MEDICATION UTILISATION EVALUATION MUE ON RIVAROXABAN USE IN

MEDICATION UTILISATION EVALUATION (MUE) ON RIVAROXABAN USE IN KHOO TECK PUAT HOSPITAL (KTPH) Jeremy Chia Pharmacist Event Name 10 Dec 2016 Date

Scope 2 A. B. C. D. E. F. Introduction to MUE and Rivaroxaban Objectives Methodology Results and Discussion Limitations Conclusion

3 A. Medication Utilisation Evaluation (MUE) What? 1 � An evaluation of the use of medications in a healthcare setting Why? 1 � Determine appropriateness of use of a particular medication in a healthcare setting � Review outcomes for safety and effectiveness of a particular medication � Ultimately: to improve patient’s quality of life 1 American Society of Health-System Pharmacists. ASHP guidelines on medication-use evaluation. Am Health-Syst Pharm. 1996; 53: 1953– 5. J

MUE For Rivaroxaban 4 Relatively new drug compared to warfarin Less need to monitor, less drug-drug or drug-food interactions But… No reversal agents (unlike warfarin) Expensive – may lead to non-compliance May be used inappropriately – not according to guidelines Need to evaluate the use of this newer drug to ensure adequate usage and monitoring

B. Objectives 5 Conduct MUE for Rivaroxaban 15 mg and 20 mg use in KTPH, to evaluate for: � Dose-appropriateness � Outcomes (for newly started patients) � Monitoring frequency

C. Methodology 6 Retrospective study Patients who collected at least 1 dose of Rivaroxaban 15 mg and/or 20 mg at KTPH Outpatient Pharmacy within 1 st Dec 2015 to 31 st Jan 2016 Information obtained from SCM and NEHR (hospital and national database records)
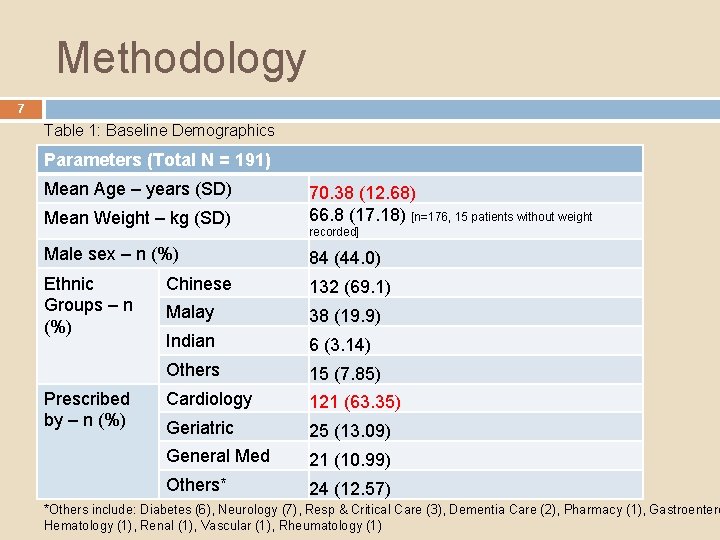
Methodology 7 Table 1: Baseline Demographics Parameters (Total N = 191) Mean Age – years (SD) Mean Weight – kg (SD) 70. 38 (12. 68) 66. 8 (17. 18) [n=176, 15 patients without weight Male sex – n (%) 84 (44. 0) Ethnic Groups – n (%) Chinese 132 (69. 1) Malay 38 (19. 9) Indian 6 (3. 14) Others 15 (7. 85) Cardiology 121 (63. 35) Geriatric 25 (13. 09) General Med 21 (10. 99) Others* 24 (12. 57) Prescribed by – n (%) recorded] *Others include: Diabetes (6), Neurology (7), Resp & Critical Care (3), Dementia Care (2), Pharmacy (1), Gastroentero Hematology (1), Renal (1), Vascular (1), Rheumatology (1)

