Medically Ill Patient Assessment of Rivaroxaban Versus Placebo

Medically Ill Patient Assessment of Rivaroxaban Versus Placebo IN Reducing Post-Discharge Venous Thrombo-Embolism Risk (MARINER) Trial: Primary Results Alex C. Spyropoulos*, Walter Ageno, Gregory W. Albers, C. Gregory Elliott, Jonathan L. Halperin, William R. Hiatt, Gregory A. Maynard, P. Gabriel Steg, Jeffrey I. Weitz, Eunyoung Suh, Theodore E. Spiro, Elliot S. Barnathan, Gary E. Raskob* On behalf of the MARINER Investigators * Study Co-Chairs

Declaration of interest -Research contracts (Janssen, Boehringer lngelheim) - Consulting/Royalties/Owner/ Stockholder of a healthcare company (Janssen, Boehringer lngelheim, Portola, Bayer, ATLAS) ESC Congress Munich 2018 •

Background • A significant proportion of ~20 million patients in the US and EU hospitalized annually for medical illness remain at risk for venous thromboembolism (VTE) after hospital discharge, but the role of extended thromboprophylaxis is controversial – Over 400, 000 VTE and fatal PE events annually with an incidence of 3% in at-risk patients – The rate of symptomatic VTE more than doubles over the first 21 days and is associated with a five-fold increase of fatal PE up to 6 weeks post-discharge – Previous trials of extended thromboprophylaxis have shown either excess bleeding or benefit based on mainly reducing asymptomatic deep vein thrombosis • The MARINER trial was designed to optimize the benefit/risk profile of extended prophylaxis with rivaroxaban at discharge in an at-risk medically ill population using clinically meaningful endpoints – VTE enrichment strategy – Reduced dosing in subjects with moderate renal insufficiency/key exclusionary criteria Anderson FA, Jr. et al. Am J Hematol 2007; 82(9): 777 -82 Cohen AT et al. Thromb Haemost 2007; 98(4): 756 -64 Heit JA et al. Arch Intern Med 2002; 162(11): 1245 -8 Spyropoulos AC et al Chest 2011; 140: 706 -14 Guyatt GH, et al. Chest 2012; 141(suppl): e 185 s-e 194 s

Objectives Primary Objective Secondary Objectives • • Prevention of symptomatic venous thromboembolism (VTE: lower extremity deep vein thrombosis [DVT] and non-fatal pulmonary embolism [PE]) and VTE-related death (death due to PE or death in which PE cannot be ruled out as the cause) • • VTE-related death Symptomatic VTE The composite of symptomatic VTE and all-cause mortality The composite of symptomatic VTE, myocardial infarction, non-hemorrhagic stroke and CV death All-cause mortality Principal Safety Objective • Major bleeding using International Society of Thrombosis and Haemostasis (ISTH) bleeding criteria Secondary Safety Objective • Non-major clinically relevant bleeding
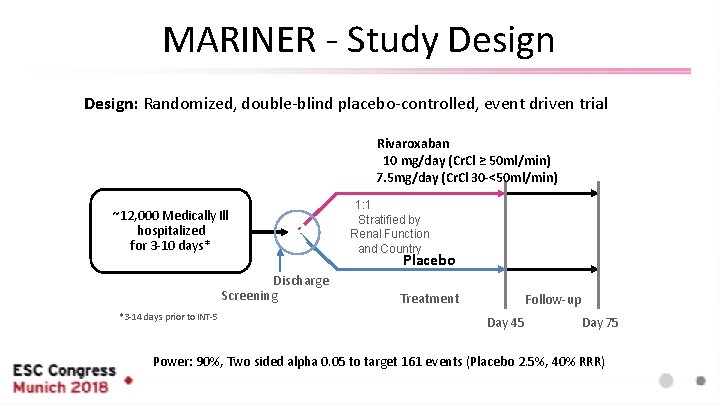
MARINER - Study Design: Randomized, double-blind placebo-controlled, event driven trial Rivaroxaban 10 mg/day (Cr. Cl ≥ 50 ml/min) 7. 5 mg/day (Cr. Cl 30 -<50 ml/min) ~12, 000 Medically Ill hospitalized for 3 -10 days* R Discharge Screening *3 -14 days prior to INT-5 1: 1 Stratified by Renal Function and Country Placebo Treatment Follow-up Day 45 Day 75 Power: 90%, Two sided alpha 0. 05 to target 161 events (Placebo 2. 5%, 40% RRR)

