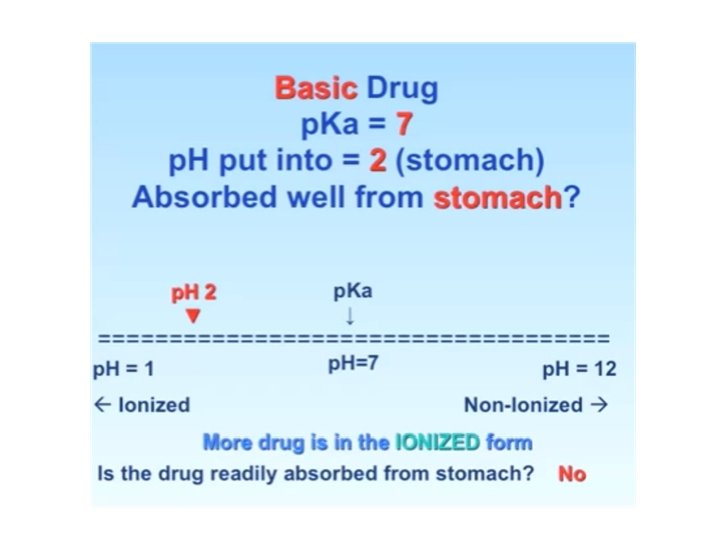
Drug Distribution and PKa Ø Acid drugs – become more NON ionized in acidic PH.
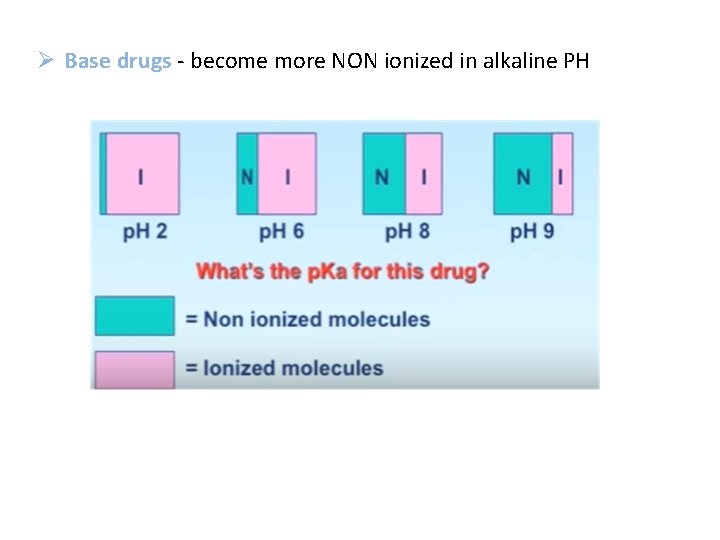
Ø Base drugs - become more NON ionized in alkaline PH

The main reason most acidic drugs absorbed from the intestinal tract rather than the stomach is that microvilli of intestinal mucosa provide a large surface area relative to that found in the gastric mucosa of the stomach



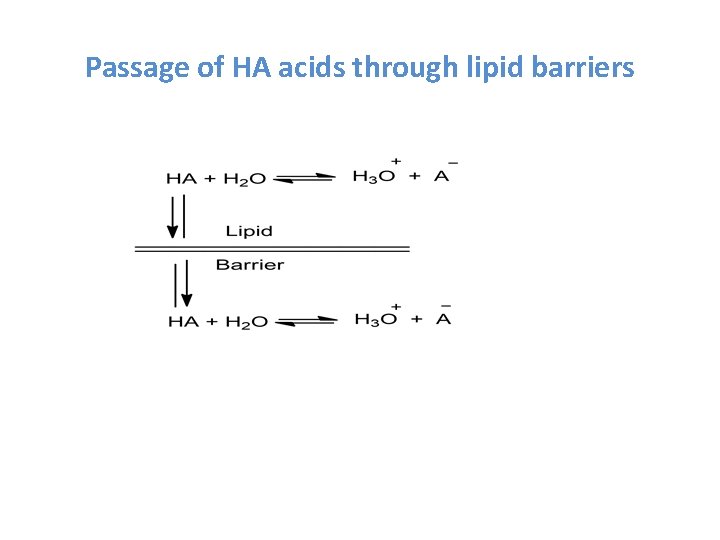
Passage of HA acids through lipid barriers
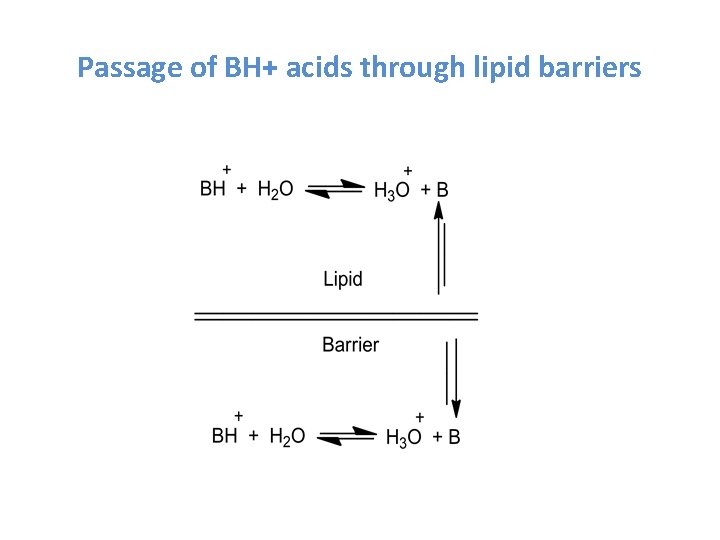
Passage of BH+ acids through lipid barriers

Summary Ø The higher Ka the stronger acid and the lower Ka weaker acid. Ø The lower Pka the stronger acid and the higher Pka weaker acid. Ø The stronger acid weaker conjugate base, the weaker acid higher conjugate base.

Home work 1. Why is Phenol considered corrosive to the skin , whereas ephedrine HCl or free ephedrine is considered innocuous when applied to the skin ?
- Slides: 33