Methodology 8 3 aspects are tabulated and discussed: � 1. Dose-appropriateness based on manufacturer’s standards for approved indications and renal function adjustments 2 � 2. Outcomes for patients newly started on Rivaroxaban from 1 st Apr 2015 to 31 st Jan 2016 � 3. Monitoring parameters and frequency appropriateness based on EHRA 2015 guidelines 3 2 Janssen Pharmaceuticals Inc. (2016, May). Xarelto (R) Package Insert. Titusville, NJ 3 Heidbuchel, H et al. (2015). Updated European Heart Rhythm Association Practical Guide on the use of non -vitamin K antagonist anticoagulants in patients with non-valvular atrial fibrillation. EUROPACE , 4 -7.

9 D. Results and Discussion
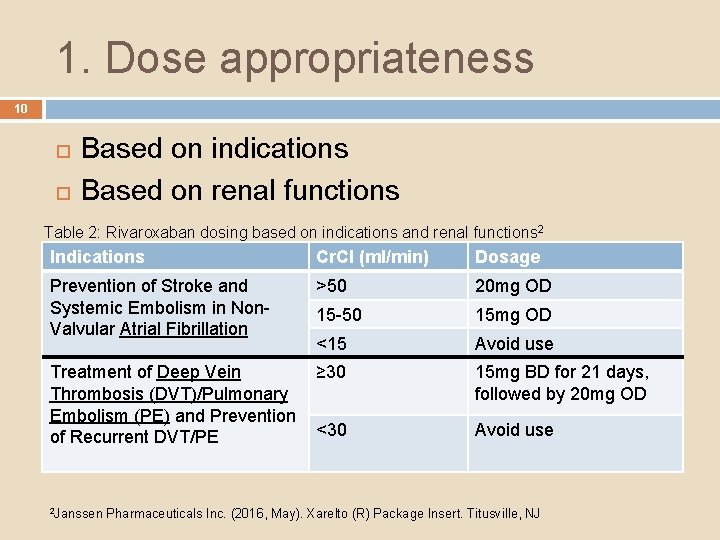
1. Dose appropriateness 10 Based on indications Based on renal functions Table 2: Rivaroxaban dosing based on indications and renal functions 2 Indications Cr. Cl (ml/min) Dosage Prevention of Stroke and Systemic Embolism in Non. Valvular Atrial Fibrillation >50 20 mg OD 15 -50 15 mg OD <15 Avoid use Treatment of Deep Vein ≥ 30 Thrombosis (DVT)/Pulmonary Embolism (PE) and Prevention <30 of Recurrent DVT/PE 15 mg BD for 21 days, followed by 20 mg OD Avoid use 2 Janssen Pharmaceuticals Inc. (2016, May). Xarelto (R) Package Insert. Titusville, NJ
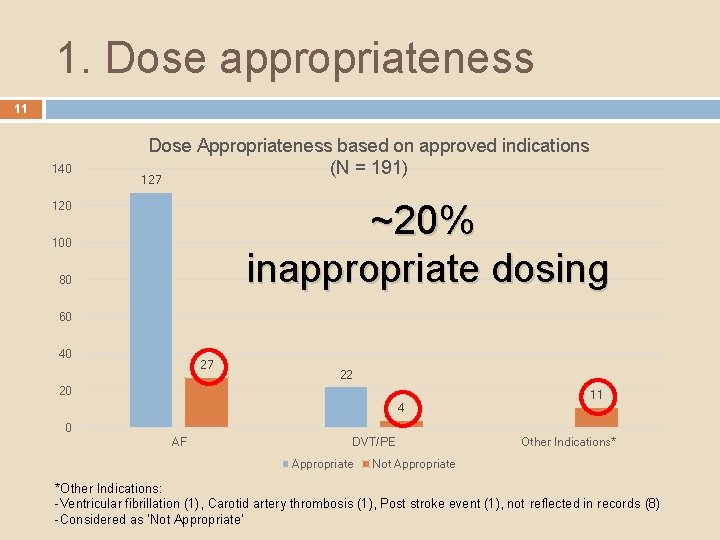
1. Dose appropriateness 11 140 Dose Appropriateness based on approved indications (N = 191) 127 ~20% inappropriate dosing 120 100 80 60 40 27 22 20 4 11 0 AF DVT/PE Appropriate Other Indications* Not Appropriate *Other Indications: -Ventricular fibrillation (1), Carotid artery thrombosis (1), Post stroke event (1), not reflected in records (8) -Considered as ‘Not Appropriate’