Key Inclusion and Exclusion Criteria Key inclusion criteria* u u Patients ≥ 40 years hospitalized for 3 -10 days with thromboprophylaxis (LMWH or UFH) prior to randomization for one of the following acute medical conditions u Heart failure u Acute respiratory insufficiency or acute exacerbation of COPD u Acute ischemic stroke u Acute infectious diseases u Inflammatory diseases, including rheumatic disease Total modified IMPROVE VTE Risk Score ≥ 4 OR total modified IMPROVE VTE Risk Score 2 or 3 and D dimer > 2 x ULN during index hospitalization * Reflects I/E Criteria as of INT-7 Key exclusion criteria* u u Bleeding Risks u Any bleeding within 3 months u Surgery, biopsy or trauma 4 weeks prior or planned u Active gastroduodenal ulcer u Active cancer Required anticoagulation after discharge Use of dual antiplatelet therapy during the index hospitalization Concomitant Medications u Combined P-gp and strong CYP 3 A 4 inhibitors u Combined P-gp and strong CYP 3 A 4 inducers
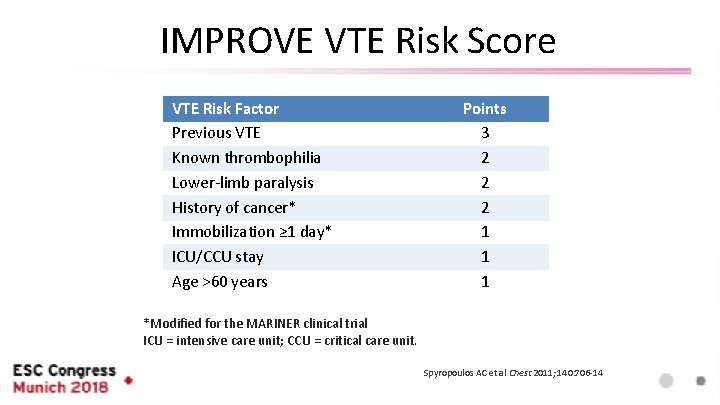
IMPROVE VTE Risk Score VTE Risk Factor Previous VTE Known thrombophilia Lower-limb paralysis History of cancer* Immobilization ≥ 1 day* ICU/CCU stay Age >60 years Points 3 2 2 2 1 1 1 *Modified for the MARINER clinical trial ICU = intensive care unit; CCU = critical care unit. Spyropoulos AC et al Chest 2011; 140: 706 -14
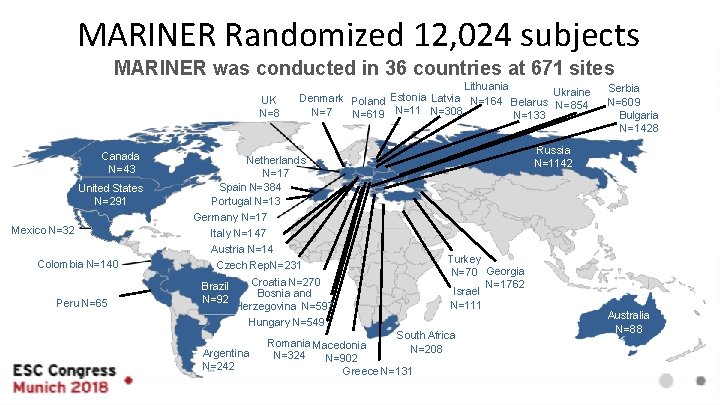
MARINER Randomized 12, 024 subjects MARINER was conducted in 36 countries at 671 sites UK N=8 Canada N=43 United States N=291 Mexico N=32 Colombia N=140 Peru N=65 Lithuania Ukraine Denmark Poland Estonia Latvia N=164 Belarus N=854 N=7 N=619 N=11 N=308 N=133 Netherlands N=17 Spain N=384 Portugal N=13 Germany N=17 Italy N=147 Austria N=14 Czech Rep. N=231 Brazil N=92 Croatia N=270 Bosnia and Herzegovina N=597 Hungary N=549 Argentina N=242 Serbia N=609 Bulgaria N=1428 Russia N=1142 Turkey N=70 Georgia N=1762 Israel N=111 South Africa Romania Macedonia N=208 N=324 N=902 Greece N=131 Australia N=88
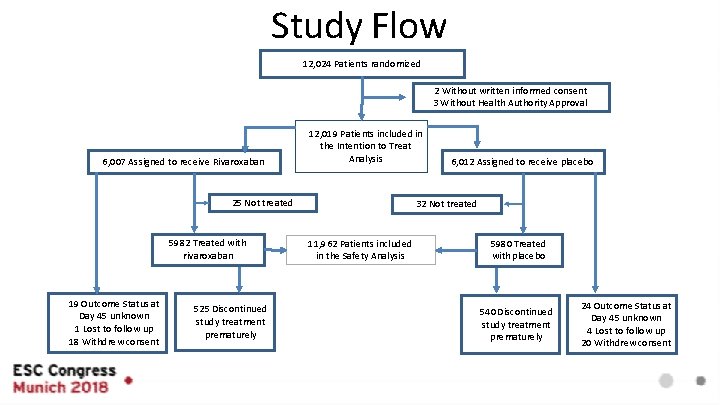
Study Flow 12, 024 Patients randomized 2 Without written informed consent 3 Without Health Authority Approval 6, 007 Assigned to receive Rivaroxaban 12, 019 Patients included in the Intention to Treat Analysis 25 Not treated 5982 Treated with rivaroxaban 19 Outcome Status at Day 45 unknown 1 Lost to follow up 18 Withdrew consent 525 Discontinued study treatment prematurely 6, 012 Assigned to receive placebo 32 Not treated 11, 962 Patients included in the Safety Analysis 5980 Treated with placebo 540 Discontinued study treatment prematurely 24 Outcome Status at Day 45 unknown 4 Lost to follow up 20 Withdrew consent
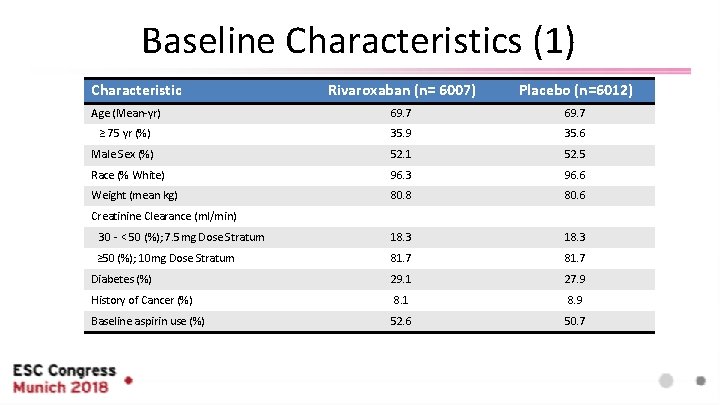
Baseline Characteristics (1) Characteristic Rivaroxaban (n= 6007) Placebo (n=6012) Age (Mean-yr) 69. 7 ≥ 75 yr (%) 35. 9 35. 6 Male Sex (%) 52. 1 52. 5 Race (% White) 96. 3 96. 6 Weight (mean kg) 80. 8 80. 6 30 - < 50 (%); 7. 5 mg Dose Stratum 18. 3 ≥ 50 (%); 10 mg Dose Stratum 81. 7 Diabetes (%) 29. 1 27. 9 History of Cancer (%) 8. 1 8. 9 Baseline aspirin use (%) 52. 6 50. 7 Creatinine Clearance (ml/min)
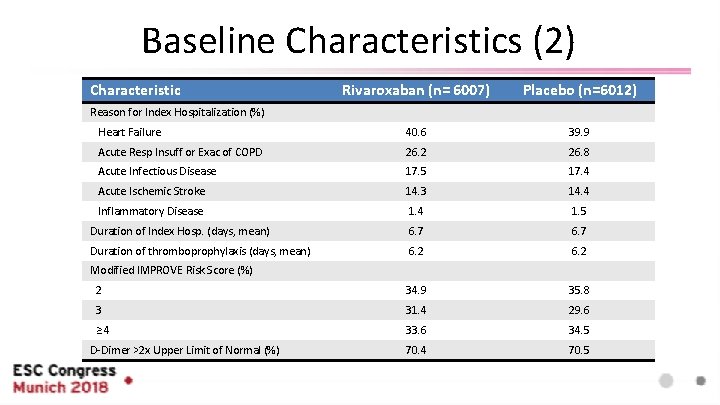
Baseline Characteristics (2) Characteristic Rivaroxaban (n= 6007) Placebo (n=6012) Heart Failure 40. 6 39. 9 Acute Resp Insuff or Exac of COPD 26. 2 26. 8 Acute Infectious Disease 17. 5 17. 4 Acute Ischemic Stroke 14. 3 14. 4 Inflammatory Disease 1. 4 1. 5 Duration of Index Hosp. (days, mean) 6. 7 Duration of thromboprophylaxis (days, mean) 6. 2 2 34. 9 35. 8 3 31. 4 29. 6 ≥ 4 33. 6 34. 5 70. 4 70. 5 Reason for Index Hospitalization (%) Modified IMPROVE Risk Score (%) D-Dimer >2 x Upper Limit of Normal (%)