1. Dose appropriateness 12 Reasons for dose-inappropriateness (n = 42) 30 25 4 20 11 15 Inappropriate dosing for this indication (38%) 10 5 Not renallyadjusted (26%) 12 11 4 0 AF DVT/PE Inappropriate dose for indication *Others -AF: no s. Cr to determine appropriate or not (4) -Other Indications: considered as ‘Not Appropriate’ (11) Not renally adjusted Other Indications Others*

2. Outcomes 15 Outcomes measured for patients newly started from 1 st Apr 2015 to 31 st Jan 2016 Efficacy – reported thrombo-embolic events Safety – reported bleeding events
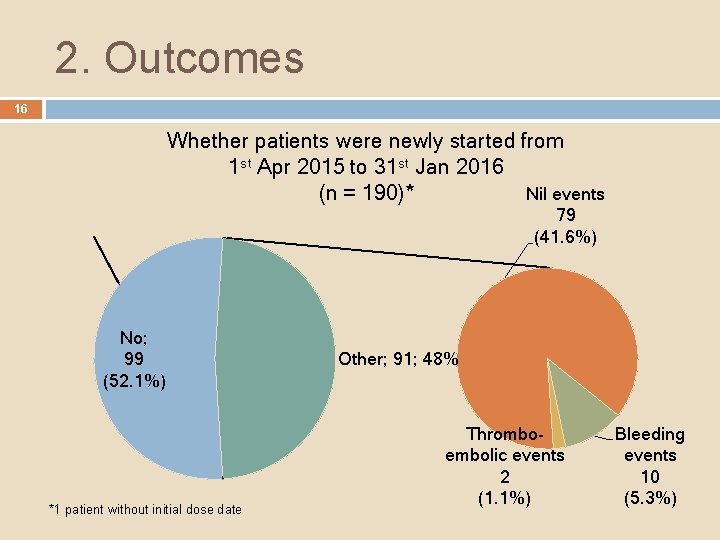
2. Outcomes 16 Whether patients were newly started from 1 st Apr 2015 to 31 st Jan 2016 Nil events (n = 190)* 79 (41. 6%) No; 99 (52. 1%) *1 patient without initial dose date Other; 91; 48% Thromboembolic events 2 (1. 1%) Bleeding events 10 (5. 3%)

2. Outcomes 17 Out of the 91 patients (100%) newly started between 1 st Apr 2015 to 31 st Jan 2016: � 2 patients (2. 2%) had reported thrombo-embolic incidents 1 patient had recurrent DVT 1 patient had lower limb pain 2 o to DVT: reported may be due to non-compliance because of cost � 10 patients (11. 0%) had reported bleeding incidents 4 patients had bleeding not directly due to Rivaroxaban use (from trauma [3], from cut [1]) 1 patient had increase bleeding risk (↑a. PTT and PT) without actual bleed

2. Outcomes 18 Bleeding outcomes in 10 patients (11. 0%), compared to that reported in clinical trials ROCKET-AF (14. 9%)4, and in EINSTEIN (9. 4%)5 � 8 patients for AF, 2 patient for DVT � Could be due to retrospective surveillance in this MUE, as compared to extensive observation during clinical trials � Number of patients recalled for this study was also much smaller than in those clinical trials 4 Patel MR, Mahaffey KW, Garg J et al (2011). Rivaroxaban versus Warfarin in Nonvalvular Atrial Fibrillation. New England Journal of Medicine (N ENGL J MED), 883 -891.