Study Results
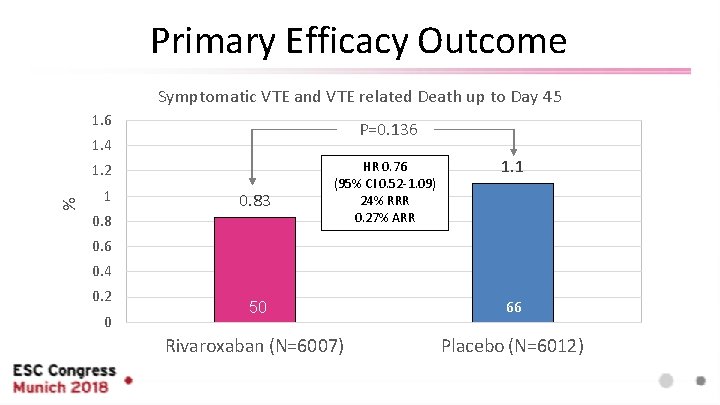
Primary Efficacy Outcome Symptomatic VTE and VTE related Death up to Day 45 1. 6 P=0. 136 1. 4 % 1. 2 1 0. 83 HR 0. 76 (95% CI 0. 52 -1. 09) 24% RRR 0. 27% ARR 1. 1 0. 6 0. 4 0. 2 0 50 66 Rivaroxaban (N=6007) Placebo (N=6012)
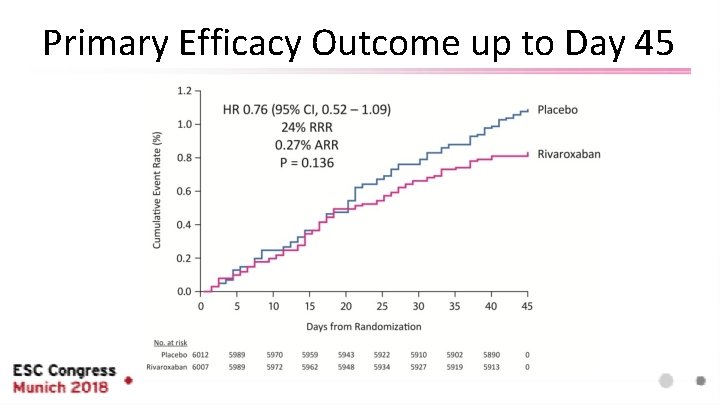
Primary Efficacy Outcome up to Day 45

Components of the Primary Efficacy Outcome up to Day 45 Outcomes Primary efficacy outcome (Sx VTE and VTE-related death) Symptomatic lower extremity DVT Symptomatic non-fatal PE VTE-related death Death (PE) Death (PE cannot be ruled out) Rivaroxaban (N=6007) Placebo (N=6012) n (%) Hazard Ratio (95% CI) [1] 50 (0. 83) 66 (1. 10) 0. 76 (0. 52, 1. 09) 0. 136 4 (0. 07) 13 (0. 22) 0. 31 (0. 10, 0. 94) 0. 039 7 (0. 12) 15 (0. 25) 0. 47 (0. 19, 1. 14) 0. 096 43 (0. 72) 46 (0. 77) 0. 93 (0. 62, 1. 42) 0. 751 3 (0. 05) 5 (0. 08) 0. 60 (0. 14, 2. 51) 0. 485 40 (0. 67) 41 (0. 68) 0. 98 (0. 63, 1. 51) 0. 912 Rivaroxaban vs Placebo p-value [2] 1 Hazard ratio (95% CI) and p-value are from the Cox proportional hazard model, stratified by baseline creatinine clearance (Cr. Cl) (30 -<50 m. L/min vs. ≥ 50 m. L/min), with treatment as the only covariate. 2 P-value (two-sided) for superiority of rivaroxaban versus placebo from the Cox proportional hazard model.
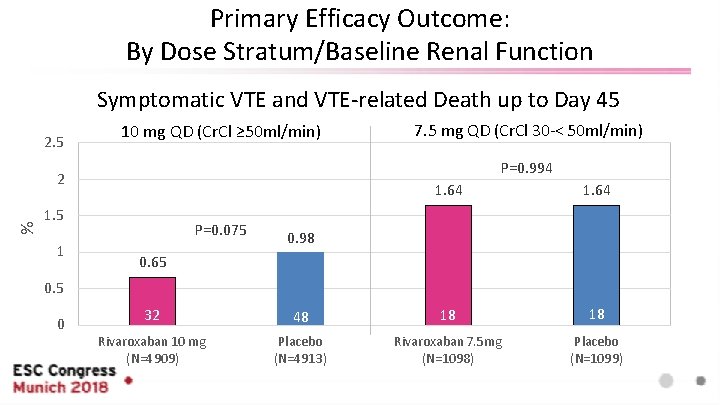
Primary Efficacy Outcome: By Dose Stratum/Baseline Renal Function Symptomatic VTE and VTE-related Death up to Day 45 2. 5 10 mg QD (Cr. Cl ≥ 50 ml/min) P=0. 994 % 2 1. 5 1 7. 5 mg QD (Cr. Cl 30 -< 50 ml/min) P=0. 075 1. 64 0. 98 0. 65 0. 5 0 32 48 18 18 Rivaroxaban 10 mg (N=4909) Placebo (N=4913) Rivaroxaban 7. 5 mg (N=1098) Placebo (N=1099)
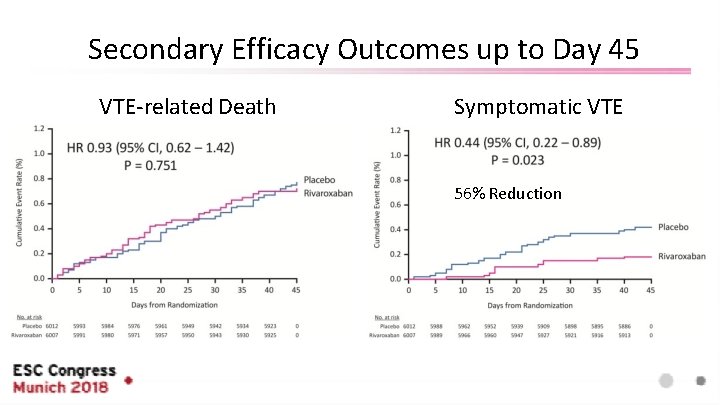
Secondary Efficacy Outcomes up to Day 45 VTE-related Death Symptomatic VTE 56% Reduction
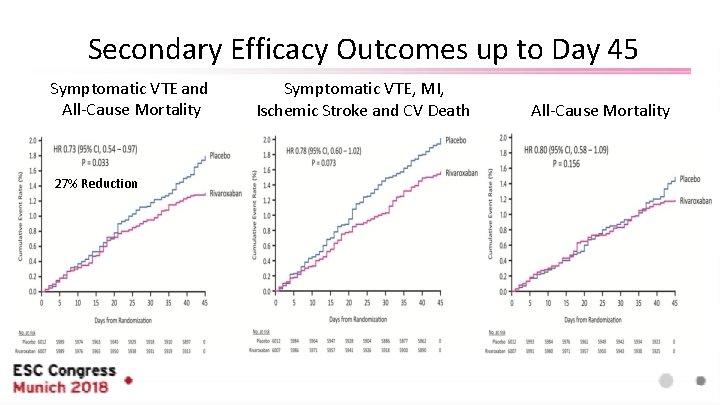
Secondary Efficacy Outcomes up to Day 45 Symptomatic VTE and All-Cause Mortality 27% Reduction Symptomatic VTE, MI, Ischemic Stroke and CV Death All-Cause Mortality