3. Monitoring Parameters 19 According to EHRA 2015 guidelines 3 � Blood Sampling (Renal functions and FBC) If ≥ 75 yo: monitor every 6 months If Cr. Cl < 60 ml/min: monitor at frequency of every (Cr. Cl/10) month Eg. Cr. Cl 40 ml/min, should monitor at every (40/10 = ) 4 months Assumption: Cr. Cl rounded to nearest 10 s to calculate for frequency � DDIs 3 Heidbuchel, H et al. (2015). Updated European Heart Rhythm Association Practical Guide on the use of non-vitamin

3. Monitoring Parameters 20 Assumption: � Monitoring parameters for non-valvular AF extend to recurrent DVT EHRA 2015 guidelines only meant for non-valvular AF
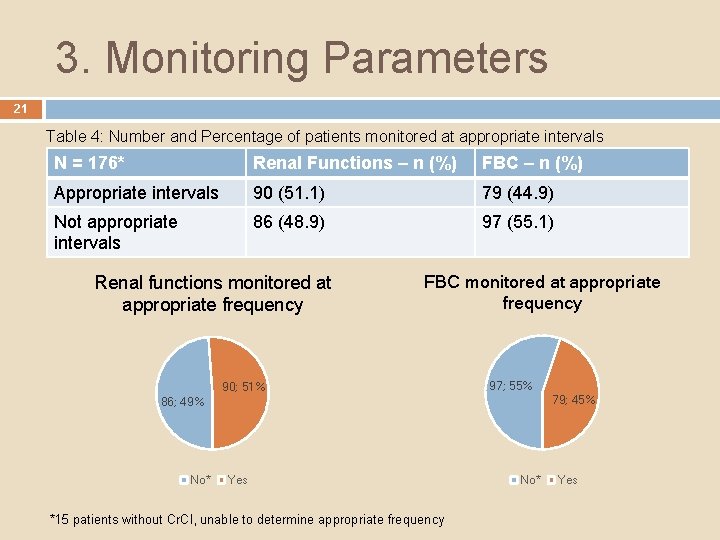
3. Monitoring Parameters 21 Table 4: Number and Percentage of patients monitored at appropriate intervals N = 176* Renal Functions – n (%) FBC – n (%) Appropriate intervals 90 (51. 1) 79 (44. 9) Not appropriate intervals 86 (48. 9) 97 (55. 1) Renal functions monitored at appropriate frequency FBC monitored at appropriate frequency 90; 51% 97; 55% 79; 45% 86; 49% No* Yes *15 patients without Cr. Cl, unable to determine appropriate frequency No* Yes
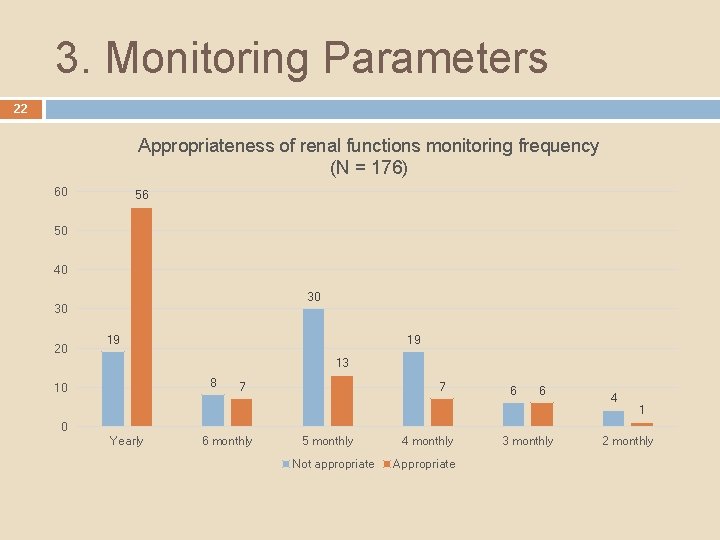
3. Monitoring Parameters 22 Appropriateness of renal functions monitoring frequency (N = 176) 60 56 50 40 30 30 20 19 19 13 8 10 7 7 6 6 4 1 0 Yearly 6 monthly 5 monthly Not appropriate 4 monthly Appropriate 3 monthly 2 monthly
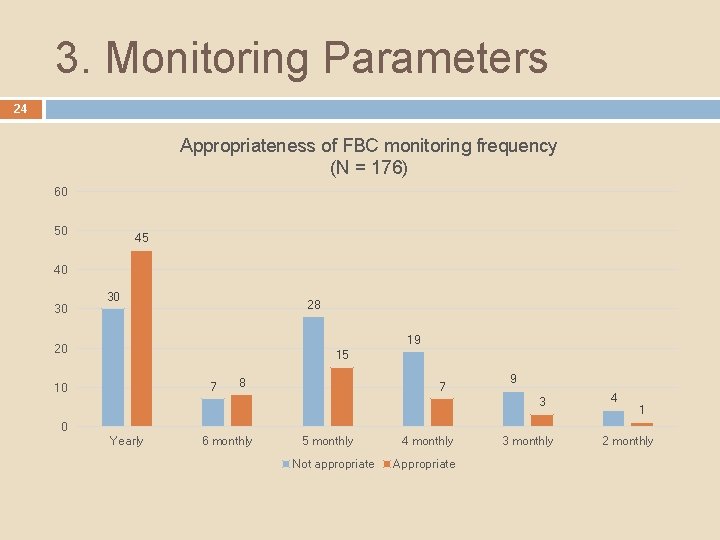
3. Monitoring Parameters 24 Appropriateness of FBC monitoring frequency (N = 176) 60 50 45 40 30 30 28 19 20 15 7 10 8 7 9 3 4 1 0 Yearly 6 monthly 5 monthly Not appropriate 4 monthly Appropriate 3 monthly 2 monthly
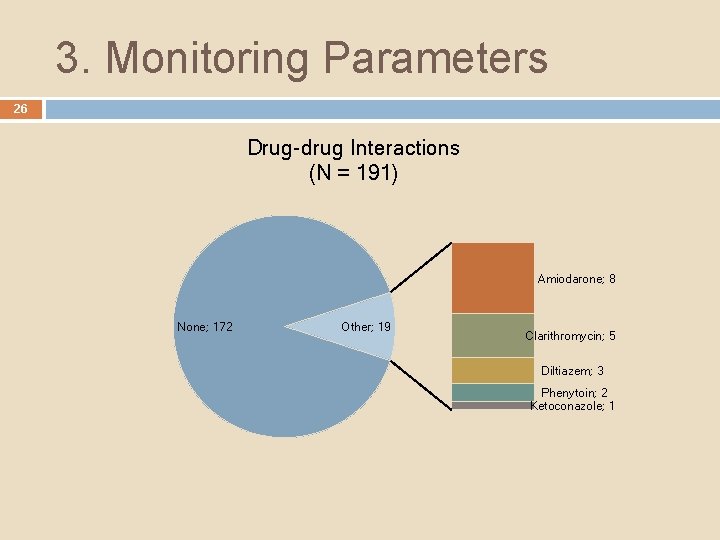
3. Monitoring Parameters 26 Drug-drug Interactions (N = 191) Amiodarone; 8 None; 172 Other; 19 Clarithromycin; 5 Diltiazem; 3 Phenytoin; 2 Ketoconazole; 1