Bleeding Outcomes (On Treatment + 2 Days) Major bleeding Rivaroxaban (N=5982) Placebo (N=5980) n (%) Rivaroxaban vs Placebo Hazard Ratio (95% CI) [1] p-value [2] 17 (0. 28) 9 (0. 15) 1. 88 (0. 84, 4. 23) 0. 124 A fall in hemoglobin of >=2 g/d. L 14 (0. 23) 6 (0. 10) 2. 33 (0. 89, 6. 05) 0. 084 A transfusion of >=2 units of packed RBC 11 (0. 18) 3 (0. 05) 3. 66 (1. 02, 13. 10) 0. 047 A critical site 3 (0. 05) 2 (0. 03) 1. 50 (0. 25, 8. 97) 0. 657 A fatal outcome 2 (0. 03) 0 (0. 0) NA (NA, NA) 85 (1. 42) 51 (0. 85) 1. 66 (1. 17, 2. 35) Non-major clinically relevant bleeding 1 Hazard ratio (95% CI) and p-value are from the Cox proportional hazard model, stratified by baseline Cr. Cl (30 -<50 m. L/min vs. ≥ 50 m. L/min), with treatment as the only covariate. 2 P-value (two-sided) for superiority of rivaroxaban versus placebo from the Cox proportional hazard model. 0. 004

Conclusions – MARINER • Rivaroxaban did not significantly reduce the composite of symptomatic VTE and VTErelated death in an at-risk medically ill population post-hospital discharge (ARR=0. 27%) – There appeared to be no effect on VTE-related death • Secondary outcomes revealed: – A 56% reduction in symptomatic VTE – A 27% reduction in symptomatic VTE and all-cause mortality • • Rivaroxaban 10 mg in subjects without significant renal impairment (Cr. CL ≥ 50 ml/min) appeared more effective than reduced dose in subjects with moderate renal impairment The incidence of major bleeding with rivaroxaban was low (0. 28%) with no significant increase in major, critical, or fatal bleeding

The results of the MARINER study are now available at NEJM. org

Acknowledgements: Committees Clinical Events Committee Jim Douketis (Chair) Alexander G. Turpie, Sam Schulman Clive Kearon Lori-Ann Linkins Sebastian Schellong Independent Data Monitoring Committee Kenneth Bauer (Chair) William Geerts Robin Roberts National Lead Investigators Patricia Casais, Alexander Gallus, Jeff Karrasch, Sabine Eichinger-Hasenauer, Vitaly Krivenchuk, Almira Hadzovic-Dzuvo, Stevan Trbojevic, Renato Lopes, Valentina Mincheva, Marc Carrier, Rodolfo Dennis, Neven Tudoric, Jindrich Spinar, Henrik Nielsen, Toomas Marandi, Tamaz Shaburishvili, Jan Beyer-Westendorf, Panos Vardas, Zoltan Boda, Benjamin Brenner, Franco Piovella, Dainis Krievins, Birute Petrauskiene, Violeta Dejanova-Ilijevska, Luis Ramon Virgen Carrillo, Saskia Middeldorp, Reynaldo Pastor Castillo Leon, Adam Torbicki, Marta Saraiva de Sousa, Maria Dorobantu, Constantin Militaru, Igor Yavelov, Biljana Vuckovic, Jindrich Spinar, Helmuth Reuter, Matthys Basson, Manuel Monreal, Serdar Kucukoglu, Alexander Parkhomenko, Raza Alikhan, David Rosenberg, Roger Yusen, Alok Khorana, Victor Tapson, Charles Pollack, Monica Hazelrigg The >1000 MARINER Investigators and Coordinators and the > 12, 000 patients and their families
- Slides: 22