3. Monitoring Parameters 27 Discussion � Only 60 patients (31. 4%) were monitored for BOTH renal functions and FBC at appropriate frequency � 19 (9. 9%) patients with DDIs 3 patients on Amiodarone and 2 patients on Diltiazem with Cr. Cl <50 ml/min: to be used with caution � Monitoring for renal functions and FBC coupled with appropriate dose adjustments is critical to avoid undesirable outcomes � Could be due to lack of awareness for the need to monitor for such parameters at such intervals

Compliance 28 20 random patients who are currently on Rivaroxaban were called and asked about compliance for the past 7 days � 19 patients had claimed compliance 7 patients had a family member to help administer � 1 patient was not compliant to prescribed instructions Experienced headache, self reduced dose to alternate days without doctor’s instructions Upon listening to friend dealing with TCM For months

Limitations/Future Directions 29 Minor thrombo-embolic events or bleeding events may not have been reported � Under-estimation of undesirable outcomes Monitoring parameters � PD interactions with Rivaroxaban may not be detected Under-estimation of DDI Future directions for project � Larger patient pool, surveying other NOACs � Compliance of patients � Transition between anticoagulants

Conclusion 30 Newer drugs such as Rivaroxaban and other NOACs are prone to inappropriate use and monitoring, leading to undesirable outcomes. Use of such drugs could be referred to pharmacist-led NOAC clinics, to better manage care of such patients.

31 Thank You!


1. Dose appropriateness 33 Assumptions: � Weight to calculate Cr. Cl based on current weight recorded in SCM, assumed to not be much different from first started Rivaroxaban � s. Cr to calculate Cr. Cl based on when Rivaroxaban was initially started. If not available, the next nearest s. Cr result when pt was on Rivaroxaban was used 34 patients (28. 3%) were not monitored for renal functions initially 1 out of these 34 patients did not have s. Cr result within 1 year prior to starting Rivaroxaban 3 out of these 34 patients do not have any s. Cr results
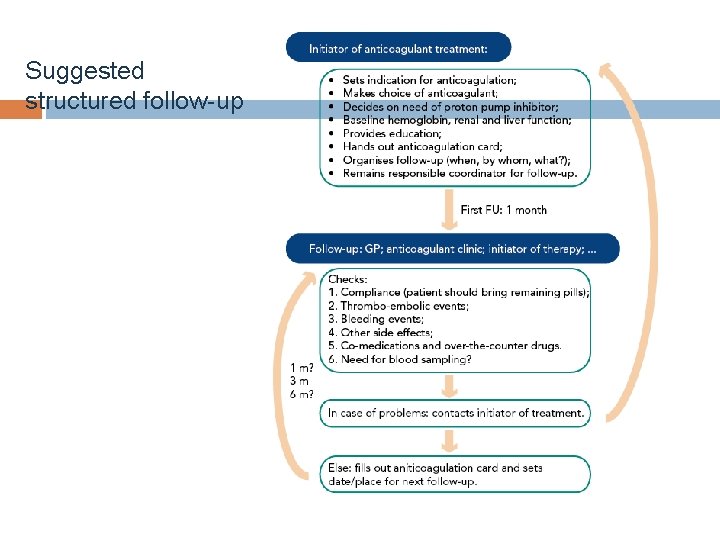
Suggested structured follow-up www. escardio. org/EHRA

At each follow-up session…. Interval Actions Compliance Each visit § Inspect remaining medication § Stress importance of compliance § Inform about compliance aids Thromboembolism Each visit Cerebral, systemic and pulmonary circulation Bleeding Each visit Side effects Each visit Co-medications Each visit § “Nuisance” bleeding – prevention possible? Try to keep on anticoag as much as possible. Any mitigation measures? § Bleeding with risk or impact on Qo. L – prevention possible? Need to revise dose? Continuation? Temporary cessation with bridging? Change of anticoagulant drug? § Prescription or over-the counter drugs, aspirin, NSAIDs § Even temporary use can be risky § Haemoglobin, renal, liver function § ≥ 75 – 80 yo, esp if on dabigatran or edoxaban; or frail# Yearly 6 -monthly § ≤ 60 ml/min: recheck interval = Cr. Cl/10 Blood sampling x-monthly § If inter-current condition may impact renal or hepatic On indication #Frailty: Three or more criteria of unintentional weight loss, self-reported exhaustion, weakness assessed by function. handgrip test, slow walking speed, or low physical activity http: //www. biomedcentral. com/1471 -2318/10/57 EHRA Practical Guide on the use of new oral anticoagulants in patients with non-valvular AF. Europace 2013; 15: 625 -51
- Slides: 31